Abstract
Approximately 15% of pregnant women experience postpartum depression (PPD). Even with currently available antidepressant treatments, many women will continue to be impaired by symptoms. Psychedelic therapy offers a promising transdiagnostic therapeutic strategy for several mental health disorders, and early results from current trials suggest that serotonergic psychedelics may represent a viable therapeutic approach for PPD. However, there is marked variability in the therapeutic response to psychedelic therapy, and the benefit–risk ratio in this population is not yet clear. To inform the rationale for the use of serotonergic psychedelics in the treatment of PPD, this review summarises the existing knowledge of immune, endocrine and neural pathways underpinning PPD and explores how serotonergic psychedelics interact with these pathways in the context of maternal motivation, bonding and caregiving behaviours. Finally, special considerations for psychedelic therapy in the postpartum period are outlined and future perspectives explored. Despite the rationale and encouraging early findings, further research is required to determine efficacy and safety profiles. Future studies, particularly longitudinal trials, should include adaptations and safeguards tailored to the unique physiological, psychological and caregiving contexts of the postpartum period.
Introduction
Globally, approximately 15% of pregnant women experience postpartum depression (PPD). 1 Poverty, migration, exposure to violence (domestic, sexual and gender-based) and low social support are particularly relevant risk factors. 2 PPD can manifest in various aberrant patterns of mother–infant interactions, including feelings of maternal inadequacy, negative perceptions of the infant, disconnection, anxiety/obsessional thoughts regarding the infant/harm to infant, together with circadian rhythm/sleep disturbance and irritability. 3
PPD overlaps substantially with major depressive disorder (MDD). Both involve a complex interaction of psychological, social and biological factors, in addition to genetic and environmental factors, with the notable added complexity of evolutionarily shaped physiological changes over the peripartum and postpartum period involving immunological, neuro-endocrine and neuronal changes, all interacting with the psychosocial environment, in preparation for the initial caring for the infant. 4
Effective treatment of PPD must account for the unique physiological, psychological and relational complexities of the perinatal period. 5 Although antidepressants remain a cornerstone of pharmacological intervention, 6 their delayed onset of action (typically 4–6 weeks) can extend maternal distress and prolong disruption of early mother–infant interaction.
The only Food and Drug Administration (FDA) approved treatments specifically for PPD are the neuroactive steroids brexanolone and zuranolone. 7 Although rapid-acting agents such as ketamine and esketamine are under investigation, 8 evidence is largely based on short follow-up periods, which may be insufficient to detect late-onset PPD.
Serotonergic psychedelics administered with psychological support/therapy, influenced by extra-pharmacological factors 9 (physical environments, music, attitudes and social interactions), induce complex and variable changes in self, sensory and emotional processing, with the potential for sustained effects/improvements in mood and social functioning. 10
There is growing evidence supporting the use of psychedelic therapy in MDD and treatment-resistant depression (TRD). 11 Although caution is warranted when extrapolating these findings to PPD, preliminary results suggest a potential therapeutic role in this condition. A phase IIa trial (n = 10) for inhalable short-acting mebufotenin (5-MeO-DMT) significantly reduced symptoms of depression from baseline to day eight, without serious adverse events. 12 This trial was conducted under the supervision of qualified healthcare professionals, providing psychological support per standard of care, but without any planned psychotherapeutic intervention before, during, or after dosing.
Recent results from a randomised, double-blind, active dose-controlled clinical trial (n = 84), which has not yet been published, evaluated the safety and efficacy of a single subcutaneous dose of a synthetic tryptamine psychedelic structurally related to psilocin for the treatment of moderate to severe PPD. 13 This study, which also did not include planned psychotherapeutic sessions, reported a 77.1% response rate at day 7, compared with 61.6% in the active control group, 13 underscoring the need for cautious interpretation given the notably high response observed in the comparator arm. Preliminary results from the same trial indicated that metabolites in breast milk accounted for <0.1% of the maternal dose, suggesting minimal infant exposure. 14
However, it is important to emphasise that these results remain preliminary, and larger, well-powered trials are required to confirm these findings and to establish the safety and efficacy of psychedelic therapy in PPD, given the unique physiological, psychological and social complexities of this condition.
These complexities include the need for enhanced support structures due to childcare, particularly if multiple dosing sessions are required, modifications to consent processes given the potential for challenging maternal experiences, including the rare but serious risk of psychosis, adaptations to breastfeeding regimens and adjustments to psychological or family-based interventions.
Drawing on evidence from both preclinical and clinical studies, we explore how psychedelic interventions impact immune function, the tryptophan-kynurenine pathway, neuroendocrine systems, neuroplasticity and neural circuits involved in reward and maternal caregiving. These mechanisms are discussed in relation to their potential role in enhancing maternal motivation, bonding and caregiving behaviours in the context of PPD.
Finally, we outline special considerations for psychedelic therapy in the postpartum period and explore future perspectives.
Search strategy and selection
A PubMed search was conducted for English-language studies published between 2018 and 2025, with earlier studies considered where relevant. Search terms included ‘immune system’, ‘endocrine’, ‘neurophysiology’, ‘cognition’ and ‘neuroimaging’, combined with keywords pertaining to PPD. Titles and abstracts retrieved from the search were screened by authors (GT, FC and DMcL) to identify the most relevant reviews and articles. Case reports, articles written in another language other than English, letters, opinion pieces, editorials, articles related to vaccinations, articles related to pregnancy complications and diseases and articles related to livestock were not considered suitable.
Endocrine pathways, PPD and serotonergic psychedelics
Psychedelics, sex steroids and PPD
The interaction between psychedelics and sex steroids has received little study. Given the abundant evidence implicating an increased brain sensitivity to changes in sex steroids in PPD pathophysiology (Table 1), the delineation of these interactions may inform therapeutic strategies. To our knowledge, no studies have directly probed the effects of psychedelic drugs on oestrogen and progesterone levels.
