Abstract
Background:
Cariprazine is a second-generation antipsychotic approved for treating schizophrenia, acute manic or mixed episodes associated with bipolar I disorder, and major depressive disorder. Antipsychotic treatment is often associated with metabolic alterations, including weight gain, dyslipidemia, and increased risk of type 2 diabetes and cardiovascular disease.
Objectives:
We performed a systematic review and meta-analysis with the overarching aim of synthesizing study results that describe the effect of cariprazine on glucose and lipid homeostasis, as well as weight, in persons living with schizophrenia.
Design:
A systematic literature review and meta-analysis was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines.
Data sources and methods:
We systematically searched three major electronic databases of medical and social science research papers (PubMed/MEDLINE, PsycINFO, and Cochrane Central Register of Controlled Trials (CENTRAL) for relevant titles and abstracts published between January 2014 and March 2025.
Results:
Using the Covidence platform, we included 12 studies for the systematic review and four randomized controlled studies for the meta-analysis. Meta-analysis revealed a significant difference in weight change between cariprazine and placebo; however, no significant differences were observed for total cholesterol or fasting glucose.
Conclusion:
This meta-analysis suggests that cariprazine, although associated with modest weight gain, does not cause significant alterations in lipid and glycemic profiles, confirming its favorable metabolic profile compared to other atypical antipsychotics.
Trial registration:
This study is registered with the International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY), 2025100014.
Plain language summary
Cariprazine, used for schizophrenia, was reviewed for its effects on weight, cholesterol, and glucose. Findings show only modest weight gain without significant changes in cholesterol or blood glucose level, suggesting a favorable metabolic profile compared to many other antipsychotics.
Keywords
Introduction
Cariprazine is a second-generation antipsychotic with a receptor profile characterized by dopamine D3-preferring D3/D2 receptor partial agonism and serotonin 5-HT1A partial agonism. 1 Cariprazine received FDA approval in 2015 for treating schizophrenia and acute manic or mixed episodes associated with bipolar I disorder. In 2022, it was approved as adjunctive therapy for treating major depressive disorder. Cariprazine is available in four doses: 1.5, 3, 4.5, and 6 mg. Cariprazine has shown efficacy across different symptoms and domains of schizophrenia.2,3 The efficacy of cariprazine in schizophrenia was observed on positive symptoms as well as negative symptoms and cognition. 4 Notably, the efficacy of cariprazine in treating acute psychotic symptoms of schizophrenia was also demonstrated in several papers, as well as the efficacy for relapse prevention. 5 Furthermore, several papers also indicated the efficacy in bipolar I depression, bipolar mania, mixed features, and anhedonia as well as in major depressive disorder.6–9
Antipsychotic treatment is often associated with metabolic alterations, including weight gain, dyslipidemia, increased risk of type 2 diabetes, and cardiovascular disease.10–12 Pillinger et al., 13 in a systematic review and meta-analysis, showed that cariprazine does not demonstrate disruption of glucose-insulin homeostasis or limit homeostasis or weight gain in replicated preclinical or clinical data. However, it can occur at significantly lower rates than other antipsychotic agents, as well as other dopamine partial agonists. Zhu et al. 14 conducted an in-depth analysis of adverse event reports associated with cariprazine using data from the FDA Adverse Event Reporting System (FAERS), demonstrating distinct characteristics of cariprazine AEs. Additionally, Masand et al. 15 conducted a retrospective study using electronic health records to analyze the metabolic profiles of psychiatric patients during the 12 months preceding cariprazine initiation (baseline) and for up to 12 months following initiation. Recently, Correll et al. 16 in a large retrospective observational study on 612 psychiatric patients in treatment with cariprazine described that cariprazine was associated with estimated annual linear trajectories of +0.91 kg/year for weight, +0.31 kg/m2/year for BMI, −2.38 mmHg/year for systolic blood pressure, and −0.57 mmHg/year.
It is well accepted that cariprazine is an effective and established treatment for schizophrenia; however, to date, no meta-analysis that synthesized metabolic alterations focused only on cariprazine treatment in this disorder has been done. Based on the hypothesis that antipsychotic treatment is often associated with metabolic alterations, including weight gain, dyslipidemia, and increased risk of type 2 diabetes, we performed a systematic review and meta-analysis with the overarching aim of synthesizing study results that describe the effect of cariprazine on glucose and lipid homeostasis as well as weight in persons living with schizophrenia.
