Abstract
In patients with Eisenmenger syndrome, life expectancy is usually longer than in patients with other forms of pulmonary arterial hypertension (PAH). We conducted a cohort study in which patients were followed over a long period of time in an attempt to identify potential predictors of clinical outcomes. Sixty-seven treatment-naïve patients were enrolled (age range = 12–60 years; median age = 33 years). Baseline demographic, diagnostic, and functional parameters, plasma levels of endothelial dysfunction markers, and treatment-related data were tested for possible correlations with event-free survival. Patients were started on oral PAH drugs at the beginning of follow-up (n = 23), during follow-up (n = 33), or remained untreated (n = 11). The duration of follow-up was 0.54–9.89 years (median = 7.13 years), with an overall survival rate of 82% and an event-free survival rate of 70%. The estimated mean for event-free survival time was 7.71 years (95% confidence interval [CI] = 6.86–8.55 years). Of the 16 variables that were analyzed, the duration of exposure to PAH drugs was identified as an independent protective factor (hazard ratio [HR] = 0.25 for quartiles, 95% CI = 0.14–0.47, P < 0.001). The initial functional class (HR = 3.07; 95% CI = 1.01–9.34; P = 0.048), the severity of right ventricular dysfunction (HR = 2.51 [mild, moderate or severe dysfunction]; 95% CI = 1.22–5.19; P = 0.013) and plasma von Willebrand factor concentration (HR = 1.74 for quartiles; 95% CI = 1.07–2.83; P = 0.026) were identified as risk factors. The length of exposure to oral PAH therapies influences survival favorably in Eisenmenger patients. This may be of interest for communities where access to medications is restricted.
Keywords
Introduction
Eisenmenger syndrome is the most advanced form of pulmonary arterial hypertension associated with congenital heart disease (PAH-CHD). 1 The overall prevalence of Eisenmenger syndrome in adults with CHD is approximately 1–6%. The prevalence varies depending on the type of cardiac anomaly present. 2 However, the number of patients with PAH-CHD seen in tertiary centers appears to be increasing even in developed nations. 3 In certain areas of developing countries, the prevalence of PAH-CHD is even higher than idiopathic PAH (IPAH) and connective tissue disease-related PAH. 4
The prognosis of patients with Eisenmenger syndrome (particularly survival) is by far better than the prognosis of patients with IPAH.5,6 Twenty-year survival rates of approximately 80% have been reported.7,8 Thus, studies aimed at characterizing predictors of prognosis for this syndrome must be based on long-term observations. Most patients die from sudden cardiac death, heart failure with hypoxemia, hemoptysis, thromboembolic events, brain abscesses or cerebrovascular accidents, complications during pregnancy, or non-cardiac surgery.
Of the several factors that have been correlated with patient survival in Eisenmenger syndrome, the complexity of cardiac defects appears to be central. 9 Iron deficiency is frequently associated with blood hyper-viscosity in these patients and represents a risk factor for adverse outcomes. 10 Data from the REVEAL registry show that for participants in the PAH-CHD cohort, a longer 6-minute walk distance (6MWD), lower mean right atrial pressure, brain natriuretic peptide level of < 50 pg/mL, and the presence of acute vasoreactivity were predictors of survival four years after enrollment; younger age and lower mean right atrial pressure were predictors of survival seven years after diagnosis. 11 In agreement with observations by Kawut et al. for idiopathic, familial and anorexigen-associated PAH, 12 we demonstrated that elevated plasma levels of the von Willebrand factor, a marker of endothelial dysfunction, were associated with an increased risk of mortality in patients with Eisenmenger syndrome who were followed for four years. 13 Access to specific PAH drugs has a significant positive impact on the survival of patients with this syndrome. 14
The purpose of the present study was to analyze factors that might predict the long-term deterioration of cardiovascular function and mortality in Eisenmenger patients. The cohort consisted of patients that had participated in previous shorter-duration studies of ours13,15 as well as naïve patients that were included afterwards. We included in the analysis not only clinical, functional, and treatment-related data but also biochemical markers of endothelial dysfunction.
Methods
Study participants
Data in this study were collected prospectively. Adolescents and adults with Eisenmenger physiology (right-to-left shunting with cyanosis) from a single center entered the study from July 2005 to February 2014 if they were in stable clinical condition and did not use specific medications for PAH. We included patients with normal systemic oxygen saturation at rest who became cyanotic with low oxygen saturation on mild exertion. At the time of entry, patients were receiving conventional therapy that often included warfarin. Prior to enrollment, patients underwent echocardiography, cardiac magnetic resonance imaging (MRI), or cardiac catheterization to confirm the diagnosis of advanced PAH-CHD. All patients were considered unsuitable candidates for the surgical repair of cardiac anomalies. Data collection was started at patient enrollment, not at the beginning of symptoms (which would be difficult to establish for this specific population of patients coming from distant regions in the country). After enrollment, patients were followed by a single group of physicians at the ambulatory care level or during hospitalization in the case of clinical deterioration. For inclusion in the study, a written informed consent was necessary (this was obtained from parents in the case of adolescents). The study was approved by the local Ethics Committee (CAPPesq #380/05).
