Abstract
Background:
Constipation is a frequent adverse effect of antipsychotic treatment in schizophrenia.
Objectives:
This study aimed to investigate the prevalence and risk factors of antipsychotic-induced constipation (AIC) in patients with schizophrenia and examine associated alterations in peripheral immune factors.
Methods:
A total of 154 patients with schizophrenia were categorized into constipation and non-constipation groups based on the Rome IV constipation criteria. Clinical assessment included the Positive and Negative Syndrome Scale, Constipation Assessment Scale, and Bristol Stool Classification Scale. A subset of patients underwent secondary confirmation of constipation status using the Radiopaque Marker. Peripheral immune factors were analyzed using ProcartaPlex in 44 patients with constipation and 44 without.
Design:
This was a two-phase, cross-sectional observational study.
Results:
The prevalence of constipation was 36.36% (56/154). Among the 88 patients assessed for immune factors, serum IL-10 levels were nominally elevated in the constipation group compared to the non-constipation group (p = 0.043, Cohen’s d = 0.32), an associated that did not remian statistically significant after false discovery rate correction (FDR-adjusted p = 0.432). In addition, the equivalent dose of clozapine showed a negative correlation with IL-1β levels. Logistic regression analysis showed a preliminary association of IL-10 with constipation.
Conclusion:
The relatively high prevalence of AIC, particularly among clozapine users, highlights the need for increased clinical awareness and monitoring. The findings may suggest a possible link between AIC and inflammatory processes. Notably, a preliminary observation of elevated peripheral IL-10 levels in patients with constipation needs.
Trial registration:
Not applicable.
Plain language summary
This study investigated constipation caused by antipsychotic drugs in people with schizophrenia. Researchers examined 154 patients, dividing them into those with and without constipation (based on standard diagnostic criteria). They assessed symptoms using different scales and confirmed constipation using a special marker method. Blood tests from 88 patients showed that those with constipation had nominally higher levels of an immune-related substance called “IL-10.” Patients taking higher doses of clozapine (a common antipsychotic) also had lower levels of another immune marker, IL-1β. Key findings: 1. 36% of patients developed constipation. 2. Patients with schizophrenia who take clozapine are more likely to experience constipation compared to those taking other antipsychotic medications. 3. IL-10 was nominally elevated in the constipation group compared to the non-constipation group, an associated that did not remian statistically significant after false discovery rate correction. 4. Clozapine dosage was linked to changes in immune markers. The study suggests doctors should monitor constipation closely in patients on antipsychotics, especially clozapine, as it may be related to inflammation. Immune factors like IL-10 could play a role in this side effect.
Introduction
Schizophrenia is one of the most common and serious mental disorders in clinical practice, typically emerging in early adulthood and often persisting over an extended period.1,2 Characterized by disturbances in perception, emotion, and behavior, schizophrenia imposes a substantial burden on patients and their families. 3 With a global prevalence of approximately 1% and a lifetime prevalence of about 0.62% in China,4,5 schizophrenia ranks among the costliest psychiatric conditions worldwide. 6 Antipsychotics, together with psychotherapy, are the cornerstone of treatment, effectively alleviating positive and negative symptoms.7,8 However, these medications frequently cause significant side effects, including constipation, which can severely impact the well-being of patients who are already in distress. 9
Antipsychotic-induced constipation (AIC), a common gastrointestinal disorder, may lead to severe complications such as intestinal obstruction, ischemic bowel disease, perforation, and bacterial septicemia, some of which can be life-threatening.10,11 Reported prevalence of AIC rates varies substantially, ranging from 1.5% to 45% for atypical antipsychotics,12–14 with clozapine demonstrating the highest risk (14%–60%).11,13 Ozbilen and Adams 14 reported that the prevalence of constipation associated with atypical antipsychotics was 1.5%–45%. Since these results are derived from a review of existing literature, the broad range may reflect heterogeneity in diagnostic criteria, study populations, and assessment methods. Compared to users of other antipsychotics, clozapine users had a 4.5-fold increased risk of constipation, with more severe symptoms and longer treatment durations. 15 A bilateral drug study found that patients treated solely with clozapine exhibited significantly prolonged colonic transit time (CTT) compared to those receiving clozapine in combination with other antipsychotics or other antipsychotics alone. 16 Furthermore, studies have indicated a dose-dependent increase in constipation risk for olanzapine and quetiapine.17–19
Mechanistic insights have suggested three primary proposed mechanisms: anticholinergic effects, 5-hydroxytryptamine receptor antagonism, and histamine H1 receptor antagonism, with the anticholinergic effect being the most prominent. Acetylcholine plays a crucial role in stimulating gastrointestinal smooth muscle activity and the interstitial cells of Cajal, thereby promoting intestinal peristalsis. 19 Anticholinergic activity suppresses duodenal contraction and motility, reduces colonic transit capacity, and disrupts autonomic regulation via antagonism of cholinergic M1 and M3 receptors, ultimately leading to prolonged gastrointestinal transit time and functional intestinal obstruction. 20 Nevertheless, the complete pathophysiology remains incompletely understood.