Summary of recent findings relating the endocrine pathways to healthy pregnancies and pregnancies associated with PPD.

ACTH, adrenocorticotropic hormone; BDNF, brain-derived neurotrophic factor; CRH, corticotropin-releasing hormone; CSF, cerebrospinal fluid; GABA, gamma-aminobutyric acid; GnRH, gonadotropin-releasing hormone; GR, glucocorticoid receptor; HPA, hypothalamic pituitary adrenal axis; mRNA, messenger ribonucleic acid; PPD, postpartum depression; SSRIs, selective serotonin reuptake inhibitors.
Gukasyan and Narayan 39 report the cases of three women describing pronounced menstrual changes after the use of classic psychedelics. Although the prevalence of such effects is unknown, this provides limited evidence for an effect of classic psychedelics on sex steroids, which play an important role in regulating the menstrual cycle. In support of this, the serotonin system has been shown to interact with the oestrogen system in vitro. 40
Acting through the 5-HT2A receptor, serotonin has been shown to enhance the expression and activity of aromatase, a critical oestrogen biosynthetic enzyme, in human placental cell lines. 41 This effect is induced by 2,5-dimethoxy-4-iodoamphetamine (DOI), a classic psychedelic, in both human placental cell lines and human primary villous trophoblast cells. 42 Such an effect on aromatase expressed in other bodily compartments, such as the ovaries or adipose tissue, could appreciably impact oestrogen biosynthesis. Hence, there is evidence to suggest that psychedelics may impact the production of sex steroids. However, human biochemical data supporting this hypothesis is lacking.
Psychedelics may also alter sex steroid production indirectly, for example, through effects on the hypothalamic–pituitary–adrenal (HPA) axis (discussed later). The HPA axis inhibits the hypothalamic–pituitary–ovarian (HPO) axis through several mechanisms. Glucocorticoids, the final product of the HPA axis, suppress the HPO axis at all levels and inhibit oestradiol signalling in target tissues.
The effect of sex steroids on the serotonin system also warrants consideration. This topic is thoroughly reviewed by Shadani et al. 43 For example, oestradiol increases serotonin levels by increasing tryptophan hydroxylase production, thereby enhancing serotonin synthesis, inhibiting transcription of the serotonin transporter (SERT; responsible for synaptic serotonin reuptake) and inhibiting monoamine oxidase A (responsible for serotonin metabolism).
Furthermore, the oestrogen receptor beta (Erβ) upregulates the 5-HT2AR, suggesting that increased oestradiol levels may be associated with increased activity of classic psychedelics. 43 These effects likely contribute to the sex differences that are observed in the response to psychedelics in preclinical models, such as cortical spinogenesis, 44 alcohol preference 45 and fear responses. 46 Hence, it is problematic that males are overrepresented in most studies of psychedelic action, both clinical and preclinical. Understanding sex differences in psychedelic response becomes particularly important as clinical trials begin to investigate psychiatric disorders which predominantly or exclusively impact women, such as anorexia nervosa 47 and PPD. 12
Neurosteroids, the GABA system and PPD
Brexanolone and zuranolone are currently the only drugs specifically licensed by the FDA for the treatment of PPD. 7 Both are neuroactive steroids with GABAAR positive allosteric modulator (PAM) properties. Brexanolone, licensed for the treatment of PPD in 2019, is a proprietary formulation of the endogenous neurosteroid allopregnanolone, which is a progesterone metabolite and a GABAAR PAM. Zuranolone, licensed in 2023, is a synthetic allopregnanolone analogue. Brexanolone and zuranolone suggest that a deficiency of gamma-aminobutyric acid (GABA) neurotransmission or neurosteroid GABAAR PAMs may be implicated in the pathophysiology of PPD (Table 1).
The interaction between psychedelics and the GABA system has received little study. Psilocybin and DOI have both been shown to increase extracellular GABA levels in the prefrontal cortex. 48 This likely occurs through action on 5-HT2AR-expressing GABAergic interneurons. 49 Psilocybin also increases extracellular GABA levels in the reticular nucleus of the thalamus. 48
Furthermore, ayahuasca, a beverage containing N,N-DMT among other compounds, has been shown to increase GABA levels in the rat hippocampus and decrease GABA levels in the amygdala. 50 The finding that ayahuasca increases GABA levels in the hippocampus is interesting because decreased hippocampal expression of several GABA receptor subunits is reported in hormone-simulated pregnancy (HSP) models. 15 Furthermore, failure to upregulate the GABAAR δ subunit may underlie PPD-like behaviour in mice. 30
Further research is needed to draw firm conclusions on the interaction between psychedelics and the GABA system, and studies could consider magnetic resonance spectroscopy to quantify and localise these effects in human subjects.
Psychedelics and the hypothalamic–pituitary–adrenal axis
It is well-established that classic psychedelics, 51 and MDMA, 52 acutely activate the HPA axis. The post-acute impact of psychedelics on the HPA axis is most obviously of interest in mental health disorders, including PPD. While this remains underexplored, preliminary insights can be gained from animal models. In a stress-induced rat model of depression, Wang et al. 53 found that psilocybin rescued depressive phenotypes and decreased blood ACTH and corticosterone concentrations at post-mortem. Nogueira et al. 54 demonstrated possible effects of 5-MeO-DMT on stress resilience. In mice 5 days post-5-MeO-DMT treatment, corticosterone was significantly lower before, although not after, acute restraint stress versus mice treated with saline. Furthermore, mice pre-treated with 5-MeO-DMT demonstrated a reduced behavioural response to restraint stress. These data suggest that 5-MeO-DMT is associated with a post-acute increase in stress resilience, which may be mediated by reductions in basal corticosterone. 54
To our knowledge, only one study has probed the post-acute effects of classic psychedelics on the HPA axis in humans. Mason et al. 55 measured plasma cortisol in the acute phase of psilocybin dosing and examined cortisol response to a psychosocial stressor 7 days later. Predictably, they found that psilocybin acutely increased plasma cortisol. However, it had no significant effect on the cortisol response to the psychosocial stressor. The authors note that the number of people who demonstrated a cortisol response to stress was lower in the psilocybin group versus the control group. However, the small sample size of 15 participants per group limits the ability to draw firm conclusions.