Specific PAH treatments and clinical events
In the study, patients were treated with oral PAH medications (the endothelin receptor antagonists bosentan or ambrisentan, and/or the phosphodiesterase-5 inhibitors sildenafil or tadalafil), as these were the only available medications in the country. Monotherapy was the initial approach for all patients. For years, sildenafil was the only drug available. Many patients therefore received sildenafil as a first-line treatment. The need for combination therapy was established on the basis of symptoms, functional class (World Health Organization [WHO]) and the 6-minute walk test (6MWT; American Thoracic Society protocol), which was performed during all ambulatory visits. Depending on the severity of symptoms and the availability of drugs from the governmental program, some individuals had access to medication from the beginning of follow-up, some were started on the drugs during the follow-up, and some did not use specific drugs at all. Because the program is based on monthly release of medication, treatment discontinuation was not a problem in the study.
Patients were followed up in a specific ambulatory unit where they were assisted by a multiprofessional team. In the absence of clinical instability or relevant events (which would require assistance in the emergency room), they were evaluated on a four-month basis, but needed to be present monthly for medication obtainment and control (this was sometimes done by relatives). In order to avoid missing visits, all of them were called-up and advised by the multiprofessional team 1–2 days before. Information about patients who died at home was obtained from relatives either directly or by telephone. The circumstances of deaths occurring in other hospitals were discussed between doctors. There were no missing cases, events, or unregistered deaths in the study.
For the specific purpose of studying event-free survival, an event was defined as acute deterioration of any cause, with clinical features indicative of low cardiac output requiring cardiovascular support drugs (dopamine, dobutamine, epinephrine, norepinephrine), regardless of full or partial recovery, or assignment to transplantation. The need for the use of intravenous PAH drugs was not included in the criteria, as these drugs were not available. In general, patients presented to the emergency room with worsening cyanosis and advanced functional class IV. Arrhythmias, infection, or simply “the cold weather” were sometimes interpreted as the cause of deterioration. More frequently, however, worsening of symptoms was attributed to a failing right ventricle, blood hyperviscosity eventually associated with iron depletion, small-vessel pulmonary arterial thrombosis (mild elevation of plasma D-dimer without large-vessel occlusion on chest tomography), or the natural history of disease.
Parameters included in the analysis
Demographic, diagnostic, functional, and general laboratory data were obtained from all patients at enrollment. Cardiac anomalies were classified into five categories: pre-tricuspid defects; post-tricuspid defects except for A-V canal; A-V canal; conotruncal defects; and complex anomalies. Although echocardiographic evaluation was not planned to provide specific indices of cardiac function, it was possible to have qualitative information about right ventricular function using multiple acoustic windows according to recommendations by the American Society of Echocardiography.16,17 Using such imaging windows to completely visualize the walls from the base to the apex, right ventricular systolic function was classified as normal or mildly impaired, moderately impaired or severely impaired. In addition, pulmonary arterial systolic pressure was estimated based on the tricuspid regurgitant jet velocity.
Plasma samples were obtained for the analysis of endothelial dysfunction markers. Concentrations of P-selectin, tissue-type plasminogen activator (t-PA), and von Willebrand factor antigen (VWF:Ag) were measured and computed for all patients at the beginning of the follow-up period (enzyme-linked assays, duplicate determinations, R&D Systems, Minneapolis, MN, USA [P-selectin] and Diagnostica Stago, Asnières, France [t-PA and VWF:Ag]). Plasma B-type natriuretic peptide (BNP) was not measured for the specific purpose of the study. Data (chemiluminescence assay) were obtained from the institutional registry. For each patient, we selected a determination performed as close as possible to the beginning of the follow-up.
Treatment-related data were collected at the time of enrollment and throughout the follow-up period. The survival status, the length of time to death or clinical deterioration (event, as defined), as well as pretreatment and treatment time were computed prospectively. All of the analyses that were carried out in the study were based on the principle of “intention to treat.”
