Abstract
Despite advances in the diagnosis and management of pediatric pulmonary hypertension (PH), children with PH represent a growing inpatient population with significant morbidity and mortality. To date, no studies have described the clinical characteristics of children with PH in the pediatric intensive care unit (PICU). A retrospective multicenter cohort study of 153 centers in the Virtual PICU Systems database who submitted data between 1 January 2009 and 31 December 2015 was performed. A total of 14,880/670,098 admissions (2.2%) with a diagnosis of PH were identified. Of these, 2190 (14.7%) had primary PH and 12,690 (85.3%) had secondary PH. Mortality for PH admissions was 6.8% compared to 2.3% in those admitted without PH (odds ratio = 3.1; 95% confidence interval = 2.9–3.4). Compared to patients admitted to the PICU without PH, those with PH were younger, had longer length of stay, higher illness severity scores, were more likely to receive invasive mechanical ventilation, cardiopulmonary resuscitation, extracorporeal membrane oxygenation, and more likely to have co-diagnoses of sepsis, heart failure, and respiratory failure. In a multivariate model, factors significantly associated with mortality for children with PH included age < 6 months or > 16 years, invasive mechanical ventilation, and co-diagnoses of heart failure, sepsis, hemoptysis, disseminated intravascular coagulation, stroke, and multi-organ dysfunction syndrome. Despite therapeutic advances, the disease burden and mortality of children with PH remains significant. Further investigation of the risk factors associated with clinical deterioration and mortality in this population could improve the ability to prognosticate and inform clinical decision-making.
Despite advances in the diagnosis and management of pulmonary vascular disease (PVD), children with pulmonary hypertension (PH) represent a growing inpatient population with significant morbidity and mortality.1,2 In the most recently revised classification system for pediatric PH, children are subclassified into groups based upon underlying etiology: pulmonary arterial hypertension (PAH; includes idiopathic, heritable, and associated with congenital heart disease [CHD]), left heart disease, lung disease or hypoxemia, chronic thromboembolic disease, and multifactorial or unclear. 3 Etiologies of PH among children are heterogeneous and vary significantly from those in adults with PH.4,5 Thus, pediatric-specific studies are needed to better characterize this population and assess factors associated with increased mortality.
Two recent US-based studies from an administrative dataset of pediatric hospital admissions (Kids' Inpatient Database [KID]) showed that children with PH had a higher average co-morbidity score and in-hospital all-cause mortality was significantly greater than for the general inpatient population.1,2 However, these studies did not describe the proportion of these patients admitted to an intensive care unit (ICU). It is critical to better understand the growing and evolving populations of infants and children with PH, especially those in an ICU setting, so strategies to improve outcomes can be identified and further investigated. Given the often life-threatening nature of PH and the likelihood for cardiorespiratory collapse if not managed quickly and appropriately, early identification of PH patients at particularly high risk may facilitate effective treatment and improve outcomes. 6
Predictors of worse outcomes for children with PH identified to date include higher baseline pulmonary vascular resistance index, higher B-type natriuretic peptide level, higher mean right atrial pressure, and lower cardiac index.7–15 Additionally, patients with PH have been shown to have greater morbidity and mortality associated with higher World Health Organization (WHO) functional class (FC)9,10,14 and Pulmonary Vascular Research Institute Functional Classification of Pulmonary Hypertension in Children (“Pediatric Functional Class”).16,17 In the inpatient pediatric PH population, sepsis, respiratory failure, acute renal failure, hepatic insufficiency, arrhythmias, and the use of extracorporeal membrane oxygenation (ECMO) were identified as predictors of mortality. 2
Given that pediatric PH is a relatively rare disease, with yearly incidence rates of 64 cases per million children, the number of patients cared for at any individual institution is relatively small. 18 In order to investigate the above-described gaps in knowledge, the use of a multicenter pediatric ICU (PICU) dataset is crucial. 5 In this retrospective multicenter cohort study, we aim to describe critical care interventions, morbidity and mortality for pediatric PH patients requiring admission to an ICU.
Methods
Study design
This retrospective multicenter cohort study uses data from the Virtual PICU Systems (VPS), LLC database. VPS is a comprehensive PICU database that prospectively tracks information about demographics, diagnoses, procedures, outcomes, and illness severity scores. A total of 153 PICUs, most of which are located in the United States, contribute to VPS. Data are entered at each site by trained VPS analysts. Extensive data quality control measures are performed. Institutional review board approval by the University of California San Francisco was obtained.
Data collection and analysis
The database query identified 498,103 patients aged under 21 years who were admitted to the PICU (670,098 admissions) between 1 January 2009 and 31 December 2015. Admissions associated with a diagnosis of primary or secondary PH were identified by querying VPS diagnosis codes and International Classification of Diseases, Ninth Revision (ICD9) codes (ICD9 416 or ICD9 416.8, respectively). The ICD9 codes for pediatric PH have not yet been updated according to the 2013 Nice Classification of PH3 and remain “primary pulmonary hypertension” (refers to idiopathic or isolated PH) and “secondary pulmonary hypertension” (refers to PH with an identifiable underlying etiology). Newborns with exclusive diagnoses of persistent pulmonary hypertension of the newborn (PPHN) or those with both a diagnosis of PPHN and secondary PH under 30 days of age at time of admission were excluded from this analysis.
For this study, a simplified 2013 Nice Classification of PH 3 was used to assign an underlying cause to cases with secondary PH: CHD only (Group 1); respiratory disorders only (Group 3); CHD and respiratory disorder (unable to differentiate between Groups 1 and 3); and other (Group 4). Left heart disease (Group 2) was not included as a separate category given that it was not possible to differentiate these cases. For patients with more than one PH diagnosis (i.e. primary and secondary PH), a study investigator reviewed all additional diagnoses, patient age, and interventions performed, and the single PH diagnosis thought to be most appropriate was selected and applied to all admissions for that patient record. Additionally, if a patient carried a diagnosis of primary PH and a diagnosis of CHD, an investigator reviewed the case to determine if the case was more likely secondary PH associated with CHD and, if so, these admissions were reclassified as such.
Outcomes
Primary outcome measure is PICU mortality, defined as death before PICU transfer or discharge. Secondary outcomes included cardiopulmonary resuscitation (CPR), invasive mechanical ventilation, and central line placement.