Emerging evidence highlighted the crucial role of neuroimmune interactions in gastrointestinal motility regulation. Inflammatory mediators can impair the interaction between interstitial cells of Cajal and smooth muscle cells, alter smooth muscle responsiveness, and disrupt pacemaker activity, contributing to gastrointestinal hypomotility.21,22 In addition, inflammation has been shown to induce enteric neuron loss, thereby reducing gastrointestinal secretion, motility, and colonic contractility.23–25 Moreover, the gastrointestinal immune system plays a key role in modulating systemic immune responses via mechanisms such as intestinal barrier dysfunction, alterations in gut microbiota, microbial by-products, food-derived antigens, and cytokine activity. 26 Antipsychotics have been found to exert anti-commensal effects on gut microbiota, leading to disruptions in the intestinal barrier, which contains immune and neural components. 27 This disruption results in immune dysregulation, impaired gut motility, and increased intestinal permeability. Consequently, the translocation of antigens, microbial byproducts, pathogens, and other proinflammatory substances into circulation may trigger localized or systemic inflammatory responses. 28 Studies have linked constipation to alterations in gut microbiota, increased intestinal permeability, and immune system activation, suggesting that antipsychotic treatment impacts not only the central nervous system but also the enteric nervous system. As outlined above, antipsychotic-induced constipation can be classified as functional constipation, a chronic condition characterized by difficult, incomplete, or infrequent bowel movements. 29 Previous research has identified altered inflammatory factor levels in functional constipation, with evidence suggesting low-grade inflammation in affected individuals. 30 Patients with functional constipation exhibit significantly elevated serum levels of proinflammatory cytokines, including IL-1, IL-6, and TNF-α, compared to non-constipated individuals. 31 Furthermore, immune system activation in constipation has been demonstrated through increased spontaneous lymphocyte proliferation and elevated levels of CD3+, CD4+, CD8+ T cells, and CD25+ cells, which express IL-2 receptors. 32 These findings suggest a potential role for peripheral immune factors in constipation pathophysiology. The specific effects of individual immune factors on gastrointestinal motility remain heterogeneous across prior studies, varying with different patient populations and etiological backgrounds. To summarize this evidence, we have compiled the immune factors included in our statistical analysis alongside their reported roles in constipation in gastroenterology from basic and clinical research (Supplement Table 1). Supplement Table 1 summarizes the gastrointestinal effects of all analyzed cytokines. These listed results highlight the urgent need to investigate the unique immune signatures or factors of AIC, thereby addressing the research gap in AIC-related immune mechanisms.
In addition, our previous study found that the FcγR-mediated phagocytosis pathway is significantly altered in patients with AIC, suggesting that the innate immune response may be involved. Meanwhile, sphingolipid metabolism is disrupted, and the decrease in its key signaling molecule S1P may impair immune cell function and intestinal barrier integrity. 33 Further gut microbiota analysis showed a significant increase in the abundance of the Synergistetes phylum, which is potentially associated with humoral immunity, in constipated patients. Functional prediction also revealed changes in the gut microbiota’s lipopolysaccharide (LPS) metabolic pathway. As an endotoxin, LPS can directly inhibit intestinal peristalsis via the enteric nervous system. 34 Taken together, these results indicate that antipsychotics may synergistically induce constipation by disrupting host immune-related pathways and microbiota-driven immune activation.
Despite these advances, critical knowledge gaps persist regarding the specific inflammatory and immune mechanisms underlying AIC. Current classification systems recognize slow transit constipation as the probable category for AIC, 35 but the precise immunopathological processes remain unclear. The interplay between antipsychotic pharmacology, gut barrier function, and systemic immune activation represents a promising yet under-explored research direction.
This study aimed to bridge these gaps by investigating the prevalence and risk factors of AIC in patients with schizophrenia and examining associated alterations in serum immune factors. We hypothesized that AIC involved distinct inflammatory signatures and immune activation patterns, which might reflect underlying pathophysiological processes. Our study combined comprehensive clinical assessment with the simultaneous analysis of multiple immune parameters to elucidate the potential mechanistic links between the development of AIC and immune dysfunction. The findings are expected to provide novel insights into AIC etiology and pathophysiology, offering guidance for improved prevention and treatment strategies in clinical practice.
Methods
Study design and setting
This was a detailed, two-phase, cross-sectional observational study that recruited participants, but with no repeated measures or follow-up conducted. It was conducted at the Department of Psychiatry, Second People’s Hospital of Jiangning District, Nanjing, between January 2020 and June 2021. The study was structured into two sequential phases to ensure a stepwise investigation: an initial broad screening phase (phase I) followed by a detailed immunological profiling phase (phase II). The reporting of this study conforms to the STROBE statement. 36
Sample size
This was a cross-sectional study with two primary aims. The first one was to estimate the prevalence of AIC, and the second one was to compare immune marker levels between constipated and non-constipated groups. A post-hoc sample size justification is provided based on these aims.
For prevalence estimation in phase I: The sample size for estimating the prevalence of AIC was calculated using the standard formula for a single-population proportion in cross-sectional studies:
Based on prior clinical reports, the anticipated prevalence (P) was set at 30%.12–14 With a 95% confidence level
For exploratory group comparison for immune factors in phase II: The sample size needed for the exploratory comparison of serum immune factors between groups was estimated for a two-independent samples t-test. Due to the lack of preliminary data for our specific population and outcome, a medium effect size (Cohen's d = 0.5) was assumed for calculation purposes. Using the standard formula for this test:
and setting a two-sided alpha (α) of 0.05
Our study screened 154 participants in phase I, which exceeded the minimum sample size of 143 required for a precise prevalence estimate (result: 36.4%, 95% CI: 29.0%–44.4%). In phase II, we enrolled 44 participants in each group for exploratory immune profiling. While this sample size provided valuable preliminary data for identifying potential immune biomarkers (such as IL-10), it is acknowledged that it is underpowered to definitively confirm associations, especially after correction for multiple comparisons, for a broad panel of immune factors. The immune findings should therefore be interpreted as hypothesis-generating.
Participants and recruitment: a two-phase approach
A total of 154 patients with schizophrenia were initially screened in phase I. All participants were long-term inpatients. Inclusion criteria for phase I were: (1) a diagnosis of schizophrenia meeting the criteria of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5); (2) at least 6 months of antipsychotic treatment, with a stable dose for the past 3 months; and (3) an age range of 18–65 years. Exclusion criteria included: (1) comorbid mental disorders meeting DSM-5 diagnostic criteria; (2) a history of neurological diseases, brain trauma, or cerebrovascular disorders; (3) presence of cardiovascular, hepatobiliary, gastrointestinal, hematological, or autoimmune diseases, or cancer; and (4) menstruating, pregnant, or lactating women.