Statistical analysis
Numeric variables are presented as medians and interquartile ranges (IQR); the criteria of closeness to normal distribution was not met for most values. Categorical variables are presented as absolute numbers of cases and percentages. Comparisons between groups were performed using the Mann–Whitney test (numeric variables) or the Chi-square family of tests (categorical variables). Cox proportional hazards regression was used to identify factors potentially impacting event-free survival. All of the numeric variables were analyzed as quartiles. Univariate analysis was carried out as the initial procedure. Once a variable (or variables) was selected as a candidate, bivariate analysis was performed to identify possible enhancers and confounders. A confounder was defined as a second variable capable of promoting a >10% change in the initial HR toward the opposite direction. Multivariate analysis was then performed as an attempt to develop a final model. We adopted the forward LR procedure for variable selection, with significance levels of 0.05 for inclusion and 0.10 for exclusion. Finally, Kaplan–Meier curves with Log-Rank (Cox-Mantel) statistics were used to illustrate survival differences. Unless otherwise specified, 0.05 was adopted as the significance level in all tests. Analysis were performed using the IBM-SPSS statistical package, version 22.
Results
Demographic, diagnostic, functional, and treatment data.

Data are presented as the number of patients and percentage, or median value and IQR.
Resting O2 saturation and 6MW O2 saturation refer to peripheral oxygen saturation measured before and at the end of a 6-min walk, respectively.
Calculated based on the tricuspid regurgitant jet velocity.
Analyzed qualitatively by a blind observer who had no access to follow-up data, according to pre-established methodology.16,17
World Health Organization classification.
From enrollment to beginning of therapy.
Only patients who were treated.
6MWD, 6-minute walk distance (American Thoracic Society protocol).
Specific laboratory analysis performed at baseline showed increased plasma VWF:Ag in patients compared with controls (123 [109–137] U/dL and 90 [71–102] U/dL, respectively; P < 0.001), increased t-PA (8.22 [6.23–10.65] ng/mL and 4.50 [3.82–5.58] ng/mL, respectively; P < 0.001) and increased P-selectin (43.9 [29.6–51.9] ng/mL and 20.6 [14.8–24.9] ng/mL, respectively; P < 0.001). Plasma level of BNP was 61.0 (28.8–183.5) pg/mL (reference normal levels < 100 pg/mL).
Univariate analysis: factors with potential impact on event-free survival.
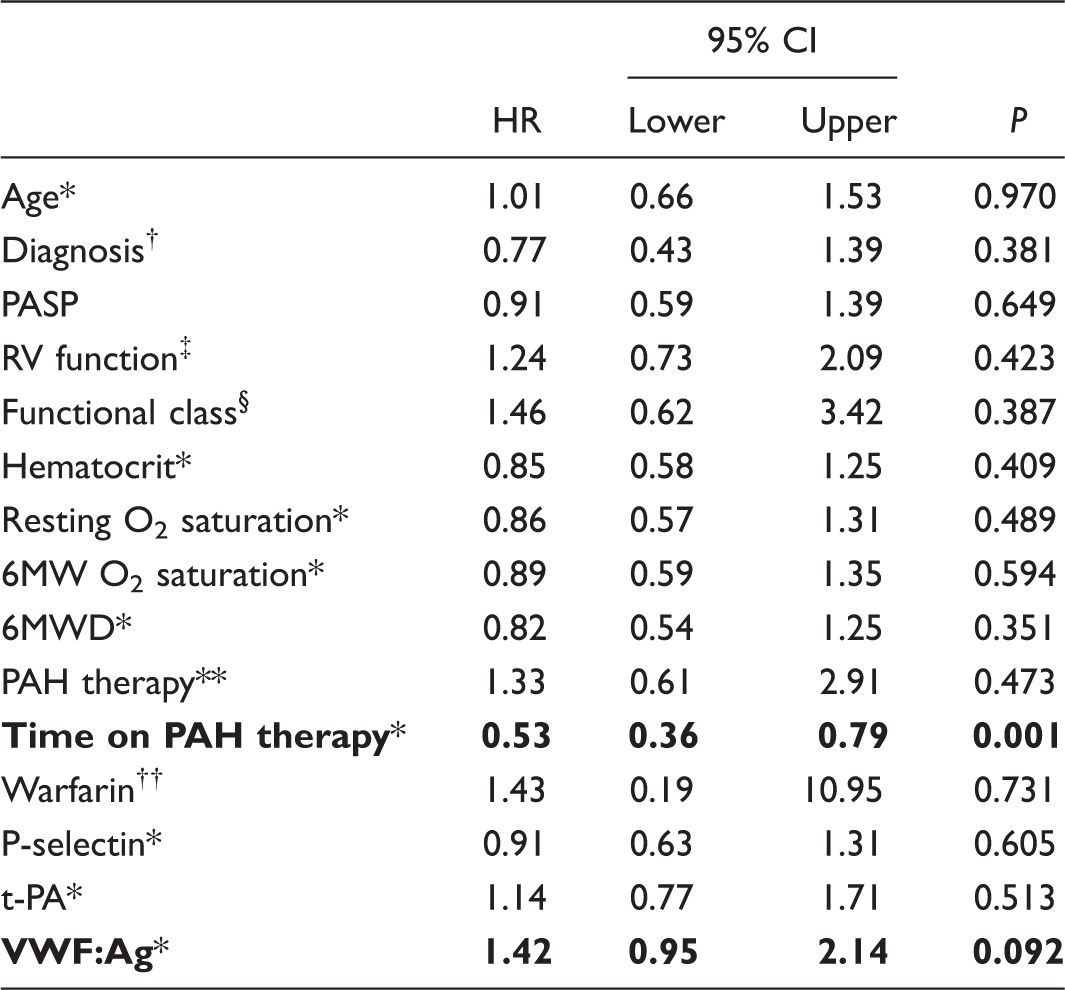
Resting O2 saturation and 6MW O2 saturation refer to peripheral oxygen saturation measured at rest and at the end of a 6-min walk, respectively.
