Abstract
Background:
Anhedonia is a key symptom of bipolar depression and a target of ketamine’s rapid antidepressant effects. However, many patients with treatment-resistant bipolar depression (TRBD) do not respond.
Objectives:
This study aimed to identify clinical and sociodemographic characteristics that are associated with nonresponse of anhedonia following short-term ketamine treatment in TRBD.
Design:
A retrospective analysis using data from two naturalistic, observational registries of 31 patients with TRBD and baseline anhedonia (Snaith-Hamilton Pleasure Scale (SHAPS) > 2). Patients received eight doses of ketamine (IV: 0.5 mg/kg; oral: 2.0–2.5 mg/kg) over a short-term treatment course.
Methods:
Patients were classified as responders or nonresponders based on a ⩾50% reduction in SHAPS score by the seventh ketamine dose. Groups were compared on baseline sociodemographic and clinical features.
Results:
Fourteen patients (45.2%) did not respond. Nonresponders had higher BMI, later illness onset, fewer hypomanic episodes, and lower employment rates.
Conclusion:
Metabolic, illness-course, and psychosocial factors may predict reduced anti-anhedonic response to ketamine in TRBD.
Introduction
Anhedonia, a core symptom of depression, manifests as deficits in interest and the ability to experience pleasure, significantly impairing quality of life and daily functioning. 1 Comparative studies indicate that patients with bipolar depression (BD) exhibit even more pronounced anhedonia than those with major depressive disorder (MDD). 2 Data from the Systemic Treatment Enhancement Programme for Bipolar Disorder have highlighted that anhedonia is an acute risk factor for suicide attempts or completed suicides in outpatients with BD. 3 Moreover, the presence of anhedonia in depressed patients also correlates with a longer time to remission. 4
Despite the critical importance of addressing anhedonia, there is currently no registered pharmacological treatment specifically targeting this condition. One promising intervention under investigation is ketamine as an add-on treatment specifically addressing anhedonia across both unipolar and BD. 5 However, the only European Medicines Agency (EMA)-approved ketamine formulation remains esketamine nasal spray, indicated as an adjunct for treatment-resistant depression (TRD) and for the acute management of depressive symptoms with suicidal ideation in MDD. 6 Findings from BD cohorts present encouraging results, showing a low risk of manic switch following ketamine interventions, promising effects in depression with mixed features, and potential mood-stabilizing properties. 7 Notably, studies evaluating anhedonia-specific outcomes report significant and rapid improvements, with reductions observed even after a single infusion. 8 Open-label and retrospective studies involving multiple ketamine infusions have consistently demonstrated a significant reduction in anhedonia.5,9
In light of the limited evidence regarding ketamine use in treatment-resistant bipolar depression (TRBD) with baseline anhedonia symptoms, this study aims to retrospectively analyze a cohort of TRBD patients who underwent short-term ketamine administration, with the aim of identifying sociodemographic and clinical characteristics that distinguish anhedonia nonresponders from responders.
Methods
This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting observational research 10 (Table S1).
Participants
The methodological framework has been previously described in detail. 9 Briefly, this study included patients with TRBD from two compatible datasets registered under ClinicalTrials.gov identifiers NCT04226963 and NCT05565352. Both study protocols adhered to similar observational guidelines for safety, tolerability, and psychometric assessments. Overall, 31 patients with TRBD and baseline anhedonia were enrolled from both datasets. The study protocols were approved by the Independent Bioethics Committee for Scientific Research at the Medical University of Gdańsk, Poland (NKBBN/172–674/2019 and NKBBN/172–447/2022) and adhered to the latest version of the Declaration of Helsinki. Written informed consent was obtained from all participants prior to study enrollment.
Study design
The registered studies aimed to assess the tolerability and safety profile of eight intravenous (IV) ketamine infusions or oral ketamine solutions administered as an adjunctive add-on to standard of care treatment. The intravenous ketamine was delivered at a dose of 0.5 mg/kg, with protocol details available elsewhere. 9 The oral ketamine regimen spanned 4 weeks, with eight administrations. Oral doses of ketamine solution were weight-based at 2.0 or 2.5 mg/kg, ensuring individualized treatment.
Outcome measures
Participants were diagnosed with BD according to Diagnostic and Statistical Manual of Mental Disorders (DSM)-5 criteria, based on a comprehensive clinical interview, with TRBD defined as a clinically unsatisfactory response following at least two presumably adequate treatments in terms of dose and duration within the current depressive episode. 11 History of trauma was defined based on the clinical interview. Anhedonia was defined as a baseline score > 2 on the Snaith-Hamilton Pleasure Scale (SHAPS). Response to ketamine intervention was defined as a ⩾50% reduction in SHAPS at the seventh ketamine administration compared to the initial score.