Patients were classified into either the constipation group or the non-constipation group based on the Rome IV criteria applied in phase I. From this initial cohort, 88 patients proceeded to phase II based on predefined eligibility criteria. The criteria for entering phase II were more stringent. For the constipation group, eligibility required: (1) fulfilling all phase-I criteria; (2) meeting the Rome IV diagnostic criteria for functional constipation; (3) having a positive Radiopaque Marker (ROM) test; (4) and having a body mass index (BMI) below 28 kg/m2. For the non-constipation group, eligibility required: (1) fulfilling all phase-I criteria; (2) no history of constipation according to the Rome IV diagnostic criteria; (3) having a negative Radiopaque Marker (ROM) test; (4) and having a BMI below 28 kg/m2. Additional exclusion criteria were applied: (1) use of laxatives within one week prior to the study; (2) history of constipation before antipsychotic treatment; (3) abdominal surgery within 3 months preceding the study; (4) antibiotic use within 3 months before study commencement; (5) significant dietary or lifestyle changes within 1 month before the study; and (6) presence of severe gastrointestinal complications, fever, vomiting, or active infections during the study period.
Participant flow and selection for phase II
The flow of participants is detailed as follows: phase I enrolled 154 patients, identifying 56 with and 98 without constipation according to Rome IV criteria. For phase-II immune exploratory, strict eligibility criteria were applied. From the constipation group (n = 56), 12 were excluded (five refused the ROM test, two used laxatives, and five had ROM-negative), leaving 44 eligible patients. From the non-constipation group (n = 98), 34 were excluded (8 refused the ROM test, 20 ROM-positive, 4 BMI > 28, and 2 recent fever), leaving 64 eligible controls.
To establish a matched case-control cohort for the exploratory analysis in phase II, we selected 44 control patients from 64 eligible individuals. To ensure baseline comparability, a purposive sampling approach was employed, matching the control group to the constipation group on the key covariates of age and sex. To assess potential bias, we compared the 44 selected controls with the remaining 20 eligible but non-selected individuals. There was no significant difference in demographic or clinical characteristics between these two subgroups (all p > 0.05, Supplemental Table 2). Thus, the final phase-II cohort comprised 44 constipation patients and 44 non-constipation patients.
Variables, measurements, and assessment tools
Primary outcome: constipation diagnosis
Patients were interviewed by registered physicians with more than 5 years of clinical experience to complete the Rome IV Diagnostic Questionnaire. Constipation was determined by physicians using the Rome IV criteria according to Items 49–59 of the Questionnaires. 37 A diagnosis of constipation required at least two of the following conditions: Straining during ⩾25% of bowel movements; Hard, lumpy, or pellet-like stools in ⩾25% of bowel movements; Sensation of incomplete evacuation in ⩾25% of bowel movements; Sensation of anorectal obstruction or blockage during ⩾25% of bowel movements; The need for manual maneuvers (e.g., digital evacuation or use of pelvic floor support) in ⩾25% of bowel movements; Fewer than three spontaneous bowel movements per week; No loose stools unless induced by laxatives; Symptoms not meeting the criteria for irritable bowel syndrome (IBS); Total duration of symptoms ⩾6 months, with symptoms meeting the criteria persistently present within the past 3 months. 37
Secondary outcomes and assessment tools
Constipation severity: The severity of constipation was evaluated using the CAS and BSS. The CAS consists of eight assessment parameters, each scored as 0, 1, or 2. The overall severity of constipation is determined by summing the parameter scores. Because of its efficiency, the CAS is widely employed in clinical settings for constipation assessment. The BSS categorizes stool consistency into seven types, serving as an indicator of constipation severity. Lower scores correspond to hard, dry stools, whereas higher scores indicate softer stools. Types 1 and 2 are associated with irregular, hardened stools, indicative of constipation. 38 In addition, the BSS can be used to estimate colonic transit time, with lower scores suggesting slower intestinal transit and higher scores indicating faster transit times.
Objective colonic transit time: The radiopaque marker (ROM) method was used. The ROM method is recognized as the gold standard for assessing colonic transit time in both clinical and research settings.30,39 It demonstrates high intra-observer reliability, inter-observer reproducibility, and a strong correlation with stool frequency. 40 To ensure accurate assessment, participants were required to discontinue any medications affecting gastrointestinal motility (e.g., prokinetics, laxatives, and antidiarrheal agents) for at least seven days prior to testing. CTT was measured as follows: On the first day (t = 0 h) at 9:00 AM, participants ingested a SITZMARKS™ capsule containing 24 radiopaque O-ring markers after breakfast. Seventy-two hours later (t = 72 h), an abdominal X-ray was taken in the supine position to determine marker retention and elimination. Two trained researchers independently analyzed the X-ray images to count the number of remaining markers and identify those excreted within 72 h. If more than 80% of the markers were expelled, the result was considered negative, indicating normal CTT. Conversely, if over 20% of the markers remained in the colon, the result was deemed positive, confirming a constipation diagnosis.
Psychiatric symptom severity: Clinical symptoms were assessed using the Positive and Negative Syndrome Scale (PANSS) and the Clinical Global Impression (CGI) scale. The PANSS and CGI scales were used to evaluate schizophrenia symptoms. PANSS is a widely used tool for assessing schizophrenia symptoms. 41 The Chinese version of PANSS demonstrates strong reliability and validity. It consists of 30 items divided into three subscales: positive symptoms, negative symptoms, and general psychopathology. Each item is rated on a seven-point scale, with 1 indicating “asymptomatic” and 7 representing “extremely severe.” Scores increase incrementally from 1 to 7 based on symptom severity. The CGI scale was used to assess overall illness severity. This scale is applicable in both clinical and research settings, employing an eight-point rating system ranging from 0 (no illness) to 7 (extremely severe illness). Due to its strong internal consistency, the CGI scale is recognized for its high reliability and validity.
