Abstract
Background:
Depression affects approximately 5.7% of adults worldwide, and around one-third of these individuals develop treatment-resistant depression (TRD). Intravenous (IV) ketamine and esketamine (administered IV or intranasally (IN)) are novel treatment options for TRD; however, only IN esketamine currently holds FDA approval.
Objectives:
Compare the acute effectiveness of IV ketamine with esketamine (IV or IN) in adults with TRD.
Design:
Mantel–Haenszel random-effects meta-analysis of head-to-head studies. Response and remission at study end point were co-primary outcomes, expressed as odds ratios (ORs) with 95% confidence intervals (CIs). Subgroup and sensitivity analyses explored the impact of diagnosis, study type, and publication format; heterogeneity was quantified with I2.
Data sources and methods:
MEDLINE, Embase, Cochrane, APA Psycinfo, and Scopus were searched from inception through 19 March 2025. Eligible studies enrolled adults with unipolar or bipolar depression directly comparing IV ketamine with esketamine and reporting response or remission.
Results:
Screening 1089 records identified eight studies (n = 978). Seven observational studies (n = 915) comparing IV ketamine with IN esketamine were included in the meta-analysis, while one randomized controlled trial (RCT) comparing IV formulations was summarized qualitatively. Pooled response from six studies gave OR = 1.26 (95% CI, 0.92–1.71; p = 0.15) and remission from seven studies gave OR = 1.31 (95% CI, 0.93–1.86; p = 0.12), both nonsignificantly favoring IV ketamine with negligible heterogeneity (I2 = 0%). Sensitivity analyses excluding bipolar depression or abstract-only reports yielded similar effect estimates, reinforcing the robustness of the findings. Evidence across three studies for faster onset with IV ketamine ranged from significant in one study to modest trends in two.
Conclusion:
Based on the currently available comparative evidence, which is almost entirely observational, IV ketamine and IN esketamine show comparable acute response and remission rates, though IV ketamine may act faster. Large head-to-head RCTs are needed to confirm these findings.
Trial registration:
The study protocol was prospectively registered on the Open Science Framework (OSF) at https://osf.io/5jzev.
Plain language summary
About one in three people with depression still feel unwell after trying at least two antidepressants, a condition called treatment-resistant depression (TRD). Two recent treatment options include intravenous (IV) ketamine and esketamine, but it remains unclear which is more effective.
We searched five large medical databases up to March 2025 and found eight studies comparing IV ketamine with esketamine for TRD. Seven studies, covering 915 participants, tested IV ketamine against intranasal (IN) esketamine; one smaller trial (n=63) compared both drugs by IV drip.
When the seven similar studies were analyzed, IV ketamine and IN esketamine both helped people feel better at about the same rate, and neither proved clearly superior; the odds of improvement slightly favored IV ketamine, but the difference was not statistically significant. Three studies reported that mood lifted sooner with IV ketamine, sometimes after only two or three infusions, whereas the IN esketamine spray usually required more sessions. Both treatments caused short-lived side effects such as temporary rises in blood pressure or feelings of disconnection, and these effects were generally mild to moderate and rarely led to stopping treatment. Because most included studies were not randomized, the certainty of these comparisons is limited.
During the first four to eight sessions, IV ketamine and IN esketamine appear equally likely to ease depression symptoms. IV infusions may work faster, but the IN spray avoids needles and is FDA approved for TRD. The choice should consider how quickly relief is needed, comfort with IV access, clinic resources, and insurance coverage. Larger, randomized, high-quality studies that track patients for long-term are required to confirm these findings and to show how long the benefits last.
Keywords
Introduction
Depression is a leading cause of disability affecting an estimated 332 million people worldwide. 1 More than 30% of these individuals with major depressive disorder (MDD) develop treatment‑resistant depression (TRD), commonly defined as persistent symptoms despite at least two adequate antidepressant trials. 2 The societal impact is substantial; in the United States alone, the incremental annual economic burden of MDD rose to $326 billion in 2019, 3 and TRD accounts for a disproportionate share of direct and indirect costs. Effective management of TRD usually requires the use of advanced strategies, including neuromodulation and rapid-acting antidepressants.