Materials and methods
A systematic literature review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, 17 see Supplemental Materials for the PRISMA checklist.
Search strategy
To ensure inclusivity, we systematically searched three major electronic databases of medical and social science research papers (PubMed/MEDLINE, PsycINFO, and Cochrane Central Register of Controlled Trials (CENTRAL) for relevant titles and abstracts published between January 2014 and March 2025. The following terms were combined to search the databases in titles/abstracts (TAs): “Cariprazine AND (Schizophrenia OR Psychosis OR Psych* OR Side effects OR Metabolic Effects OR Weight Gain OR Dyslipidemia OR Glucose OR Cholesterol).”
Filters were used to limit results to English. The search identified 912 papers. See the flowchart for details of the study selection process (Figure 1).

PRISMA 2020 flow diagram for new systematic reviews, which included searches of databases and registers only.
Eligibility criteria
This review included original articles that explicitly discussed the presence of metabolic effects of the use of cariprazine in schizophrenia. When the title or abstract indicated an eligible study, the full-text article was obtained and carefully examined to assess its relevance to the review’s aims. Articles needed to meet the following eligibility criteria: (a) include the use of cariprazine and (b) include the presence of a diagnosis of schizophrenia according to Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria; (c) include the presence of side effects, and parameters to evaluate metabolic effect as laboratory values of cholesterol total, high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglyceride, glucose and weight. In addition, (d) participants needed to be adults (aged 18 years and older), and (e) the study needed to be a quantitative research paper with a case-control, cohort, or cross-sectional design or randomized controlled trials. The exclusion criteria were as follows: (a) studies published before 2014; (b) absence of side effects evaluation; (c) studies that did not investigate the efficacy and safety of cariprazine; (d) studies not published in peer-reviewed journals; (e) not published in English; (f) case studies, meta-analytical reviews, systematic reviews, narrative reviews, or book chapters.
Study selection and data collection
The authors independently extracted and reviewed the studies using a two-step process: (1) screening and selecting based on the article’s title and abstract and (2) screening and selecting based on the full text. A data extraction spreadsheet was developed, adding the author(s), publication year, country, sample characteristics (population type and sample size), study design, outcome measures, and main results. Discussions among the senior authors, who also independently read all the articles, resolved potential disagreements regarding article inclusion and data collection. Furthermore, we managed screening, data extraction, and risk-of-bias assessment in Covidence (Veritas Health Innovation, Melbourne, Australia). All search results were exported to Covidence (Veritas Health Innovation, Melbourne, Australia) for record management. Covidence automatically removed duplicates prior to screening. Two reviewers independently screened titles/abstracts and then full texts against pre-specified eligibility criteria within Covidence; conflicts were resolved by consensus or, when necessary, a third reviewer. Reasons for full-text exclusion were logged in Covidence and are reported in the PRISMA 2020 flow diagram. Furthermore, the assessment of inter-rater reliability yielded an overall Cohen’s kappa coefficient of 0.92, indicating almost perfect agreement between the two independent reviewers across all domains.
Meta-analyses methods
Three meta-analyses focused on patients with schizophrenia, evaluating changes in weight, cholesterol, and glucose for both cariprazine 1.5 and 3 mg. Egger’s test and funnel plots were used to assess the presence of publication bias, while the homogeneity test was conducted to evaluate variability among the studies examined. The meta-analyses were performed using the difference between the mean values in the placebo group and the mean values in the cariprazine 1.5 mg/3 mg groups as the effect size. We did not evaluate other doses of cariprazine in the meta-analysis process due to the paucity of randomized controlled trials. In all the aforementioned analyses, an alpha significance level of 0.05 was used. IBM SPSS Statistics software version 29 was used for the statistical analysis.
For the meta-analysis, the following studies included in the review part of the article were excluded: Barabássy et al., 18 Cutler et al., 19 Durgam et al., 20 Durgam et al., 21 Earley et al., 22 Nakamura et al., 23 Nasrallah et al., 24 Németh et al. 25 as metabolic parameters were not reported or there were aggregate analyses, or they were open-label studies.
This study is registered with the International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY) under protocol number 2025100014.
