Abstract
Background:
Intravenous ketamine is effective in treatment-resistant bipolar depression (TRBD) with dosing typically based on actual body weight (ABW).
Objective:
This study examined whether alternative normalization formulas are associated with treatment response.
Design:
A retrospective exploratory analysis of a naturalistic registry for short-term ketamine use.
Methods:
A total of 22 TRBD inpatients received short-term intravenous ketamine. Doses were recalculated using the Boer and Devine formulas for lean body mass (LBM) and ideal body weight (IBW), and the Mosteller formula for body surface area (BSA). Calculated doses were compared with ABW dosing in responders and nonresponders.
Results:
Using the Mosteller formula, BSA-normalized doses ranged from 17.63–23.09 mg/m2 in nonresponders and 15.73–23.89 mg/m2 in responders. LBM- and IBW-based recalculations at 0.5 mg/kg yielded lower relative doses, particularly among nonresponders, suggesting potential underdosing.
Conclusion:
These preliminary findings do not support alternative dosing formulas over ABW, but replication in larger controlled studies is warranted.
Plain language summary
This study looked at how best to calculate the right dose of ketamine for people with treatment-resistant bipolar depression (TRBD). Ketamine is known to work quickly for some people with hard-to-treat depression, but it’s not officially approved for bipolar depression. There’s debate about whether the dose should be based on a person’s actual body weight, lean body mass, ideal body weight, or body surface area. Data from 22 patients who received ketamine infusions while continuing their usual psychiatric care was reviewed. The goal was to see if using different ways to calculate body size would affect whether patients responded to treatment. Patients were considered responders if their depression scores dropped by half after seven infusions. The comparison between the actual dose each patient received (based on their current weight) with what they would have received if the dose was based on lean body mass, ideal weight, or body surface area. The study found no advantage in using these alternative methods over actual body weight. It was concluded that ketamine dosing should be based on a patient’s actual body weight rather than formulas that adjust for weight or height. Using formulas like lean body mass or ideal body weight might lead to insufficient dosing, potentially reducing ketamine’s effectiveness. This is the first study to explore these different dosing methods in bipolar depression and highlights the importance of accurate, individualized dosing in maximizing ketamine’s benefit for those with treatment-resistant conditions.
Keywords
Introduction
Treatment-resistant bipolar depression (TRBD) has historically presented a substantial diagnostic challenge. For many years, the absence of a universally accepted definition impeded both the precise estimation of its prevalence and the formulation of treatment strategies aligned with regulatory standards. 1 This stands in contrast to major depressive disorder (MDD), for which a regulatory definition—albeit imperfect—has been established. 2 Recently, however, Vieta and the International Society for Bipolar Disorders (ISBD) Task Force have proposed and validated a consensus definition, marking a significant step toward greater diagnostic clarity, research standardization, and the advancement of evidence-based therapeutic strategies. 1 Despite diagnostic inaccuracies, there is growing evidence that intravenous ketamine is effective in TRBD. Recent meta-analyses report rapid and clinically meaningful reductions in depressive symptoms after infusion, although data on the durability of response remain limited.3,4 This accumulating evidence suggests that ketamine may represent a valuable option for patients who do not respond to conventional therapies, despite the absence of formal regulatory approval for bipolar depression.