Galvão et al. 56 studied the effects of ayahuasca in patients with TRD and healthy controls. Interestingly, patients demonstrated basal hypocortisolaemia and a blunted salivary cortisol awakening response (CAR). Ayahuasca administration was associated with acutely increased salivary cortisol in both patients and controls; however, 2 days after ayahuasca dosing, patients had similar levels of salivary CAR to healthy controls. This effect was not seen in the placebo group. The combined findings of Mason et al. 55 and Galvão et al. 56 may point towards an ability of classic psychedelics to normalise HPA axis activity, although there is currently insufficient evidence to support this.
It is possible that acute activation of the neuroendocrine stress response also plays an important role in the lasting therapeutic efficacy of psychedelic agents. This is tentatively supported by murine experiments demonstrating that the post-acute anxiolytic effects of psilocybin may be mediated by acute HPA axis activation, insofar as they are abolished by the glucocorticoid antagonist mifepristone as well as by chronic CORT administration. 57 Acute stress facilitates the formation of memories of the stressful context, which is thought to be evolutionarily advantageous. For this reason, De Wit et al. 58 have hypothesised that acute activation of the neuroendocrine stress response by psychedelic drugs both lends salience to the experience and facilitates later recollection of insights derived from the experience. 58
Taken together, classic psychedelics acutely stimulate the HPA axis, early preclinical evidence suggests potential post-acute normalisation and possible enhanced stress resilience. However, the precise clinical relevance for PPD (Table 1) is not yet clear.
Psychedelics and peptide hormones
DOI administration in rats acutely increases oxytocin and prolactin release in a 5-HT2AR-dependent manner. 59 In healthy humans, circulating oxytocin and prolactin are acutely increased by LSD 60 and psilocybin. 61 Ley et al. 62 additionally found that mescaline increased circulating oxytocin levels, although they found no effect of psilocybin. LSD-mediated increases in oxytocin were abrogated by co-administration of the 5-HT2AR antagonist ketanserin, providing human evidence that the effect is largely 5-HT2AR-mediated. 60
Interestingly, Straumann et al. 63 found that the increase in oxytocin induced by LSD, although statistically significant, was minimal compared with that induced by MDMA. Similarly, Holze et al. 64 found that MDMA, but not LSD, significantly increased plasma oxytocin concentrations. These data may help explain why pro-social effects are particularly pronounced during MDMA experiences, whereas they are less intense, though still reliably observed, with LSD.
Psychedelics, peptide hormones and PPD
The effect of psychedelics on oxytocin may have clinically relevant implications for the mother–infant bond, which is often adversely affected in PPD. A circuit involved in maternal behaviour has been delineated in rodents, over which oxytocin has many modulatory functions. 65 The medial preoptic area (MPOA) is the core node of the maternal circuit. MPOA projections stimulate dopaminergic ventral tegmental area (VTA) projections to the nucleus accumbens (NAc) – the classical mesolimbic ‘reward pathway’ – to mediate reward associated with pup interactions. Indeed, mother-offspring interactions are highly rewarding in both rodents and humans. 66 For example, mothers exhibit positron emission tomography (PET) evidence of increased NAc dopamine release in response to their own infant’s affective signals. 67
The rewarding aspects of mothering are likely of great functional significance, as synchronous mothers – those that coordinate their maternal behaviour with infant signals – demonstrate greater NAc activation in response to videos of their infants, while intrusive mothers do not. Furthermore, synchronous mothers demonstrate an increased NAc dopamine response to their infant. 68 Interestingly, plasma oxytocin was corelated with NAc activity in synchronous, but not intrusive, mothers. 69 Oxytocin modulates several areas of the maternal circuit, including the reward pathway. 70 Oxytocin enhances the activity of VTA dopaminergic neurons, 71 and specific disruption of oxytocin signalling in either the VTA or the NAc prevents the onset of maternal behaviours. 72 These data indicate that mesolimbic oxytocin signalling is a key factor in the onset of maternal behaviours.
Animal models of PPD indicate an important role for disruptions in the mesolimbic reward pathway. For example, decreased dendritic length, number of branch points and spine density have been noted in the NAc shell in gestational stress models, 73 which were reversed by citalopram. 73 Rincón-Cortés and Grace 74 demonstrated reduced activity of VTA dopamine neurons in early pup separation and scarcity-adversity models of PPD. They found that VTA dopamine neuron activity was positively correlated with social sniff time, a measure of social motivation. 75 The authors specifically assayed for maternal behaviours in their scarcity-adversity model and found them to be impaired, 76 hypothesising that this may be underpinned by disrupted activity of VTA dopamine neurons.
Women with PPD demonstrate impaired ventral striatal response to positive or rewarding stimuli, 77 including mother–infant interactions. The ventral striatum is a limbic reward structure that encompasses the NAc. This may therefore represent a neural substrate for depressive symptoms such as anhedonia, and for the impaired mother–infant interactions seen in PPD. Given that reduced circulating oxytocin is reported quite consistently in postpartum depression, these deficits may partly relate to impaired oxytocin signalling in the mesolimbic pathway, namely the VTA and the NAc.
Nardou et al. 78 showed that classic psychedelics (psilocybin, LSD), ketamine and ibogaine – durably reopen a critical period for social reward learning in mice. They previously reported similar findings using MDMA and were able to directly link this phenomenon to an oxytocin receptor-dependent increase in oxytocin-mediated neuroplasticity in the NAc. 79 Therefore, it is possible that psychedelics may facilitate social reward learning through mesolimbic oxytocinergic mechanisms. This finding is interesting in the context of PPD, given that existing evidence raises the possibility of dysfunctional mesolimbic oxytocin signalling.
