Abstract
Iloprost, an inhaled synthetic prostacyclin analogue, improves hemodynamic and clinical status with minimal systemic adversity in patients with pulmonary arterial hypertension. Our single-site, prospective case series aimed to determine the effects of iloprost in subjects with group 2 pulmonary hypertension and heart failure with preserved ejection fraction. Patients referred to Boston Medical Center for initial evaluation of suspected pulmonary hypertension received a test dose of 2.5 μg inhaled iloprost, followed by two subsequent doses of 5 μg. Hemodynamic measurements were recorded for each inhalation after 15, 30, 60, and 90 minutes. Results were analyzed via paired t test and signed-rank test. Eight subjects fulfilled criteria and elected to enter the study. There was a reduction of pulmonary arterial pressure (by an average of 7.0 mmHg [P = 0.005] and 4.7 mmHg [P = 0.021] with the first and second 5-μg inhalations, respectively) and pulmonary vascular resistance (by an average of 161.9 dyn· s/cm 5 [P = 0.019] and 95.0 dyn · s/cm 5 [P = 0.014] with the first and second 5-μg inhalations, respectively). There were trends for increased cardiac output and decreased oxygen saturation. There were no changes in other vital or hemodynamic parameters, including pulmonary capillary wedge pressure. All patients completed each cycle of iloprost administration without preestablished termination criteria. In patients with pulmonary hypertension and heart failure with preserved ejection fraction, inhaled iloprost resulted in acute reduction of pulmonary arterial pressure and pulmonary vascular resistance. Further evaluation of iloprost in this subset of patients is warranted.
In community-based studies, the echocardiographic prevalence of pulmonary arterial systolic pressure elevation above accepted community norms ranges from 6.6% to 28%.1–3 If further evaluated, nearly two-thirds of such individuals will have pulmonary venous hypertension from left heart disease (World Health Organization [WHO] group 2 pulmonary hypertension [PH]). 4 Existence of PH is a poor prognostic indicator among patients with left heart disease.5–7
Although there are extensive data with which to develop current guidelines for treatment of pulmonary arterial hypertension (PAH; WHO group 1), 8 there are few data regarding treatment of WHO group 2 PH. 9 Even fewer data exist for patients with pulmonary venous hypertension associated with left ventricular diastolic dysfunction (heart failure with preserved ejection fraction [HFpEF]). Treatment of patients with group 2 PH with systemic prostanoids is fraught with adverse effects and, in some cases, increased mortality. 10 However, systemic adverse effects may be reduced with inhaled prostacyclin analogues, compared with their parenteral counterparts. 11
Iloprost, an inhaled synthetic prostacyclin analogue, improves hemodynamic and clinical status with minimal systemic adversity in patients with PAH. 12 Because of these attributes, we evaluated the acute effects of iloprost in patients with group 2 PH associated with HFpEF.
MATERIAL AND METHODS
Subjects
The study was approved by the institutional review board of Boston Medical Center. Patients referred to the Pulmonary Hypertension Center at Boston Medical Center for initial evaluation of suspected PH were considered for inclusion. Initial screening criteria were age 21–90 years, New York Heart Association functional class III or IV, and an echocardiogram demonstrating a pulmonary artery systolic pressure >50 mmHg and left ventricular ejection fraction >50%. Exclusion criteria were pregnancy or lactation; significant valvular heart disease; clinically significant hypotension (systolic blood pressure <90 mmHg); thromboembolic disease; acute coronary syndrome; coagulopathy (international normalized ratio >1.5); allergy to prostacyclins; previous diagnosis of PH; and/or treatment with phosphodiesterase-5 inhibitors, endothelin receptor antagonists, or prostacyclin analogues or concomitant use of investigational drugs within the previous month. Eligible patients were informed of the study before diagnostic right heart catheterization and invited to participate if right heart catheterization revealed a mean pulmonary artery pressure (PAP) >25 mmHg, pulmonary capillary wedge pressure (PCWP) >18mmHg and <30 mmHg, and pulmonary artery diastolic pressure–PCWP gradient <10 mmHg. These criteria were chosen to restrict the population to patients in whom left heart disease (in this case, HFpEF) was responsible for the PH. The study initiated with signature of the informed consent form.