Ketamine is a racemic N‑methyl‑D‑aspartate (NMDA) receptor antagonist originally developed as an anesthetic. 4 Given intravenously, it has demonstrated rapid and robust antidepressant effects in individuals with TRD. 5 While its modern application in psychiatry is well established, 6 ketamine’s exploration for psychiatric indications dates back to 1973, when Khorramzadeh and Lotfy reported that IV ketamine, used as an “abreactive” aid to psychotherapy in psychiatric inpatients in Iran, facilitated therapy and symptom relief in most participants. 7 In 2000, a pilot double-blind crossover trial by Berman et al. showed that a single sub-anesthetic intravenous (IV) ketamine infusion led to clinically meaningful mood improvement within 72 h; this study was the first randomized controlled trial (RCT) to demonstrate ketamine’s rapid antidepressant effects in MDD, establishing a crucial proof of concept for subsequent clinical investigation. 8 Since then, the evidence supporting ketamine’s antidepressant effect has grown substantially.6,9 This increasing interest in ketamine spurred the development of esketamine, its S-enantiomer with a higher affinity for the NMDA receptor, 10 and in 2019, the FDA approved SPRAVATO™ (esketamine) nasal spray, classified as a Schedule III controlled substance, for use in combination with an oral antidepressant to treat TRD in adults. 11 Although intranasal (IN) esketamine is FDA-approved for TRD, and IV racemic ketamine remains an off-label treatment; IV ketamine is still extensively utilized in specialized clinical settings due to compelling evidence of its rapid antidepressant efficacy.12–16
While both agents are believed to enhance glutamatergic signaling and neuroplasticity,17,18 they differ in administration route, pharmacokinetics, and regulatory status. 9 A 2022 meta-analysis by Bahji et al., 19 which included 36 RCTs (n = 2903), found that IV ketamine was associated with higher risk ratios for clinical response (RR = 3.01; 95% confidence interval (CI), 2.24–4.03) and remission (RR = 3.78; 95% CI, 2.44–5.78) compared to IN esketamine (response: RR = 1.20; 95% CI, 0.96–1.49; remission: RR = 1.28; 95% CI, 1.11–1.47). However, none of the RCTs compared IV ketamine to IN esketamine in a head-to-head comparison.
We aimed to conduct a comprehensive systematic review and meta-analysis to compare the effectiveness of IV racemic ketamine with esketamine (IN or IV). Our review focused on studies that include both an IV ketamine group and an esketamine group within the same study, enabling direct within-study comparison of the two treatments.
Methods
This systematic review and meta-analysis were conducted and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines. 20 The study protocol was registered with the Open Science Framework (OSF) 21 prior to the start of the review (https://osf.io/5jzev).
Search strategy and sources
A comprehensive search of MEDLINE (via PubMed), Embase, Cochrane CENTRAL, APA Psycinfo, and Scopus was conducted from database inception to 19 March 2025, without restrictions on publication date or language. An experienced medical librarian (GW) developed and executed the search strategy in collaboration with the study’s authors. The search strategy combined controlled vocabulary (e.g., MeSH terms) and keywords for intravenous ketamine, esketamine, and depressive disorders. In addition, the reference lists of potentially eligible articles were manually reviewed to identify any additional relevant studies. The detailed search strategy for each database is provided in the Supplemental Material.
Study selection and eligibility criteria
Studies were eligible for inclusion if they directly compared IV racemic ketamine with esketamine (IV or IN) in adults (⩾18 years) diagnosed with unipolar or bipolar depression. Eligible studies were required to report at least one of the primary outcomes of remission rate or response rate. Secondary outcomes of interest included time to remission, number of treatments required, adverse effects, dropout rates, relapse rates, or quality-of-life measures. Eligible study designs included RCTs and observational studies (e.g., cohort studies). Conference abstracts that provided sufficient data for analysis were included. Studies were excluded if they were nonhuman studies, case reports, case series, reviews, commentaries, or editorials; did not include both ketamine and esketamine groups; or failed to report relevant outcome data. Three reviewers (AE, IT, and BS) independently screened the titles and abstracts of all identified records using Covidence systematic review software. 22 The same three reviewers then assessed the full texts of potentially eligible articles against the inclusion criteria. Any disagreements during the screening and selection process were resolved through discussion and consensus.