Results
The PRISMA flowchart of the study selection process is presented in Figure 1. Electronic searches identified 912 publications. After excluding duplicates (n = 267), 645 abstracts and titles were screened for suitability. After non-RCT studies (n = 89), studies not in English language (n = 25), and non-pertinent studies (n = 491) were removed (n = 605), 40 full-text titles were assessed for eligibility, with 28 records excluded because they did not align with the aims and inclusion criteria of our review (see Figure 1). Any disagreements regarding study eligibility were resolved following consensus discussions among the authors. (for the PRISMA checklist see Supplemental Materials).
Safety and tolerability of cariprazine in patients with schizophrenia
Twelve randomized controlled studies investigated metabolic alterations associated with cariprazine treatment in patients affected by schizophrenia (Table 1).3,18–28
Randomized controlled studies investigated metabolic alterations associated with cariprazine treatment in patients affected by schizophrenia.
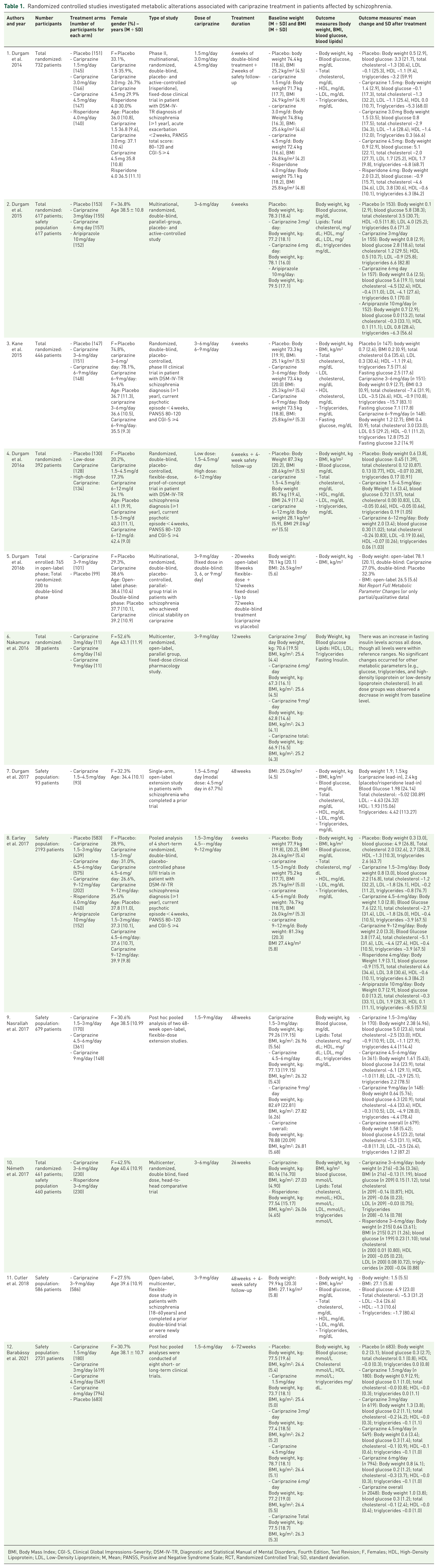
BMI, Body Mass Index; CGI-S, Clinical Global Impressions-Severity; DSM-IV-TR, Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision; F, Females; HDL, High-Density Lipoprotein; LDL, Low-Density Lipoprotein; M, Mean; PANSS, Positive and Negative Syndrome Scale; RCT, Randomized Controlled Trial; SD, standard deviation.
Durgam et al. 26 reported on a 9-week, multinational, randomized, double-blind, placebo- and active-controlled phase II trial in patients with acute exacerbation of schizophrenia. Participants were randomized to five different groups: placebo, cariprazine 1.5, 3.0, 4.5 mg/day, or risperidone 4.0 mg/day. The most frequent AEs included insomnia, akathisia, extrapyramidal disorder, nausea, sedation, dizziness, and constipation, while the metabolic changes were small and similar across all the groups. Durgam et al. 27 conducted a double-blind, parallel-group, placebo- and active-controlled study, showing that body weight increased by 0.8 kg for cariprazine 3 mg/day, 0.6 kg for cariprazine 6 mg/day, 0.1 kg for placebo, and 0.7 kg for aripiprazole.