Psychedelics may be able to enhance mother–infant bonding and help to ameliorate feelings of maternal disconnection, partly through upregulation of oxytocin-mediated NAc neuroplasticity. Future studies exploring the impact of classic and non-classic psychedelics on these peptide hormones and their relationship to neural circuitry 80 and how this affects bonding/attachment in PPD would be an interesting prospect.
Psychedelics and neuroplasticity
Extensive preclinical work has identified psychedelics – including classic serotonergic agents, MDMA and ketamine – as potent promoters of rapid and sustained neuroplasticity. This has been hypothesised to underlie their sustained therapeutic effects and has earned them the moniker of psychoplastogens. 81 Several studies corroborate the ability of serotonergic psychedelics to rapidly and robustly induce structural neuroplasticity in cortical neurons. Several signalling pathways appear to play a role in psychedelic-induced neuroplasticity, including TrkB receptors, 5-HT2A receptors, AMPA receptors and downstream mTOR signalling. 82
It is hypothesised that 5-HT2AR activation on cortical neurons enhances glutamate release, which, in turn, activates AMPA receptors, causing secretion of BDNF. Secreted BDNF activates TrkB receptors and downstream mTOR signalling to produce neuroplastic antidepressant effects. 83 Interestingly, it appears to be specifically intracellular 5-HT2ARs that mediate the plasticity-promoting effects of serotonergic psychedelics, which are lipophilic and membrane-permeable. 84
Serotonin, a hydrophilic molecule, does not promote structural neuroplasticity in rat cortical neurons unless manipulations are undertaken to permit serotonin uptake, such as electroporation or SERT overexpression. 84 Hence, the structural modifications of existing psychedelics that enhance membrane permeability may enhance their efficacy. 85 It is hypothesised that the neuroplasticity induced by psychoplastogens underlies their sustained therapeutic efficacy.
In the case of ketamine, its neuroplasticity-promoting effects are required for its sustained antidepressant-like actions. 86 Although this has not been definitively demonstrated in serotonergic psychedelics, existing evidence indicates that antidepressant-like effects co-occur with plasticity-like effects. 44 The finding that BDNF-TrkB signalling is required for both the neuroplastic and antidepressant-like effects of LSD and psilocin constitutes the most direct evidence of the mechanistic role of neuroplasticity. 87
The degree to which psychedelic-induced neuroplasticity maps onto the behavioural effects is not yet fully clear. 88 In support of this is evidence that 5-HT2AR antagonists abolish the head twitch response but not psilocybin- or LSD-induced plasticity or antidepressant-like effects in mice. 89 Moliner et al. 87 provide evidence that psychedelic-induced plasticity and antidepressant-like effects are 5-HT2AR-independent and mediated through directly binding TrkB to allosterically facilitate BDNF activity. However, there is conflicting evidence that pre-treatment with the 5-HT2AR antagonist ketanserin completely blocks psychedelic-induced structural neuroplasticity in vitro. 81
Furthermore, Cameron et al. 90 found that ketanserin pre-treatment completely blocked the antidepressant-like effects of 5-MeO on the forced swim test. The authors reasoned that inter-study discrepancies may relate to incomplete receptor occupancy with lower doses of ketanserin and longer pre-treatment intervals. They additionally showed that complete 5-HT2AR knockout abolished the antidepressant-like effects of psilocybin on the sucrose preference test. 90 Hence, studies conflict as to whether the antidepressant effects of psychedelics, which are presumably underpinned primarily by neuroplasticity, can be disentangled from their 5-HT2AR-mediated hallucinogenic effects. Given that the purest available evidence – genetic 5-HT2AR knockout – supports a role for the 5-HT2AR in antidepressant efficacy, it seems most plausible to interpret conflicting studies in light of the limitations of pharmacological 5-HT2AR antagonists, which may produce incomplete receptor blockade or act at other receptors.
Furthermore, it may be that 5-HT2AR signalling does not necessarily entail hallucinogenic effects. Recently, several non-hallucinogenic psychedelic analogues have been described, which act at 5-HT2ARs and produce antidepressant-like effects but not head twitching. 91 The mechanism is thought to relate to biased agonism, with preferential recruitment of β-arrestin versus G-protein signalling.
Serotonergic psychedelics, neuroplasticity and PPD
Measuring neuroplasticity in humans is limited by indirect methods. During pregnancy, there is a global reduction in brain volume and grey matter volume. 92 This is accompanied with a decrease in global cortical thickness, surface area, gyrification, sulcal depth, sulcal length and an increase in global sulcal width. 93 Ventricle size and cerebrospinal fluid (CSF) volume is also increased during pregnancy. 93 Numerous brain regions experience grey matter loss during pregnancy, and these include, but are not limited to, the posterior cingulate cortex (PCC), precuneus, insula, hippocampus and ventral striatum.
In the postpartum period, these ‘negative’ changes that are observed during pregnancy begin to reverse, with increases in global brain size. 94 This is accompanied by decreased ventricle size and CSF volume. 94 Regional increases in the hippocampus, amygdala and auditory cortex have also been reported. 95 Voxel-wise increases in grey matter volume have been observed in many regions, including the ventromedial prefrontal cortex (vmPFC), nucleus accumbens, parahippocampal gyrus, insula and precuneus. However, some of these changes that occur during pregnancy are present 2 to 6 years after parturition and include the medial prefrontal cortex (mPFC) and precuneus. 96 Structural MRI studies in PPD are inconsistent.
