Abstract
Chronic lung disease (CLD), including pulmonary fibrosis (PF) and chronic obstructive pulmonary disease (COPD), is the fourth leading cause of mortality worldwide. Both are debilitating pathologies that impede overall tissue function. A common co-morbidity in CLD is vasculopathy, characterized by deregulated angiogenesis, remodeling, and loss of microvessels. This substantially worsens prognosis and limits survival, with most current therapeutic strategies being largely palliative. The relevance of angiogenesis, both capillary and lymph, to the pathophysiology of CLD has not been resolved as conflicting evidence depicts angiogenesis as both reparative or pathologic. Therefore, we must begin to understand and model the underlying pathobiology of pulmonary vascular deregulation, alone and in response to injury induced disease, to define cell interactions necessary to maintain normal function and promote repair. Capillary and lymphangiogenesis are deregulated in both PF and COPD, although the mechanisms by which they co-regulate and underlie early pathogenesis of disease are unknown. The cell-specific mechanisms that regulate lung vascular homeostasis, repair, and remodeling represent a significant gap in knowledge, which presents an opportunity to develop targeted therapies. We have shown that that ABCG2pos multipotent adult mesenchymal stem or progenitor cells (MPC) influence the function of the capillary microvasculature as well as lymphangiogenesis. A balance of both is required for normal tissue homeostasis and repair. Our current models suggest that when lymph and capillary angiogenesis are out of balance, the non-equivalence appears to support the progression of disease and tissue remodeling. The angiogenic regulatory mechanisms underlying CLD likely impact other interstitial lung diseases, tuberous sclerosis, and lymphangioleiomyomatosis.
Keywords
Vascular dysfunction in chronic lung disease
Microvascular dysfunction and/or structural remodeling (vasculopathy) is frequently, if not ubiquitously, observed in most chronic lung diseases (CLD), including pulmonary fibrosis (PF), chronic obstructive pulmonary disease (COPD)/emphysema and interstitial lung disease associated with systemic sclerosis (SSc). This vasculopathy may be characterized by deregulated angiogenesis, pathologic remodeling, and/or loss of microvessels.
The complex, orchestrated formation of blood vessels that occurs during lung development provides insights into the origins of vasculopathy later in life. The growth of new vessels in the adult can occur through angiogenesis, which is new vessel growth from existing vessels and vasculogenesis, which is new vessel growth independent of existing vessels.1–6 The coordination of both processes is important in lung development and it also plays a key role in adult-onset lung disease.
During the fourth week of lung development in humans, hematopoietic progenitor cells initiate vasculogenesis to form vascular lakes.1,7,8 These structures will ultimately become capillaries and connect with the pulmonary circulation, which begins to develop separately during the seventh and eighth week of gestation. These connections enable the lung to carry out its primary function, which is to deliver blood flow to alveolar capillaries where oxygen absorption and carbon dioxide elimination occurs. The bronchial circulation is a distinct system from the pulmonary circulation. It functions to supports the airway structure of the lung and the bronchial blood supply develops via angiogenesis from the aorta during the ninth and 12th week of gestation.7–9 When there is obstruction of pulmonary blood flow, ischemic stimuli induce rapid and extensive growth of the bronchial circulation. Indeed, it is the bronchial circulation that supplies the blood vessels that permit tumor growth in lung cancer. 10 In addition to tissue hypoxia, the bronchial vasculature is also sensitive to local inflammatory responses. 11 Inflammation promotes the release of mediators like IL-1 and IL-6 that stimulate the release of vascular endothelial growth factor (VEGF). 12 This increases bronchial vascular density and leads to airway remodeling in asthma.9,11–15 Though its role in asthma is well-established, the impact of these factors on other inflammatory lung diseases like emphysema and PF is less clear. 16 Some studies have reported increased bronchial vascular density in COPD17,18 and this may contribute to the mucus production and thickened airways that occur in this disease. However, other studies have shown decreased or no changes in the bronchial vasculature.19–21 Thus, the role of the bronchial blood vessels in COPD is not as clearly defined as it is in asthma.22,23 Increases in the bronchial vasculature are noted in bleomycin-induced PF in mice. 24 Despite these findings, most studies have concentrated on alveolar capillaries because the alveolar parenchyma is the region most affected by fibrotic remodeling in this disease.25–27 This review will largely focus on factors affecting the alveolar microvasculature 28 since alterations in this region have a well-established role in the pathogenesis of both PF and emphysema.
In most forms of CLD, development of secondary pulmonary hypertension (PH) is associated with poor prognosis and worsened survival, 29 underscoring the importance of better understanding the role of the pulmonary vasculature in disease pathogenesis. The etiology of microvascular remodeling and mechanisms through which it contributes to the development and severity of various CLD remain unknown, due in part to a lack of models recapitulating early stage vasculopathy before environmental injuries, disease, or aging. The relevance of vasculopathy to the pathophysiology of CLDs has not been resolved as conflicting evidence depicts angiogenesis as both reparative or pathologic;30–41 it is possible that both of these are true in specific contexts.
Most work to date investigating the pulmonary vasculature has focused on arterial and capillary networks in the lung; the role of lymphangiogenesis has been largely overlooked. As a balance of existing and de novo capillary and lymphatics is necessary for tissue homeostasis, one might argue that their collective deregulation is paramount to the pathophysiology of disease.
Capillary and lymph angiogenesis
The pulmonary lobule, which measures 2–3 cm in size, is the basic functional unit of the lung. It comprises several terminal bronchioles which run with pulmonary arteries and deep lymphatics in the center. This structural unit is enveloped by interlobular septa containing pulmonary veins and independent superficial lymphatics that are in communication with the pleural lymphatic system. The deep lymphatics travel in the bronchovascular interstitium and drain the airways while the superficial lymphatics are present in the interlobular septa and drain alveolar regions. The structure and function of lymphatic vessels is very different from the circulatory system and they have been shown to be extremely sensitive to interstitial stresses. 42
In contrast to the circulatory system, lymphatic flow is regulated by the movements of the body and muscle contraction. 42 These structures are interlaced among arterioles and venules of most soft connective tissues of the body to aid in the regulation of fluid balance.42–47 In the adult lung, lymphatics are localized to the bronchiolar wall and bronchiole associated arteries. 48 Intra-alveolar lymphatics are more difficult to distinguish but are also present, interwoven within the alveolar-capillary interstitium.45,49 Of note, disorders of these lymphatic systems could promote airway or alveolar injury by impairing the clearance of antigens and lung biomediators.