By contrast, esketamine nasal spray has received approval by the U.S. Food and Drug Administration (FDA) for treatment-resistant depression (TRD) and MDD with acute suicidal ideation or behavior, and by the European Medicines Agency (EMA) for TRD in adults.5,6 Evidence supporting the use of arketamine in bipolar depression remains limited, despite literature suggesting a superior safety and tolerability profile, with fewer sedative and dissociative effects compared to other ketamine formulations.7,8 It should also be noted that both ketamine and esketamine demonstrate an acceptable safety and tolerability profile.9,10
Despite encouraging efficacy data, there is no consensus on the optimal ketamine dosing strategy for TRBD. Most clinical protocols use actual body weight (ABW) to calculate the standard 0.5 mg/kg dose. 11 However, ABW-based dosing may expose patients with obesity to higher total doses and increased hemodynamic risk, while potentially underdosing leaner patients. It has also been hypothesized that acute responses may partly reflect higher doses in patients with elevated body mass index (BMI), whereas poorer long-term outcomes could relate to metabolic syndrome. 12 Some studies further suggest that higher total doses are associated with better short-term response in obese patients. 13
These observations raise the question of whether alternative dosing strategies—such as those based on lean body mass (LBM), ideal body weight (IBW), or body surface area (BSA)—may yield more consistent and clinically appropriate dosing. Determining whether alternative body weight–based formulas influence treatment response may help optimize the precision, safety, and efficacy of ketamine dosing in patients with TRBD.
This retrospective, exploratory analysis investigates whether dose calculation based on ABW, LBM, IBW, or BSA correlates with treatment response in a naturalistic sample of TRBD inpatients.
Methods
Patients
This retrospective exploratory analysis comprises data from two registries reporting on the safety and tolerability of intravenous (IV) ketamine use in psychiatric inpatients. Detailed methodology and clinical and demographic characteristics are discussed elsewhere. 14 Initially, thirteen individuals were identified from a naturalistic observational registry (NCT04226963), a population that was also included in a prior study. 15 Subsequently, nine subjects were enrolled in a follow-up naturalistic registry protocol (NCT05565352). Both registries adhered to similar observation protocols concerning safety, tolerability, and psychometrics.
Inclusion criteria for this study required the presence of a depressive episode within the course of bipolar disorder as defined by Diagnostic and Statistical Manual of Mental Disorders, 5th Edition, along with treatment resistance, defined as the inability to achieve sustained symptomatic remission after undergoing two approved and appropriate interventions for bipolar depression with sufficient doses and duration, following the criteria set forth by Poon et al. 16 Exclusion criteria included pregnancy, lactation, hypersensitivity to ketamine, uncontrolled hypertension, and other unmanaged somatic conditions deemed by investigators to potentially impact safety. The study followed the guidelines for intravenous ketamine administration outlined by Sanacora et al. 11
The present study involves a cohort of 22 patients experiencing depressive episodes without psychotic features in the context of bipolar disorder. Both research protocols were approved by the institutional review board NKBBN/172–674/2019 and NKBBN/172–447/2022. All participants provided informed consent after confirming their comprehension of the study procedures. The study was conducted in compliance with the most recent iteration of the Declaration of Helsinki.
Study design
The registry adopted an observational design to assess safety and tolerability, during which participants maintained their baseline psychotropic medications and received standard care for coexisting chronic somatic conditions throughout ketamine infusions. The therapeutic intervention comprised eight intravenous ketamine infusions administered over 4 weeks as an adjunct to standard care. Ketamine was administered at a dosage of 0.5 mg/kg adjusted according to the patient’s ABW, delivered via IV infusion over a period of 40 min. Safety oversight was conducted by the managing psychiatrist, who monitored patients for safety indicators before, during, and after the infusion, including assessment of vital signs.
The main endpoint measured the alteration in the Montgomery–Åsberg Depression Rating Scale (MADRS) score following the seventh ketamine infusion. Response was characterized by a reduction of 50% or more in the MADRS score from the baseline.
Dose calculations
Three distinct formulas for dosage calculation, based on various factors, were employed to determine appropriate dosing regimens. The Mosteller formula, a commonly utilized method, estimates BSA through the equation: BSA = √((height (cm) * weight (kg))/3600). Subsequently, the actual administered dose was divided by the patient’s BSA to determine the dosage per square meter. Another formula calculates LBM, representing body weight minus fat weight, such as the Boer formula. LBM dosage calculation differs between males and females. For males, estimated LBM = 0.407 (weight) + 0.267 (height) – 19.2, while for females, estimated LBM = 0.252 (weight) + 0.473 (height) – 48.3. The third calculation method is based on IBW, an estimate of healthy weight based on height. An example of IBW estimation is the Devine formula. For males, the equation is IBW = 50 kg + 0.9 kg × (height (cm) − 152); for females, it is IBW = 45.5 kg + 0.9 kg × (height (cm) − 152). For the Boer and Devine formulas, theoretical doses were calculated based on a regimen of 0.5 mg/kg.