All numeric variables were analyzed as quartiles.
Analyzed as categories: pre-tricuspid defects; post-tricuspid defects except for AV canal; AV canal; conotruncal defects; complex anomalies.
Right ventricular systolic function, analyzed as categories: normal or mildly impaired; moderately impaired; severely impaired.
World Health Organization classification.
Analyzed as categories: initiated at the beginning of follow-up; initiated during the follow-up; never used.
Chronic oral anticoagulant therapy.
HR: hazard ratio (Cox Proportional Hazards regression); 6MWD, 6-minute walk distance (American Thoracic Society protocol); PAH, pulmonary arterial hypertension; PASP, pulmonary arterial systolic pressure; t-PA, plasma tissue-type plasminogen activator; VWF:Ag, plasma von Willebrand factor antigenic concentration.
Bivariate analysis: time on PAH therapy as a protective factor and potential modifiers.
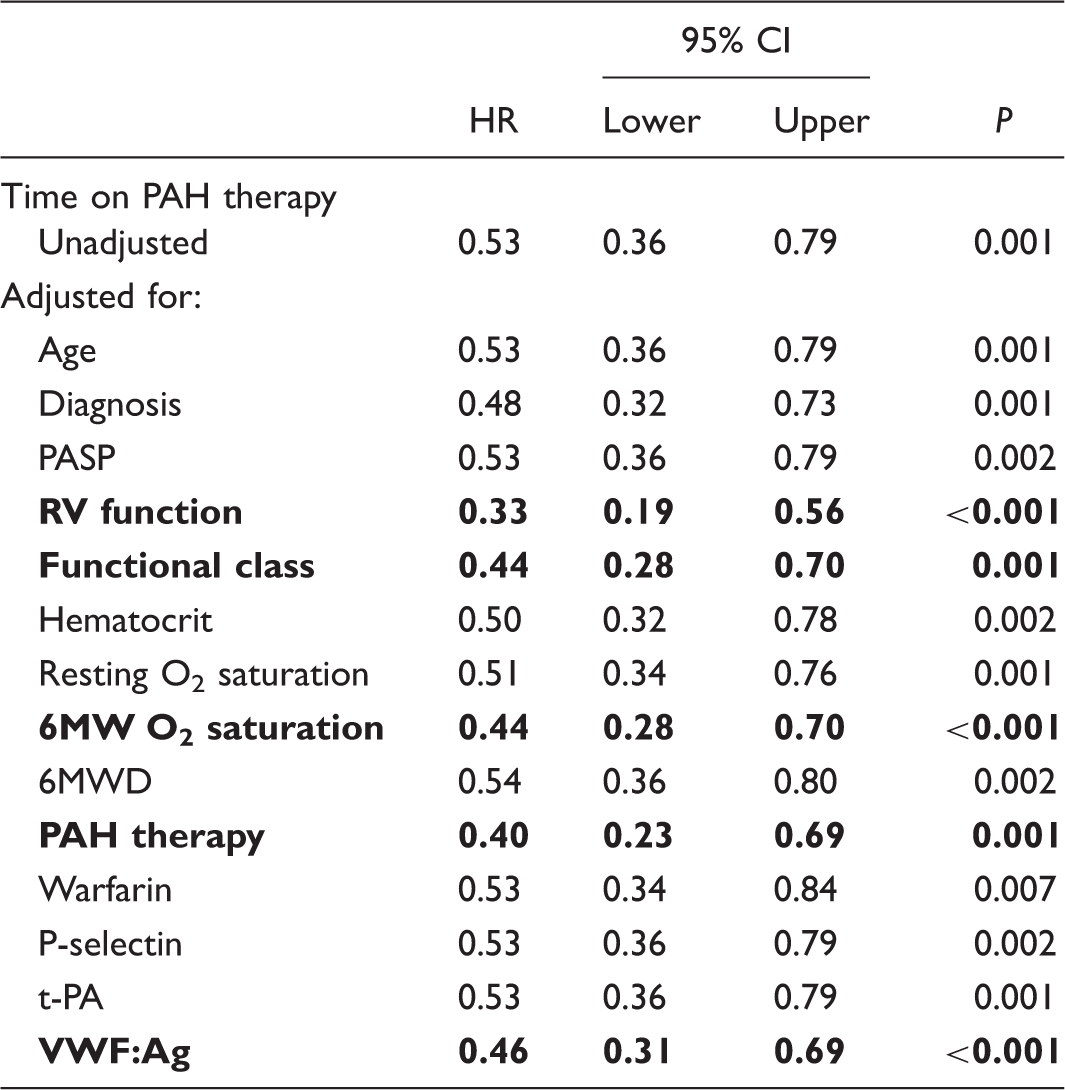
For details about the variables, see the legend of Table 2.