The reported incidence of body weight increase ⩾7% was 6% for cariprazine 3 mg/day, 5% for cariprazine 6 mg/day, 3% for placebo, and 6% for aripiprazole. The incidence of orthostatic hypotension was 18% with cariprazine 3 mg/day, 12% with cariprazine 6 mg/day, 12% with placebo, and 11% with aripiprazole. In a separate 6-week, randomized, double-blind, placebo-controlled, flexible-dose, proof-of-concept study, the efficacy and safety of cariprazine were evaluated at doses of 1.5–4.5 mg/day and 6–12 mg/day in participants with an acute exacerbation of schizophrenia. 3 In a multinational, randomized, double-blind, placebo-controlled study, the authors evaluated the efficacy, safety, and tolerability of long-term cariprazine treatment in relapse prevention among participants with schizophrenia. 20 The study lasted up to 97 weeks, consisting of a 20-week open-label phase (8 weeks of flexible-dose run-in and 12 weeks of fixed-dose stabilization), followed by double-blind randomization for up to 72 weeks. Participants were treated with cariprazine 3–9 mg/day in the open-label phase. Metabolic changes and weight gain were minimal, and no significant changes in cardiovascular parameters were observed. Kane et al. 28 in a 6-week, randomized, double-blind, placebo-controlled, parallel-group, phase III clinical trial investigated the efficacy and safety of cariprazine 3–6 mg/day and 6–9 mg/day in patients with acute schizophrenia exacerbation. The most common AEs were akathisia, Extrapyramidal Symptoms (EPS), and tremors, while metabolic changes were small and comparable between groups. A 28-week, multicenter, randomized, open-label, parallel group, fixed-dose clinical pharmacology study by Nakamura et al. 23 on Japanese patients with schizophrenia evaluated three parallel therapeutic doses of cariprazine: 3, 6, or 9 mg/day. For most laboratory parameters, changes from baseline were small, and no major differences were observed between dose groups. There was an increase in fasting insulin levels across all dose groups from baseline to the end of the study, though all levels were within reference ranges. Furthermore, in all dose groups, there was a decrease in weight from baseline. Durgam et al. 21 in a 48-week, single-arm, open-label extension study showed the long-term safety and tolerability of cariprazine (1.5–4.5 mg/day) in patients with schizophrenia. Mean body weight increased by 1.9 kg from the start of the lead-in study to the end of this extension. Changes in cholesterol, glucose, and prolactin were small and not clinically relevant. In a post hoc pooled safety analysis of four phase II/III, randomized, double-blind, placebo-controlled trials involving acute schizophrenia treatment with cariprazine, Earley et al. 22 confirmed that cariprazine was generally well tolerated across all dose groups (1.5–3, 4.5–6, and 9–12 mg/day). Also, weight gain was modest, averaging 1–2 kg across dose groups. No clear differences were seen in metabolic parameters or weight change in a phase IIIb randomized, double-blind trial including 66 study centers in 11 European countries. 25 Nasrallah et al. 24 observed that in the group of patients treated with cariprazine 9 mg/day, mean total cholesterol, fasting LDL, and HDL cholesterol levels decreased more than in the 1.5–3 mg/day group. The percentage of patients with a ⩾7% increase in body weight was similar in the 1.5–3 and 4.5–6 mg/day dose groups, and slightly lower in the 9 mg/day group.
In a 53-week, multicenter, open-label study designed to evaluate the long-term safety and tolerability of cariprazine in adult patients with schizophrenia, Cutler et al. 19 observed that the average weight gain was 1.5 kg, and changes in metabolic (total cholesterol, HDL cholesterol, LDL cholesterol, triglycerides, and fasting glucose), were generally small and not clinically significant. A pooled analysis of eight phase II/III Studies. Eight schizophrenia studies (2731 patients) revealed that in the overall cariprazine dose group, levels of total cholesterol, high-density lipoprotein cholesterol, and fasting triglycerides decreased from baseline; 18 no dose response was noted for any parameter. Increases in fasting glucose were small and similar to those in the placebo group across all cariprazine dose groups and overall. Slightly more total cariprazine-treated patients (3.4%) than placebo-treated patients (2.0%) shifted from normal to high levels of fasting glucose (<5.6–>7 mmol/L), although levels for cariprazine patients remained stable over time. The incidence of hyperglycemia/diabetes mellitus-related AEs was low and similar in all treatment groups. The mean increase in weight from baseline was 1 kg for overall cariprazine; the AE of weight increase was greater in cariprazine-treated patients than in placebo, with no observed dose–response relationship for cariprazine. Finally, the authors noted a higher rate of Treatment-Emergent Adverse Events (TEAEs) in the total cariprazine group (77%) compared with the placebo group (68%), with a higher percentage of events observed for cariprazine 6 mg/day compared with lower doses.