Data extraction
Two reviewers (AE and IT) independently extracted data from all included studies using a predefined and standardized data extraction form within Covidence. The following data points were systematically extracted: study characteristics, including first author, publication year, study design, and country; participant characteristics, including the total number of participants in each treatment arm, specific psychiatric diagnosis, mean age, and percentage of female participants; intervention and comparison details, including drug formulation, dosage, frequency, and duration of the treatment course; and outcome data, including the number of participants achieving remission and response in each arm, the specific rating scales used to define outcomes and the time point of assessment. Any discrepancies in extracted data were resolved through discussion and adjudication by a third reviewer (BS). Where data were missing or reported in a way that precluded meta-analysis, attempts were made to contact the corresponding authors for clarification and additional information.
Methodological quality and risk of bias assessment
The methodological quality and risk of bias of included studies were independently assessed by two reviewers (AE and IT). For any included RCTs, the assessment involved the utilization of the Cochrane Risk of Bias tool (RoB). 23 This tool evaluates seven key domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other potential sources of bias. For each domain, a judgment of “low risk,” “high risk,” or “unclear risk” of bias was assigned. A “high risk” judgment was given if the study protocol raised significant concerns about bias, “unclear risk” was assigned if there was insufficient information to make a judgment, and “low risk” was assigned if the protocol was adequate to minimize bias. For nonrandomized observational studies, methodological quality was assessed using the Newcastle-Ottawa Scale (NOS), 24 which evaluates studies on three dimensions: the selection of the study groups, the comparability of the groups, and the ascertainment of the exposure or outcome of interest. The NOS awards up to nine stars in total, with studies receiving seven to nine stars classified as low risk of bias, those scoring four to six stars as moderate risk of bias, and those scoring fewer than four stars as high risk of bias.
Data synthesis and statistical analysis
All statistical analyses were performed using Review Manager (RevMan Version 5.4) 25 The primary outcomes, remission and response, were treated as dichotomous variables. We calculated pooled odds ratios (ORs) with their corresponding 95% CIs using the Mantel–Haenszel statistical method. A random-effects model was selected for all analyses to account for clinical and methodological diversity across the studies. Sensitivity analyses were performed when necessary to evaluate the influence of individual studies or methodological choices on the overall results. Statistical heterogeneity was evaluated using the I2 statistic, with interpretation guided by the Cochrane Handbook 26 : I2 values of 0%–30% suggest heterogeneity might not be important, 30%–50% may indicate moderate heterogeneity, 50%–75% may indicate substantial heterogeneity, and 75%–100% suggest considerable heterogeneity. We also reported the Tau2 statistic, which estimates the between-study variance, and the chi-squared (χ2) statistic with its degrees of freedom, a p-value < 0.10 on the chi-squared test was considered to indicate statistically significant heterogeneity. We could not conduct a meta-analysis of continuous outcomes (e.g., quantitative change in depression scores) because the standard deviations of the change scores were not consistently available or derivable, and the measurement instruments were not consistent across study arms.
Publication bias
We planned to assess small-study and publication bias by visual inspection of funnel plots 26 for each primary outcome, and to apply Egger’s regression test 27 to assess funnel plot asymmetry when at least ten studies were available per outcome. 28
Results
Study selection
The systematic literature search identified 2248 records from databases. After removing 1159 duplicates, 1089 titles and abstracts were screened for eligibility. This process led to the retrieval of 20 full-text articles for detailed assessment. Of those, 12 were excluded for not meeting inclusion criteria, resulting in a final cohort of eight studies16,29–35 for qualitative synthesis. Of these eight studies, seven were included in the quantitative meta-analysis.16,30–35 Refer to Figure 1.