Predictors
Predictor variables included diagnosis of and type of PH (primary vs. secondary), patient factors (age and sex), critical care interventions, co-diagnoses, and standardized illness severity scores. Critical care interventions were grouped based on widely accepted terminology and included invasive mechanical ventilation, endotracheal intubation, non-invasive mechanical ventilation (continuous or bilevel positive airway pressure), renal support (renal replacement therapy or dialysis), nitric oxide, CPR, ECMO, arterial lines, and central lines. Co-diagnoses assessed included arrhythmia, heart failure, sepsis, respiratory failure, hemoptysis/pulmonary hemorrhage, disseminated intravascular coagulation (DIC), intracranial hemorrhage (ICH) or stroke, and multi-organ dysfunction syndrome (MODS). Illness severity scores included the Pediatric Index of Mortality-2 (PIM-2) probability of death (POD) 19 and the Pediatric risk of Mortality-3 (PRISM-3) raw score with POD,20,21 calculated within the first hour and first 12 hours of PICU admission, respectively. Age was stratified into six categories based upon those used in the Pediatric FC guidelines (0–6 months, > 6–12 months, > 12 months–2 years, > 2–5 years, > 5–16 years, and > 16 years). 17 As documentation of several variables was optional for centers (including PRISM-3, non-invasive mechanical ventilation, nitric oxide, and renal support), predictor-specific denominators are reported accordingly and analyses of these variables excluded centers not collecting the respective variable.
Statistics
Comparisons between distributions of categorical variables were performed by chi-square tests. Continuous normally distributed variables were presented as mean and standard deviation (SD) and compared by t-tests. Non-normally distributed continuous data were presented in median and interquartile range (IQR) and compared with Wilcoxon rank-sum tests. Univariate and multivariate logistic regression models were used to analyze the independent effect of predictors on PICU mortality. Factors for inclusion in the multivariate analysis were chosen a priori. Non-independence for multiple admissions of the same patients was accounted for by clustering by unique patient identifier code. Results were presented in odds ratios (OR) and 95% confidence intervals (CI). P < 0.05 was considered statistically significant. Data analysis was performed using Stata (version 13, StataCorp, College Station, TX, USA).
Results
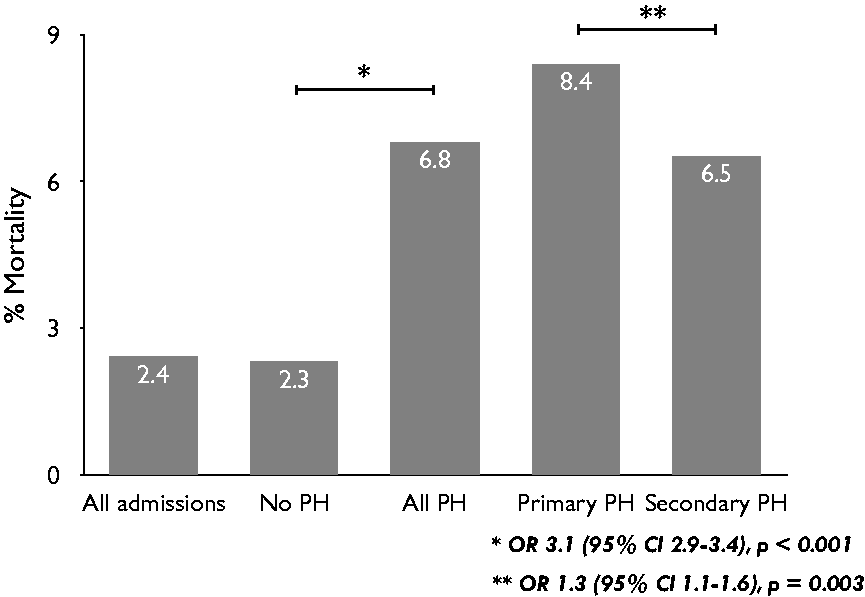
A total of 7573 of 498,103 patients (1.5%) were identified with a diagnosis of PH. These patients accounted for 2.2% of all admissions (14,880/670,098) (Fig. 1). Median number of admissions per patient was one (range = 1–38; IQR = 1–2). Of all PH admissions, 2190 (14.7%) were classified as primary PH and 12,690 (85.3%) as secondary PH (Fig. 1). PH admissions had a 6.8% mortality rate compared with 2.3% mortality for those admitted without PH (OR = 3.1; 95% CI = 2.9–3.4). PH patients accounted for 6.4% (1013/15,908) of all PICU mortalities. Mortality for admissions with primary PH was 8.4% compared to 6.5% for those with secondary PH (OR = 1.3; 95% CI = 1.1–1.6) (Fig. 2).
Composition of study population. PH, pulmonary hypertension; PPHN, persistent pulmonary hypertension of the newborn. Mortality by admission type. PH, pulmonary hypertension; OR, odds ratio.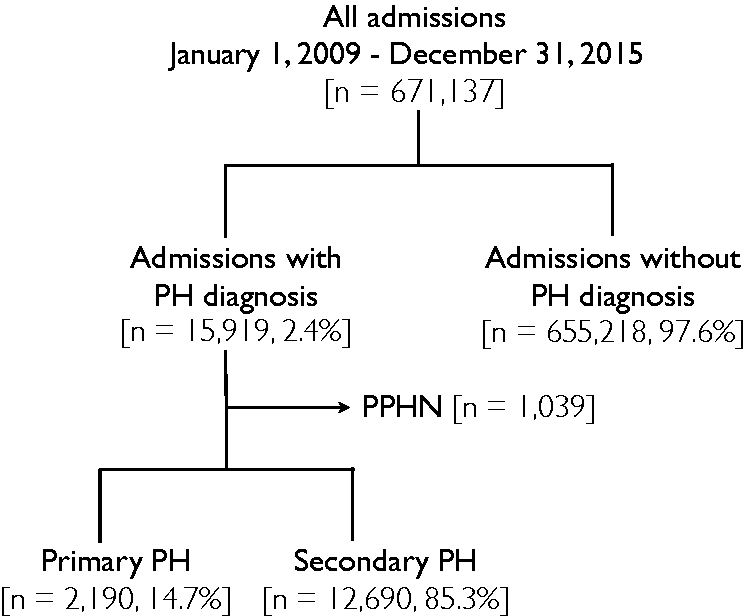
Characteristics of admissions with and without PH.