PET imaging of synaptic vesicle protein 2A (SV2A) can characterise changes in synaptic density in patients. 97 A study of the effect of ketamine on SV2A density in humans at 24-h post-administration failed to show a significant increase; however, when stratified into subgroups according to baseline SV2A density, those with low baseline SV2A density showed a significant increase in the dorsolateral prefrontal cortex (dlPFC) and ACC and a trend increase in the hippocampus. 98 This technique has not yet been employed in the study of classic psychedelics in humans and PET in the postpartum period requires temporary cessation of breast feeding. However, Raval et al. 99 found that psilocybin administration in pigs was associated with significantly increased SV2A density in the hippocampus and PFC (prefrontal cortex), which was persistent at 7 days post-administration.
In terms of peripheral proxy markers of neuroplasticity, several studies suggest that women with PPD may have lower circulating levels of BDNF (Table 1). Several psychedelic studies, though limited by relatively small sample sizes, have measured circulating BDNF, however, a recent meta-analysis found no evidence of increased peripheral BDNF associated with psychoplastogen use in humans. 100 In general, the extent to which peripheral BDNF is a valuable marker of central neuroplasticity is unclear. Given the weight of preclinical evidence supporting a role for psychedelics in inducing plasticity, it seems more likely that peripheral BDNF is perhaps too crude a marker to capture subtle central changes.
It remains to be seen whether the ability of psychedelics to induce molecular and cellular changes that promote neuroplasticity and changes in functional connectivity (FC) (discussed below) map onto the subjective and therapeutic effects in PPD, particularly related to rumination, perfectionism, intrusive thoughts, low self-esteem/maternal adequacy and loss of maternal role gratification.
Immune pathways, PPD and serotonergic psychedelics
Serotonergic psychedelics and immunomodulation
Preclinical and clinical studies have reported that serotonergic psychedelics, such as psilocybin, have direct and indirect immunomodulatory properties. 101 This is driven by the fact that serotonergic receptors are widely expressed across different immune cells, including T-cells, B-cells, macrophages and dendritic cells. These receptors have been shown to contribute to the modulation of key immune functions like cell proliferation, differentiation, migration, adhesion, phagocytosis and cytokine production. 102
In vitro studies have found that psychedelics, including the full 5-HT2AR agonist (R)-DOI, psilocybin, LSD, N,N-DMT, or psilocybin-containing mushrooms, modulate various immune functions in stimulated and unstimulated conditions. In studies using a range of immune cell types, 5-HT2AR psychedelics have been found to reduce the expression levels of pro-inflammatory cytokines including TNF-α, IL-6, IL-1β and increase anti-inflammatory cytokines like IL-10 and IL-2. 101
Additionally, serotonergic psychedelics been found to reduce nitric oxide (NO) release in microglia culture, 103 inhibit inflammasome activation and downregulate key inflammation-associated transcription factors. 104
In vivo studies in animal models and in humans have found that with an unstimulated immune system, psilocybin and (R)-DOI increase levels of pro-inflammatory cytokines, namely TNF-α and IL6 accompanied with an increase in plasma corticosterone. 101 Whereas LSD was found to decrease kynurenine levels and the kynurenine/5-HT ratio. 105 Mixed results were found in trials involving human participants. N,N-DMT and 5-MeO-DMT have been reported to increase cortisol levels, and modulate immune markers such as decreased CD3 and CD4 positive T cells, and decrease salivary IL6. 101
Ayahuasca has also been found to decrease CRP levels in humans, which was correlated with lower depressive scores. 106 However, preliminary studies of psilocybin in healthy humans,55,107 and in those with depression, anxiety and cancer suggest that in the context of an unstimulated immune system, serotonergic psychedelics may have subtle effects. 108
In vivo studies have also explored the immunomodulatory effect of psychedelics in the context of a pre-stimulated immune system suggest that psychedelics have an anti-inflammatory effect. Psilocybin was found to decrease expression levels of TNFα, IL-2, IL-6, IL1-β and IBA1 after lipopolysaccharide (LPS) injection, or in models of colitis or middle cerebral arterial occlusion. 101 Psilocybin also reduced stress-induced fear in mice by blocking the migration of inflammatory monocytes to the brain and suppressing neuroimmune signalling linked to fear responses, with parallel findings in human tissue samples indicating potential clinical relevance for anxiety and depression treatments. 109
N,N-DMT and Ayahuasca were reported to decrease production of proinflammatory cytokines TNFα, IL1-β, IL6, TLR4, TLR6 and TLR7, while increasing production of the anti-inflammatory cytokine IL10 and decreasing nitric oxide signalling. 101 (R)-DOI has been reported to decrease expression levels of pro-inflammatory cytokines, including IL6, TNFα, IL9, IL13 and VCAM1, in various models that lead to immune activation. 101
Among the multiple overlapping contributors to PPD (immune, HPA axis, hormonal shifts), emerging evidence also highlights the microbiota-gut-brain (MGB) axis as an exploratory interface between psychedelics and peripheral immune signalling. 110 Perturbations in the gut microbiome are linked to stress-related psychiatric disorders, including perinatal depression, possibly via pro-inflammatory bacterial shifts and reduced anti-inflammatory metabolites. 110 Psychedelics may indirectly modulate inflammation by interacting with the MGB axis, which regulates stress responses, tryptophan metabolism and immune pathways.
Further preclinical and clinical studies are required to assess the molecular and cellular mechanisms underlying the immunomodulatory properties of psychedelics and could incorporate the MGB axis and blood–brain barrier (BBB). 111 Dam-offspring separation in rats is a valuable model to assess PPD in animals and could be used to test whether psychedelics influence the depressive-like phenotype. 112 Interestingly, in a recent preclinical mouse model of peripartum stress, psilocybin-treated dams displayed increased anxiety and greater behavioural impairments. 113
Serotonergic psychedelics, the immune system and PPD
The promising results for the use of psilocybin, 11 5-MeO-DMT 114 and N,N-DMT 115 in the treatment of MDD/TRD and the aforementioned immunomodulatory properties of psychedelics suggest potential benefits for PPD. During pregnancy, the immune system undergoes significant changes, culminating in an acute pro-inflammatory state during labour (Table 2). In some individuals, non-resolution of this state may transition into a chronic low-level inflammatory state, which could have adverse implications for mood and anxiety levels.