Study design and hemodynamic measurements Patients who met the inclusion criteria and agreed to participate received a test dose of inhaled iloprost (Ventavis; 2.5 μg) in a seated position over a 10-minute period via a Prodose automatic activation device. Preinhalation baseline variables were collected, including finger systemic arterial oximetry (SaO2); heart rate (HR); systolic blood pressure; diastolic blood pressure; mean arterial pressure (MAP); right atrial pressure; pulmonary artery systolic pressure; pulmonary artery diastolic pressure; PAP; PCWP; cardiac output (CO) and cardiac index by thermodilution; pulmonary vascular resistance (PVR); and systemic vascular resistance (SVR). Mixed venous oxygen saturation data were not collected. All vital and hemodynamic measurements were performed in the supine position.
The aforementioned hemodynamic measurements were recorded 15, 30, 60, and 90 minutes after inhalation. Patients were monitored throughout for predetermined hemodynamic changes (5-mm increase in PCWP, >10-mm decrease in MAP, or decrease in SaO2 to <90%) or intolerable adverse effects that would terminate the study. At 120 minutes after initial inhalation, when all parameters had returned to baseline, patients who tolerated the test dose of iloprost received an increased dose of 5 μg. The hemodynamic and clinical parameters were again measured at the same intervals after inhalation. At 120 minutes after this inhalation, when all hemodynamic and clinical parameters had again returned to baseline, patients who tolerated the 5-μg dose of iloprost received a second dose of 5 μg, and values were measured in a similar fashion.
The study was completed at the end of the second 5-μg cycle. Outpatient medications were adjusted as directed by the hemodynamic parameters measured at baseline.
Statistical analysis
Summary statistics, including the mean, median, standard deviation, and range were calculated for the initial 2.5-μg dose and each 5-μg dose cycle. The 95% confidence intervals for the mean are presented. Paired t tests were performed on change from baseline to 15 minutes along with the signed-rank test for confirmation. Analyses were performed using SAS, version 9.3. Statistical significance was assessed at the 0.05 level.