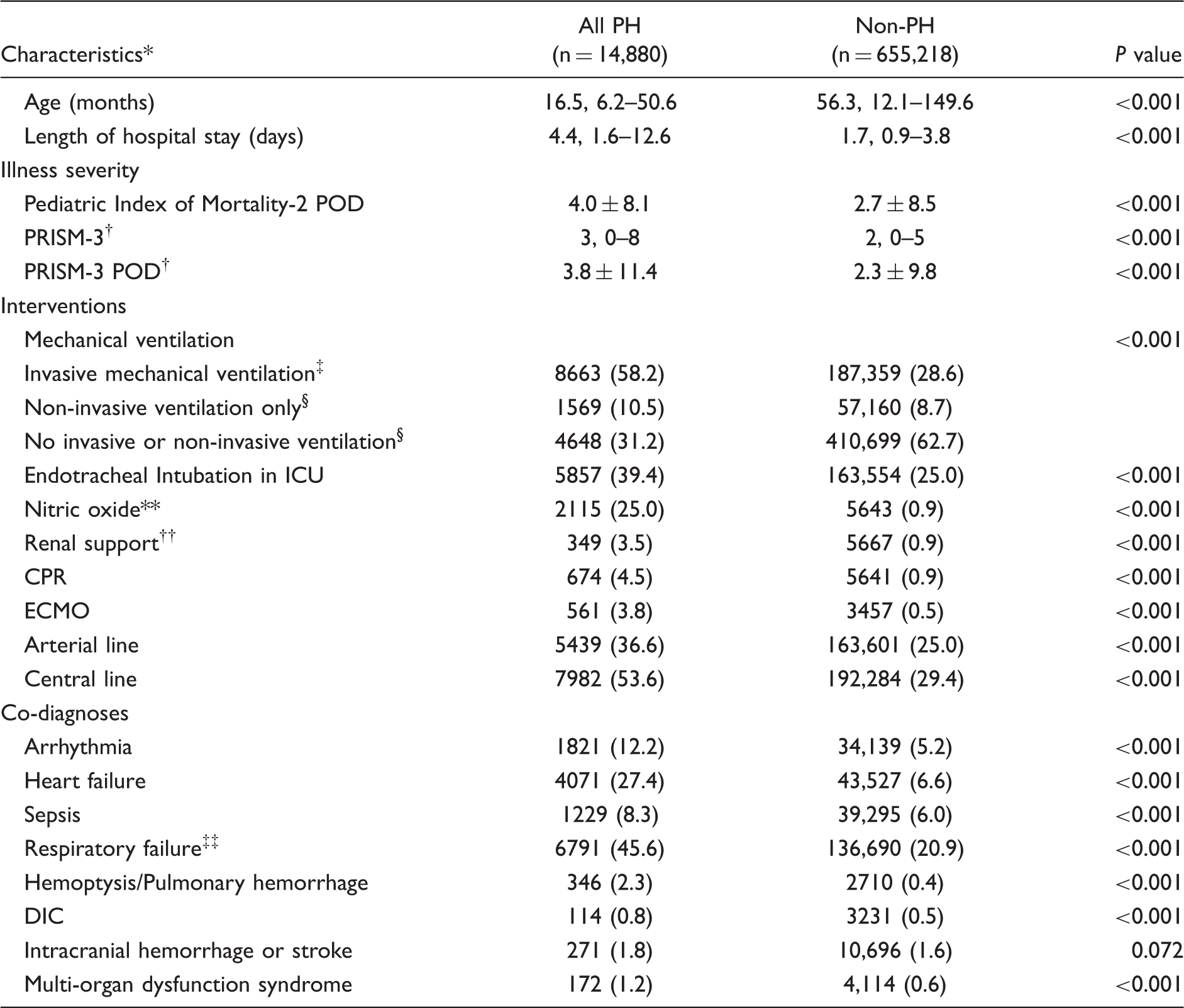
Results displayed as median, IQR; n (%); or mean ± SD.
Missing in 743 and 36,013 cases for PH and non-PH patients, respectively.
Includes “Endotracheal intubation,” “Mechanical ventilation, conventional,” and “HFOV (high frequency oscillator ventilation).”
Includes “BiPAP,” “CPAP,” and “Humidified high flow oxygen.” Non-invasive mechanical ventilation n = 10,108 for PH patients, n = 453,179 for non-PH patients, not reported at all centers.
Nitric oxide n = 8473 for PH patients, n = 340,903 for non-PH patients, not reported at all centers.
Renal support, defined as continuous renal replacement therapy or dialysis, n = 9862 for PH patients, n = 402,930, not reported at all centers.
Defined as “Pulmonary insufficiency/Shock lung, Adult respiratory distress syndrome (ARDS), Acute hypoxic respiratory failure (AHRF), Acute lung injury (ALI).”
PH, pulmonary hypertension; POD, probability of death; PRISM, Pediatric Risk of Mortality; ICU, intensive care unit; CPR, cardiopulmonary resuscitation; ECMO, extracorporeal membrane oxygenation; DIC, disseminated intravascular coagulation.
Univariate predictors of mortality in children with PH, by admission (n = 14,880).