Flow diagram.
Characteristics of included studies
A total of eight studies met the inclusion criteria, providing data on 978 patients. The search strategy was deliberately broad, accepting both randomized and nonrandomized studies that compared IV ketamine with either IV or IN esketamine. The final set comprised one RCT and seven observational studies. The only available RCT (n = 63) compared IV ketamine with IV esketamine for TRD. 29 Because this design differed substantially from the IV versus IN comparison examined in the other included studies, the RCT was retained for qualitative description but excluded from the quantitative synthesis in order to maintain a consistent analytic framework. The seven observational studies (n = 915) compared IV ketamine with IN esketamine; two of these reports were available only as conference abstracts (n = 234)30,34 but contributed sufficient data on at least one primary outcome, such as response or remission. Across the seven studies that reported sex distribution, female participants represented approximately 56.7% of the sample. Based on six studies reporting group-specific mean ages and standard deviations, the pooled mean age for participants receiving IV ketamine was 49.4 ± 15.3 years, whereas for those receiving esketamine was 47.4 ± 14.3 years. Further details on study methodology and participant characteristics are provided in Table 1.
Characteristics of the included studies.

Abstract only.
AE, Adverse event; BD, bipolar disorder; BD-I, bipolar disorder type I; BD-II, bipolar disorder type II; BDI, Beck Depression Inventory; CGI-S/I, Clinical Global Impression Severity and Improvement scales; ESKET, esketamine; KET, ketamine; MADRS, Montgomery Asberg Depression Rating Scale; MDD, Major Depressive Disorder; N/A, not available; PHQ-9, Patient Health Questionnaire-9; QIDS-SR 16, 16-item Quick Inventory of Depressive Symptomatology Self Report; TRBD, Treatment-Refractory Bipolar Disorder; TRD, Treatment-Resistant Depression.
The included patient populations varied, though most studies evaluated adults with TRD. Santucci et al. 35 focused exclusively on a cohort with “refractory bipolar depression.” Nikayin et al. 31 included patients from a tertiary care setting treated for depression more generally; the authors suggested that this clinical environment likely managed a more severely ill and treatment-resistant patient population, although a “formal” TRD diagnosis was not required for inclusion. 31 There was also some variability in the definition of TRD across studies. For example, the study by Correia-Melo et al. 29 used different criteria, defining TRD as a failure to respond to at least one antidepressant at a sufficient dose for 12 weeks. In contrast, the most commonly used definition across the other studies required a failure to respond to at least two adequate trials of antidepressants, typically administered for 4–6 weeks at a sufficient dosage.
Meta-analysis of response and remission rates
Six observational studies that included both IV ketamine and IN esketamine treatment groups provided response data for the meta-analysis (Figure 2). Using a random-effects model, the synthesis produced a pooled odds ratio (OR) of 1.26 (95% CI, 0.92–1.71). The overall effect test yielded Z = 1.46 (p = 0.15), indicating a numerical advantage for IV ketamine that does not reach statistical significance. Between-study variability was minimal (Tau2 = 0.00; χ2 = 4.58, df = 5, p = 0.47; I2 = 0%), suggesting that the observed dispersion is compatible with random sampling error rather than true heterogeneity.

Forest plot of pooled odds ratios for acute response to IV ketamine versus IN esketamine.
Seven observational studies reported remission outcomes and were synthesized in Figure 3. The pooled OR was 1.31 (95% CI, 0.93–1.86) in favor of IV ketamine, with Z = 1.54 (p = 0.12). Heterogeneity again remained negligible (Tau2 = 0.00; χ2 = 2.85, df = 6, p = = 0.83; I2 = 0%).

Forest plot of pooled odds ratios for remission following IV ketamine versus IN esketamine.
Overall, the two analyses point in the same direction, showing an aligned though statistically nonsignificant tendency toward higher response and remission rates with IV ketamine compared with IN esketamine. Since these are observational cohorts, the pooled ORs should be interpreted solely as associations. Larger, well-powered RCTs will be required to determine whether this emerging signal translates into a definitive clinical benefit.