Meta-analysis of cariprazine versus placebo in schizophrenia
Cholesterol
Initially, Egger’s test and a funnel plot were used to assess publication bias. Egger’s test indicated no evidence of publication bias (t = −0.050, p-value = 0.964); consequently, the analysis proceeded to consider all four of the initial articles (Figure 2s).
The test of homogeneity was not statistically significant (p-value > 0.05); therefore, a fixed-effects model was used. Specifically, the I2 heterogeneity index was 32.5% (chi-square (Q statistic) = 4.447, df = 3, p-value = 0.217).
The effect size was not statistically significant (p-value = 0.237), with an effect size of −0.129 (95% CI (−0.344, 0.085); Figure 2).
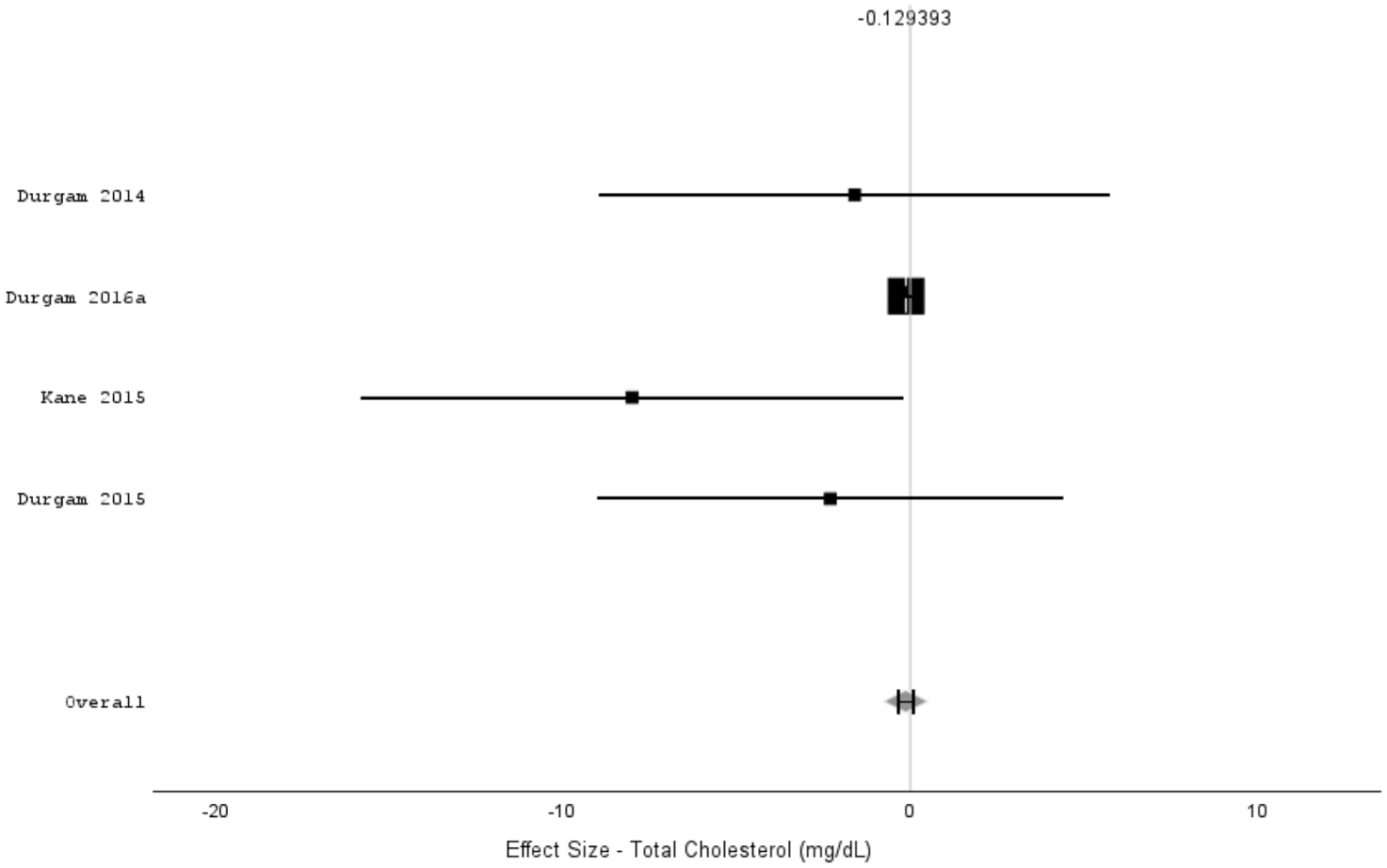
Random-effects meta-analysis of four short-term (6 weeks) trials of Cariprazine (1.5–3 mg) versus placebo for the treatment of schizophrenia on blood cholesterol levels. Weight of each study is indicated by the size of each box, and 95% confidence intervals are indicated by horizontal lines. Detailed data are provided in Table 1.