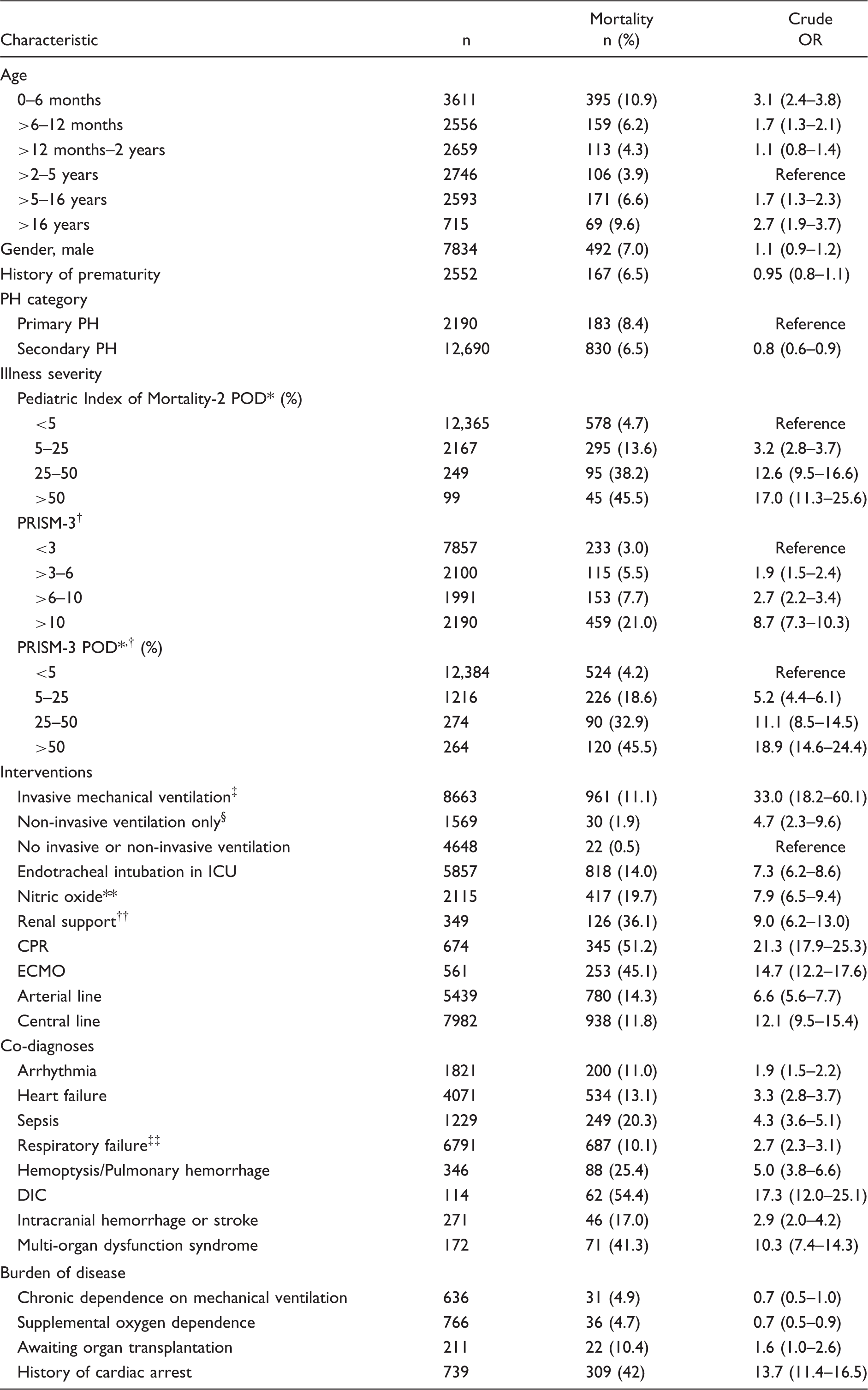
% mortality scores.
Missing in 743 and 36,013 cases for PH and non-PH patients, respectively.
Includes “Endotracheal intubation,” “Mechanical ventilation, conventional,” and “HFOV (high frequency oscillator ventilation).”
Includes “BiPAP,” “CPAP,” and “Humidified high flow oxygen.” Non-invasive mechanical ventilation n = 10,108 for PH patients, n = 453,179 for non-PH patients, not reported at all centers.
Nitric oxide n = 8473 for PH patients, n = 340,903 for non-PH patients, not reported at all centers.
Renal support, defined as continuous renal replacement therapy or dialysis, n = 9862 for PH patients, n = 402,930, not reported at all centers.
Defined as “Pulmonary insufficiency/Shock lung, Adult respiratory distress syndrome (ARDS), Acute hypoxic respiratory failure (AHRF), Acute lung injury (ALI).”
OR, odds ratio; PH, pulmonary hypertension; POD, probability of death; PRISM, Pediatric Risk of Mortality; ICU, intensive care unit; CPR, cardiopulmonary resuscitation; ECMO, extracorporeal membrane oxygenation; DIC, disseminated intravascular coagulation.
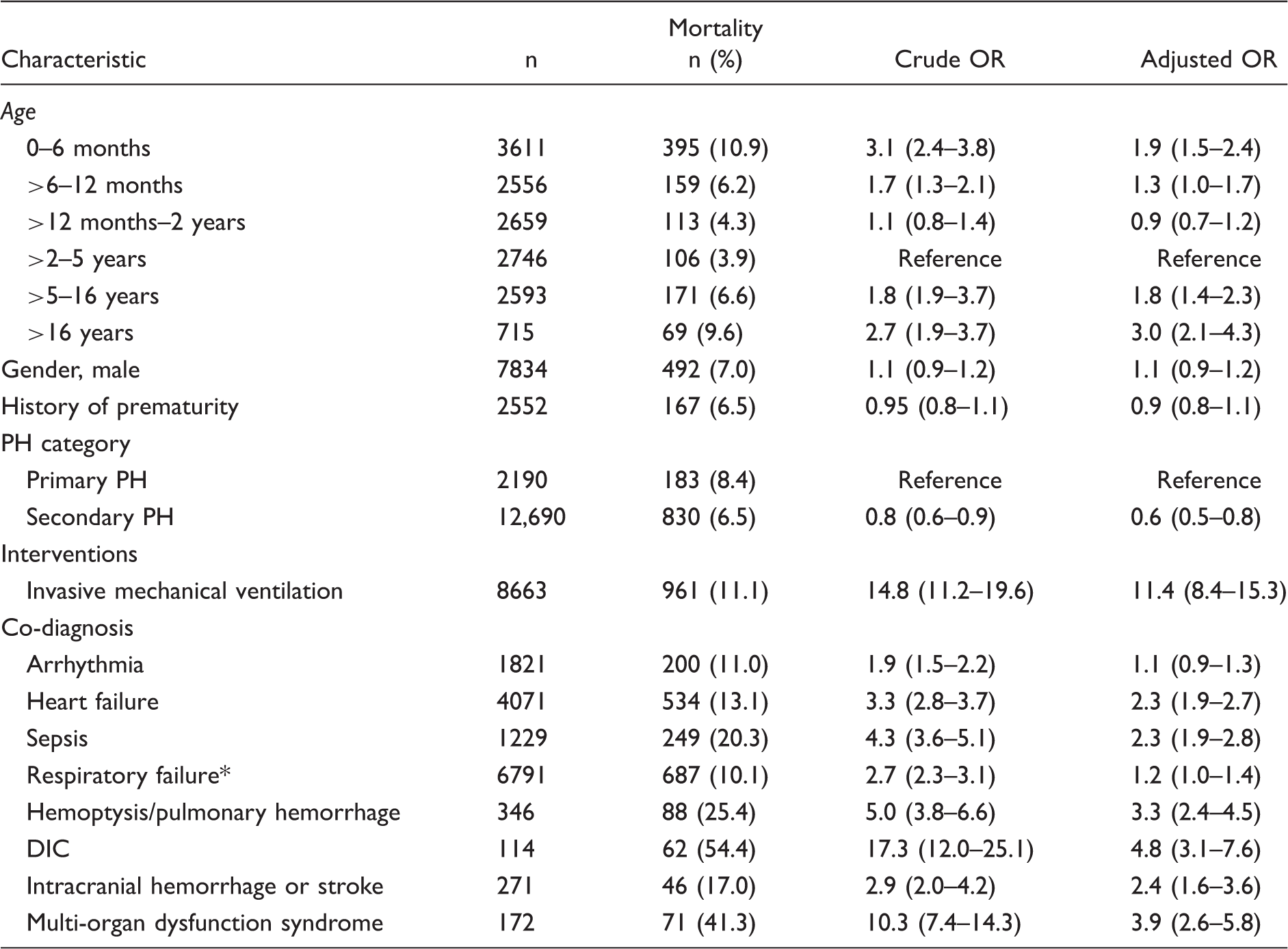
Each of the co-diagnoses assessed was associated with an increased risk of mortality for admissions with PH compared to admissions without that co-diagnosis (Table 2). DIC and MODS demonstrated the greatest increase in mortality risk with ORs of 17.3 (95% CI = 12.0–25.1) and 10.3 (95% CI = 7.4–14.3), respectively.