Peripheral immune profile: In phase II, additional fasting blood samples were drawn at 6:00 AM for peripheral immune factor analysis. A panel of 18 immune factors, including granulocyte-macrophage colony-stimulating factor (GM-CSF), interferon-gamma (IFN-γ), tumor necrosis factor-alpha (TNF-α), and interleukins (IL)-1β, IL-2, IL-4, IL-5, IL-6, IL-9, IL-10, IL-12p70, IL-13, IL-17A, IL-18, IL-21, IL-22, IL-23, and IL-27, was quantified from fasting plasma samples using the ProcartaPlex Multiplex Immunoassay on the Luminex 200 platform.42,43 The ProcartaPlex assay, based on the sandwich ELISA principle, was utilized. This method employs two highly specific antibodies that bind to different epitopes of the target protein, allowing simultaneous quantification of multiple proteins using Luminex technology. The ProcartaPlex test uses matched antibody pairs to identify proteins of interest, with differently labeled protein-specific antibodies being bound to different beads, allowing for analyses of multiple targets in one well. Immune factors with more than 30% of sample concentrations below the lower limit of quantification (LLOQ) were excluded from the final analysis to ensure data reliability and avoid the bias introduced by imputing non-quantifiable values. Specifically, the following cytokines were excluded due to a high proportion (>30%) of samples with concentrations below the assay’s LLOQ: GM-CSF (LLOQ: 18.4 pg/mL, 98.86% below), IL-2 (LLOQ: 9.1 pg/mL, 98.86% below), IL-6 (LLOQ: 12.4 pg/mL, 98.86% below), IL-17A (LLOQ: 2.8 pg/mL, 98.86% below), IL-23 (LLOQ: 16.4 pg/mL, 100% below), and IL-27 (LLOQ: 23.2 pg/mL, 100% below). The ProcartaPlex™ MAP Plex kit (catalog number: EPX180-12165-901) was used to measure specific immune markers. Laizee Biotech Co., Ltd., Shanghai, China, provided technical support for protein chip component analysis.
Covariates and potential confounders
We systematically collected extensive demographic and clinical data to control for potential confounding effects. This included age, sex, height, weight, waist circumference, BMI, equivalent dose of clozapine (EDC), smoking status, duration of illness, duration of antipsychotic use, and length of hospitalization. Fasting blood samples were collected at 6:00 AM for metabolic parameter analysis, including C-reactive protein (CRP), glucose (Glu), total cholesterol (TC), triglycerides (TG), high-density lipoprotein (HDL), and low-density lipoprotein (LDL). Antipsychotic doses were standardized to the EDC using the defined daily dose (DDD) method recommended by the World Health Organization Collaborating Centre for Drug Statistics Methodology. 44
Data collection procedures and quality assurance
Fasting venous blood samples were collected from all participants at 6:00 AM. All clinical assessments, including the PANSS and CGI, were conducted by at least two experienced psychiatrists or deputy chief physicians. Prior to the study, raters were trained and standardized, achieving an intra-class correlation coefficient (ICC) for internal consistency reliability exceeding 0.85.45,46 The analysis of ROM X-rays was performed independently by two trained researchers who were blinded to the participants’ group assignment to prevent assessment bias. In the laboratory, all immune factor analyses incorporated both high- and low-concentration quality control samples, and each patient sample was analyzed in duplicate to ensure the reliability and reproducibility of the results.
Statistical analysis
Demographic and clinical data were analyzed using SPSS Version 23 (Chicago, IL, USA) and Matlab Version R2013b (Natick, MA, USA). Categorical variables were compared using the chi-square test or Fisher’s exact test. For continuous variables, normally distributed data were analyzed using Student’s t-test (values reported as mean ± SD), whereas the Mann–Whitney U test was applied to non-normally distributed data (values reported as median with interquartile range). Multiple testing for immune factors was corrected using the Benjamini-Hochberg method for false discovery rate (FDR), with a q-value of 0.05. The effect sizes for the significant continuous variables were estimated using Cohen’s d. The relationships between variables were assessed using Pearson’s correlation coefficient and logistic regression analysis. A p-value ⩽ 0.05 was considered statistically significant. Immune factors were considered significantly different if they met a p-value < 0.05.
Results
Demographics and clinical characteristics
From January 2020 to June 2021, a total of 154 hospitalized patients with schizophrenia were screened based on the Rome IV criteria for constipation in phase I. Among these, 56 patients were diagnosed with constipation, representing a prevalence of 36.36%.
Compared to the non-constipation group, patients with constipation exhibited significantly higher CAS scores (t = 4.008, p = 0.000), longer disease duration (t = 2.096, p = 0.038), and a greater number of hospitalizations (t = 2.107, p = 0.037). Conversely, they had significantly lower weight (t = 2.500, p = 0.013), BMI (t = −2.512, p = 0.013), and BSS scores (t = −4.300, p = 0.000). No statistically significant differences were observed between the constipation and non-constipation groups regarding height, age, sex, equivalent clozapine dose, PANSS total score, positive symptom factor score, negative symptom factor score, or general psychopathology score (all p > 0.05; see Table 1).
Comparison of baseline demographic and clinical characteristics between constipated and non-constipated patients in phase I.