RESULTS
Nine consecutive patients met demographic and clinical conditions for study inclusion. Eight subjects fulfilled the hemodynamic criteria and elected to enter the study; one patient was excluded at right heart catheterization due to PCWP >30 mmHg. Of note, the transpulmonary gradient was >15 mmHg in 6 (75%) of 8 patients, and the PVR was >240 dyn· s/cm 5 in 5 (62.5%) of 8 patients. All patients had received a diagnosis of systemic hypertension, 3 (37.5%) of 8 had received a diagnosis of dyslipidemia, 3 (37.5%) of 8 had received a diagnosis of diabetes, 3 (37.5%) of 8 had received a diagnosis of obstructive sleep apnea, 2 (25%) of 8 had received a diagnosis of coronary artery disease, and 1 (12.5%) of 8 had received a diagnosis of atrial fibrillation. Additional baseline demographic and hemodynamic data are summarized in Table 1.
Baseline demographic and hemodynamic characteristics
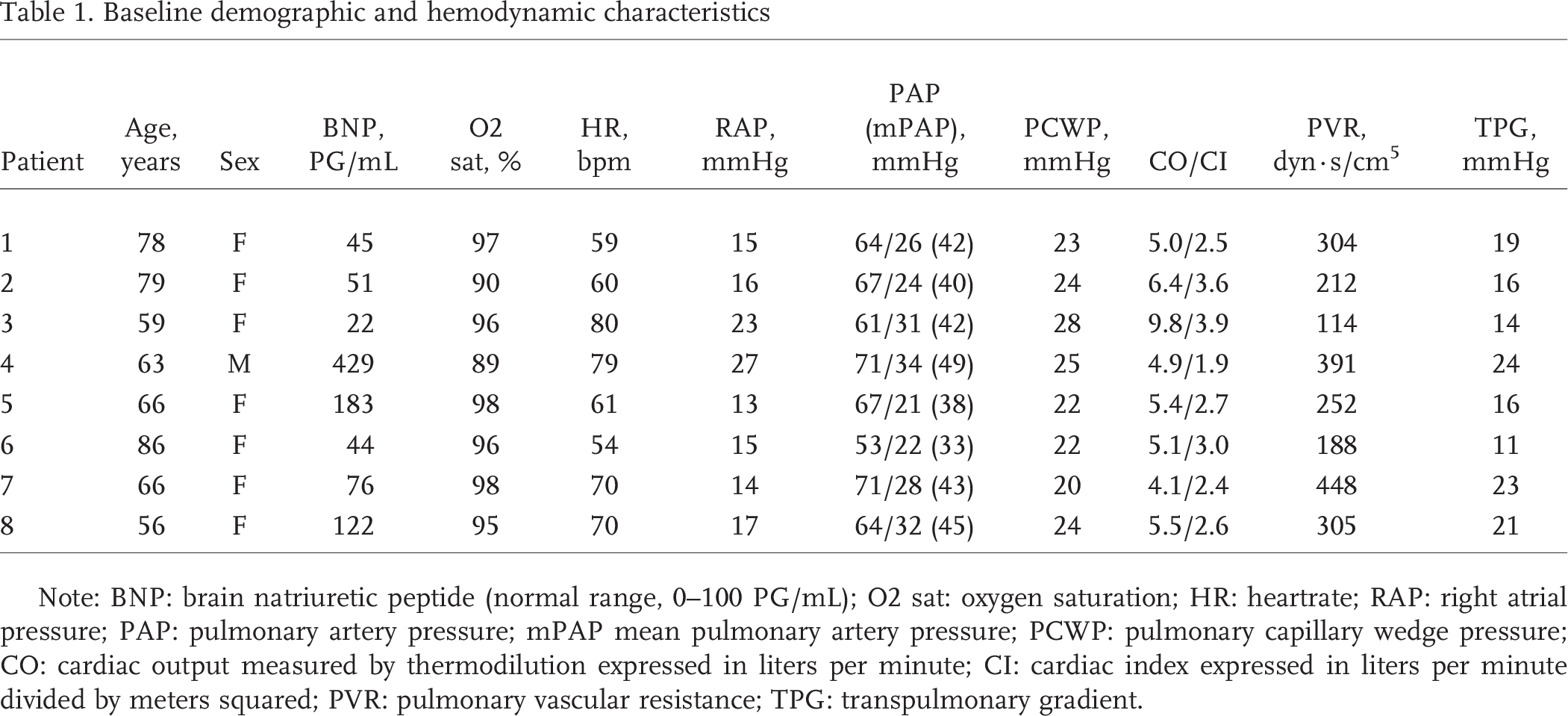
Note: BNP: brain natriuretic peptide (normal range, 0–100 PG/mL); O2 sat: oxygen saturation; HR: heartrate; RAP: right atrial pressure; PAP: pulmonary artery pressure; mPAP mean pulmonary artery pressure; PCWP: pulmonary capillary wedge pressure; CO: cardiac output measured by thermodilution expressed in liters per minute; CI: cardiac index expressed in liters per minute divided by meters squared; PVR: pulmonary vascular resistance; TPG: transpulmonary gradient.
Mean hemodynamic effects of iloprost inhalation are summarized in Figure 1. The greatest combined change from baseline in PAP and CO was observed soon after inhalation of iloprost at 15 minutes; hemodynamic changes were either less robust or insignificant at other time points. Cohort changes in MAP and PCWP were observed throughout the 90-minute observation period. Of note, the time of maximal change from baseline for each hemodynamic parameter varied among the individual subjects; this is highlighted by the standard error bars in Figure 1 and again summarized in Figures S1–S3 (available online).