Angiogenesis is the sprouting and growth of new vasculature from existing vascular structures. Lymphangiogenesis is similar to capillary angiogenesis in that during development and in the maintenance of adult tissue homeostasis, the formation of de novo lymphatics may occur from sprouting of lymphatic endothelial cells (LEC) from existing vessels as well as single LEC or mesenchymal progenitors migrating to a zone where they later connect and form vascular structures.4,43,44,50–54 Currently, distinguishing between LEC and angiogenic mesenchymal progenitors in vivo is technically challenging due to the promiscuity of lymphatic markers (prox1, lyve1, flt4). In vitro, definition between the cell types relies on functional characteristics and expression of multiple lineage specific markers.55,56 The regulation of both angiogenic processes involves VEGF signaling.43,44,50,57 A comparison of the distinct processes of capillary and lymphangiogenesis is reviewed by Adams and Alitalo. 43
During development, lymphangiogenesis is closely linked with capillary angiogenesis. 43 Their interaction and balance is necessary for maintenance of tissue homeostasis.43,44,49,51,58 During disease, angiogenesis of one may exceed the other.4,31,32,41,42,45–47,58–71 Both angiogenesis and lymphangiogenesis are required for wound healing and functional tissue repair, processes deregulated in CLD.43,50,72 Typically, lymphatic capillaries do not contain mesenchymal cells/pericytes. However during disease, the lymphatic capillaries may attract these cells. 73 In both developmental and pathological instances, mesenchymal cells may also form new lymphatic structures, devoid of endothelium, either connected or separate from existing lymphatic circulation.52,53,74 These abnormal mesenchymal structures have been described in tumor angiogenesis and termed vascular mimicry.75–78
Pulmonary fibrosis
PF (familial, idiopathic [IPF], and associated with SSc) is a debilitating disease characterized by excessive matrix deposition, angiogenesis, and epithelial cell hyperplasia that impedes overall tissue function. Abnormal neovascularization in fibrosis was first characterized in 1963 by Turner-Warwick. 79 The underlying cause of fibrotic remodeling and microvascular dysfunction in the early stages of fibrosis is unknown.30–32,35,45,49,58,62,70,80–84 Research suggests that impaired cross-talk between endothelial cells and mesenchymal progenitors during disease drives a functional switch of the mesenchyme to a pro-remodeling phenotype, modulating both vascular regression as well as fibrosis. 85 Additionally, the relevance of angiogenesis, microvascular remodeling, and lymphangiogenesis to the pathophysiology of disease are an area of intense investigation. 31
A heterogenous pattern of microvascular remodeling has been reported in PF. Microvascular density (as defined by CD34+ cells) was reported to be greater during fibrosis in areas of mild remodeling compared to normal lungs, whereas fibroblast foci are described as devoid of vessels.24,30,31,84,86,87 Similarly, fibrosis in SSc is associated with an initial increase in microvessels, followed by a progressive decrease.62,65,80–82 Heterogeneity of the microvasculature is largely dependent on the localization and severity of fibrosis.30–32,84 It is not entirely clear whether these discrepancies in microvascular density represent reactive/compensatory changes or reflect the temporal heterogeneity of the lesions that occur in PF. For example, alveolar capillaries are increased and dilated in non-fibrotic regions but absent in fibrotic interlobular septa.88–90 These analyses are complicated by the promiscuity of lineage marker expression; for example, CD34 may be expressed on some epithelial cell populations including bronchoalveolar stem cells (BASCs); 91 as such there is a need to more comprehensively define patterns of vascular remodeling in PF using multi-marker strategies.
Abnormal lymphangiogenesis is also present in PF and typically associated with the degree of disease severity.45,49,58,92 El-Chemaly correlated the diameter of lymphatics to disease severity in IPF patients, 45 while the diameter has also been correlated to survival. 93 They further demonstrated that angiogenesis in IPF patients was regulated by short fragment hyaluronan, the extracellular matrix protein, present in the bronchioalveolar lavage fluid and macrophages, not found in healthy controls. 45 Lymphatic microvasculature is localized to areas of remodeling where capillaries are typically absent. Fibrosis disrupts lymphatics in the interlobular septa 94 and this impairs alveolar clearance in the lung. 95 These changes could perpetuate lung fibrosis by impairing the elimination of inflammatory cells that express TGF-β, a profibrotic cytokine that inhibits lymphangiogenesis.96,97 Abnormal lymphatics have been proposed as a unifying mechanism for fibrogenesis. 58 Intriguingly, in a murine model of PF induced by intratracheal or intraperitoneal bleomycin, the development of abnormal lymphatic structures was driven by platelet-derived growth-factor-beta (PDGF-β), and pharmacologic inhibition of PDGF-β attenuated lymphatic remodeling and improved fibrosis. 98 This is of particular interest as the recently approved antifibrotic treatment nintedanib, among its actions, inhibits PDGF-β/PDGFR-β signaling, 99 raising the possibility that its efficacy may be due in part to effects on lymphatic organization.
COPD/Emphysema
Emphysema is a form of COPD, characterized by abnormal enlargement of the distal airspaces/alveoli, and is an important contributor to reduced lung function in patients with COPD. 100 Emphysema is a progressive disease that destroys alveolar septa over time and causes a decrease in functional alveolar surface area that impairs the absorption of oxygen. Along with small airways disease and chronic bronchitis, emphysema contributes to persistent airflow obstruction in patients with COPD, resulting in persistent obstruction to expiratory airflow, chronic dyspnea, and death in approximately one-third of affected patients. 101 The narrowing of the airway and the elaboration of thick mucus causes breathlessness by impeding flow in the airways. COPD was ranked sixth among the causes of death globally in 1990 but is projected to be the third most common cause of death by 2020. 102 Current treatment is limited and focuses on halting further lung destruction and preserving lung function and includes smoking cessation, bronchodilators, steroids, and supplemental oxygen. 103 A limited understanding of the cellular pathogenesis of COPD has impeded the development of effective treatments. Recent evidence has identified alterations to the lung microvasculature during the early pathogenesis and heterogeneity of COPD,104–107 although the underlying mechanisms are not defined.
Emphysematous loss of the alveolar capillary network was first described by Liebow in 1959. 108 However, the relevance of the microvasculature during the early stages of COPD has not been resolved. The basis for a vascular component contributing to emphysema was suggested by Wiebe and Laursen in 1998, via quantitation of significantly decreased capillary length and density in COPD patients (68%), relative to controls. 109 A functional basis for vasculopathy as a contributor to disease pathogenesis has been demonstrated in clinical studies comparing FEV1 to severity of tissue remodeling and vascular perfusion. Barr et al. defined a relationship between endothelial function, FEV1, and percentage of emphysema using computed tomography (CT) in ex-smokers and demonstrated that endothelial dysfunction was associated with a significant decrease in FEV1. 110 McAllister et al. defined a relationship between emphysema and systemic vascular dysfunction 105 and Sabit et al. showed that evidence of increased arterial stiffness and endothelial dysfunction was related to the severity of airflow obstruction increasing the risk for COPD. 104 Alford et al. documented increased areas of decreased perfusion in individuals with early visual CT evidence of emphysema, relative to emphysema-free smokers and persons who had never smoked. 106 Most recently, imaging of the pulmonary vasculature in COPD patients demonstrated that the extent of loss of vascular function correlates to the degree and heterogeneity of emphysema/COPD. 107 Many studies exploring the mechanism(s) of vasculopathy in COPD have focused on endothelial apoptosis. 111 Alpha-1-antitrypsin has been shown to inhibit caspase-3 activation and apoptosis in endothelial cells, providing a potential mechanism through which vasculopathy contributes to emphysema in patients with alpha-1-antitrypsin deficiency. 112 In addition, SERPINF1 has been shown to be elevated in patients with COPD and may contribute to increased endothelial cell apoptosis. 113 In addition to the associative studies in humans, animal studies have suggested a causal link between endothelial dysfunction and emphysema. Antagonizing vascular endothelial growth factor receptor (VEGFR) in rodents induced endothelial apoptosis and the subsequent destruction of alveolar lung tissue. 114
Clinical studies have also linked enhanced lymphangiogenesis to the pathogenesis of COPD. Hardavella et al. correlated the lymphatic microvessel density, as determined by Lyve1 stain, to the degree of airway obstruction, measured by FEV1, in COPD patients vs. non-COPD smokers. 46 Mori et al. followed these proof-of-principle studies with a detailed histochemical quantification of lymphatic distribution and morphological characteristics in healthy control vs. COPD lung tissue. 48 Pathologic, de novo lymphatics were localized in the alveolar parenchyma, not associated with smooth muscle actin positive vessels. They were also found at a higher density in areas of alveolar parenchymal fibrosis. Taken together, these studies suggest that while de novo capillary angiogenesis is halted in COPD, lymphangiogenesis progresses, representing an imbalance of angiogenesis. How these lymphatic vessels contribute to the pathogenesis of COPD remains to be determined. However, it has been speculated that they may channel inflammatory signals to regional lymph nodes where T cell activation occurs. The coordination of T cell activation by this lymphatic system could have a major impact on COPD. T cells promote emphysema by directly injuring the lung epithelium 115 and they promote airway disease by elaborating cytokines like IL-4, -5, and -13, which induce airway obstruction and mucus secretion.