Their inclusion was intended to capture different aspects of body composition relevant to drug disposition. Specifically, the Boer and Devine formulas estimate lean or adjusted body weight, which are particularly important for drugs such as ketamine, where fat distribution and lean mass can influence the volume of distribution and clearance. The Mosteller formula, in contrast, estimates body surface area, which has long been applied as a scaling factor in pharmacokinetic modeling and drug dosing. By incorporating these approaches, we aimed to explore how different anthropometric measures might relate to ketamine’s pharmacokinetics and clinical effectiveness.
Statistical analysis
In view of the exploratory design of this study and the limited sample size, formal statistical analyses were not undertaken. Instead, the findings are reported in a descriptive format.
Results
Baseline characteristics
Demographic and clinical variables are detailed in Table 1. Based on the MADRS scores at baseline and after the seventh infusion, the 22 patients were categorized into responders (n = 14) and nonresponders (n = 8).
Clinical and sociodemographic characteristics.

BD, bipoler disorder; MADRS, Montgomery–Åsberg Depression Rating Scale.
Dose calculations
Dosage adjustments based on specific formulas are outlined for both individuals who do not exhibit a response and those who do in Tables 2 and 3. Utilizing the Mosteller formula, we initially computed the BSA, subsequently dividing the actual administered dose by the BSA. In this computation, the minimum dose per square meter was determined to be 17.63 mg/m2 for nonresponders and 15.73 mg/m2 for responders, with maximum doses reaching 23.09 and 23.89 mg/m2, respectively. For the Boer and Devine formulas, LBM and IBW were initially computed, followed by dose calculations using a dosage of 0.5 mg/kg. Using the LBM formula, nonresponders were estimated to receive between 55.4% and 75.6%, and responders between 58.7% and 90.2% of the actual administered dose. Comparable calculations were carried out using IBW, resulting in estimated doses ranging from 47% to 96% for nonresponders and from 57.4% to 124% of the actual dose for responders, respectively.