Time to response and remission
Three observational studies16,31,33 evaluated the time or number of treatment sessions required to achieve clinical response or remission, finding that IV ketamine reached these end points with fewer sessions than IN esketamine. Notably, Singh et al. 16 provided the most in-depth analysis, reporting both response and remission time metrics: the median number of treatments to achieve response was 2.0 (Interquartile range [IQR] 1.0–3.0) for IV ketamine versus 4 (IQR 3.0–6.0) for IN esketamine, and to achieve remission was 2.0 (IQR 1.75–3.0) versus 7.0 (IQR 5.3–8.0) treatments, respectively (p ⩽ 0.01); time to remission was significantly shorter with IV ketamine (Hazard Ratio (HR) = 5.0; 95% CI, 1.0–24.3; p = 0.02). Gutierrez et al. 33 found that 47% of IV ketamine patients responded by week 1 compared with 7% of IN esketamine patients. In the Yale cohort (Nikayin et al.), 31 IV ketamine produced greater symptom reduction over the first six treatments, with an adjusted mean difference in MADRS score reduction of 2.49 points (95% CI, 0.01–4.98; p < 0.05).
Sensitivity analysis
Excluding the single bipolar depression study produced only modest shifts in both pooled estimates. For response, the OR increased from 1.26 (95% CI, 0.92–1.71; p = 0.15) to 1.32 (0.96–1.82; p = 0.08). For remission, the pooled effect moved from 1.31 (0.93–1.86; p = 0.12) to 1.35 (0.95–1.92; p = 0.10). In each case, the confidence interval continued to cross 1, the p values remained above the conventional 0.05 threshold, and heterogeneity stayed negligible (I2 = 0%). Although the shifts are small and all point in the same direction, excluding the bipolar cohort does not meaningfully change our overall conclusions.
Excluding studies that were available only in abstract form resulted in minor changes to the overall estimates. For response, the OR changed from 1.26 (95% CI, 0.92–1.71, p = 0.15, I2 = 0%) to 1.30 (0.93–1.83, p = 0.13, I2 = 3%). For remission, the pooled effect moved from 1.31 (0.93–1.86, p = 0.12, I2 = 0%) to 1.35 (0.90–2.02, p = 0.14, I2 = 0%). In both outcomes, the direction of effect remained consistent, the confidence intervals still crossed 1, and heterogeneity stayed negligible, indicating that omitting abstract-only records does not materially alter the overall conclusion.
Methodological quality and risk of bias assessment
The RCT by Correia-Melo et al. 29 was assessed using the Cochrane RoB 23 tool and was generally of good quality, though domains concerning allocation concealment and other potential sources of bias were rated as unclear risk. The five observational cohort studies with full publications were assessed16,32–35 using the NOS 24 and were of moderate-to-high quality. Four of the studies were determined to have a low risk of bias.16,31,33,35 One study was found to have a moderate risk of bias, 32 primarily because the two treatment arms consisted of distinct cohorts from different countries and used different primary outcome scales. Full details of these quality assessments are available in Figure 4 and Table 2.