Combined pulmonary fibrosis and COPD (CPFE)
The presence of both emphysema and PF in the same patient is a disorder known as combined pulmonary fibrosis and emphysema (CPFE). Whether this reflects a distinct disease or the chance co-occurrence of two processes is a matter of some debate. This syndrome is characterized by upper-lobe emphysema, lower-lobe fibrosis, and abnormalities of gas exchange that result in dyspnea. 116 Pulmonary function tests (PFTs) differ from the obstructive pattern with increased lung volumes in COPD and the restrictive pattern with reduced lung volumes in lung fibrosis. CPFE patients typically present with near normal (pseudonormalized) lung volumes and with a significantly reduced diffusing capacity for carbon monoxide (DLCO). This reduction reflects the severity of the gas exchange abnormalities that occur in this disorder. Cigarette smoke exposure is a well-recognized risk factor for the development of PF and COPD/emphysema.117–119 In fact, smoking exhibits deleterious effects on the systemic circulation 118 and exacerbates systemic sclerosis.117,120 PH is prevalent in CPFE and is the main co-morbidity affecting survival of CPFE patients. 121 In contrast to patients with COPD, CPFE is frequently associated with profound system hypoxemia, suggesting that in these patients, shunt rather than ventilation/perfusion mismatching is a primary driver of hypoxemia. This suggests that there are some distinctions in the patterns of vascular remodeling in CPFE and COPD.
Extracellular matrix remodeling plays an essential role in COPD and PF but the character of the remodeling in these syndromes differs significantly. Alveolar elastin and type III collagen are destroyed in emphysema and replaced by fibrils that are thickened and disorganized. 122 This degrades alveolar tissue and redistributes mechanical forces to disrupt alveolar interdependence and perpetuates lung tissue destruction.123,124 In contrast, PF is characterized by an excessive deposition of lung collagen that obliterates distal lung tissue structures. Though the pathogenesis is distinct, the loss of alveolar capillary units induces similar clinical symptoms including cough and shortness of breath.
Inflammation, deregulated vascular remodeling, pruning of microvascular structures, and excessive lymphangiogenesis are present in both disorders. These shared mechanisms likely account for the coexistence of both diseases in CPFE. Subtle differences in these processes are probably responsible for the development of fibrosis in some regions and emphysema in others. For example, studies suggest that inflammation plays a lesser role in the pathogenesis of IPF and this disease does not respond to steroid therapy.125–127 The inflammatory cells present in IPF appear to contribute to disease pathology by coordinating fibroblast activation and epithelial mesenchymal transition.128,129 In addition, auto-antibodies produced by B cells attack the endothelium to induce microvascular injury that contributes to PH and lung fibrosis.130–133 In contrast to IPF, COPD responds clinically to anti-inflammatory therapy with improved lung function and decreased exacerbations. 134 The inflammatory cells in COPD, such as neutrophils, macrophages, lymphocytes, and eosinophils, release mediators that increase mucus production, airway hyper reactivity, and lung tissue destruction. 135 Thus, they play a more direct role in the disease onset and progression. Like PF, auto-immunity triggers the vascular injury that occurs in COPD.136,137 Since both diseases involve immune-mediated damage to the microvasculature, it is not surprising that PH is so prevalent in CPFE.
How these distinct biological processes are induced in different regions in CPFE is unknown but likely involves changes in inter- and intra-cellular signaling. Kusko et al. recently published a comprehensive study comparing the transcriptome networks of lung tissue between COPD and IPF patients, relative to normal controls. They identified activation of the p53/hypoxia pathway as well as alternative splicing of PDGFA as a common factor in both diseases. 138 This is intriguing since regional variations in alveolar oxygen tension are present in the lung. The apex has comparatively high ventilation to perfusion ratio resulting in increased alveolar oxygen tension. On the other hand, the lung base has the highest blood flow and lowest ventilation-to-perfusion ratio. 139 Thus, oxygen tension at the base is lower than the apex. These regional differences could have differing effects on the p53/hypoxia pathway, potentially explaining why fibrosis occurs at the bases and emphysema at the apex.
Balance of developmental signaling pathways may also play a role. In PF, it has been suggested that there is increased canonical Wnt signaling, while in COPD this is suppressed. 140 We have recently shown that in the bleomycin model of PF, persistent Wnt/B-catenin signaling leads to enhanced proliferation of mesenchymal progenitor cells but impairs differentiation. Since canonical Wnt signaling is required for pulmonary angiogenesis, it may be that in COPD, suppressed Wnt signaling leads to failure of progenitor cell function, while in PF, failure to suppress Wnt signaling culminates in a hyperproliferative but dysfunctional microvascular network. Further work will be required to better elucidate the shared and divergent mechanisms of microvascular remodeling in COPD and PF; however, taken together, the literature supports that there is an inability to sustain functional tissue repair in epithelial, vascular, and mesenchymal compartments in both diseases.
VEGF: friend or foe?
The basis for a vascular component contributing to fibrosis and emphysema has also been demonstrated in rodents via manipulation of VEGF. VEGF is a survival factor for lung endothelial and mesenchymal cells and decreased signaling through its receptors results in loss of distal lung tissue structure.141–143 Levels of VEGF are increased in early COPD patients,144–146 and decreased in late COPD. 67 VEGF is overexpressed in the skin of SSc patients 80 despite impaired angiogenesis. It is also abundant in the type 2 pneumocytes and myofibroblasts in IPF lungs.31,70
The use of the VEGF receptor tyrosine kinase inhibitor, SU5416, as a VEGF antagonist induces vascular injury, remodeling, subsequent PH, and loss of alveolar tissue structure.114,147–151 Targeted knockdown of VEGF gene expression in the lung drives septal wall destruction. 152 To date, studies have focused on endothelial cells and VEGF deregulation as the basis for microvascular dysfunction during COPD, while additional contributing cell types have been largely overlooked.