Statistical methods
Binary variables were analyzed using Fisher’s exact test. For categorical variables with more than two levels, Fisher’s exact test with the Freeman-Halton extension was applied. The normality of quantitative variables was assessed using the Shapiro-Wilk test. For normally distributed variables, comparisons were made using the t-test; otherwise, the Mann–Whitney U test was applied. A p-value of 0.05 was considered statistically significant. No data imputation was performed.
Results
Sociodemographic characteristics of the whole study population are presented in Table 1. Table 2 provides clinical features of participants. Among the 31 patients with TRBD and baseline anhedonia, 14 (45.2%) failed to respond to ketamine. Those classified as nonresponders manifested a higher body mass index (BMI) (30.7 vs 26.4) and a distinct employment profile—with markedly lower employment (14.3% vs 35.3%), coupled with higher incidences of pension dependency (42.9% vs 11.8%) and retirement (35.7% vs 11.8%; p = 0.0477). Clinically, nonresponders experienced a later onset of BD (36.4 vs 30.5 years, p < 0.001) and a reduced frequency of hypomanic episodes (1 vs 2, p = 0.0103). Figure S1 presents the time course of Montgomery-Asberg Depression Rating Scale (MADRS) and SHAPS score changes during ketamine administrations.
Demographical characteristics of anhedonia responders and nonresponders.
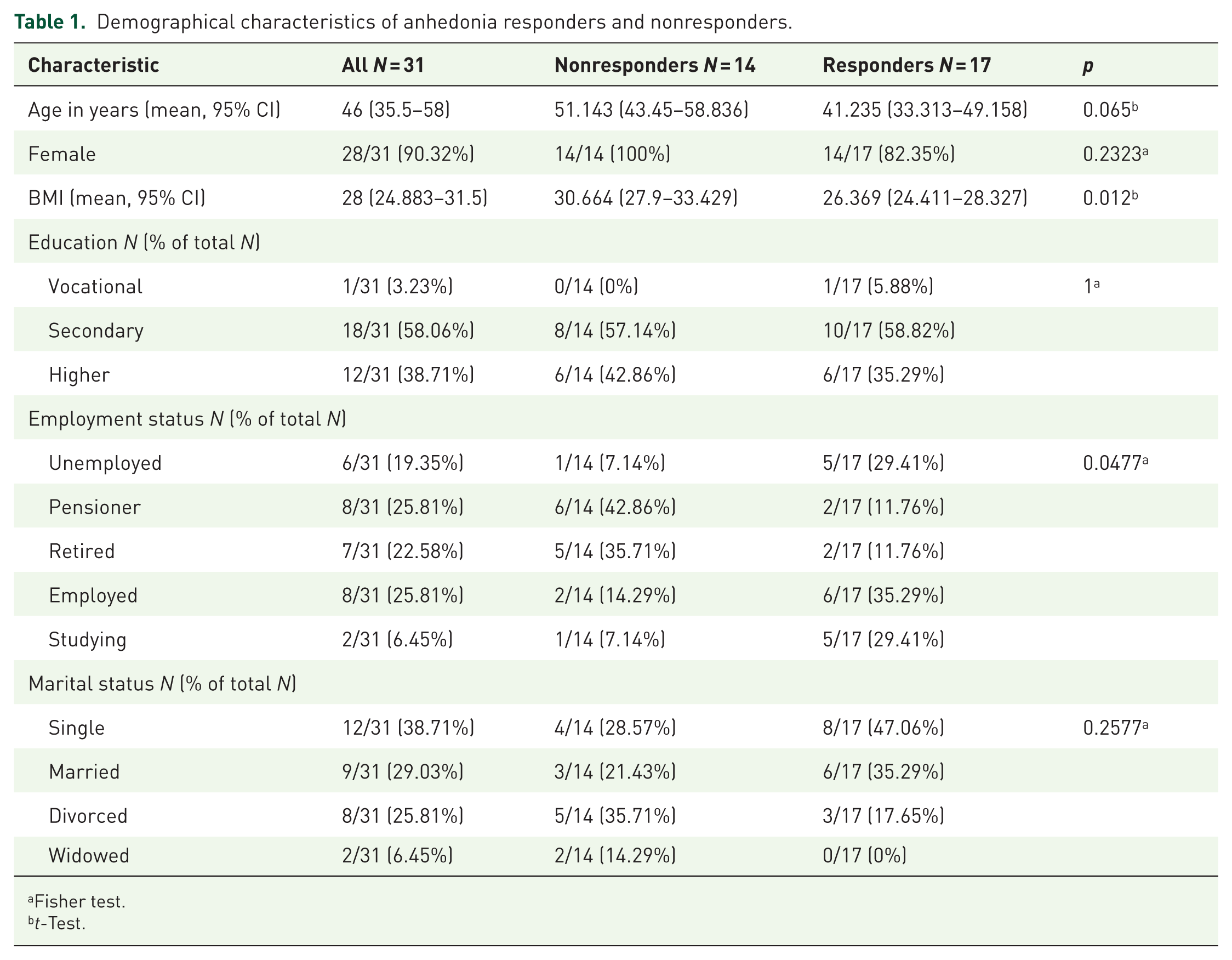
Fisher test.
t-Test.