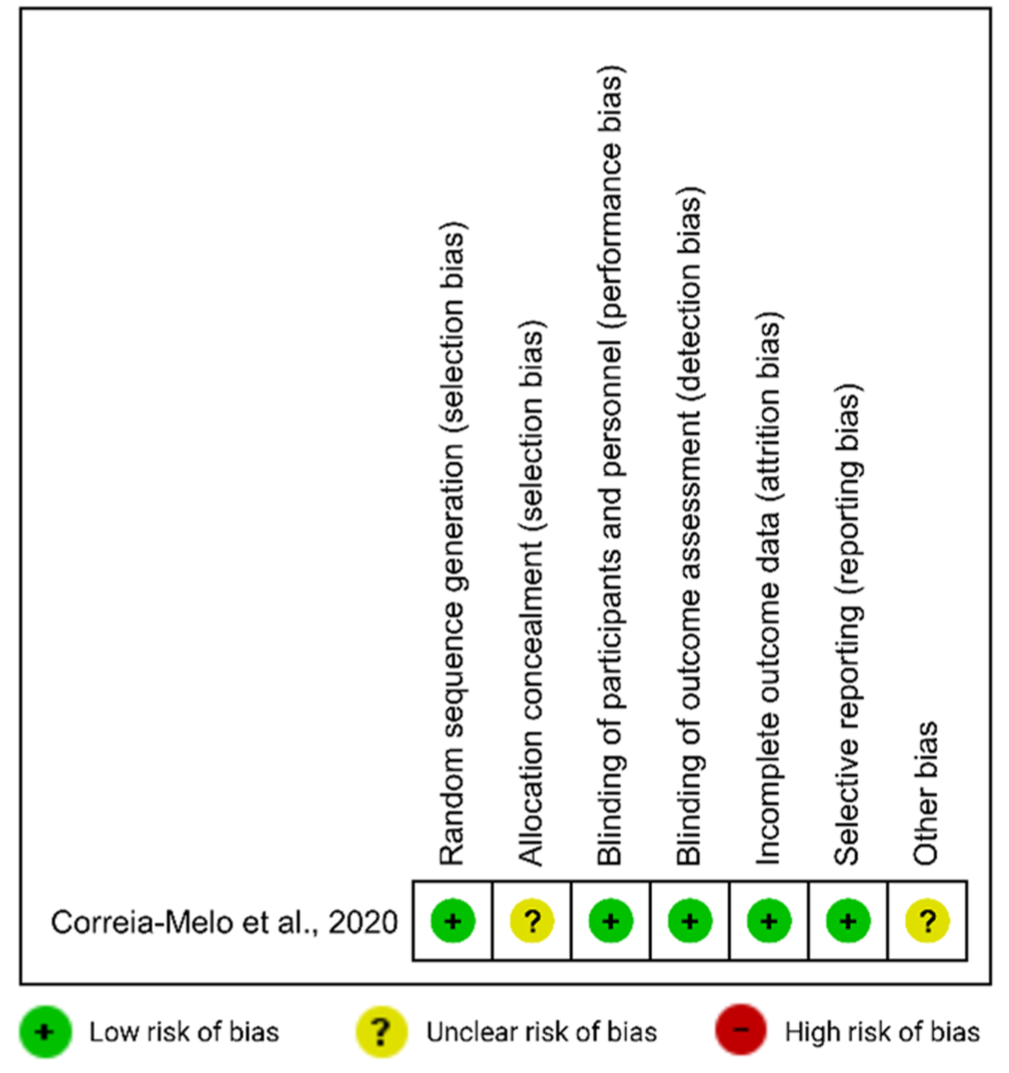
Cochrane risk of bias assessment for the included randomized trial.
Methodological quality assessment using the Newcastle-Ottawa Scale.

A formal risk of bias assessment could not be conducted for the two studies by Davila et al. and Hutchison et al., as they were only available in abstract form. The risk of bias for these studies is therefore considered unclear due to insufficient methodological detail.
Funnel plots for response and remission (Figures 5 and 6) did not show clear asymmetry on visual inspection; with fewer than 10 studies per outcome, formal asymmetry tests, including Egger’s regression, were not performed.