Additionally, VEGF-A splice variant b has been described as anti-angiogenic. Increased levels of VEGF165b have been correlated with PH, fibrosis, peripheral artery disease, and SSc,65,80,82,146,153–155 while its significance in lymphangiogenesis has not been described. VEGF-A and its receptors are significantly upregulated in asthmatic airways and this expression correlates positively with submucosal vascularity and negatively with FEV1 and airway hyper-responsiveness. 156 Thus, it is conceivable that alternative splicing of VEGF-A could dictate whether fibrotic remodeling or airway obstruction occurs in the lung. Indeed, alternative splicing has been implicated in a number of respiratory diseases including COPD, IPF, and lung cancer.138,157
Adult lung mesenchymal progenitors, angiogenesis, and tissue remodeling
There is a precedent for multipotent adult mesenchymal stem or progenitor cells (MPC) to stimulate capillary angiogenesis, as well as lymphangiogenesis. Both bone marrow derived and adipose MPC promote lymphangiogenesis in models of tumor metastases as well as regulate the proliferation of LEC.158,159 MPC have been hypothesized to be the precursor to pericytes.160,161 Interestingly, pericytes have also been hypothesized to be mesenchymal stem cells (MSC) in adult tissue. 162 However, this hypothesis was recently challenged by elegant lineage tracing analyses.163,164 It is likely that pericytes and MSC express similar cell surface determinants and co-localize in the microvasculature, yet are functionally distinct.
The current understanding of the role MPC/pericytes play in adult lung disease, de novo angiogenesis, and vascular remodeling is controversial.30–32 Pericytes are the “smooth muscle cell” of the microvessels/capillary beds 73 and provide stability to the vasculature by direct contact with endothelium and regulation of vascular tone. 73 Mice lacking pericytes die before birth due to hemorrhage. 165 They participate in wound healing, tissue repair, vascular remodeling/vasculopathy, and fibrosis.33,35,37–40,73,87,166–183
While these hypotheses are intriguing, the studies of tissue-resident stromal progenitor cells as well as the origin of pericyte lineages in the adult have been complicated by a lack of unique markers to define specific cell types within heterogeneous mesenchymal and lineage specified pericyte populations. The lack of suitable markers to trace mesenchymal progenitor subpopulations and their differentiation in the adult lung has limited our understanding of their role in homeostasis and disease. However, the importance of mesenchymal precursors during disease has been underscored by current limitations. Agha et al. recently published a summary of MSC and their roles in fibrotic diseases of multiple organs. 184
Labeling pulmonary mesenchymal subpopulations.
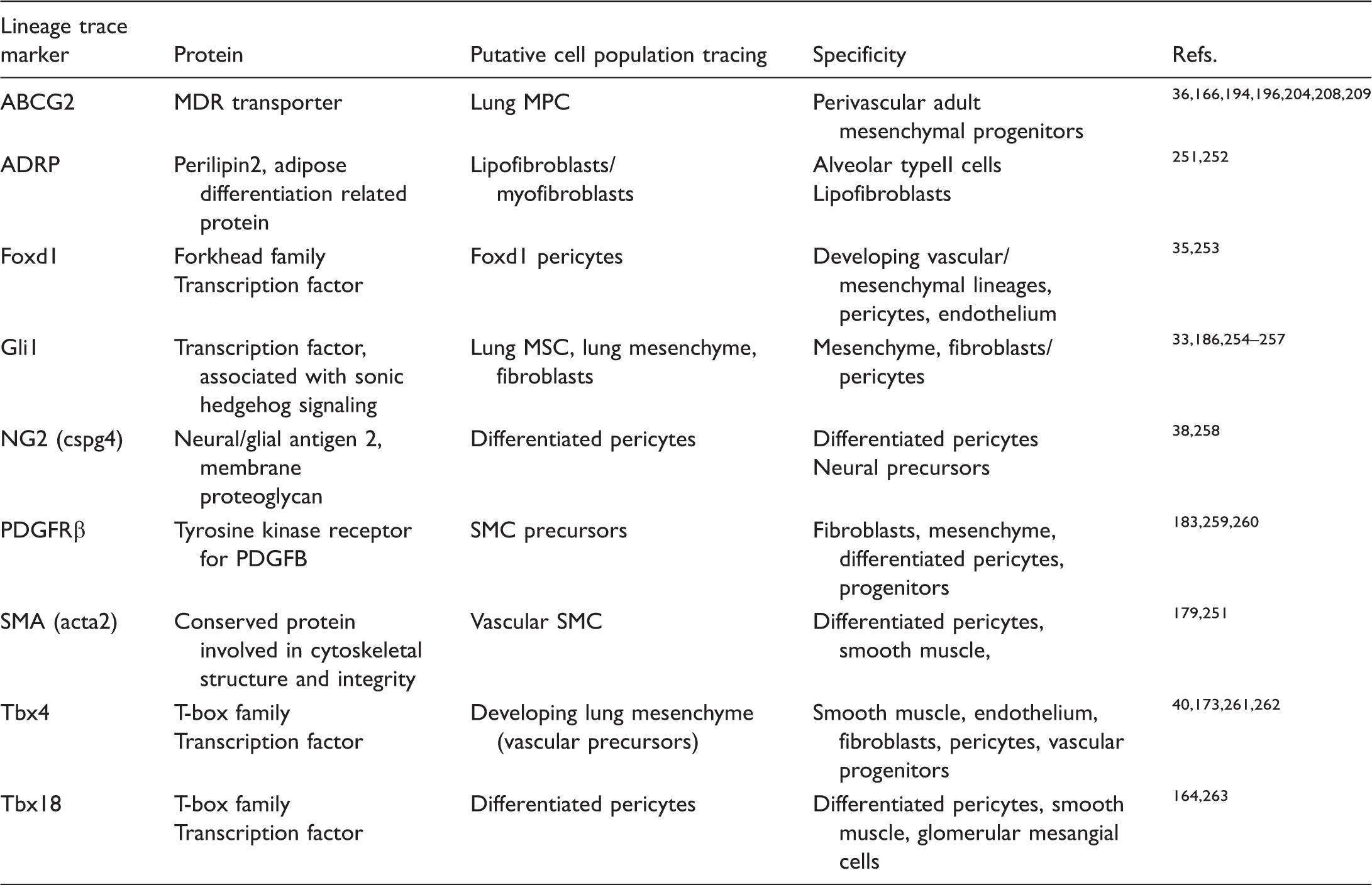
A second limitation to understanding the function of multipotent mesenchyme in the adult lung is the ability to translate the findings from populations identified in rodent tissues to a comparable population of primary patient cells. Because ABCG2 is present at the cell surface, we have been able to isolate populations of adult lung MPC from lung tissue explants from normal and disease lungs and characterize them in parallel to the murine models and cells.36,55,194,195 We have been able to define tissue-specific signatures of MPC as well as pathways related to signaling, matrix, inflammation, and angiogenesis disrupted in disease.194–196
ABCG2 mesenchymal progenitor cells and angiogenesis
The validation of ABCG2 as a marker of adult mesenchymal progenitors enabled us to determine their relationship to pericytes as well as putative function using an inducible murine lineage tracing model system.36,166,194 The selection of ABCG2 as a marker to define this lung mesenchymal progenitor population was based on historical evidence that the expression of this multi-drug resistance transporter selects for a “side population” of cells that demonstrate stem cell-like properties in adult tissues.191,197–203 Side population selection was used to identify multipotent mesenchymal and vascular precursors in the lung.194,204–211
Transitioning from the so-called “side population” (SP) of cells to lineage tracing and expression of ABCG2 in adult lung facilitated lineage-tracing studies to elucidate MPC location and function.36,194,195 ABCG2 MPC are a perivascular population of cells that share properties of differentiated NG2 pericytes and are more closely related to pericytes than fibroblasts. 36 However, MPC significantly differ from NG2 pericytes in functional properties including contraction.36,194 We chose to evaluate naïve vs. canonical Wnt activated MPC 194 because Wnt/β-catenin signaling is biologically relevant, due to its association with tissue homeostasis and many adult pulmonary and vascular diseases.36,212–218 While Wnt signaling in adult CLD has been well studied in terms of the epithelium, little is understood about its role in MPC regulation of the microvasculature and pathological angiogenesis.