Doses determined through distinct formulas in nonresponders.
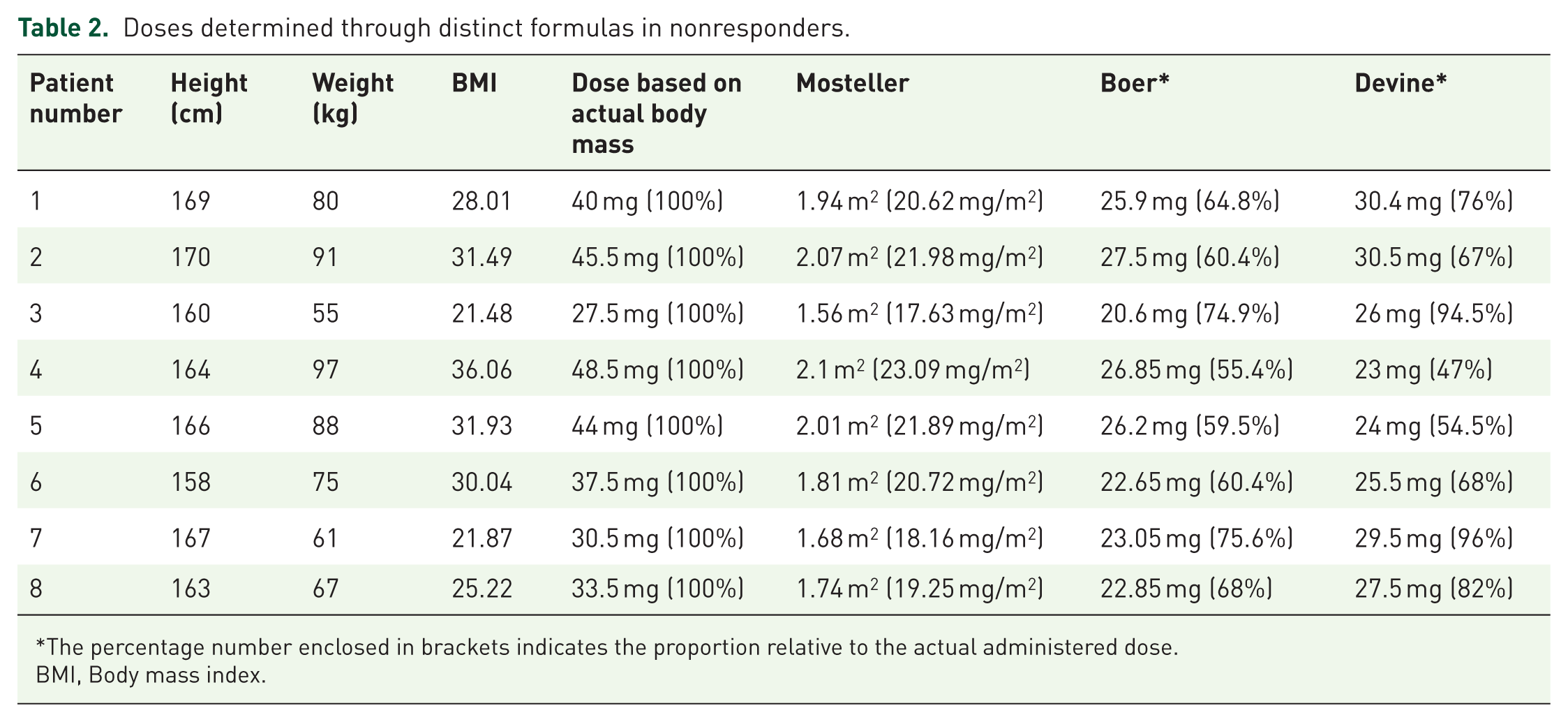
The percentage number enclosed in brackets indicates the proportion relative to the actual administered dose.
BMI, Body mass index.
Doses determined through distinct formulas in responders.

The percentage number enclosed in brackets indicates the proportion relative to the actual administered dose.
BMI, Body mass index; M, male.
The BMI of each patient was calculated and correlated with the percentage of dose adjustment as determined by the Boer and Devine formulas. The results are presented in scatter plots, with individual data points representing each patient, categorized into responders and nonresponders (Figures 1 and 2). Both scatter plots demonstrate discrepancies between the BMI inhomogeneity of the studied population and dose adjustments per the Boer and Devine formulas.

Scatter plot illustrating the distribution of participants by body mass index and dose adjustment according to the Boer formula. Each point corresponds to an individual participant: blue points indicate responders (⩾50% reduction in MADRS score), while orange points denote nonresponders (<50% reduction in MADRS score).

Scatter plot illustrating the distribution of participants by body mass index and dose adjustment according to the Devine formula. Each point corresponds to an individual participant: blue points indicate responders (⩾50% reduction in MADRS score), while orange points denote nonresponders (<50% reduction in MADRS score).
Discussion
This exploratory analysis indicates that dosing formulas based on LBM, IBW, or BSA do not confer clinical advantages over ABW–based dosing in TRBD. Recalculations using LBM and IBW resulted in lower relative doses, raising concerns regarding potential underdosing in both subgroups. Moreover, scatter plot analyses highlighted the difficulty of establishing a definitive relationship with BMI. In contrast, normalization by BSA yielded overlapping dose ranges between responders and nonresponders, further reinforcing the absence of a clear benefit compared with ABW-based dosing. To our knowledge, this represents the first study to directly compare multiple dosing paradigms within a TRBD cohort.