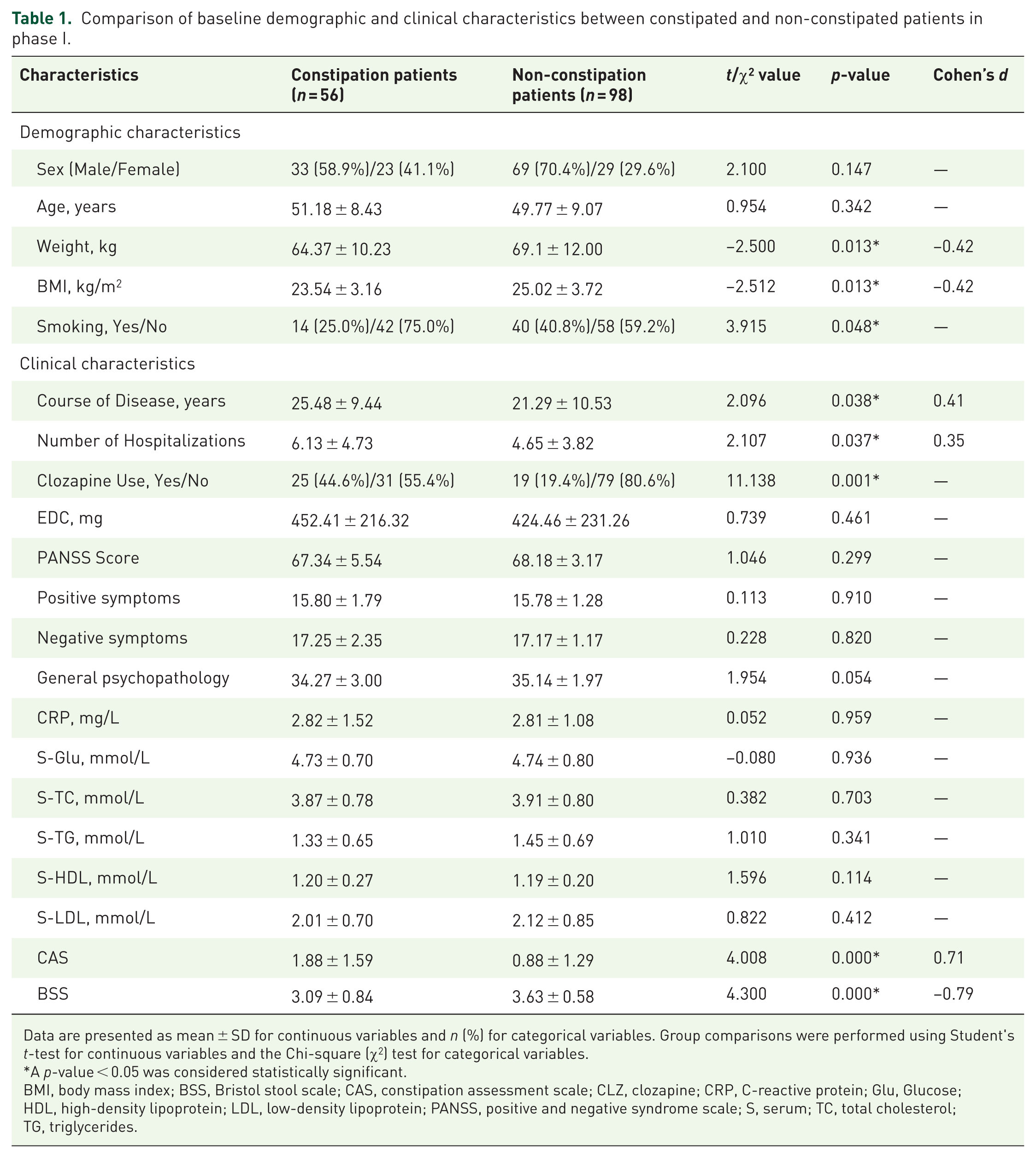
Data are presented as mean ± SD for continuous variables and n (%) for categorical variables. Group comparisons were performed using Student's t-test for continuous variables and the Chi-square (χ2) test for categorical variables.
A p-value < 0.05 was considered statistically significant.
BMI, body mass index; BSS, Bristol stool scale; CAS, constipation assessment scale; CLZ, clozapine; CRP, C-reactive protein; Glu, Glucose; HDL, high-density lipoprotein; LDL, low-density lipoprotein; PANSS, positive and negative syndrome scale; S, serum; TC, total cholesterol; TG, triglycerides.
Among the study participants, constipation was observed in 56.82% (25/44) of clozapine-treated patients, compared to 28.18% (31/110) of non-clozapine-treated patients. This indicated a significantly higher prevalence of constipation in the clozapine-treated group than in non-clozapine users (χ² = 11.138, p < 0.01). In addition, the proportion of smokers was lower among constipation patients than among non-constipation patients (χ² = 3.915, p = 0.048).
Immune factors and constipation
Test results for immune factors
Following the eligibility assessments and matching procedure described in detail in the Methods, a final cohort of 44 patients with constipation and 44 matched patients without constipation were included in the phase-II analysis. The average ages of participants in the constipation and non-constipation groups were 51.02 ± 9.11 and 50.68 ± 9.11 years, respectively (p = 0.861).
Based on previous studies, six immune factors (GM-CSF, IL-2, IL-6, IL-17A, IL-23, and IL-27) were excluded due to missing data exceeding 30% beyond the detection range, leaving 12 inflammatory factors for analysis.47,48 Comparisons of immune factor levels between the constipation and non-constipation groups revealed a nominal and uncorrected increase in interleukin-10 (IL-10) (p = 0.043, Cohen’s d = 0.32) in the constipation group. After FDR correction for multiple comparisons, the IL-10 association did not remain statistically significant (FDR-adjusted p = 0.432). No significant differences were observed for the remaining factors (see Table 2).
Inflammatory factor levels in constipated and non-constipated patients from phase II.

Mann–Whitney U tests were applied to analyze non-normally distributed data (i.e., IL-21 and IL-22), which are presented as the median (interquartile range). Continuous normally distributed variables were compared between groups with Student’s t-tests and are presented as the mean ± SD. The concentration unit of immune factors is picograms per milliliter (pg/mL).