From the forest plot, it is evident that the effect of the Kane 2015 article 28 is the only one that is significant; the remaining articles (Durgam 2014, 26 Durgam 2015, 27 Durgam 2016a 3 ) show a non-significant effect. The overall effect size, as previously mentioned, is not significant; therefore, there is no statistically significant difference between cariprazine and placebo in terms of change in total cholesterol.
Glucose
Initially, Egger’s test and the funnel plot were utilized to assess the presence of publication bias. Egger’s test indicated no evidence of publication bias (t = 1.222, p-value = 0.346); consequently, the analysis proceeded to consider all four of the initial articles (Figure 3s).
The test of homogeneity was not statistically significant (p-value > 0.05); therefore, a fixed-effects model was used. Specifically, the I2 heterogeneity index was 53.1% (chi-square (Q statistic) = 6.396, df = 3, p-value = 0.094).
The effect size was not statistically significant (p-value = 0.147), with an effect size of 0.274 (95% CI (−0.096, 0.645); Figure 3).

Random-effects meta-analysis of four short-term (6 weeks) trials of cariprazine (1.5–3 mg) versus placebo for the treatment of schizophrenia on blood glucose levels. Weight of each study is indicated by the size of each box, and 95% confidence intervals are indicated by horizontal lines. Detailed data are provided in Table 1.
From the forest plot, it is evident that the effect of the Kane 2015 article 28 is the only one that is significant; the remaining articles (Durgam 2014, 26 Durgam 2015, 27 Durgam 2016a 3 ) show a non-significant effect. The overall effect size, as previously mentioned, is not significant; therefore, there is no statistically significant difference between cariprazine and placebo in terms of change in fasting glucose.
Weight
Initially, Egger’s test and a funnel plot were used to assess publication bias. Egger’s test indicated no evidence of publication bias (t = −0.967, p-value = 0.436); consequently, the analysis proceeded to consider all four of the initial articles (Figure 4s).
The test of homogeneity was not statistically significant (p-value > 0.05); therefore, a fixed-effects model was used. Specifically, the I2 heterogeneity index was 16.8% (chi-square (Q statistic) = 3.607, df = 3, p-value = 0.307).
The effect size was statistically significant (p-value < 0.001), with an effect size of 0.649 (95% CI (0.301, 0.998); Figure 4).

Random-effects meta-analysis of four short-term (6 weeks) trials of Cariprazine (1.5–3 mg) versus placebo for the treatment of schizophrenia on body weight. Weight of each study is indicated by the size of each box, and 95% confidence intervals are indicated by horizontal lines. Detailed data are provided in Table 1.
From the forest plot, it is evident that the effect of the Kane 2015 article 28 is the only one that is not significant; the remaining articles (Durgam 2014, 26 Durgam 2015, 27 Durgam 2016a 3 ) show a significant effect. The overall effect size, as previously mentioned, is significant; therefore, there is a statistically significant difference between cariprazine and placebo in terms of weight change.
Discussion
This systematic review and meta-analysis assessed the metabolic effects of cariprazine in patients with schizophrenia, focusing on three key parameters: cholesterol, glucose, and body weight, which are considered the most frequent metabolic alterations associated with second-generation antipsychotic treatment.29,30 The results of the systematic review, based on 12 randomized controlled trials, provide a consistent picture of the absence of metabolic disruption in key parameters of interest, including fasting and non-fasting glucose, total cholesterol and its fractions, and anthropometrics such as body weight.
The meta-analysis on body weight found a statistically significant weight gain in patients treated with cariprazine compared to placebo. However, the magnitude of the increase was modest (approximately 1–2 kg in most studies) and less clinically relevant than that of other atypical antipsychotics known for their metabolic impact. Furthermore, it is essential to note that no clear dose–response relationship was observed for cholesterol, glucose, and body weight. Weight gain did not exceed critical risk thresholds for most patients. The literature on body weight changes with cariprazine treatment is mixed, and some studies23,31 have even reported a slight weight reduction.