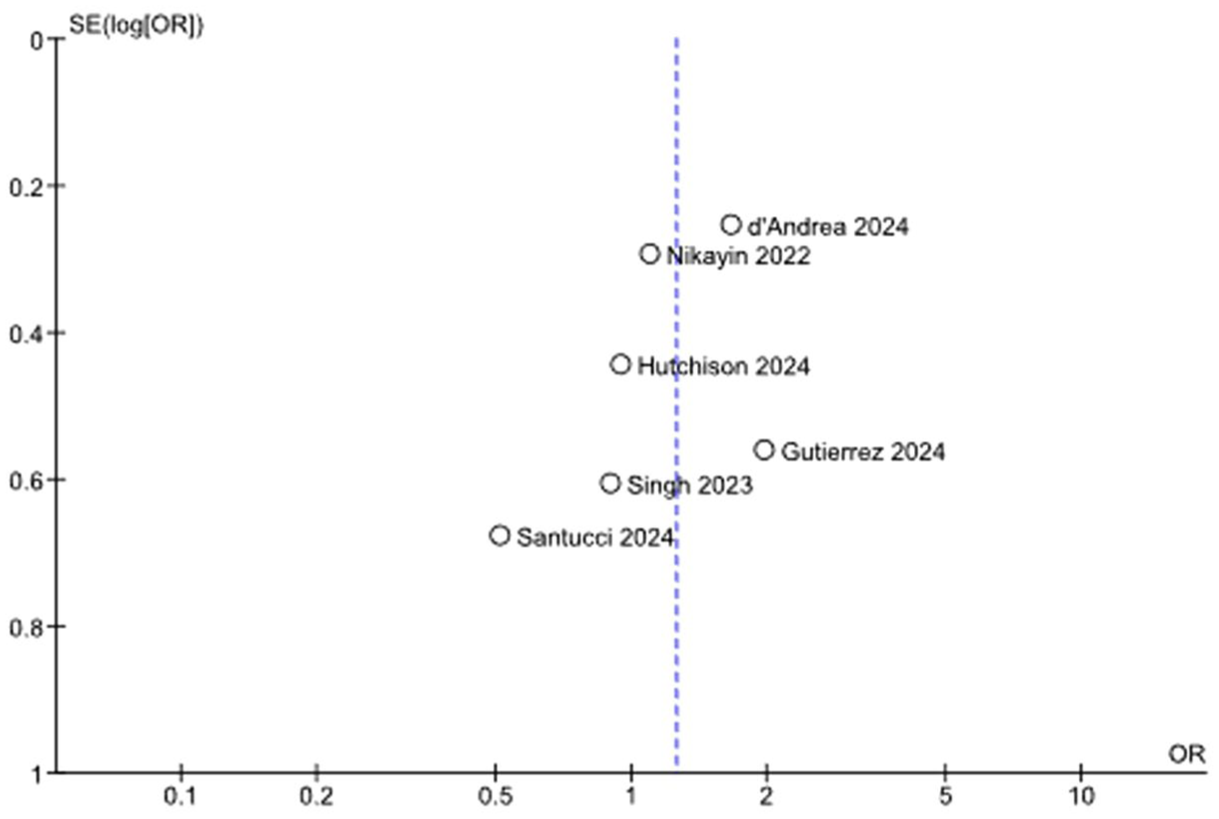
Funnel plot for response (IV ketamine vs IN esketamine).

Funnel plot for remission (IV ketamine vs IN esketamine).
Adverse effects and tolerability
Cardiovascular
Across the included studies of TRD, IV ketamine produced larger acute hemodynamic shifts than IN esketamine. In the study by Singh et al., 16 mean systolic blood pressure rose 8.66 mm Hg at 40 min with IV ketamine versus 0.97 mm Hg with IN esketamine, and 5.88 versus 1.02 mm Hg by the monitoring end point. Heart rate increased by 1.66 bpm at 40 min and by 0.42 bpm at the monitoring end point with IV ketamine; with IN esketamine, heart rate decreased by 3.12 bpm at 40 min and by 2.80 bpm at the monitoring end point. Diastolic blood pressure changes were similar between groups at both time points, and no patients discontinued during monitored cardiac observation. In a larger pooled comparison, d’Andrea et al. 32 reported a higher frequency of treatment-emergent hypertension with IV ketamine, 45% (78 of 171), than with IN esketamine, 9.2% (13 of 140). Gutierrez et al. 33 reported session-level cardiovascular complaints: elevated heart rate in 5.05% with IV ketamine versus 3.05% with IN esketamine, and increased blood pressure in 14.14% versus 25.19%, respectively.