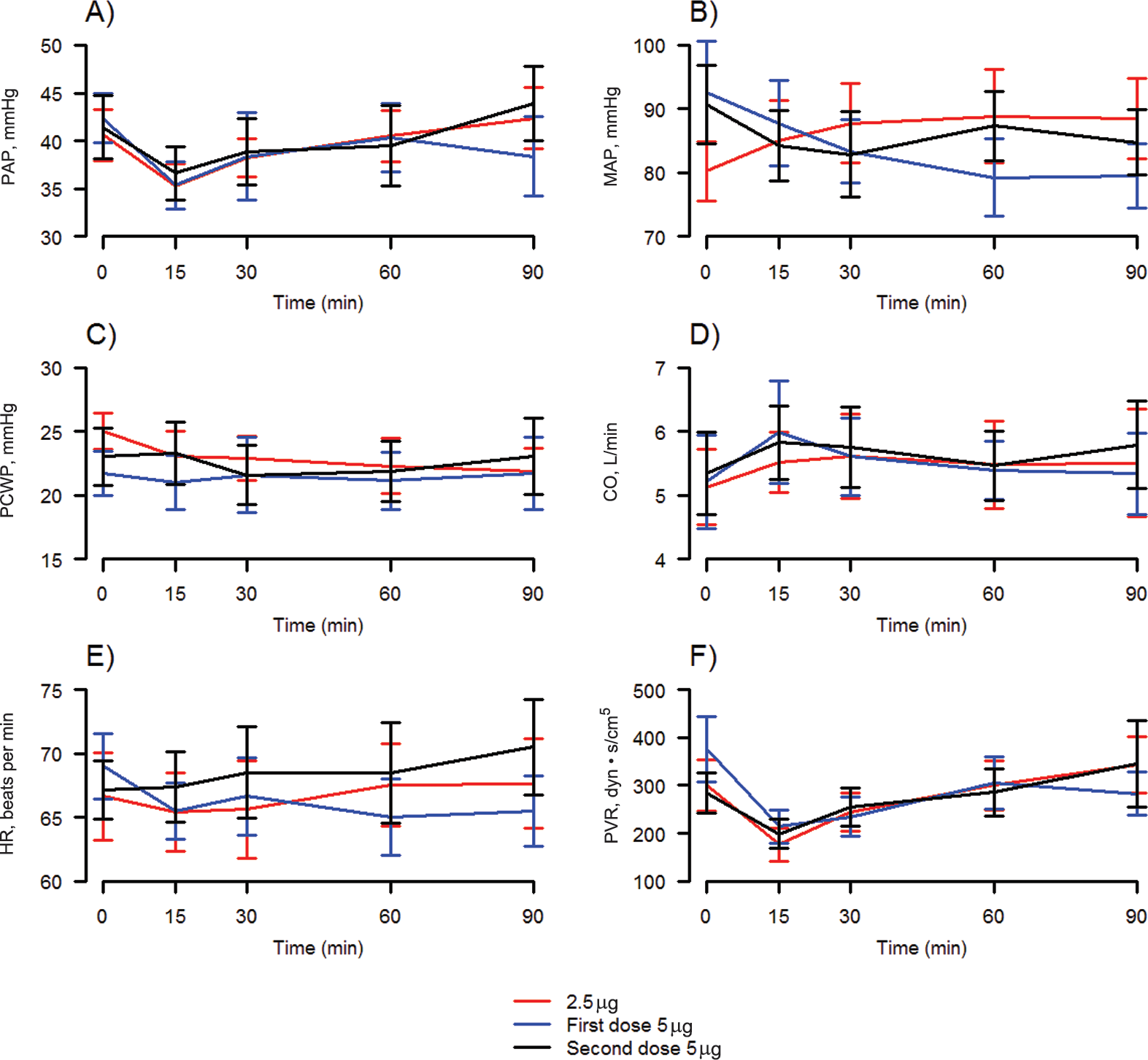
Mean hemodynamic effects of iloprost inhalation. CO: cardiac output; HR: heart rate; MAP: mean arterial pressure; PAP: mean pulmonary artery pressure; PCWP: pulmonary capillary wedge pressure; PVR: pulmonary vascular resistance.
Because of these observations, we targeted our analysis to 15 minutes after inhalation of iloprost (Table 2). Reduction of PAP and PVR were noted with all doses of iloprost, with the greatest effects observed after the first 5-μg inhalation: PAP and PVR decreased by an average of 7.0 mmHg (P = 0.005) and 161.9 dyn· s/cm 5 (P = 0.019), respectively, after the first 5-μg inhalation. They decreased by an average of 4.7 mmHg (P = 0.021) and 95.0 dyn · s/cm 5 (P = 0.014) after the second. There was a trend toward an increase in CO and a decrease in SaO2. No significant change was noted in HR, PCWP, or SVR.