Resting O2 saturation and 6MW O2 saturation refer to peripheral oxygen saturation measured at rest and at the end of a 6-minute walk, respectively.
Highlighted are the variables that promoted a > 10% change in the initial HR associated with time on PAH therapy.
HR, hazard ratio (Cox Proportional Hazards regression); 6MWD, 6-minute walk distance (American Thoracic Society protocol); PAH, pulmonary arterial hypertension; PASP, pulmonary arterial systolic pressure; t-PA, plasma tissue-type plasminogen activator; VWF:Ag, plasma von Willebrand factor antigenic concentration.
Multivariate analysis: covariates in the final models for the prediction of event-free survival.
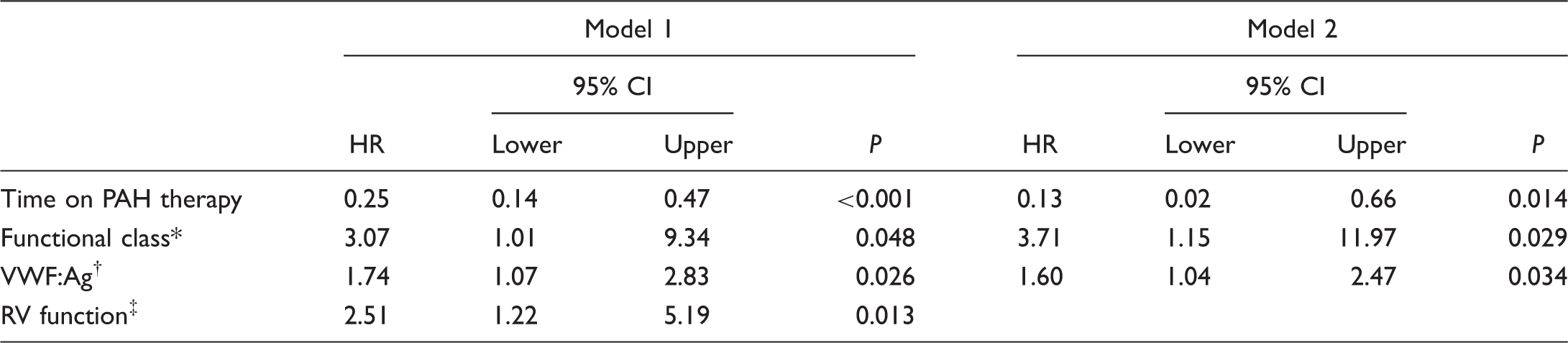
Model 1: Variables were tested using the Forward LR procedure for inclusion. Time on PAH therapy was analyzed as quartiles
Model 2: Time on PAH therapy was analyzed relative to total follow-up time (treatment time/follow-up time).
World Health Organization classification.
Plasma VWF:Ag concentration was analyzed as quartiles in both models.
Right ventricular systolic function, analyzed as categories: normal or mildly impaired; moderately impaired; severely impaired.
HR, hazard ratio (Cox Proportional Hazards regression); PAH, pulmonary arterial hypertension.
Kaplan–Meier survival curves and Log-Rank statistics for the duration of exposure to PAH drugs and baseline levels of VWF:Ag are presented in Fig. 1. For time on PAH therapy, patients in quartiles 3 and 4 had a significantly better survival compared with quartiles 1 and 2. For baseline VWF:Ag, a borderline advantage in terms of survival was observed for participants in quartile 1 compared with quartile 4.