The following predictors of mortality were entered in a multivariate model: age; history of prematurity; type of PH; invasive mechanical ventilation; and co-diagnoses of arrhythmia, heart failure, sepsis, respiratory failure, pulmonary hemorrhage, DIC, ICH/stroke, and MODS (Fig. 3, Table 3) . In the multivariate model, the following variables remained statistically significantly associated with increased odds of mortality: age (0–6 months, > 5–16 years, > 16 years); type of PH; invasive mechanical ventilation; and co-diagnoses of heart failure, sepsis, pulmonary hemorrhage, DIC, ICH/stroke, and MODS.
Multivariate predictors of mortality in children with pulmonary hypertension. mos, months; yrs, years; PH, pulmonary hypertension; DIC, disseminated intravascular coagulation; ICH, intracranial hemorrhage; MODS, multi-organ dysfunction syndrome. Multivariate predictors of mortality in children with PH, by admission (n = 14,880). Defined as “Pulmonary insufficiency/Shock lung, Adult respiratory distress syndrome (ARDS), Acute hypoxic respiratory failure (AHRF), Acute lung injury (ALI).” OR, odds ratio; PH, pulmonary hypertension; DIC, disseminated intravascular coagulation.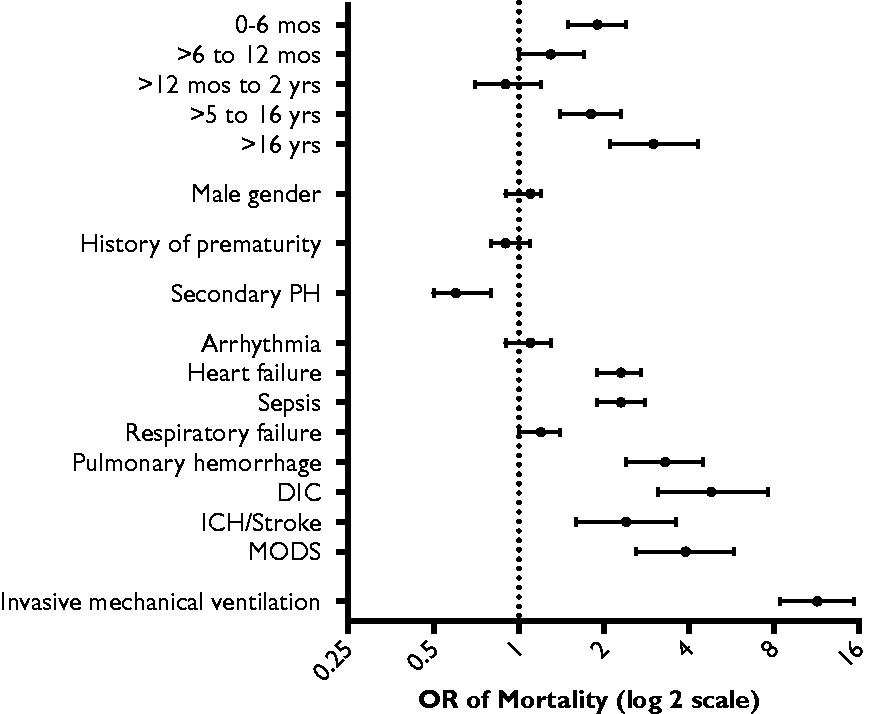
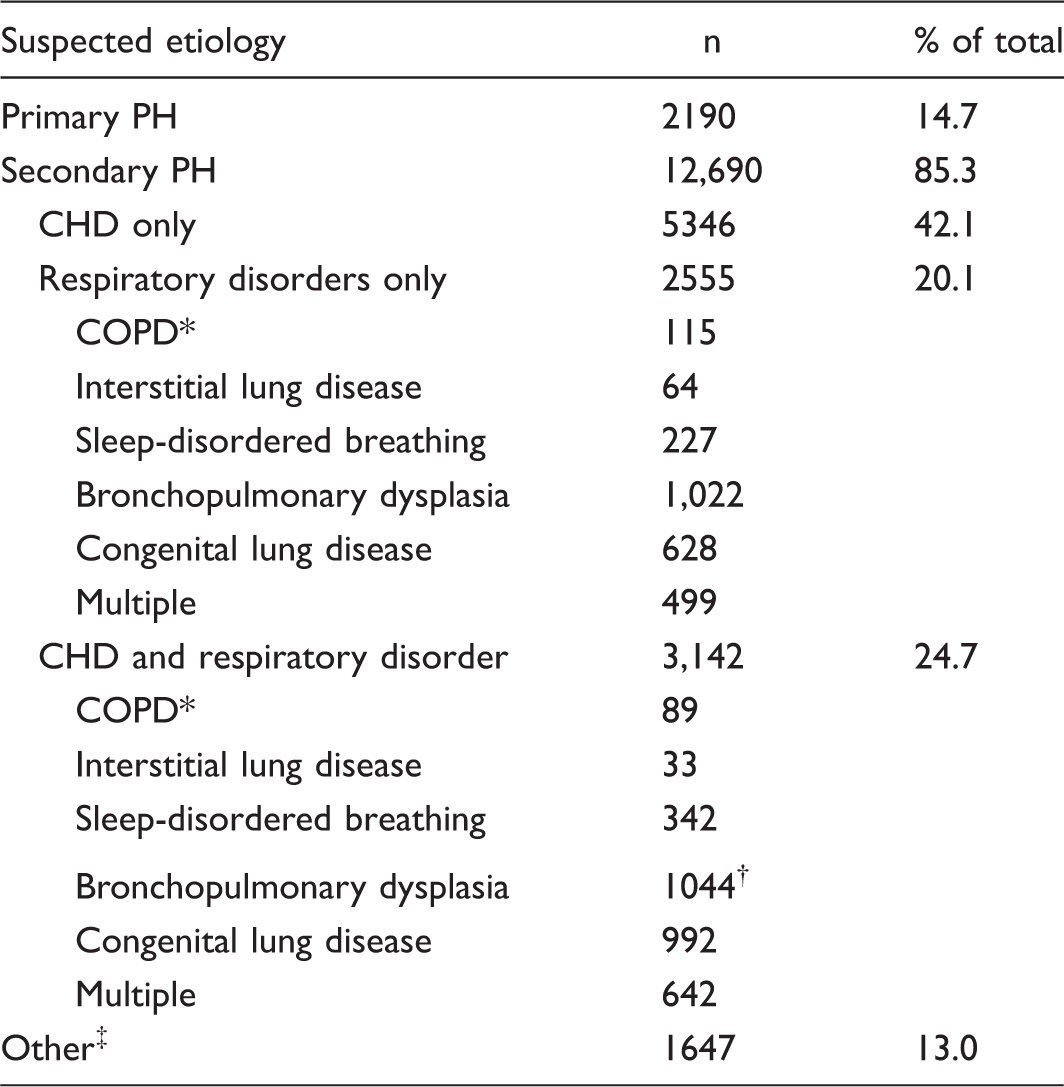
Suspected etiologies of PH, by admission (n = 14,880).