Mean cohort change from baseline at 15 minutes after inhalation of iloprost
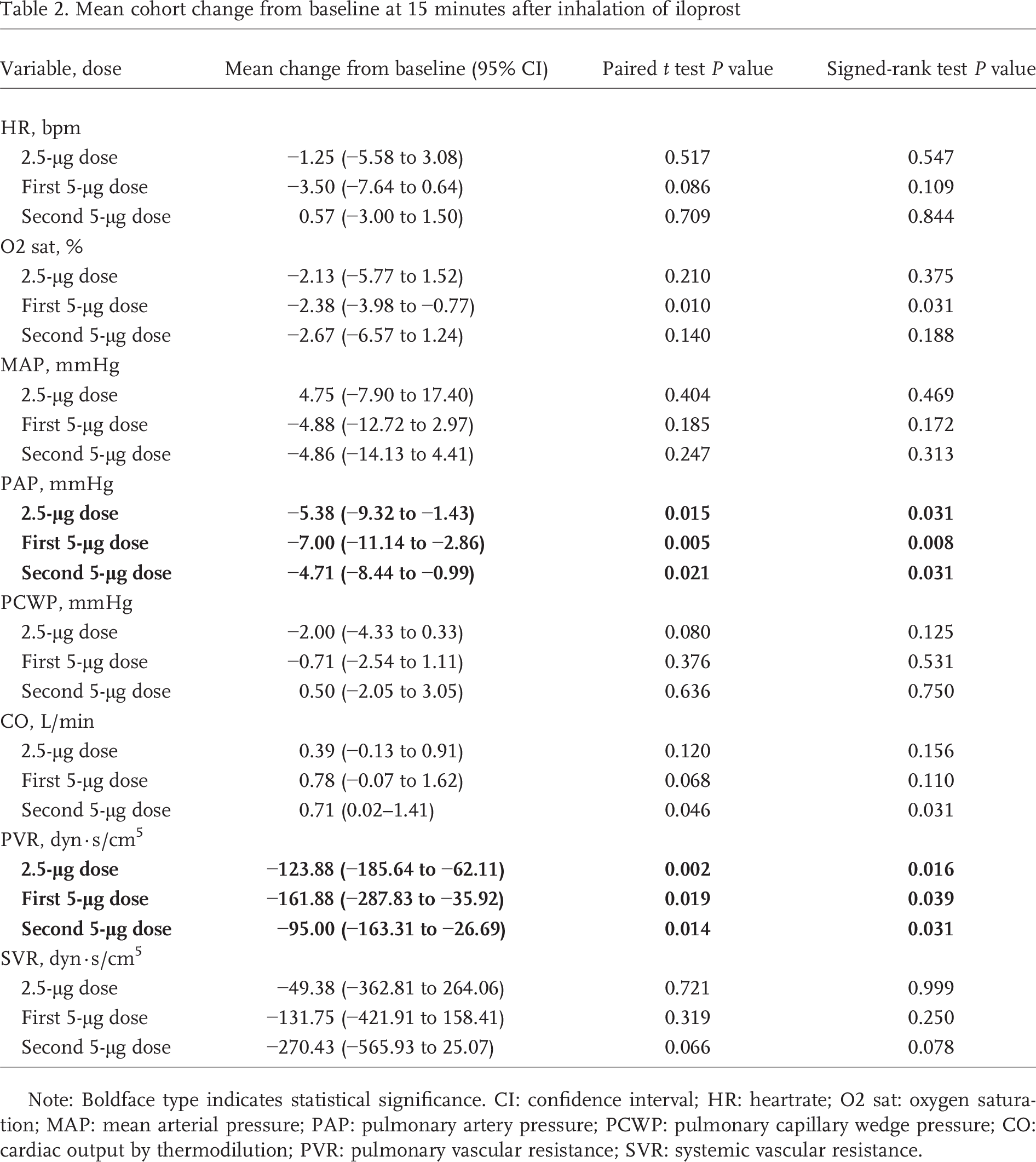
Note: Boldface type indicates statistical significance. CI: confidence interval; HR: heartrate; O2 sat: oxygen saturation; MAP: mean arterial pressure; PAP: pulmonary artery pressure; PCWP: pulmonary capillary wedge pressure; CO: cardiac output by thermodilution; PVR: pulmonary vascular resistance; SVR: systemic vascular resistance.
All patients completed the three cycles of iloprost administration; no patient experienced preestablished criteria or adverse effects sufficient to terminate the protocol with any dose of iloprost. The most commonly noted adverse effect was mild, self-limited headache.