β-catenin signaling regulates MSC/MPC cell specification, cell differentiation, renewal, proliferation, and angiogenesis.4,166,194,213,219–235 We found that conditional genetic stabilization of β-catenin in ABCG2pos MPC resulted in expansion of this progenitor pool in the lung. However, the MPC did not assume contractile function or expression of smooth muscle alpha actin, while microvessel expression of smooth muscle alpha actin decreased. Alteration of proper MPC function in the lung, via dysregulated Wnt/β-catenin signaling, facilitated the finding that MPC regulate lung microvascular homeostasis and function (Fig. 1).
194
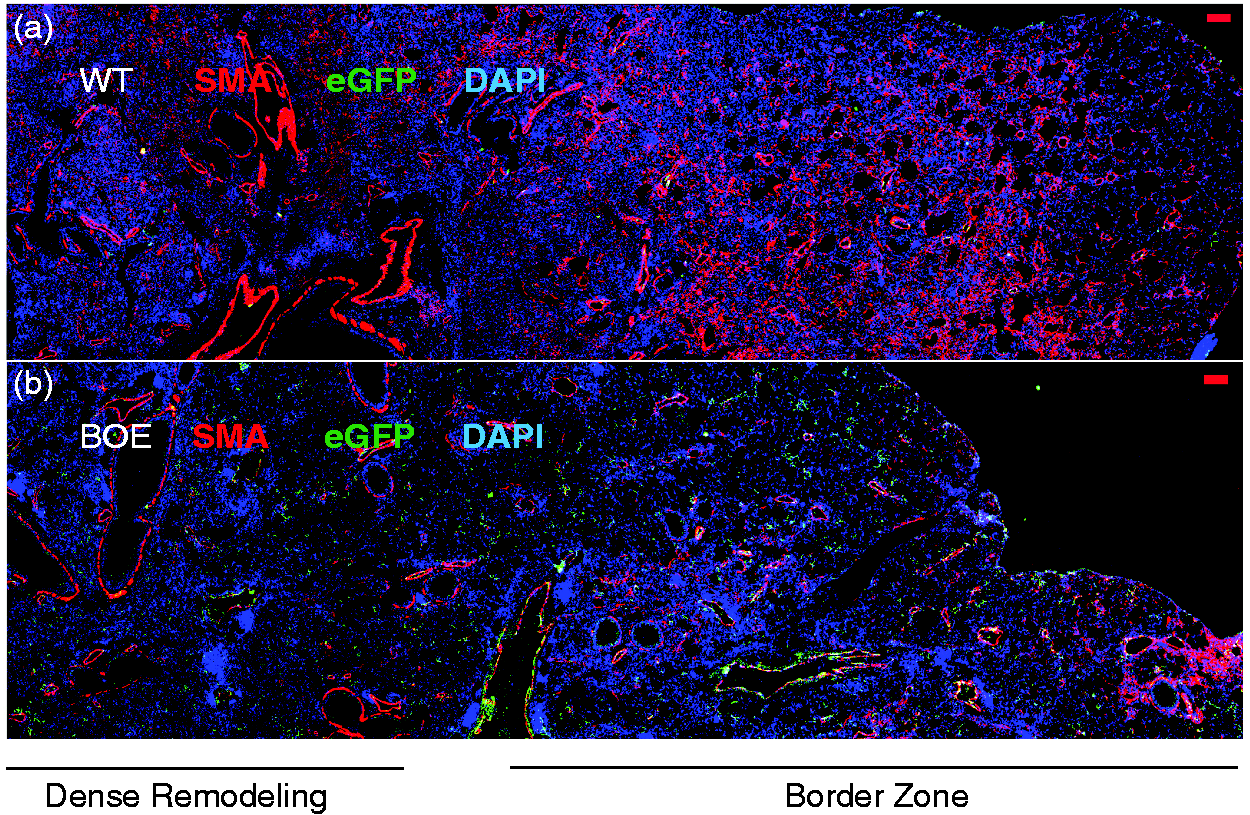
Loss of MPC function resulted in a decreased microvascular density, contractility, and smooth muscle cell homoeostasis. In a murine model of bleomycin-induced fibrosis, wild-type ABCG2pos MPC associated with both smooth muscle alpha actin or Factor VIII positive microvessels in alveolar tissue peripheral to the actively remodeling regions (Figs. 2 and 3).
194
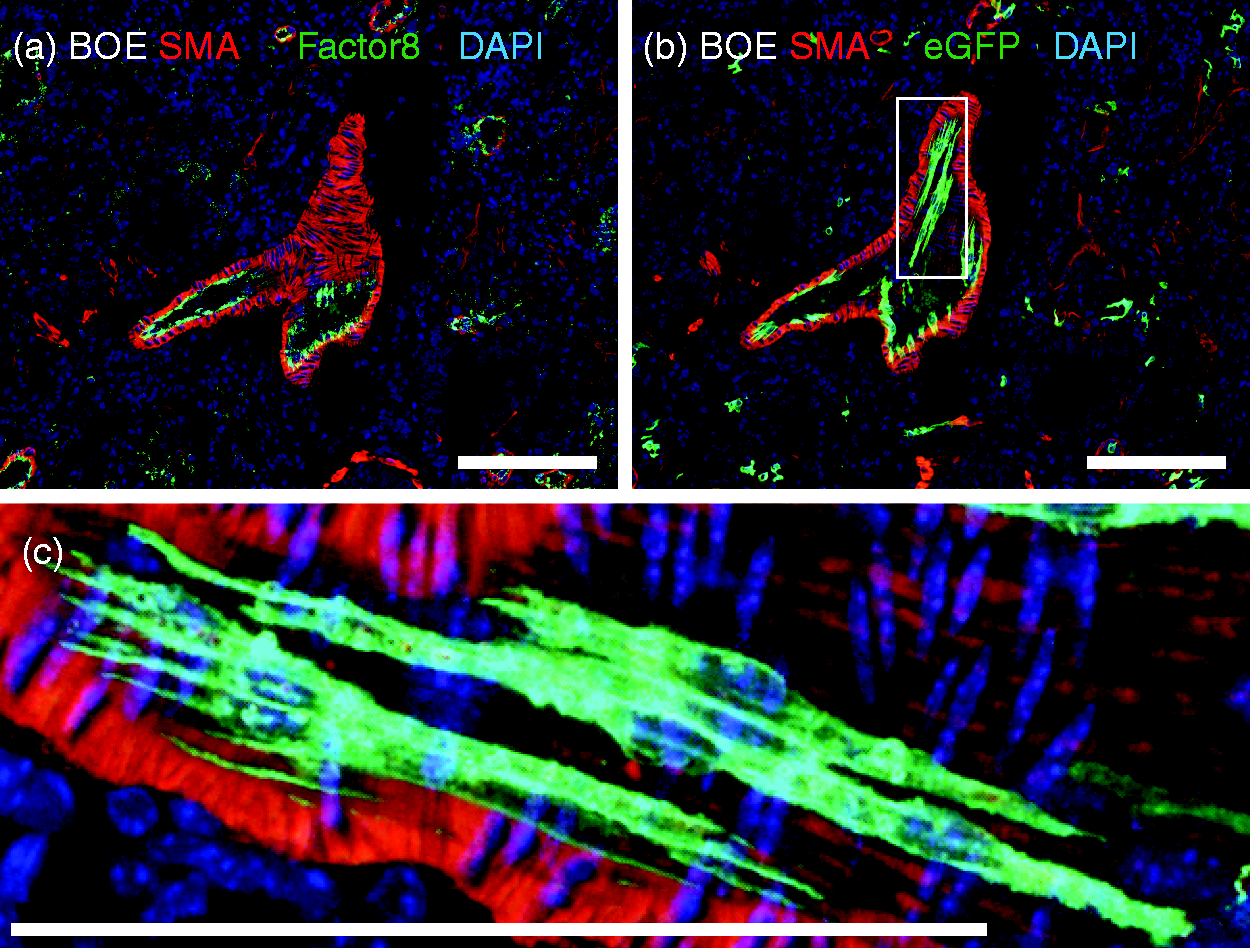
However, in the lung tissue of Wnt activated MPC (termed βOE), we detected MPC contribution to atypical vascular structures in the fibrotic and peripheral areas of remodeling. These microvessels were atypical because they were devoid of both Factor VIII expressing endothelium and smooth muscle alpha actin (Fig. 3).