Kaplan–Meier event-free survival curves according to the duration of exposure to PAH therapies and baseline plasma levels of VWF:Ag analyzed as quartiles. (a) Time on PAH drugs for quartiles 1 through 4 (Q1–Q4) was 0.00–0.54 years, 0.84–2.13 years, 2.19–4.15 years, and 4.19–9.58 years, respectively. The respective means for survival time (and 95% CI) were 5.55 (3.72–7.38) years, 5.27 (3.06–7.48) years, 8.53 (7.19–9.87) years and 9.47 (9.10–9.85) years. Survival was significantly better for Q3 and Q4 compared with Q1 and Q2 (P < 0.01, Log-Rank pairwise comparisons). (b) Plasma VWF:Ag for Q1–Q4 was 73–108 U/dL, 109–122 U/dL, 123–136 U/dL, and 137–177 U/dL, respectively. The respective means for survival time (and 95% CI) were 8.89 (8.47–9.32) years, 7.17 (5.54–8.90) years, 7.81 (6.18–9.44) years, and 6.68 (4.93–8.43) years. There was a trend toward a better survival in Q1 compared with Q4 (P = 0.055). The overall event-free survival time (all patients) was 7.71 years (95% CI = 6.86–8.55 years).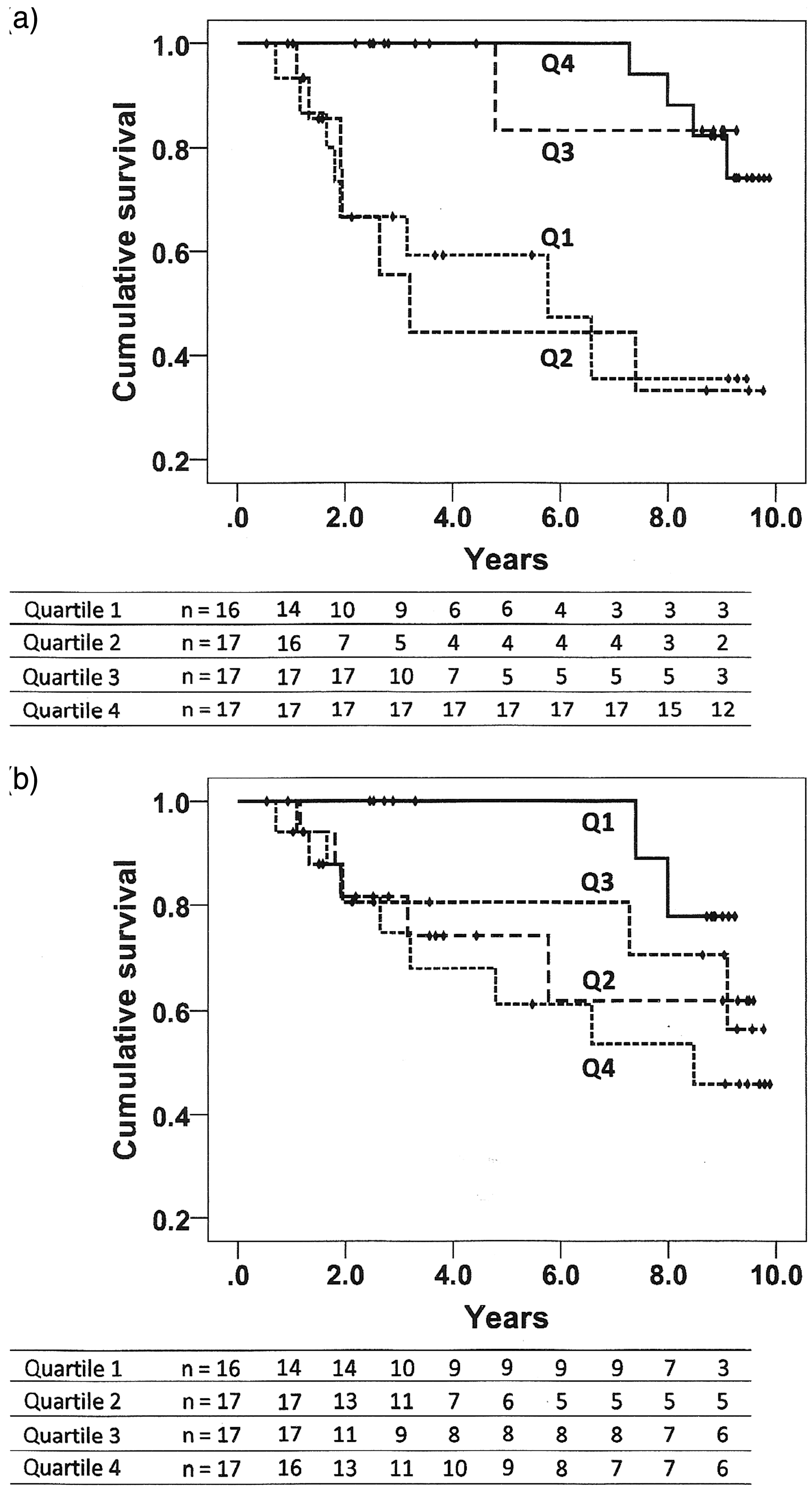
Additional analyses were performed including only participants with resting oxygen saturation of < 90% (n = 39). Time on PAH therapy remained as a protection factor in univariate and multivariate analysis (respectively, HR = 0.43 for quartiles, 95% CI = 0.25–0.75, P = 0.003 and HR = 0.10, 95% CI = 0.03–0.35, P < 0.001). In multivariate analysis, right ventricular dysfunction (HR = 3.85, 95% CI = 1.47–10.13, P = 0.006), the initial functional class (HR = 4.52, 95% CI = 1.19–17.15, P = 0.026) and peak-exercise oxygen saturation (HR = 0.20 for quartiles, 95% CI = 0.05–0.85, P = 0.029) also influenced event-free survival.