Summary of recent findings relating immune pathways to healthy pregnancies and pregnancies associated to postpartum depression.

CRP, C-reactive protein; CSF, cerebrospinal fluid; ILs, interleukins; Kyn, kynurenine; TNF, tumour necrosis factor; uNK, uterine natural killer cells; ↑, increase; ↓ decrease.
As discussed above, serotonergic psychedelic studies in humans, thus far, have suggested subtle effects and it remains to be determined whether the immunomodulatory properties of psychedelic will be of clinical relevance in PPD.
Neural pathways, PPD and serotonergic psychedelics
The default mode network
The default mode network (DMN) is a group of interconnected brain regions with increased temporal coherence at rest. This network is made up of four functional hubs: mPFC, the PCC, precuneus and the angular gyrus. 143 The activity between these brain regions is generally decreased when an individual is paying attention to an external stimulus, such as conducting an externally oriented task. In the absence of an external stimulus, the network defaults to internally oriented processes such as self-reflection, mind wandering, envisioning the future, recalling personal experiences and social interactions. 143 However, the underlying mechanisms are not fully understood. 144 Alterations in DMN activity have been observed in MDD, anxiety, post-traumatic stress disorder, attention deficit hyperactivity disorder and addiction. 145
The DMN is one of the most extensively studied networks in PPD (Table 3), with a trend of altered activity in key DMN hubs, namely the PCC, precuneus and mPFC. 146
Summary table listing key neuroimaging findings from studies comparing women with PPD to healthy postpartum women, highlighting differences in brain function and connectivity associated with PPD.

dmPFC, dorsomedial prefrontal cortex; FC, functional connectivity; HPW, healthy postpartum women; mPFC, medial prefrontal cortex; PCC, posterior cingulate cortex; PPD, postpartum depression; ReHo, regional homogeneity; SampleEN, sample entropy; ↑, increase; ↓ decrease.
Neural pathways and serotonergic psychedelics
Many studies have investigated how psychedelics modulate the DMN. 152 Psilocybin has been shown to robustly decrease FC within the DMN, particularly between the mPFC and the PCC. This decoupling of DMN nodes is associated with ego dissolution and altered states of consciousness. Psilocybin also increases global brain connectivity, leading to a more integrated brain state. 153 Additionally, psilocybin induces spatial desynchronisation cortical activity as measured by global spatial complexity, which indicates how similar the time series of each brain voxel is. 154 Furthermore, psilocybin’s induced loss of subjective self-identity, termed ego dissolution was shown to be predicted by region-dependent alterations in glutamate, specifically in the mPFC and hippocampus. 155
Similarly, LSD decreases within-network connectivity in the DMN, while increasing connectivity between the DMN and other networks, such as the salience network, visual network and sensorimotor network. 156 This increased between-network connectivity is associated with ego dissolution and altered states of consciousness. 157
Ayahuasca disrupts normal DMN function during the psychedelic experience, leading to decreased activity in the mPFC and PCC. Long-term ayahuasca use has been associated with structural brain changes, such as increased thickness in the anterior cingulate cortex (ACC) and decreased thickness in the PCC. 158
Can the neuroimaging findings in MDD be applied to PPD?
From a neuroimaging perspective, MDD and PPD exhibit both shared and distinct neural signatures. 146 The most common network feature of MDD is hyperactivity of the DMN, 159 with a recent study reporting that the frontostriatal salience network is expanded in the cortex of most individuals with MDD. 160 This is further supported by the ability of antidepressants to restore normal DMN activity. Similarly, hyperactivity in the DMN is the most observed PPD-related change, with several studies reporting changes in the PCC, precuneus and mPFC. 146
Baseline resting state network features in those with TRD have been shown to be associated with treatment response. 161 Baseline FC of the DMN, visual and executive networks were associated with early positive response to treatment, while FC of the salience network was associated with late response to treatment.
While similarities between MDD and PPD exist, some features seem to be exclusive to PPD, namely, dampened amygdala responses and corticolimbic activity. 162 Impaired mother–infant interaction and less empathetic behaviour during mother–infant interaction has been linked to disrupted PCC and amygdala connectivity at rest. 150 Additionally, impaired top-down dmPFC-amygdala connectivity is observed when viewing threatening content. 163 Blunted responsivity of the amygdala itself is associated with infant-related hostility. 163 Attenuated amygdala activation is also observed during the emotional processing of words of negative valence. 164
Other parts of the mesolimbic system show dysfunction in mothers with PPD, which may have implications for mother–child interactions. The use of infant cue-based tasks is useful to assess alterations in child–mother interactions. Healthy mothers display increased activity in reward relevant regions including the VTA, ventral striatum and orbitofrontal cortex when shown the smiling face of their own infant versus that of an unknown infant. 165 On the other hand, women with PPD exhibit blunted responses to smiling pictures of their own child, with lower activity in striatal and orbitofrontal regions. 166 Interestingly, women with PPD struggle to recognise happy affect based on their infant’s facial expression, which may explain the above findings. 167 This highlights the maternal–child disconnect in PPD.