194
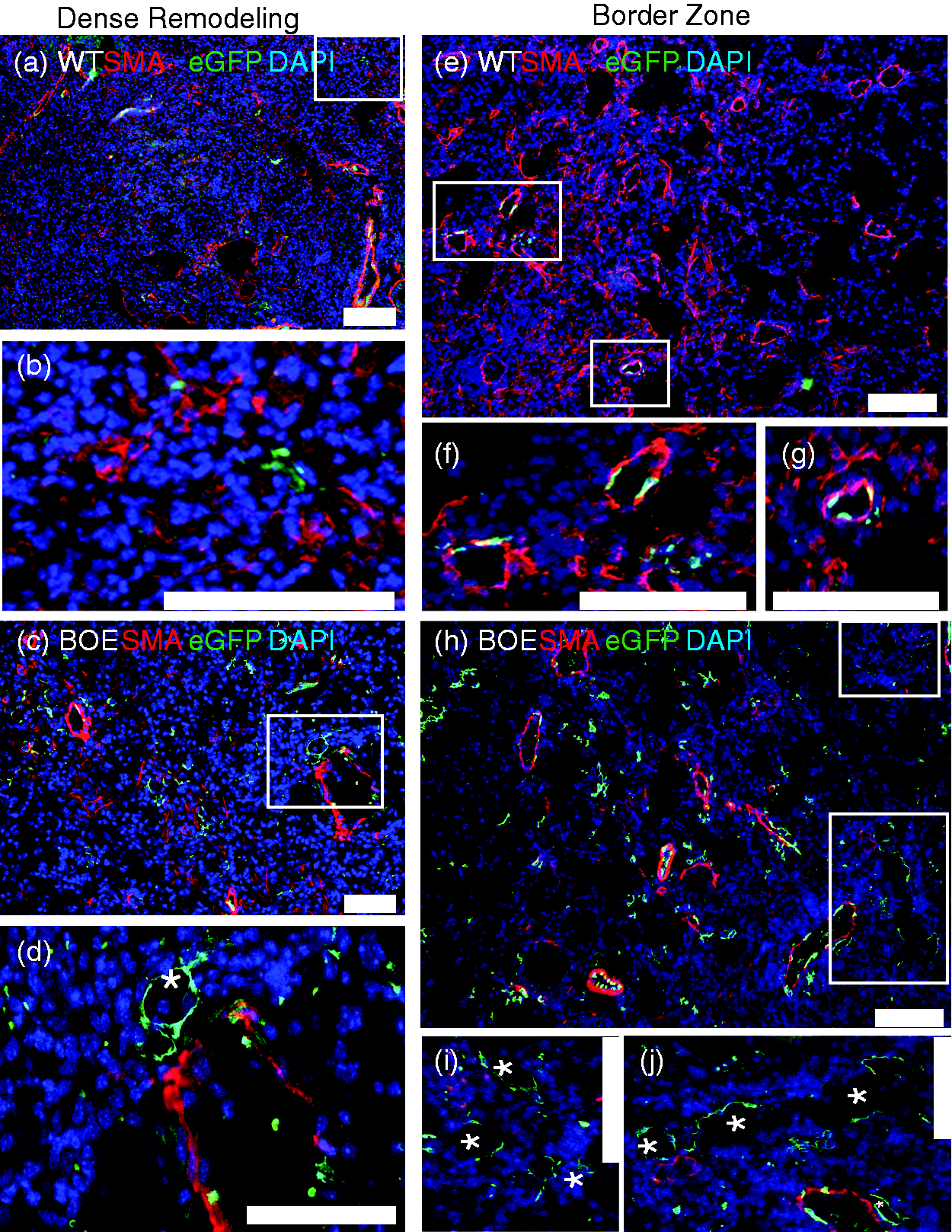
Of note, in response to bleomycin injury we detected migration of MPC, or derived cells, along the existing alveolar vasculature (arterioles, capillaries, and veins; Figs. 3 and 4).
Hypothetical model showing a role for MPC during the development of vasculopathy in disease. During tissue homeostasis, MPC participates in the regulation of capillary microvessels. Injury results in increased MPC β-catenin activity, and loss of MPC–MVEC interactions. MVEC respond with decreased barrier function. MV density decrease while MPC increase migration and abnormal angiogenesis. We speculate that prolonged vascular remodeling at the expense of repair results in CLD. Overview of remodeling and angiogenesis in bleomycin injured mouse lung tissue. β-catenin stabilization in MPC was achieved by engineering a conditional activator [β-catenin lacking degradation sites; Catnbloxp(ex3)], targeted to lung MPC using ABCG2CreERT2 with reporters:Rosa26 mtomato/mGFPlox-stop.36,166,200,249,250 Mice were induced with intraperitoneal low dose tamoxifen (0.5 mg total).
194
Groups: All room air exposure; Control, or Wnt activated/β-catenin over-expressors (βOE). Two weeks following induction, 0.15 U of bleomycin or PBS vehicle was administered intratracheally and mouse lung tissue harvested on day 14 peak fibrosis and analyzed. Immunostaining was performed on lung tissue sections to localize smooth muscle alpha actin (SMA) and eGFP-labeled MPC lineage cells. SMA-labeled myofibroblasts in areas of remodeling as well as muscularized microvessels, airways and vasculature. (a) Representative WT or (b) β–catenin over-expressor (βOE) mouse lung tissue sections. DAPI was used to stain nuclei (blue). Scale bars = 100 μM. MPC contribution to abnormal angiogenesis in bleomycin injured mouse lung tissue. Mice were induced with intraperitoneal low dose tamoxifen (0.5 mg total).