Regarding cholesterol, the change in total cholesterol did not show a statistically significant difference between the cariprazine and placebo groups. The homogeneity test indicated moderate heterogeneity, but it was insufficient to justify a random-effects model. Some studies2,18 have documented slight reductions in LDL, HDL, and total cholesterol levels, but within physiological limits. The observed changes do not appear to have significant clinical relevance, stressing the safety and tolerability of cariprazine in treating patients with schizophrenia.
Also, for fasting blood glucose, the meta-analysis did not identify statistically significant differences between cariprazine and placebo. In line with FAERS data and studies by Barabássy et al., 18 most patients did not exhibit evidence of dysglycemia or dyslipidemia. Only a small percentage of patients showed a shift from normal to elevated levels, but without a clear causal association with the drug.
Schizophrenia is associated with a significant reduction in life expectancy, mainly attributable to cardiovascular and metabolic comorbidities. Atypical antipsychotics are the standard of care for this disorder, but metabolic adverse effects, such as weight gain, hypertension, and overall cardiovascular risk burden many. 12 The common occurrence of weight gain in metabolic alterations associated with antipsychotic agents has provided the impetus to discover and develop antipsychotics with an attenuated weight gain and metabolic profile. 32 Furthermore, a study by Correll et al. 16 used retrospective data from a large US health database (Symphony Health IDV), including adults diagnosed with schizophrenia (n = 75), bipolar I disorder, or major depressive disorder, receiving a treatment with cariprazine and demonstrated that the stabilization or reduction of weight gain in patients with schizophrenia, is a clinically significant result in terms of secondary cardiovascular prevention. Results of this study accord with other lines of evidence that adverse events related to metabolic disorders were rare in patients with schizophrenia treated with cariprazine, and the metabolic profile of cariprazine is more favorable compared to many other second-generation antipsychotics.
The low incidence of hyperglycemia, dyslipidemia, and significant weight gain suggests that cariprazine may represent a safe and sustainable therapeutic choice for schizophrenic patients at metabolic risk. In a context in which overweight, obesity, and hypertension are predictive factors of poor adherence to treatment and worse functional outcome, the choice of an antipsychotic such as cariprazine may represent an advantageous therapeutic strategy. The relatively benign effect or minimal effect that this drug has on metabolic as well as anthropometric parameters and schizophrenia accords with what has also been reported in adults with major depressive disorder and bipolar disorder. 32
The present study presents several limitations: the limited number of studies for each metabolic outcome (only four included in each of the three meta-analyses). The variability in experimental designs and doses administered. Another limitation is the relatively few long-term studies examining the long-term effects of cariprazine on anthropometric and/or metabolic parameters. Although available evidence does not suggest progressive disruption in these outcomes in individuals receiving maintenance treatment, 33 further research is needed. Furthermore, only cariprazine 1.5 and 3 mg were meta-analyzed due to the paucity of RCTs. Finally, we have not subjected this particular agent to more sophisticated measures of effects on glucose and insulin homeostasis, including HOMA-IR.
In conclusion, this meta-analysis suggests that cariprazine, although associated with modest weight gain, does not cause significant alterations in lipid and glycemic profiles, confirming its favorable metabolic profile compared to other atypical antipsychotics. Considering the greater vulnerability of patients with schizophrenia to metabolic disorders, cariprazine is proposed as an appropriate therapeutic choice effective on the symptomatic level and not expected to exacerbate the risk of metabolic conditions in this population of persons living with mental illness. However, further studies with larger samples and longer follow-up will be essential to consolidate this evidence and better define the management of metabolic risk in cariprazine treatment.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253251408651 – Supplemental material for Effects of cariprazine on cholesterol, glucose, and weight in patients with schizophrenia: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tpp-10.1177_20451253251408651 for Effects of cariprazine on cholesterol, glucose, and weight in patients with schizophrenia: a systematic review and meta-analysis by Isabella Berardelli, Mariarosaria Cifrodelli, Attilio Valerio Mammoliti, Federico Formica, Riccardo Iannazzo, Roger S. McIntyre and Maurizio Pompili in Therapeutic Advances in Psychopharmacology