Positive words may result in lower activation of the striatum in women with PPD compared to control women. 168 Women with greater PPD severity displayed lower striatal activity. 77 Women with PPD also display altered ventral striatum activation in response to a monetary reward task. In contrast, healthy mothers displayed a sustained response to reward, activation of the ventral striatum quickly attenuated in women with PPD. 169 However, this decrease in ventral striatum activity was not observed in young women with less severe PPD. 170
Together, these studies have led to the proposal of a putative ‘parental caregiving brain network’. 171 Currently, six key regions have been suggested to form part of the parental care giving network: the dmPFC, vmPFC, parahippocampal gyrus, posterior cingulate cortex, amygdala and nucleus accumbens. Studies from healthy mothers have identified that increased FC between the amygdala, nucleus accumbens and dmPFC at rest is associated with positive maternal care. 172 Furthermore, increased FC between the dmPFC and the PCC during the postpartum period is associated with greater cognitive performance when measured during tasks or at rest. 173
Women with PPD display altered connectivity between the PCC, parahippocampal gyrus, amygdala, vmPFC and dmPFC. 150 This suggests that regions involved in maternal–child interactions are also implicated in PPD. 174
Many models have been proposed to explain the actions of psychedelics with examples including the cortico-striatal thalamo-cortical loop (CSTC) model, cortico-claustro-cortical model (CCC) model and the relaxed beliefs under psychedelics (REBUS) model. 175
Psychedelics induce a global change in brain connectivity, so it’s not surprising that many regions involved in the parental caregiving network are modulated by these substances. For instance, psilocybin decreases connectivity between the vmPFC and the right amygdala during face processing. This change was associated with reduced rumination 1-week post-treatment in patients with TRD. 176 A possible explanation is that psilocybin reduces inhibitory input from the vmPFC to the right amygdala during emotional face-viewing, thereby increasing amygdala responsiveness.
Additionally, psilocybin significantly decreases connectivity between the vmPFC and the PCC. It also reduces activity in the mPFC, with the magnitude of this decrease predicting the intensity of subjective effects. 177
While findings indicating changes in the reward system such as the NAc are limited. Preclinical data have shown that DOI has been shown to increase dopamine levels in the NAc of rats. 178 Additionally, data in healthy controls suggest low doses of LSD (13 or 26 μg) increase reward-related brain activity in humans as measured by electroencephalogram (EEG) during a monetary incentive delay task. 179 This impact on the reward system may be useful in treating the unique presentation of PPD.
The parahippocampal gyrus is central in the parental caregiving network and plays an important role as a lower-level region involved in bottom-up signalling in the REBUS model of psychedelics. 180 LSD has been found to reduce FC between the parahippocampal gyrus, the PCC and the retrosplenial cortex. Conversely, it enhances connectivity between the parahippocampal gyrus and both the dmPFC and the dlPFC. 157 Furthermore, LSD increased resting-state connectivity between the vmPFC and both the bilateral caudate and inferior frontal gyrus. On the other hand, decreased connectivity was noted between the vmPFC and the PCC. 157
Notwithstanding the small sample sizes and methodological heterogeneity, collectively these studies suggest that the core symptoms of PPD, some of which overlap with MDD/TRD, can be mapped onto neural correlates. 181 It is therefore reasonable to speculate that psychedelic-induced expansion of global FC, modulation of the DMN 154 and altered activity in the amygdala 176 and maternal caregiving network (Figure 1) are linked to shifts in maternal self-referential processing, psychological flexibility and rumination, which, when combined with psychological support, could enhance emotional bonding with the infant.
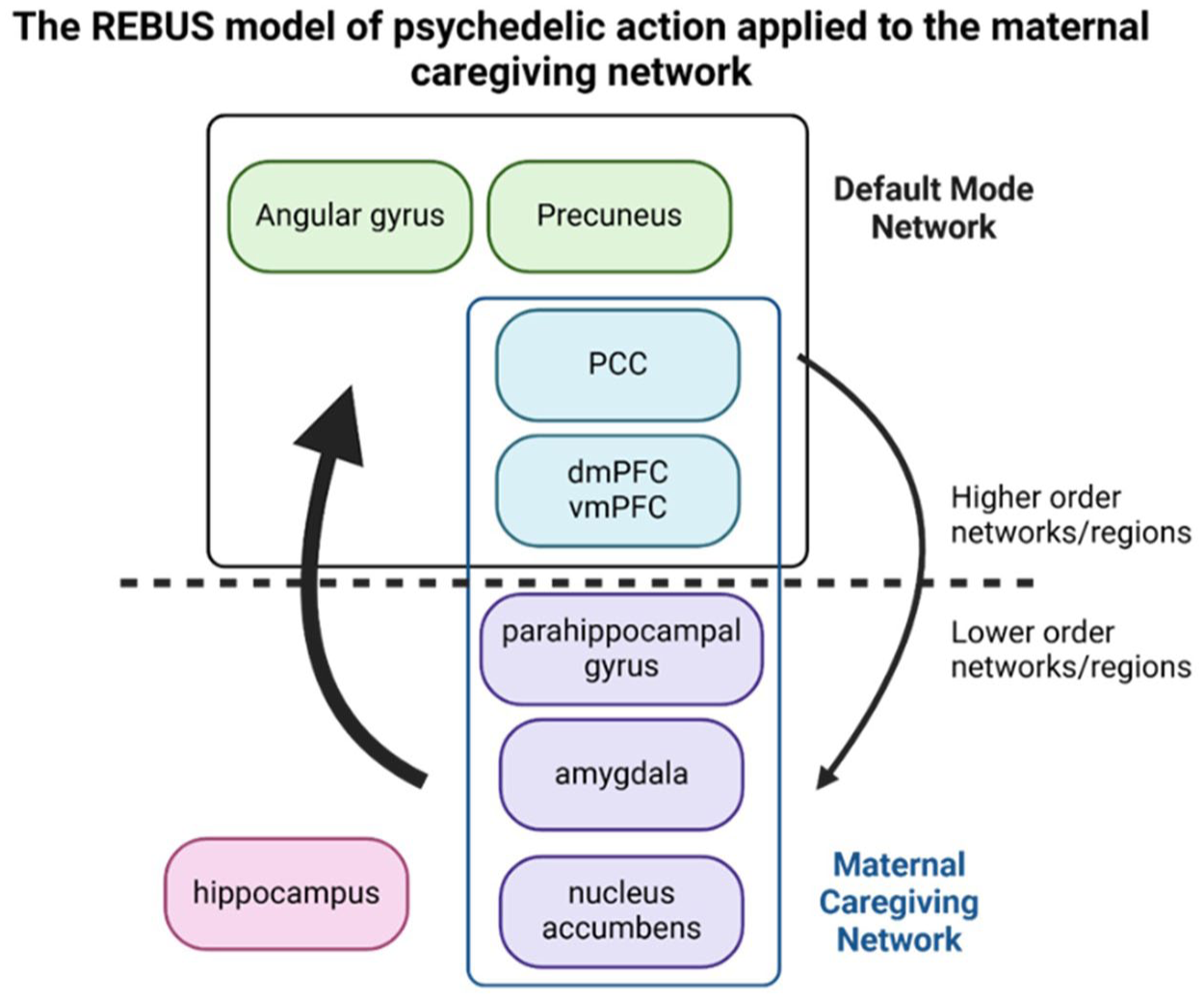
The REBUS model of psychedelic action applied to the maternal caregiving network.