Includes patients with cystic fibrosis, emphysema, chronic obstructive bronchitis, and chronic airway obstruction not otherwise specified.
85 out of 1044 had a patent ductus arteriosus without any other CHD.
Included patients with rheumatologic disease, portal hypertension, etc.
CHD, congenital heart disease; COPD, chronic obstructive pulmonary disease.
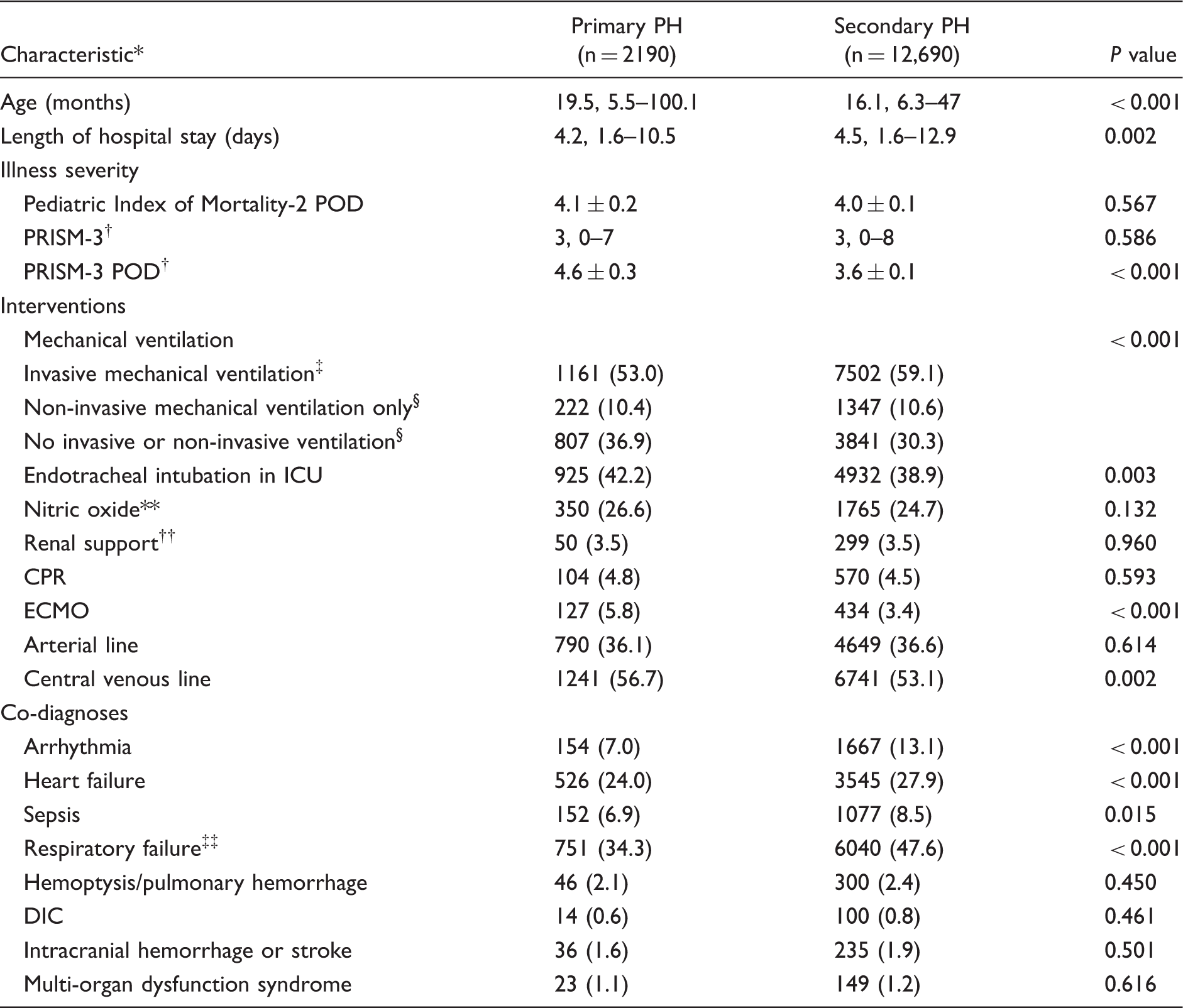
Characteristics of admissions with primary vs. secondary PH.
Results displayed as median, IQR; n (%); or mean ± SD.
Missing in 743 cases.
Includes “Endotracheal intubation,” “Mechanical ventilation, conventional,” and “HFOV (high frequency oscillator ventilation).”
Includes “BiPAP,” “CPAP,” and “Humidified high flow oxygen.” Non-invasive mechanical ventilation n = 10,108 for PH patients, n = 453,179 for non-PH patients, not reported at all centers.
Nitric oxide n = 8473 for PH patients, n = 340,903 for non-PH patients, not reported at all centers.
Renal support, defined as continuous renal replacement therapy or dialysis, n = 9862 for PH patients, n = 402,930 for non-PH patients, not reported at all centers.