Clinical characteristics of anhedonia responders and nonresponders.

Fisher test.
t-Test.
Mann–Whitney U test.
Defined as SHAPS > 7.
BP, bipolar disorder; CI, confidence interval; ECT, electroconvulsive therapy; IQR, interquartile range; SHAPS, Snaith-Hamilton Pleasure Scale.
Discussion
This retrospective cross-sectional study outlines the clinical and sociodemographic characteristics of TRBD patients with baseline anhedonia who failed to respond to ketamine. Our findings provide preliminary evidence that anhedonia nonresponse to adjunctive ketamine use is associated with an elevated BMI, lower employment engagement, a later BD onset, and a diminished incidence of hypomanic episodes.
Accumulating evidence suggests that baseline anhedonia predicts higher ketamine response rates in depression regardless of polarity. 12 However, little is known about how patient-specific characteristics influence ketamine’s anti-anhedonic effects when anhedonia is targeted as a primary symptom rather than just one aspect of depression. This distinction seems important, as anhedonia, not overall depression severity, predicts prolonged remission and delayed recovery, thereby altering the long-term course of depression. 4 In staging models of TRD, which conceptualize depression as a progressive disorder, reducing symptom severity, such as anhedonia, is essential for assessing treatment effectiveness. 13 Furthermore, beyond its role in TRD, anhedonia occupies a prominent position in BD; recent network and machine-learning studies have reinforced its centrality as a bridge symptom linking depressive and manic states. 14
Higher BMI (mean 30.7, indicating obesity) was linked to a diminished anhedonia response following ketamine treatment. This observation contributes to an evolving narrative in which prior research on BMI and ketamine response has yielded mixed outcomes—some studies reported no association, while others noted a short-term positive effect—but none have specifically targeted anhedonia as an outcome.12,15 Within bipolar disorder, elevated BMI has been linked to more severe clinical features, including higher rates of suicidality, comorbidities, and core depressive symptoms. 16 Obesity is causally connected to lower glutamine levels and heightened severity of depressive symptoms, including anhedonia. 17 Although ketamine enhances glutamate transmission, its therapeutic effect may be attenuated in obese individuals due to chronic metabolic dysfunction, neuroinflammation, and oxidative stress. 18 Moreover, a straightforward explanation for the reduced response observed in obese patients could be related to dosing: the standard dose of 0.5 mg/kg based on total body weight, as employed in the present study and in most others, may not adequately account for differences in body composition, potentially resulting in suboptimal dosing for patients with higher BMI. 19
Employment is often linked to better treatment outcomes in depression, likely reflecting the stabilizing effects of social engagement and daily structure. Conversely, unemployment is not only more common in individuals with MDD but is particularly prevalent in those experiencing anhedonia. 20 In this study, ketamine nonresponders with baseline anhedonia were more likely to be on a pension or retired and had lower rates of employment and academic enrollment. While fewer nonresponders were unemployed (7.1% vs 29.4%), a broader categorization—including unemployment, pension, and retirement as indicators of social benefit reliance—revealed that nonresponders had significantly higher rates of work inactivity (85.7% vs 53%) compared to responders. This aligns with findings from a recent systematic review and individual patient data meta-analysis, which reported that depressive symptom severity at 3 to 4 months was 47.3% higher in unemployed patients than in those employed, regardless of treatment. 21 Similarly, a post hoc analysis of two esketamine studies found that response and remission rates were significantly higher among employed patients. 22
Late-onset BD is characterized predominantly by depressive episodes. 23 In the present study, later BD onset was found among ketamine nonresponders with baseline anhedonia. This observation extends a recent systematic review that examined age at first MDD episode across unipolar and bipolar depression in relation to ketamine response. In that review, six of eight studies found no significant association, one study associated younger age of onset with ketamine response, and another linked later onset with a better treatment response. 12 Notably, none of these studies categorized patients by their primary diagnosis of unipolar or bipolar depression. One study that did report a negative relationship included 26% TRBD patients and found that those with a later onset (>30 years) were significantly less likely to respond (48.3%) compared to those with an earlier onset (76.5%). 24 This association may stem from both the severity of anhedonia and distinct neurobiological mechanisms complicating anhedonia in this patient population. 25 Although direct evidence is lacking, anhedonia severity appears to be a critical determinant of ketamine nonresponse, particularly in BD, as patients experience more severe anhedonia than those with MDD, as reflected in higher SHAPS scores. 2 Furthermore, later-onset BD with marked anhedonia may share similarities with late-onset depression, which is often characterized by a higher prevalence of cognitive impairment and structural brain alterations—both of which have been associated with delayed and insufficient treatment response. 26