Special considerations for psychedelic therapy in the postpartum period
While psychedelic therapy has potential in PPD, the convergence of neuroendocrine shifts, identity/role transitions and caregiving demands in the postpartum period can represent a heightened window of vulnerability, which necessitates tailored considerations for the potential delivery of psychedelic therapy in PPD.
It is, therefore, even more important to carefully appraise both the general and unique physiological, emotional, psychological and sociocultural/ethical complexities of the mother and her infant over the postpartum period to ensure safety and therapeutic integrity of any potential psychedelic therapy.
Adaptation of psychedelic therapy to the postpartum context with additional supports may minimize destabilisation during this vulnerable period and may mitigate risks and improve outcomes for the mother–infant dyad. Rigorous screening and monitoring in the preparation phase, with particular awareness/vigilance for the risk factors of postpartum psychosis is necessary (see Box 1).
Interim clinical checklist.

Enhanced supports, such as appropriate childcare arrangements, must be considered for PPD patients undergoing psychedelic therapy. Arrangements such as this may be a substantial undertaking depending on the duration and number of the therapeutic sessions.
Notably, the preliminary trials conducted to date in PPD did not include any planned psychotherapeutic intervention before, during, or after dosing. Given the variable and complex experiences induced by psychedelics, which may further intensify emotionally salient experiences such as bonding and attachment, grief, identity reconstruction and unresolved trauma, integrating structured therapeutic support may enhance treatment outcomes. However, the optimal format, frequency and framework required to support and enhance psychedelic treatment outcomes in PPD have yet to be precisely established (Table 4).
Special considerations for psychedelic therapy in the postpartum period.

More broadly, the development of a psychedelic therapy care pathway integrating general adult mental health and perinatal mental health services could reduce risks and optimise therapeutic outcomes.
Discussion
As discussed in this review, psychedelic therapy represents a promising prospect for the treatment of PPD. While awaiting the extension of preliminary findings12,13 into larger clinical trials with longer follow-up periods and robust safety data, several priorities for future research in PPD should be considered.
Advancing and refining personalised approaches to psychedelic therapy represents an important subjective for optimising outcomes in PPD. The complex multimodal mechanisms underpinning psychedelic therapy, the variability in experiences and therapeutic response rates, 195 together with the complexities of PPD, provides an impetus to develop a more comprehensive understanding and personalised paradigms to more efficiently identify those women who may preferentially benefit, while at the same time mitigating the potential risks.
Future studies incorporating a combination of immune/kynurenine pathway, endocrine and neural network profiles, alongside mother–infant markers (e.g., saliva), biometric/wearable and smartphone data, could progress understanding of the variability in therapeutic responses.
Sex steroid and endocrine profiling could help identify women with hormonal hypersensitivity, such as dysregulated HPA axis reactivity or sustained postpartum cortisol elevation. Blunted cortisol responses or elevated placental corticotrophin-releasing hormone may reflect neuroendocrine dysfunctions that are potentially amenable to psychedelic-induced plasticity within stress-related neural circuits. When combined with maternal subjective insights and assessments of the mother–infant developmental trajectory, these approaches could support the stratification of PPD into subgroups more likely to benefit from psychedelic therapy, while also informing clinical decisions about women for whom such treatment may not be appropriate.
A systems approach that encompasses environmental and socioeconomic factors (Figure 2) could deepen the understanding of the complex mechanisms underpinning psychedelic therapy, improve outcomes for both mother and infant and help reduce systemic and psychosocial inequities.

Biopsychosocial factors associated with postpartum depression.
Alongside efforts to advance system-level understanding of psychedelic therapy and associated personalised approaches, the development of novel compounds with more favourable side effect profiles may provide additional therapeutic value in the coming years.
Future PPD-specific research will need to clarify the optimal postpartum dosing window (early vs late), particularly for multiple dosing sessions, and establish compound-specific safety guidelines for breastfeeding alongside more robust consent procedures (Table 4).
More broadly, implementing evidence-based psychedelic therapy for PPD within public health systems will require collaborative care models that link perinatal, primary care and adult mental health services, underpinned by research-embedded centres of expertise. Combined with pragmatic regulatory frameworks and the collection of real-world long-term outcomes, including cost-effectiveness data, these measures could enable national outcomes registries and support the equitable, sustainable integration of psychedelic therapy into public health systems.
Limitations
While this review provides a comprehensive overview, it was not conducted as a formal systematic review and therefore may not have captured all relevant studies. It is important to emphasise that trials in PPD conducted to date remain preliminary. Caution is required when extrapolating results from MDD/TRD trials to PPD.
Conclusions
Psychedelic therapy shows promise as a treatment for PPD, but current evidence remains preliminary. Larger, adequately powered clinical trials are needed to confirm and extend these initial findings and to optimise both safety and efficacy.