Initial clinical parameters in patients receiving PAH drugs for different periods of time.
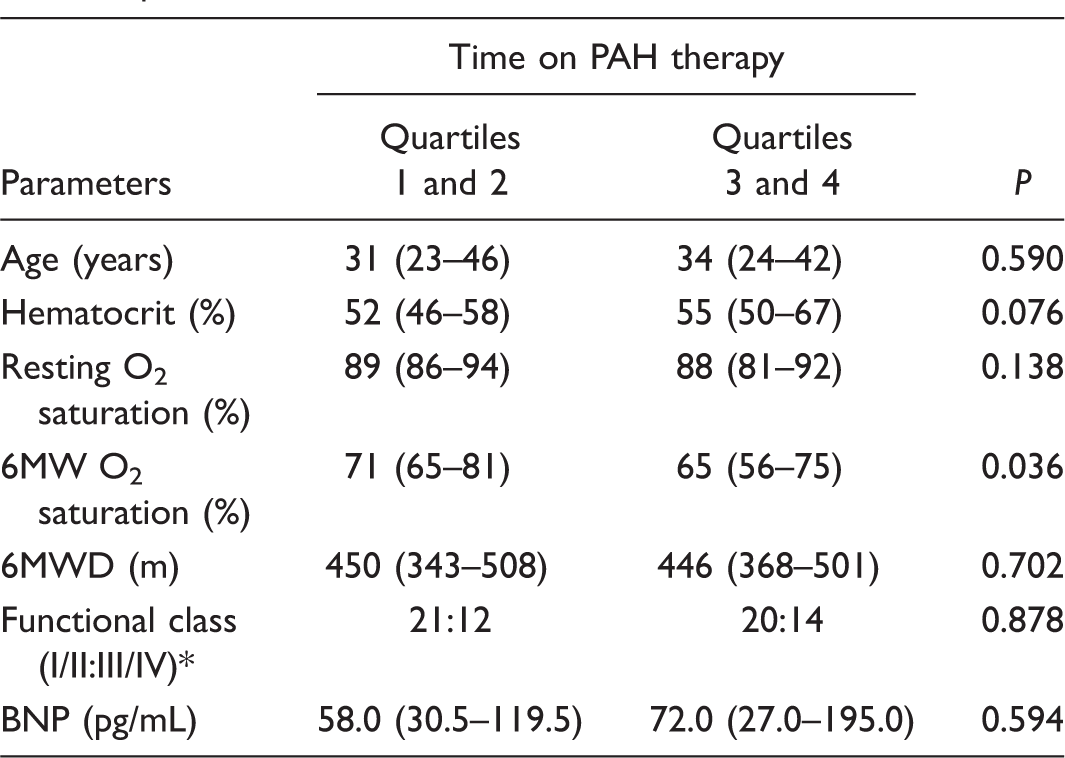
Numeric variables are presented as median value and IQR.
Resting O2 saturation and 6MW O2 saturation refer to peripheral oxygen saturation measured at rest and at the end of a 6-min walk, respectively.
World Health Organization classification.
6MWD, 6-minute walk distance (American Thoracic Society protocol); BNP, plasma B-type natriuretic peptide; PAH, pulmonary arterial hypertension.