194
Two weeks following induction, 0.15 U of bleomycin or PBS vehicle was administered intratracheally and mouse lung tissue harvested on day 14 peak fibrosis and analyzed. Immunostaining was performed on lung tissue sections to localize smooth muscle alpha actin (SMA) and eGFP-labeled MPC lineage cells. Representative images were taken in the areas of (a–d) dense remodeling and (e–j) border zones outlined in Fig. 2. (a, b, e, g) WT or (c, d, h, j) β–catenin over-expressor (βOE) mouse lung tissue sections. DAPI was used to stain nuclei (blue). Scale bars = 100 μM. Enhanced intravascular migration of βOE MPC following bleomycin injury. Immunostaining was performed on day 14 bleomycin injured β–catenin over-expressor (βOE) mouse lung tissue sections to localize smooth muscle alpha actin (SMA) expressing vasculature (a–c), Factor VIII positive endothelium (a) and eGFP-labeled MPC lineage cells (a–c). DAPI was used to stain nuclei (blue). Scale bars = 100 μM.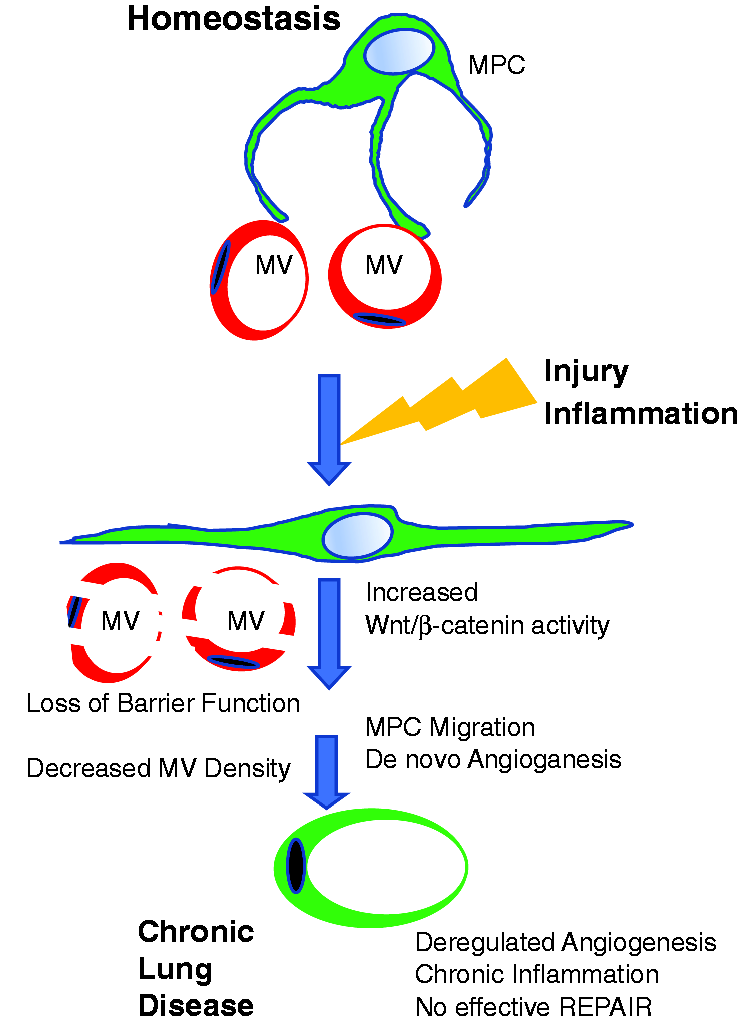
Because lymphangiogenesis is deregulated in CLD, it is reasonable to hypothesize that MPC may also influence this process during injury and disease. We therefore analyzed the MPC lineage labeled cells and abnormal vascular structures for the expression of lyve-1.4,43,45,49,51–53,58,158 Lyve-1 is a receptor for hyaluronan, necessary for cell migration and metastases as well as a typical marker for lymphangiogenesis and existing lymphatic vasculature (Fig. 5a–c).44,52,53,74,192,236 However, lyve-1 expression is not limited to lymphatic endothelium.
56
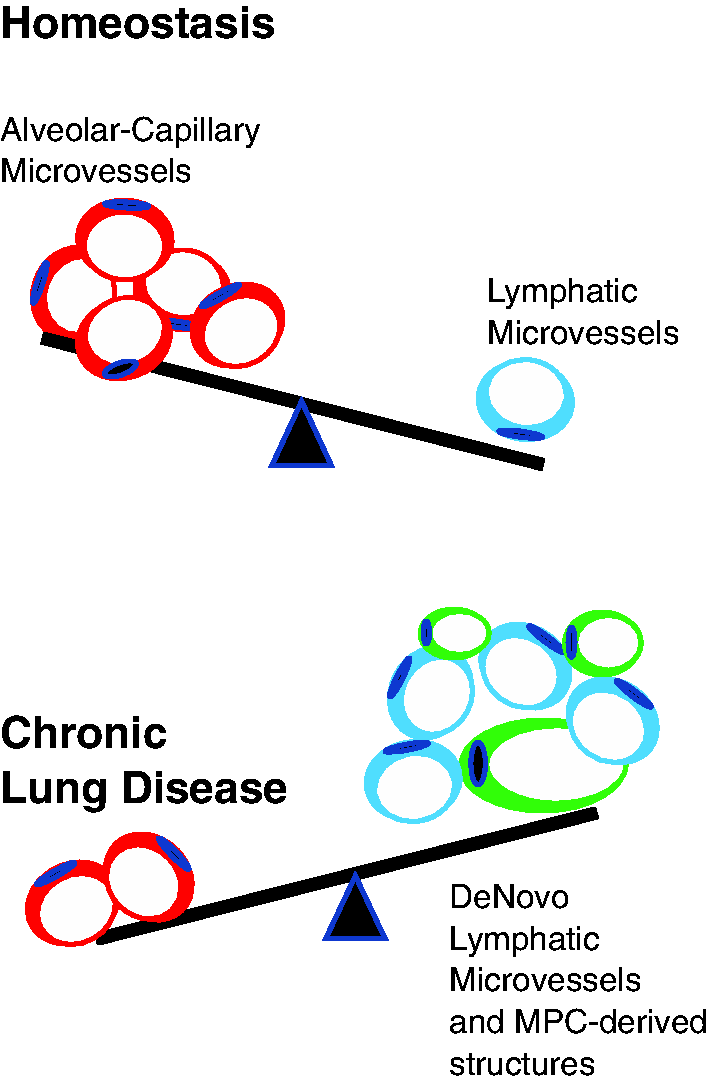
We found that the MPC lineage and the de novo vascular structures exhibited heterogeneous expression of lyve-1 (Fig. 5d and e). Heterogeneous expression of lyve-1 may be due to stage of vessel formation and cell (MPC or endothelial) migration.43,49–53 We therefore speculate that during disease MPC contribute to abnormal angiogenesis that contributes to the imbalance of capillary and lymphatic microvasculature (Fig. 6).