Evidence from the literature indicates that BD II tends to follow a more chronic course—with more prolonged depressive episodes and severe anhedonia—than BD I. 27 However, in this study, the prevalence of BD I versus BD II was similar between responders and nonresponders. What is striking is that nonresponders experienced fewer hypomanic episodes, hinting at a depressive-predominant course marked by persistent anhedonia. While the overall count of depressive, manic, and mixed episodes did not differ significantly, nonresponders seemed to endure a continuous, unremitting depressive state—one defined not by the number of episodes but by the relentless severity of their symptoms. However, in this study, baseline anhedonia severity was almost equally distributed between ketamine responders and nonresponders. This finding may be due to the limitations of the SHAPS, which fail to capture the full complexity of anhedonia. The SHAPS primarily measures consummatory pleasure and does not account for aspects such as reward-based decision-making. 28 Moreover, it is plausible that nonresponders experience a more enduring, trait-like form of anhedonia that remains persistent over time, a nuance that the SHAPS is not well-equipped to detect.
Whether the factors associated with nonresponse identified in this study are specific to anhedonia or reflect broader resistance to antidepressant treatment warrants consideration. In a previous analysis conducted within the same cohort, 29 overall depressive symptom responses, as measured by MADRS, did not differ significantly between responders and nonresponders in terms of BMI, employment status, age of BD onset, or BD type. Statistically significant differences were observed for psychiatric burden, including a higher median number of psychiatric comorbidities, a greater likelihood of having at least one psychiatric comorbidity, and increased prior benzodiazepine use. These findings suggest that while some general indicators of treatment resistance are present, factors for anhedonia nonresponse in TRBD do not overlap with those for overall depression symptom nonresponse.
Limitations and strengths
The presented study has several notable limitations. First, it is a post hoc study based on data from open-label observational trials, which lacked placebo control groups, randomization, or blinded raters. The relatively small sample sizes may raise concerns regarding statistical power. In addition, some clinical variables, such as concomitant medications other than benzodiazepines, were not recorded, while others, such as the number of previous affective episodes, were based partly on retrospective patient reports, which may be subject to recall bias. The follow-up period was limited to 1 week after the last infusion, precluding assessment of ketamine’s long-term effectiveness in alleviating anhedonia. Differences in the route of administration and dosage of ketamine may also have influenced the final results. Since the observed intervention was used as an adjunctive treatment to ongoing oral treatment, variability in baseline pharmacological interventions could have affected the measured outcomes. Given these factors and the exploratory, post hoc nature of this analysis without adjustments for multiple comparisons, which may increase the risk of false-positive findings due to the number of variables examined, the present findings should be considered preliminary and interpreted cautiously as hypothesis-generating, with future studies incorporating larger samples, standardized routes of ketamine administration, and appropriate statistical corrections warranted to validate and expand upon these results.
Despite these limitations, this study has notable strengths. It is the first to delineate a unique population—patients with TRBD and baseline anhedonia who did not respond to ketamine. This specificity allows for a more precise understanding of ketamine’s anti-anhedonic effects within the bipolar spectrum. In addition, by emphasizing real-world evidence, our approach captures the complexities of clinical practice, contrasting with the constrained environments of randomized controlled trials.
Conclusion
The clinical characteristics of nonresponders, notably a later onset of bipolar disorder and a reduced number of hypomanic episodes, appear to delineate nonresponse to short-term ketamine use. Furthermore, the link between nonresponse, an elevated BMI, and lower employment engagement emphasizes the role of demographic and psychosocial factors in shaping anhedonia treatment outcomes among these patients. Identifying such patients early might mitigate their exposure to ineffective treatments and accelerate the shift to alternative therapeutic strategies.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253251412629 – Supplemental material for Anhedonia nonresponse to short-term ketamine administration for treatment-resistant bipolar depression
Supplemental material, sj-docx-1-tpp-10.1177_20451253251412629 for Anhedonia nonresponse to short-term ketamine administration for treatment-resistant bipolar depression by Zofia Kachlik, Wiesław Jerzy Cubała, Michał Walaszek, Michał Pastuszak, Krzysztof Pastuszak and Aleksander Kwaśny in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