Discussion
This prospective study was focused on a cohort of Eisenmenger patients followed for up to 9.89 years. Of the variables that were analyzed with potential impact on outcomes, duration of exposure to specific PAH drugs was identified as an independent determinant of prognosis by univariate, bivariate, and multivariate analyses. Pre-treatment time (from enrollment to the initiation of therapy) was taken into consideration indirectly, by further analyzing time on PAH drugs as a fraction of the total follow-up duration. There is growing evidence suggesting benefits of PAH therapies in patients with Eisenmenger syndrome.18,19 The benefits of bosentan administration were demonstrated in the BREATHE-5 randomized placebo-controlled study. 20 Improved survival has been reported for Eisenmenger patients who received PAH-specific therapies, namely bosentan, sildenafil, epoprostenol, or combinations of these drugs. 14 In the present study, the time on oral PAH drugs influenced prognosis. Medications for PAH management are only available in the oral dosage form in this country, as is probably the case in many developing countries. Thus, our observations may be relevant to patients of communities where access to medications is limited.
Of the variables investigated in this study, three were markers of endothelial dysfunction. At the beginning of the follow-up period, plasma levels of all three markers were elevated compared with controls. Circulating markers of microvascular dysfunction and inflammation have been investigated in patients with PAH in general, and specifically in patients with PAH-CHD. These include selectins, thrombomodulin, VWF:Ag, D-dimer, soluble CD40 ligand, C-reactive protein, osteoprotegerin, circulating endothelial cells, and a number of cytokines, chemokines, and related molecules.21–25 Preliminary observations of ours suggested an association between increased plasma VWF:Ag and one-year mortality in a small group of patients that included patients with PAH-CHD. 26 More recently, in the search for factors that could be associated with prognosis in Eisenmenger patients, we observed that elevated VWF:Ag was an independent predictor of four-year mortality (cohort of 46 individuals). 13 In the present study, with an extended patient population (n = 67) and a considerably longer follow-up, we observed that baseline VWF:Ag remained associated with outcomes to some extent.
Plasma BNP has been considered a standard biomarker in studies of survival in PAH in general and Eisenmenger syndrome in particular.27,28 We speculate that the lack of correlation with outcomes in the present study was probably due to the fact that BNP levels very likely changed with PAH therapies. This has already been demonstrated. 27 We did not plan the study to test the effects of therapies on biomarkers and correlate improvements with survival. It would be interesting to investigate this in the future in the Eisenmenger population.
The overall survival of 82% computed for our patients who were monitored for up to 9.89 years is not much different from ten-year survival rates reported for Eisenmenger patients from excellent tertiary care centers. 29 The presence of complex cardiac anomalies is associated with shortened survival time in these patients. 9 However, in our study, only five patients exhibited conotruncal or more complex anomalies. The functional capacity has been shown to have prognostic significance in Eisenmenger patients, 14 although its relevance may change depending on the duration of follow-up. In this study, the initial functional class had some relevance in bivariate and multivariate analysis, but the 6MWD did not. Of note, for patients in the PAH-CHD cohort of the REVEAL registry, 11 a longer 6MWD was a predictor of survival at four years after enrollment but not at seven years after diagnosis. Finally, 42% of our patients exhibited moderately or severely impaired right ventricular function, which seemed to influence outcomes. This finding is in agreement with previous observations that right ventricular dysfunction does occur in Eisenmenger patients and has prognostic relevance.30,31
Study limitations
An important limitation of the study was the non-inclusion of specific right ventricular function parameters in the analysis. Cardiovascular MRI is routinely used in our institution, but data were not available for many patients at the beginning of the follow-up period. Echocardiographic analysis was performed in all patients to visualize the detailed anatomy of cardiovascular anomalies and to confirm the presence of Eisenmenger physiology, but specific indices of right ventricular function were not systematically obtained. However, one of the authors (CRPC) reviewed and revised all echocardiographic analyses that were performed at the beginning of follow-up, and was able to qualitatively classify right ventricular function in 90% of cases according to pre-established methodology (American Society of Echocardiography). Right heart catheterization could help, but in most tertiary centers, it is no longer routinely performed in Eisenmenger patients. Finally, in view of the small number of patients with complex cardiac anomalies in our cohort, we were unable to analyze the impact of this factor.
Conclusion
We demonstrated a direct correlation between longer exposure to specific PAH drugs and better outcomes in patients with Eisenmenger syndrome. Functional status, right ventricular function, and circulating levels of the endothelial marker von Willebrand factor also appeared to influence patient outcomes. Because the current study was carried out using oral PAH therapies, these results may be of value in countries or areas where oral dosage forms are exclusively available. Additional studies with an extended number of biological, functional, and treatment-related parameters to improve risk stratification are needed in this population.
Footnotes
Acknowledgements
The authors thank Mrs. Roseli Polo for her technical assistance in the preparation of this manuscript.
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This work is supported by the Foundation for Research Support of the State of São Paulo (FAPESP), Brazil, grants nos. 50320/05 and 2012/10739-0.