MPC contribute to de novo lymphangiogenesis. Immunostaining was performed on day 14 bleomycin vehicle control (a, b) or injured (c–e) β–catenin over-expressor (βOE) mouse lung tissue sections to localize smooth muscle alpha actin (SMA) expressing vasculature, lyve-1 expressing cells/lymphatics and eGFP-labeled MPC lineage cells. DAPI was used to stain nuclei (blue). Scale bars = 100 μM. Balance of capillary and lymphangiogenesis: hypothetical model of vasculopathy in CLD. Abnormal MPC vascular structures = green.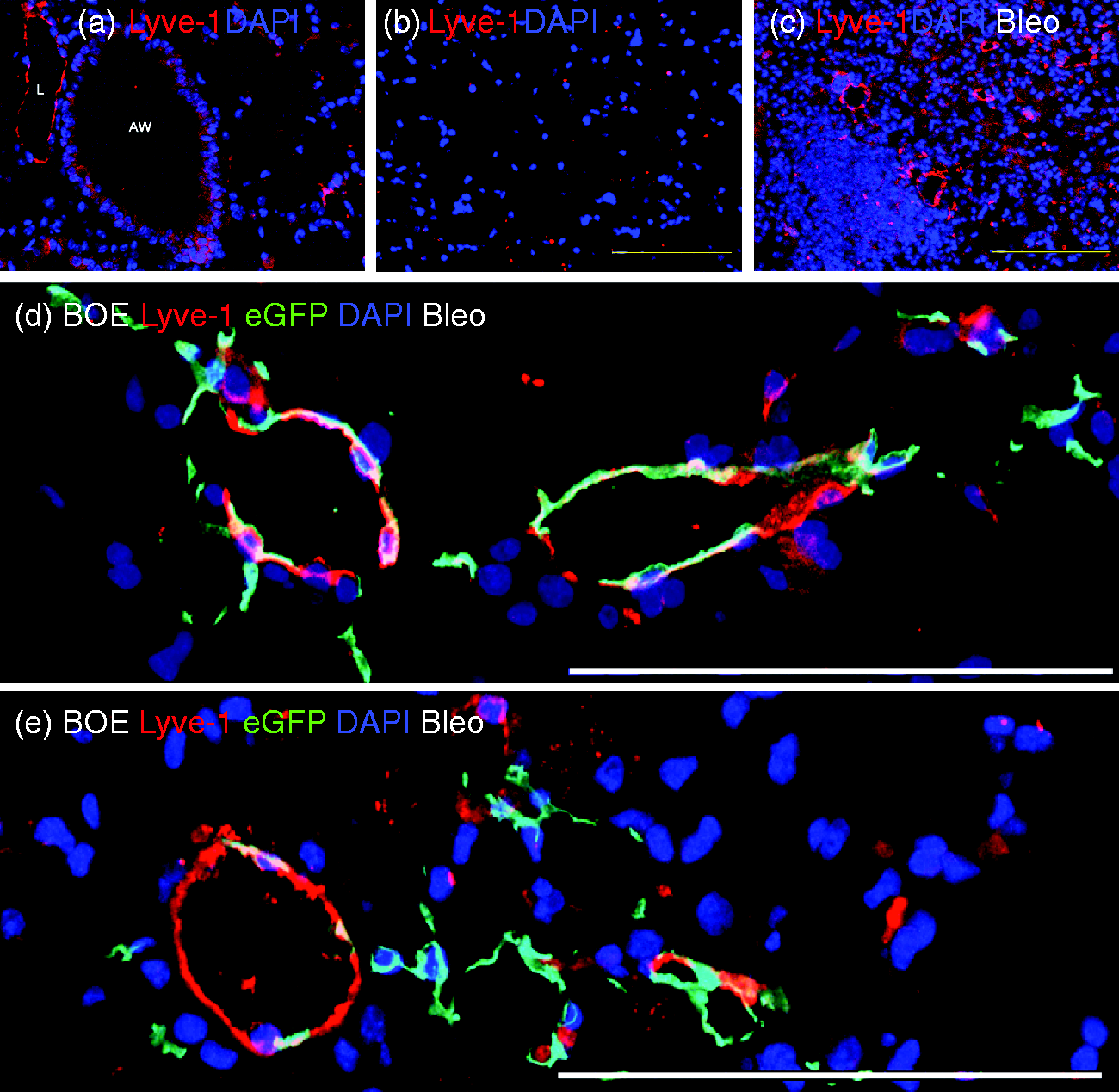
Summary
Conclusions
Capillary and lymphangiogenesis are deregulated in both IPF and COPD, although the mechanisms by which they co-regulate and underlie early pathogenesis of disease are unknown. We have shown that that ABCG2pos MPC influence both the capillary microvasculature and lymphatic angiogenesis. A balance of both is required for normal tissue homeostasis and repair. Our current models suggest that when lymph and capillary angiogenesis are out of balance, the non-equivalence appears to support the progression of disease and tissue remodeling. The angiogenic regulatory mechanisms underlying both COPD/emphysema, IPF, and SSc likely impact additional CLD including other interstitial lung diseases, tuberous sclerosis (TSC), and lymphangioleiomyomatosis.
Future directions
Ongoing studies are designed to elucidate the mechanisms by which MPC influence capillary and lymphangiogenesis as well as understanding the balance between them in normal lung tissue, following injury, and during repair. The MPC niche and environment regulates their function which in turn impacts the vascular microenvironment (Fig. 1). This is a seemingly complex interaction because the niche involves endothelium, epithelium, vascular smooth muscle, extracellular matrix, and environmental influences.
Cigarette smoke is a major risk factor for CLD including COPD/emphysema and IPF, and is therefore a reasonable candidate factor to deregulate the MPC niche. Both cigarette smoke and hypoxia upregulate the expression of ABCG2.237–239 Additionally, the MPC express the hyaluronan receptors CD44 and lyve-1 which function to regulate cell migration, Wnt, mTOR, and VEGF signaling as well as phenotype and function.68,192,240–243 Hyaluronan is cleaved into high and low molecular weight fragments differentially during disease and has been characterized as altered in COPD and IPF.46,56,68,240,241,244–246 These receptors cluster with ABCG2 at the cell surface 247 and likely regulate cell-specific niches and their responses to the microenvironments, including proliferation, apoptosis, response to oxidant stress, and metabolism.247,248
A fundamental question that remains is how MPC reprogrammaing disrupts capillary and lymphatic networks to hinder the physiologic function of the lung. Establishing these mechanisms would provide key new insights into the pathogeneis of lung disease. Therefore, it is essential to examine these aspects of the MPC niche, including ABCG2 regulation of cell signaling, proliferation, and migration, using murine models of emphysema and fibrosis as well as isolated human MPC. The ultimate goal of these studies is to identify novel therapeutic targets to restore MPC and vascular function and promote pulmonary tissue repair.
Footnotes
Acknowledgments
The authors thank Dr. M.M. Taketo for providing the flΔEx3 catnb mice.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This work was funded by grants to S.M. Majka from the NIH/NHLBI R01HL116597. Additional funding was provided by NIH/NHLBI K08HL130595 and the Francis Family Foundation to J.A. Kropski.
2017 Grover Conference Series
This review article is part of the 2017 Grover Conference Series. The American Thoracic Society and the conference organizing committee gratefully acknowledge the educational grants provided for the support of this conference by Actelion Pharmaceuticals US, Inc., Gilead Sciences, Inc., and United Therapeutics Corporation. Additionally, the American Thoracic Society is grateful for the support of the Grover Conference by the American Heart Association, the Cardiovascular Medical Research and Education Fund, and the National Institutes of Health.