A p-value < 0.05 was considered statistically significant.
The FDR-adjusted p-value = 0.432.
IFN, interferon; IL, Interleukin; TNF, tumor necrosis factor.
Correlation and logistic regression analyses for peripheral immune factors
Pearson correlation analysis indicated a significant positive correlation between IL-10 levels and CAS scores among the 88 patients. In addition, a significant negative correlation was found between equivalent clozapine dosage and IL-1β levels (r = −0.348, p < 0.001, Figure 1). Exploratory multivariate logistic regression analysis indicated a nominal association between IL-10 levels and constipation because of long-term antipsychotic use (OR = 1.606, 95% CI: 1.066–6.323, p = 0.036), prior to multiple comparison correction. In light of the FDR result, these findings should be considered preliminary and hypothesis-generating.

Correlation between peripheral immune factors and clinical indicators in 88 patients. (a) Correlation analysis between peripheral blood IL-10 concentration and constipation severity (CAS). The horizontal axis represents peripheral blood IL-10 concentration, while the vertical axis denotes constipation severity (CAS score). (b) Correlation analysis between peripheral blood IL-1β and EDC. The horizontal axis represents peripheral blood IL-1β concentration, while the vertical axis denotes EDC. Constipated subjects are indicated by green circles, whereas non-constipated subjects are represented by blue circles.
Diagnostic model based on inflammatory factors
A logistic regression model was employed to assess the ability of IL-10 levels to differentiate between constipation and non-constipation patients. The resulting area under the Receiver Operating Characteristic (ROC) curve was 0.602 (95% CI: 0.482–0.722), indicating a limited discriminative ability for a standalone diagnostic biomarker at this stage (Figure 2).

ROC curves for IL-10 and CAS in diagnosing antipsychotic-induced constipation.
Discussion
To the best of our knowledge, this is the first study to investigate alterations in peripheral immune factors in patients with AIC. Constipation is a common adverse effect of antipsychotic medications in schizophrenia treatment, yet its underlying mechanisms remain incompletely understood. Currently, there is a lack of clinically valuable biomarkers for diagnosis, and management is often symptomatic. Existing literature suggests a connection between the immune system and gastrointestinal function. Immune system activation and mucosal inflammation may influence gastrointestinal motility by affecting smooth muscle activity and pacemaker cell function. This study posits that an imbalance between proinflammatory and anti-inflammatory cytokines may contribute to constipation in schizophrenia patients receiving antipsychotics.
The prevalence of AIC among patients enrolled in this study was 36.36%. Notably, patients treated with clozapine exhibited a significantly higher prevalence of constipation (56.82%) compared to those receiving other antipsychotics (28.18%), which is consistent with previous reports of clozapine-associated constipation rates ranging from 14% to 60%. 49 A recent systematic review further confirmed that clozapine users experience higher constipation rates than those taking other antipsychotics (OR = 3.02, 95% CI: 1.91–4.77), 50 aligning with our findings. Clozapine serves as the first-line antipsychotic for treatment-resistant and refractory schizophrenia.51,52 Despite its robust efficacy in alleviating positive psychotic symptoms, it is also associated with non-negligible adverse effects. Constipation is among the top 4 most commonly reported adverse effects, 50 and clozapine presents a high risk of inducing constipation. The mechanisms underlying clozapine-induced constipation might include not only acetylcholine receptor antagonism, but also its antagonistic effects on 5-hydroxytryptaminergic, histaminergic, and opioid receptors.53–55 These effects can inhibit smooth muscle contraction, delay intestinal transit, and result in functional intestinal obstruction. In addition, clozapine-induced weight gain or obesity may also be associated with constipation. Therefore, timely assessment of bowel movement status is essential for clinicians in routine practice to avoid life-threatening risks caused by severe constipation.
The BSS and CAS scales were employed in this study to assess constipation severity. The BSS scale categorizes stool consistency based on patient-reported observations. Our findings revealed that constipated patients exhibited lower BSS scores and higher CAS scores compared to non-constipated individuals, effectively validating the clinical grouping. 16 It is noteworthy that schizophrenia patients may experience cognitive deficits and altered pain perception,56,57 potentially leading to biased self-reporting. Therefore, the use of such structured scales is particularly important in this population for obtaining more objective assessments.
In phase I, there was a lower smoking rate observed in the constipation group. While there was no statistically significant difference in smoking status between the two groups (Pearson’s chi-square = 1.725, p = 0.193) in the phase-II intergroup analysis. The potential influence of smoking on gastrointestinal motility warrants consideration. Research indicates that smoking may influence digestive function and small intestinal transit rate, but may not directly alter colonic transit rate.58–60 However, tobacco may induce small intestinal bacterial overgrowth by prolonging small intestinal transit time, which in turn causes diarrhea. 61 Nicotine may also trigger diarrhea by reducing pancreatic secretion. 62 A population-based study of 2560 community individuals in Sweden reported a dose-response relationship between cigarette smoking category and the odds of irritable bowel syndrome with diarrhea (IBS-D). 63 Therefore, we hypothesize that the lower smoking rate initially found in the constipation group in phase-I might be due to tobacco or nicotine partially counteracting the effects of constipation.