Defined as “Pulmonary insufficiency/Shock lung, Adult respiratory distress syndrome (ARDS), Acute hypoxic respiratory failure (AHRF), Acute lung injury (ALI).”
PH, pulmonary hypertension; POD, probability of death; PRISM, Pediatric Risk of Mortality; ICU, intensive care unit; CPR, cardiopulmonary resuscitation; ECMO, extracorporeal membrane oxygenation; DIC, disseminated intravascular coagulation.
Characteristics of admissions with and without CPR, invasive mechanical ventilation, and central line placement.
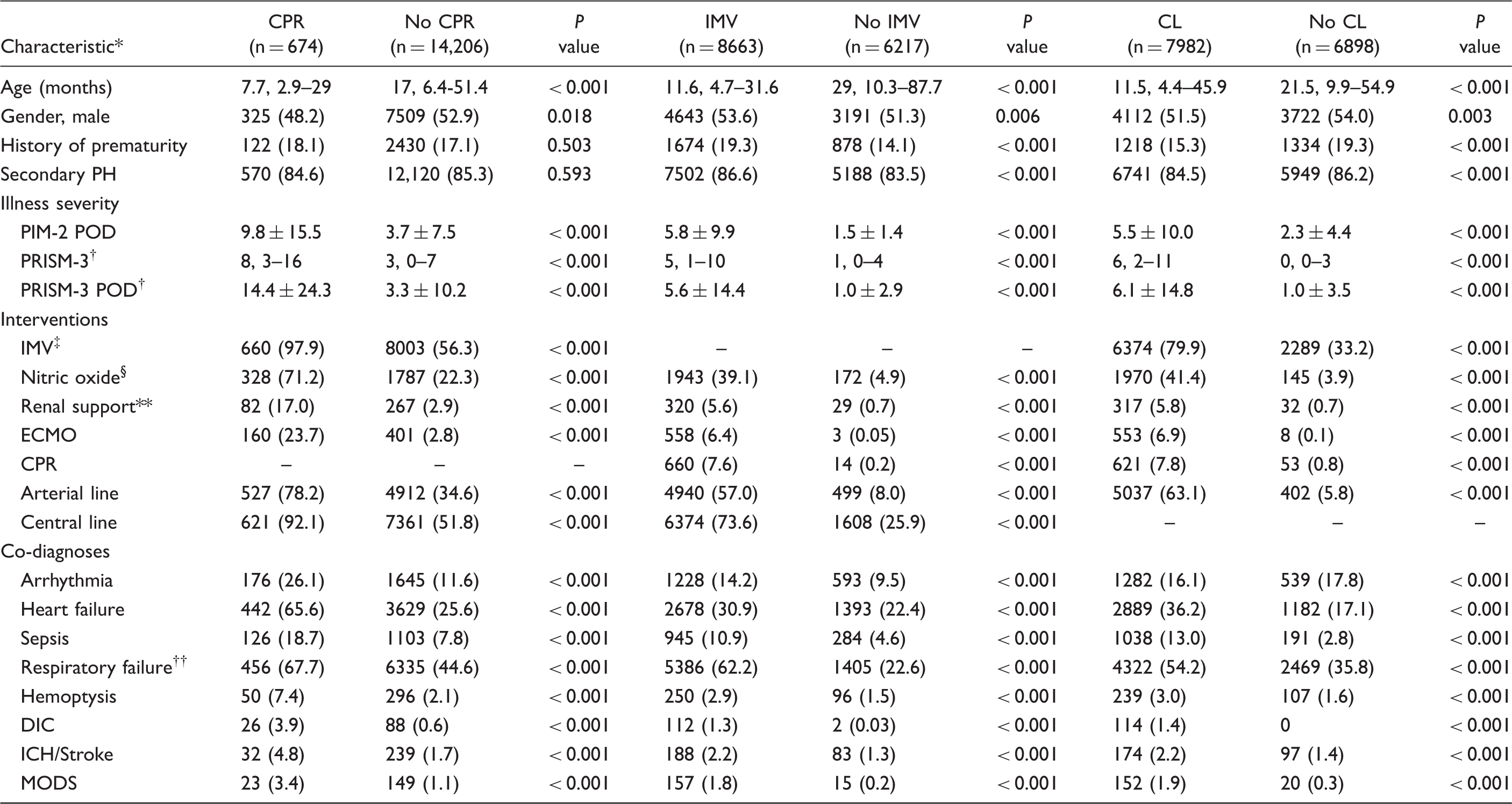
Results displayed as median, IQR; n (%); or mean ± SD.
Missing in 743 and 36,013 cases for PH and non-PH patients, respectively.
Includes “Endotracheal intubation,” “Mechanical ventilation, conventional,” and “HFOV (high frequency oscillator ventilation).”
Nitric oxide n = 8473 for PH patients, n = 340,903 for non-PH patients, not reported at all centers.
Renal support, defined as continuous renal replacement therapy or dialysis, n = 9862 for PH patients, n = 402,930, not reported at all centers.
Defined as “Pulmonary insufficiency/Shock lung, Adult respiratory distress syndrome (ARDS), Acute hypoxic respiratory failure (AHRF), Acute lung injury (ALI).”
CPR, cardiopulmonary resuscitation; IMV, invasive mechanical ventilation; CL, central line; ICH, intracranial hemorrhage; PH, pulmonary hypertension; POD, probability of death; PIM, Pediatric Index of Mortality; PRISM, Pediatric Risk of Mortality; ICU, intensive care unit; ECMO, extracorporeal membrane oxygenation; DIC, disseminated intravascular coagulation; MODS, multi-organ dysfunction syndrome.
Discussion
Children with PH represent a growing population of patients admitted to pediatric hospitals and have a significantly higher mortality rate compared to the general inpatient pediatric population.1,2 This multicenter study is the first to describe the clinical characteristics and predictors of mortality of children with PH in the PICU. We demonstrated that pediatric PH patients represent only 2.2% of all PICU admissions but account for 6.4% of all PICU deaths. Further, mortality for PH admissions was significantly higher than for the general PICU population. Secondary PH was more common than primary PH but patients with primary PH had significantly greater ICU mortality. Children with PH in the PICU are younger, have longer length of stay, and have markers of more severe illness. Finally, predictors of mortality for children with PH included extremes of age, invasive mechanical ventilation, and co-diagnoses of heart failure, sepsis, pulmonary hemorrhage, DIC, stroke, and MODS.