The evidence regarding dosing strategies or paradigms for ketamine administration in treatment-resistant mood disorders remains limited and is primarily focused on TRD. Kwaśna et al. 17 note that the use of LBM or IBW formulas generally results in lower calculated doses compared to those based on ABW, a finding consistent with our results. A post-hoc analysis of a ketamine clinical trial identified obesity as a predictor of favorable response, a finding that was echoed by a retrospective study, though limited by its small sample size.13,18 However, evidence has not been entirely consistent. In our recent exploratory analysis with 35 TRBD patients (in which currently analyzed participants are included), we did not find significant differences in BMI between responders and nonresponders; however, various formulations and dosing paradigms were utilized. 19 A separate post-hoc analysis pooling data from multiple studies suggested that higher BMI was associated with acute improvement following a single ketamine infusion, but this effect did not persist with continued treatment. 20 In contrast, a larger study of 230 patients receiving four ketamine infusions found no association between BMI and treatment response. 21 Likewise, an earlier randomized controlled trial (RCT) reported no relationship between body weight and either maximum plasma concentration or the area under the concentration–time curve over 6 h following ketamine administration. 22
There is a noted inconsistency among researchers and practitioners regarding the dose calculation for ketamine. Evidence supports the clinical efficacy, safety, and favorable tolerability profile of IV ketamine in the range of 0.5–1.0 mg/kg, with some data in favor of higher doses (i.e., 0.75–1.0 mg/kg) for patients with multiple treatment resistances.23,24 This dosage range is empirically established, although it may be attributable to calculations based on different body mass metrics, including adjustments per BSA. Lipsitz et al. 21 point out that most reports do not clarify whether ideal or actual body weight was used for dosing. The authors also highlight that ketamine’s lipophilic nature could influence its pharmacokinetics and pharmacodynamics, making safety considerations essential, especially in patients with high BMI. 21 Our findings, albeit preliminary, suggest that recalculating doses with LBM or IBW formulas could underestimate exposure, which might contribute to underdosing in certain patients, though the clinical implications of this remain uncertain.
Intravenous ketamine’s short-term efficacy necessitates repeated dosing for sustained therapeutic benefit. However, dose calculation is of prime clinical importance. Studies suggest higher dosing (0.5–1.0 mg/kg) is more effective compared to lower dosing (0.1–0.2 mg/kg) 1 day post-administration. The upper dosing limit in TRD is not established, as both 0.5 and 1.0 mg/kg show efficacy without superiority observed for 1.0 mg/kg. Still, for doses exceeding 1.0 mg/kg, the less favorable safety and tolerability profile is seen as dose-dependent, and treatment-related adverse events, such as elevated blood pressure, dissociation, and sedation, are reported. 25 Importantly, a recent systematic review concluded that the majority of patients receiving ketamine did not develop dependence on the drug. 26 Thus, from the clinician’s perspective the higher propensity for adverse events and safety concerns with relatively higher doses of IV ketamine is of importance. 25
Limitations
This study has several important limitations. It was a retrospective analysis of open-label observational data with no statistical testing, and the small, predominantly female sample precludes subgroup analyses and limits generalizability. All analyses were descriptive and exploratory, and ketamine serum concentrations were not measured. Despite these constraints, this is the first study to systematically evaluate multiple dosing formulas in TRBD. Its unique contributions may guide clinicians and underscore the need for prospective controlled trials that stratify patients by BMI or body composition, incorporate pharmacokinetic modeling, and directly compare dosing strategies with respect to efficacy, safety, and long-term tolerability.
Conclusion
This preliminary analysis did not support alternative ketamine dosing formulas over ABW–based calculations in this sample of patients with TRBD. While LBM- and IBW-based approaches appeared to yield lower relative doses, particularly in nonresponders, these findings are descriptive and should be interpreted with caution. Prospective, adequately powered trials are needed to determine whether dose normalization strategies based on LBM, IBW, or BSA can improve efficacy, safety, or tolerability in TRBD.