A key finding of this study was the nominally elevated serum level of IL-10 in the constipated group. IL-10 is a crucial anti-inflammatory cytokine essential for maintaining immune balance. 64 The broader context of immune modulation by antipsychotics must be considered when interpreting this result. Evidence suggests that IL-10 is implicated in the pathophysiology of schizophrenia, with increased IL-10 expression alleviating abnormal aggressive behaviors in schizophrenia model mice. 65 Genome-wide association studies have identified differences in the distribution frequencies of IL-10 single-nucleotide polymorphism (SNP, rs1800872) and two haplotypes (A-C-An and G-C-C) between individuals with schizophrenia and healthy controls. 66 In addition, elevated serum IL-10 levels have been reported in schizophrenia patients compared to healthy individuals, 67 with peripheral IL-10 levels positively correlated with the severity of negative symptoms. 68
We hypothesize that antipsychotic treatment may initiate a localized, low-grade proinflammatory state in the gut, potentially through gut barrier disruption or microbiota alterations. In response to this primary insult, the body may upregulate IL-10 as a counter-regulatory mechanism to mitigate inflammation and limit tissue damage.69,70 This paradigm, where anti-inflammatory cytokine release follows initial inflammation, is well-documented in other gastrointestinal conditions, such as irritable bowel syndrome (IBS), where elevated IL-10 has also been observed and is considered a potential therapeutic target for modulating low-grade inflammation.71,72
This compensatory hypothesis is supported by the two findings from our study. First, we observed a positive correlation between IL-10 levels and constipation severity (CAS scores). This suggests that the IL-10 response scales with the clinical burden. In addition, exploratory regression analysis suggested a nominal association for IL-10 (OR = 1.606), highlighting its association with the condition. Therefore, IL-10 may display a nominal association with AIC, although its current standalone predictive ability, as indicated by the ROC curve (AUC = 0.602), which indicates poor to modest discriminative ability, far from the level required for a standalone clinical diagnostic or predictive biomarker. While our results suggest clinical validity through association with constipation severity, future studies must establish analytical validity (assay standardization) and clinical utility (cost-effectiveness in preventing complications) before clinical implementation. 73 While the compensatory response is our leading interpretation, we cannot rule out the possibility that the elevated IL-10 itself may directly contribute to reduced gut motility through its potent immunosuppressive effects. Future mechanistic studies are needed to distinguish between these possibilities. The clinical monitoring of such immune markers is not yet routine in patients with AIC, but our findings highlight a potential future direction where establishing clinically meaningful thresholds could aid in risk stratification. This approach aligns with the broader principles of precision medicine in neuropsychiatry, which advocate for integrating diverse biological data, such as immune markers, with clinical and environmental factors to create individualized treatment and monitoring plans. 74
Our study’s identification of IL-10 as a candidate biomarker aligns with initial steps of established principles for reliable biomarker development. 73 However, given the cross-sectional design, the loss of statistical significance after FDR correction, and the modest AUC, any claim for its utility as a predictive biomarker is premature. While our findings suggest clinical validity through association with constipation severity, future studies should address analytical validity (e.g., assay standardization across platforms) and clinical utility (e.g., cost-effectiveness of routine IL-10 monitoring in preventing complications). The identification of IL-10 as a potential biomarker for antipsychotic-induced constipation contributes to the growing field of routine blood biomarkers in clinical monitoring. 75 Unlike trauma biomarkers used for acute triage, IL-10’s role here would be for chronic risk stratification. Its integration into clinical practice would require validation against established constipation assessment tools (like CAS or BSS) and demonstration of added value beyond current monitoring strategies. The phase-II study selected the non-constipated control group using purposive sampling, which ensured age and gender matching with the constipation group and guaranteed intergroup comparability. In addition, comparability existed between the non-constipated individuals selected for inclusion in phase II and those not selected. This finding alleviates concerns about major selection bias (Supplemental Table 2). Given that the phase-II sample size provides limited statistical power for the multi-immunological factor analysis, the immunological findings of this study should be considered preliminary and exploratory, primarily aimed at generating hypotheses for future research. In conclusion, although the elevated IL-10 provides a notable view for understanding the immune mechanism of AIC, the loss of significance after correction for multiple comparisons and limited discriminatory power requires a highly cautious interpretation of this finding.
Furthermore, our study identified a significant negative correlation between the EDC and IL-1β levels in schizophrenia patients. IL-1β is a proinflammatory cytokine whose levels have been reported to fluctuate throughout the course of schizophrenia and antipsychotic treatment.76–78 Moreover, current research on the effects of antipsychotics on cytokines has yielded inconsistent results.79–83 For instance, some studies indicate that risperidone reduces plasma levels of IL-1β, IL-2, IL-4, IL-6, and IL-10 while increasing IL-12 levels.79,80 Olanzapine has been shown to decrease peripheral IL-2, TNF-α, and IFN-γ levels, whereas clozapine increases sIL-2R and TNF receptor levels, and haloperidol decreases IL-2 levels. 80 Conversely, other studies suggest that atypical antipsychotics, including clozapine, olanzapine, and risperidone, upregulate blood IL-10 levels. 84 The discrepancy between our finding and some previous research, that clozapine treatment leads to elevated IL-1β levels,85,86 might be explained by differences in study population, treatment duration, or the complex, dynamic nature of immune adaptation to long-term medication. The inverse correlation between the EDC and IL-1β levels observed in this study may reflect a broader, dose-dependent immune-modulatory effect of antipsychotics. This finding is consistent with prior reports indicating that antipsychotic treatment can suppress specific inflammatory markers. For example, atypical antipsychotics have been shown to reduce the anti-inflammatory capacity of serum and lower the levels of proteins such as Clara Cell protein (CC16). 87 Similarly, repeated administration of antipsychotics has been reported to suppress plasma concentrations of pro-inflammatory cytokines like IL-12 in patients. 88 Therefore, our observation could align with a pharmacological adaptation in which higher drug exposure modulates the inflammatory response. However, given the exploratory and cross-sectional nature of the study, this interpretation remains preliminary and requires validation in prospective research designed to investigate the causal dose-response relationship between antipsychotics and immune markers. This highlights the need for further research to clarify the precise interactions between specific antipsychotics and immune markers, such as IL-1β. Against this backdrop of complex and drug-specific effects, our finding of elevated IL-10 in AIC patients gains significance.