These findings corroborate and build upon prior studies utilizing another national retrospective inpatient dataset (KID).1,2 Maxwell et al. showed that children with PH comprise 0.13% of pediatric hospitalizations with an increasing trend over the 15-year study period. 1 Therefore, based on our study's findings, children with PH represent a greater proportion of PICU admissions than they do in the general inpatient pediatric population (2.2% vs. 0.13% of all admissions). This likely reflects the greater clinical severity of PH compared to other pediatric diagnoses necessitating inpatient admission and therefore the increased likelihood for requiring higher-level care. Consistent with our study's findings, children with PH in their cohort had greater overall mortality (5.9% vs. 0.6%), were younger, had longer length of stay, and higher co-morbidity scores. 1
This study is the first to assess factors associated with mortality in children with PH admitted to the PICU. We demonstrated a bimodal distribution in mortality by age with the youngest (0–6 months) and oldest ( > 16 years) patients having the greatest mortality. While a prior study of inpatient pediatric admissions also found younger age ( < 1 year) to be associated with increased mortality, they found that older age was associated with reduced mortality, although neither remained significant in their multivariate model. 2 In our study, children admitted with primary PH were older than those with secondary PH, which perhaps explains the greater mortality in those > 16 years of age. However, further interpretation of these findings is limited by our lack of detailed data regarding the underlying etiology of PH.
We found the interventions associated with greatest odds of mortality to be invasive mechanical ventilation, CPR, ECMO, and central line placement (Table 2). Not surprisingly, absence of any ventilator support (invasive or non-invasive) was associated with a significantly lower mortality rate. However, these findings should not encourage reluctance to intubate PH patients. In this retrospective cohort study, they likely reflect the underlying severity of clinical illness; children who require these invasive procedures are more likely to die from their underlying disease process. Consistent with this hypothesis, we found sepsis, heart failure, DIC, pulmonary hemorrhage, stroke, and MODS to be predictors of mortality in our multivariate model. While respiratory failure was associated with mortality in univariate analyses, it was no longer significant in the multivariate model. Meanwhile, invasive mechanical ventilation remained a strong predictor of mortality. We suspect that this likely reflects the variability in definition of respiratory failure by data entry analysts along with strong collinearity between respiratory failure and invasive mechanical ventilation in our multivariate model (of the 687 admissions with respiratory failure who died, 669 also received invasive mechanical ventilation). As such, invasive mechanical ventilation emerges as the more important factor, which highlights the fact that the requirement for invasive respiratory support is more significant than the diagnosis of respiratory failure alone. In their analysis of inpatient admissions, Frank et al. similarly identified sepsis, ECMO, and respiratory failure as predictors of mortality; however, acute renal failure and arrhythmias also retained statistical significance in their multivariate analyses. 2
Given that CPR, invasive mechanical ventilation, and central line placement were associated with significantly higher patient mortality, these were assessed independently as secondary outcomes (Table 6). Patients who received each of these interventions were significantly younger than those who did not. This is consistent with the fact that younger age predicts mortality but does not capture the bimodal age distribution. Not surprisingly, these invasive interventions were all associated with greater illness severity scores, other interventions, and co-diagnoses. History of prematurity was only associated with need for invasive mechanical ventilation, which likely reflects the role of underlying chronic lung disease associated with prematurity.
Secondary PH was more common than primary PH in our cohort, which is consistent with several large registry studies.18,22 Among those with secondary PH, 42% had diagnoses suggestive of CHD as the underlying etiology (Group 1 PH) while 20% were thought to be secondary to respiratory disorders (Group 3 PH); another 25% had both cardiac and respiratory diagnoses and could not be differentiated in this dataset. Several other published cohorts have reported a much greater predominance ( > 90%) of Group 1 PH (summarized by Ploegstra et al. 14 ). However, recent studies have suggested a growing population of Group 3 and multifactorial PH 23 and a trend in increased inpatient admissions for children with PH who do not have CHD.1,2 In that sense, our results are likely reflective of the heterogeneous population of children with PH that continues to evolve as treatment teams' identification of PVD in a variety of disease conditions increases.
In our ICU-based cohort, admissions with primary PH had greater mortality (8.4% vs. 6.8%). Secondary PH patients admitted to the PICU were younger and had longer PICU stays. While these patients more often received invasive mechanical ventilation, they were less likely to undergo intubation while in the PICU. While not previously described in the literature, this is not surprising given that many of these patients have CHD and therefore a large proportion of them arrive already intubated from the operating room or catheterization laboratory.
This study has several limitations. The study was performed retrospectively and relied on accurate database entry based on chart review; some data were not reported by all centers. Additionally, the VPS database does not include dedicated pediatric cardiac intensive care units (PCICUs) but may include some combined units. Therefore, our patient sample likely excludes a large number of PH patients with disease secondary to CHD, especially those requiring cardiac surgery. This may have artificially lowered secondary PH-related mortality by selecting for those patients with CHD who are less sick. PH admission practice patterns related to PCICUs likely vary with some centers admitting all PH patients to dedicated PCICUs, others admitting only surgical PH patients to PCICUs, and others admitting all PH patients to combined units. Nonetheless, this study provides a broad multicenter assessment of pediatric PH admissions to the PICU setting.
This study relied on VPS diagnosis codes and ICD9 codes for identification of patients with PH. It is not known how these diagnoses were made as no echocardiography or catheterization data are available for confirmation. Moreover, we were limited in our ability to delineate the underlying cause of PH and had no information on disease severity or therapies for PH. Given this, we greatly simplified our attempt to categorize patients with secondary PH. Our post hoc reclassification of patients with multiple PH diagnoses or with primary PH and CHD was likely subject to some misclassification, although a strict and consistent method was used across all admissions. Finally, this study does not capture deaths that occurred following transfer or discharge from the PICU. Overall, these limitations are offset by the large, diverse national sample, which unlike many registry studies is not limited to PH referral centers.
This is the first study to comprehensively describe PICU admissions for pediatric patients with PH and provides valuable new insight into the morbidity and mortality of these children in the ICU setting. Despite therapeutic advances, the disease burden and mortality of children with PH are significantly higher than for the general PICU population. Risk factors for mortality identified include primary PH, the extremes of age, invasive interventions, and co-diagnoses suggestive of greater illness severity. Early identification of the risk factors associated with mortality in this population could help to inform clinical decision-making, quickly initiate appropriate treatment, and improve the ability to prognosticate for patients and families. Prospective, multicenter studies are needed to confirm and further investigate these findings.
Footnotes
Acknowledgements
VPS data were provided by the VPS, LLC. No endorsement or editorial restriction of the interpretation of these data or opinions of the authors has been implied or stated.
Conflicting interests
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