DISCUSSION
In these subjects with PH and HFpEF, inhalation of iloprost caused a significant reduction in PAP and PVR after 15 minutes. There were trends toward an increase in CO and a decrease in SaO2. No effect on HR, PCWP, or SVR was observed. There were no limiting hemodynamic or clinical adverse effects.
Despite these promising observations, there are several limitations to the current study. First, because the patients represent a spectrum of the types of PH commonly seen in practice, they did not all have just one class of PH. Some had elevated PCWP alone, and others had both elevated PCWP and PVR. Second, although the referring physician considered each of the subjects to be euvolemic, the elevated PCWP in each of our subjects may be a reflection of inadequate diuresis. Third, the trend toward a decrease in SaO2 with iloprost inhalation might suggest a worsening in V/Q matching that must be further investigated with a larger cohort. Lastly, we investigated the acute effects of iloprost in only eight patients; the potential long-term adverse effects and hemodynamic benefits of iloprost on PAP, PVR, and vascular remodeling remain unknown in patients with PH associated with HFpEF. Long-term clinical and hemodynamic benefits of iloprost have been inconsistent, depending on the group of PH patients studied. Thus, further study of the acute and chronic effects of inhaled prostanoids on the cardiopulmonary hemodynamics and clinical status of euvolemic group 2 PH patients with HFpEF and elevated PCWP and PAP are merited to determine whether there is an effect on quality of life, risk of a clinical event, and morbidity and mortality.
Randomized clinical trials have demonstrated efficacy of therapy with phosphodiesterase-5 inhibitors, endothelin receptor antagonists, and prostanoids in group 1 PH; however, approved treatment of group 2 PH remains limited to primary treatment of the underlying cardiac disease. Few data suggest efficacy of advanced therapy in the treatment of group 2 PH, and even fewer data exist for those with pulmonary venous hypertension associated with HFpEF.
Studies that examine the efficacy and safety of phosphodiesterase-5 inhibition in patients with group 2 PH and patients with diastolic dysfunction (HFpEF) have reached conflicting conclusions. Single-center observational and randomized controlled cohort studies in patients with PH and HFpEF suggest that the phosphodiesterase-5 inhibitor sildenafil may reduce PAP and improve right ventricular function.13,14 However, the RELAX trial, a recent multicenter, prospective, randomized, placebo-controlled trial, demonstrated no benefit of sildenafil in patients with diastolic dysfunction and unknown PH status. 15 The RELAX trial also demonstrated a trend toward renal insufficiency and death in subjects who received sildenafil. Because of these conflicting results, further study is necessary to determine the potential role of phosphodiesterase-5 inhibition in patients with PH and HFpEF.
The safety and benefit of endothelin receptor antagonists in subjects with heart failure is also unclear. The Research on Endothelin Antagonism in Chronic Heart Failure (REACH) and Endothelin Antagonist Bosentan for Lowering Cardiac Events in Heart Failure (ENABLE) trials demonstrated no clinical improvement in patients with depressed systolic function and unknown PH status.16,17 In fact, both studies were prematurely stopped because of increased prevalence of adverse events (early risk of worsening heart failure necessitating hospitalization) in patients who received the endothelin receptor antagonist. There have been no trials to investigate the role of endothelin receptor antagonists in patients with PH associated with HFpEF.
Lastly, the potential use of systemic prostaglandins in patients with group 2 PH is still unknown, because intravenous epoprostenol has been associated with increased mortality in subjects with PH and reduced left ventricular systolic function. 10 To our knowledge, this is the first study to examine the potential role of an inhaled prostanoid, in this case iloprost, in the management of patients with PH associated with HFpEF.
The current pilot study suggests that alteration of the delivery (systemic vs. inhaled) of these molecules may make a substantial difference in their effect and tolerability. Our study is the first to demonstrate that inhaled iloprost results in acute reduction of PAP and PVR in patients with PH associated with HFpEF without effect on PCWP or SVR. Subjects tolerated the inhaled drug without significant adverse effects. Thus, iloprost may be both beneficial to and tolerated by patients with PH and HFpEF. Further study of iloprost in patients with PH associated with HFpEF over a longer time course is warranted to better determine the risks and benefits of inhaled prostanoid therapy in this population.
Footnotes
ACKNOWLEDGMENTS
We acknowledge the invaluable assistance of Cynthia Garcia, RN, who gathered and recorded the hemodynamic values for each patient.