Beyond physical complications, AIC significantly impacts patient well-being and treatment adherence. As demonstrated in conditions such as eating disorders, gastrointestinal symptoms profoundly affect quality of life. 89 Our findings suggest that immune-mediated constipation may represent an underrecognized contributor to reduced QoL in schizophrenia. Future interventions targeting IL-10 pathways should evaluate not only physiological outcomes but also patient-reported QoL metrics.
Limitations
This study has several limitations. First, the cross-sectional design limits causal inferences. Therefore, the observed association between elevated IL-10 levels and AIC could not determine whether IL-10 is a cause, a consequence, or a concurrent biological phenomenon of constipation. Future longitudinal studies may provide more definitive insights. Second, the relatively small sample size for the immune factor analysis in phase II was not based on a pre-specified power calculation for the immune factor analysis because of the relatively limited number of participants. This may have limited the ability to detect small, while potentially important, associations for some cytokines and increases the risk of type II errors. Future research with larger sample sizes is necessary to validate these results. Moreover, advanced computational methods, such as artificial intelligence, represent a promising frontier for developing more robust predictive models. AI algorithms could potentially integrate complex datasets—including immune profiles, genetic information, medication types, and clinical variables—to more accurately identify patients at high risk for AIC than single-marker approaches. 90 Third, although we controlled several key clinical and demographic variables, the potential influence of unmeasured confounding factors, such as detailed dietary habits or comorbidities, such as diabetes, cannot be fully ruled out. Specifically, the presence of diabetes, a highly prevalent comorbidity in schizophrenia and a condition exacerbated by metabolic side effects of antipsychotics, was not systematically assessed or controlled for. Diabetic enteropathy or enteric neuropathy that affects colonic transit has been associated with both diarrhea and constipation,91–93 which might independently contribute to or modify the presentation of AIC. Moreover, the potential confounding effect of the observed smoking difference in phase I, despite its absence in phase II, should be acknowledged. For example, smoking had a potential confounding effect in the phase I study. Although no significant between-group difference was observed in phase II, this potential effect should still be acknowledged. Smoking affects gastrointestinal motility and immune parameters. Future studies should explicitly account for and evaluate its potential confounding role. Fourth, the interpretation of our immune marker analysis is limited by multiple comparisons. After applying a FDR correction, the association for IL-10 was attenuated to non-significance (q = 0.432), suggesting that future studies with larger sample sizes and a specified focus on a narrower set of immune markers (e.g., IL-10 and related pathways) are necessary to conclusively determine its role in AIC. Fifth, even though we intentionally used “equivalent clozapine dose” to standardize the use of all antipsychotics for the patients, standardized dosages cannot fully reflect the drugs’ potential to induce constipation. In future studies, it will be necessary to further control the types of antipsychotics administered to participants and improve the detection of drug plasma levels. In addition, the exclusion of six cytokines from the analysis due to the lower limit of quantification of the assay’s standard curve narrows the spectrum of immune factors evaluated, which may limit the comprehensiveness of the immune profile. Finally, all participants in this study were chronic schizophrenia patients with long-term hospitalization. The characteristics of this specific population may limit applicability to outpatients or those with shorter illness duration or treatment.
Conclusion
This was the first study to investigate the clinical characteristics and peripheral immune factors in schizophrenia patients with AIC. This study confirms the high prevalence of constipation among patients receiving long-term antipsychotic treatment, with clozapine representing a major risk factor. Our findings also identified a preliminary association between AIC and elevated peripheral IL-10, suggesting a possible involvement of immune regulation in its pathophysiology. However, this finding is preliminary and hypothesis-generating. Before any clinical application can be considered, further, larger longitudinal studies are required to validate IL-10, explore potential causality, assess a broader panel of immune markers, and control for important confounders such as diet and diabetes. Further research is needed to elucidate the mechanisms of immune modulation and its potential to reduce adverse drug reactions, ultimately improving the quality of life and treatment adherence in schizophrenia patients. This goal is at the heart of integrative care, which recognizes that managing physical side effects is as crucial as psychosocial interventions for ensuring patient well-being and long-term engagement with treatment. 94
Supplemental Material
sj-docx-1-tpp-10.1177_20451253261432764 – Supplemental material for Clinical hallmarks and peripheral immune factors associated with antipsychotic-induced constipation in schizophrenia
Supplemental material, sj-docx-1-tpp-10.1177_20451253261432764 for Clinical hallmarks and peripheral immune factors associated with antipsychotic-induced constipation in schizophrenia by Miaomiao Shao, Tingting Zhu, Xiuxiu Hu, Xinyu Fang, Xinming Pan, Lu Wen, Peng Dai, Nousayhah Amdanee, Lin Zhang, Yue Xu and Xiangrong Zhang in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253261432764 – Supplemental material for Clinical hallmarks and peripheral immune factors associated with antipsychotic-induced constipation in schizophrenia
Supplemental material, sj-docx-2-tpp-10.1177_20451253261432764 for Clinical hallmarks and peripheral immune factors associated with antipsychotic-induced constipation in schizophrenia by Miaomiao Shao, Tingting Zhu, Xiuxiu Hu, Xinyu Fang, Xinming Pan, Lu Wen, Peng Dai, Nousayhah Amdanee, Lin Zhang, Yue Xu and Xiangrong Zhang in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-3-tpp-10.1177_20451253261432764 – Supplemental material for Clinical hallmarks and peripheral immune factors associated with antipsychotic-induced constipation in schizophrenia
Supplemental material, sj-docx-3-tpp-10.1177_20451253261432764 for Clinical hallmarks and peripheral immune factors associated with antipsychotic-induced constipation in schizophrenia by Miaomiao Shao, Tingting Zhu, Xiuxiu Hu, Xinyu Fang, Xinming Pan, Lu Wen, Peng Dai, Nousayhah Amdanee, Lin Zhang, Yue Xu and Xiangrong Zhang in Therapeutic Advances in Psychopharmacology