Dissociation and other side effects
In d’Andrea et al., 32 dissociative adverse events occurred in 57.5% with IV ketamine (99 of 171) versus 34% with IN esketamine (48 of 140), with events described as transient. Singh et al. 16 assessed dissociation using the Clinician-Administered Dissociative States Scale (CADSS) 41 during monitoring and found no significant difference between modalities (p = 0.06). Gutierrez et al. 33 quantified depersonalization using the Clinician-Administered Dissociative States Scale 6-item depersonalization subscale (CADSS-6): 42 34.33% of IV ketamine patients and 58.02% of IN esketamine patients experienced depersonalization during treatment, with mean first-treatment session scores of 1.12 and 1.44, respectively.
Overall tolerability
AE-related discontinuations were uncommon and not significantly different between modalities in d’Andrea et al. 32 15.2% with IV ketamine (26 of 171) versus 8.5% with IN esketamine (12 of 140). No serious AE was reported in either group, and events resolved after the peak exposure period. Gutierrez et al. 33 found that 88.89% receiving IV ketamine and 81.68% receiving IN esketamine reported at least one side effect during the acute series, with an average of 2.39 and 2.34 side effects per patient, respectively; side effects were described as mild and transient, subsiding within 15–20 min after administration. In bipolar depression, Santucci et al. 35 observed no mania or hypomania during the acute twice-weekly induction series. During maintenance, 13 of 45 patients 28.9% experienced hypomanic or manic symptoms, totaling 16 events over 518 patient-months (43.1 patient-years), or approximately 1 event per 2.7 patient-years. Six of the 16 events occurred during IV ketamine exposure across 270 patient-months, and 10 occurred during esketamine exposure across 247 patient-months. One event was severe and required hospitalization.
Discussion
IV Ketamine and IN esketamine are increasingly used for depression; however, no RCT has directly compared their effectiveness. This comprehensive systematic review and meta-analysis of observational data found no statistically significant difference in acute response and remission rates between IV ketamine and IN esketamine, although IV ketamine may be associated with a faster onset of effect. IV ketamine is typically administered 2–3 times per week for up to 3–6 treatments during the acute phase, 12 whereas IN esketamine is usually given twice weekly for 4 weeks. 43 Pharmacokinetic differences between these interventions may contribute to the faster onset of response observed with IV ketamine. However, by the end of the acute or induction phase, response and remission rates, although slightly favoring IV ketamine, were not significantly different from those with IN esketamine. It is also important to note that some included studies had smaller sample sizes, which could have affected the results; larger studies could help build on this evidence.
The findings from our systematic review and meta-analysis differ from those reported by Bahji et al., 19 who found higher response and remission rates with IV ketamine compared to IN esketamine. Several factors may explain these differences. In the study by Bahji et al., none of the included RCTs directly compared IV ketamine with IN esketamine; thus, their results were based on indirect comparisons. In contrast, our meta-analysis included only studies with direct, head-to-head comparisons, which could have influenced the pooled estimates. Additionally, there were differences in study populations. The only RCT that compared IV ketamine with IV esketamine was not included in our meta-analysis due to methodological differences related to drug delivery routes.
Data from studies included in our systematic review, particularly Singh et al., 16 indicate that individuals with TRD treated with IV ketamine tend to reach remission in fewer sessions than those treated with IN esketamine. Although a few other studies point to a similar pattern,31,33 the question of how quickly each intervention produces remission or response remains underexplored. Given the observational design, limited number of relevant studies, and small sample sizes, these findings must be interpreted cautiously. The existing evidence, though suggestive of a potential efficacy advantage for IV ketamine, remains insufficient to draw definitive conclusions.
Several pharmacological factors may underlie the numerical trend favoring IV ketamine. 44 IV racemic ketamine provides near-complete, immediate bioavailability with rapid CNS penetration and a brisk glutamatergic surge, proposed to initiate neuroplastic cascades relevant to antidepressant effects. 45 By contrast, IN esketamine is absorbed more slowly through the nasal mucosa with a mean absolute bioavailability around 54% and a formulation peak at approximately 40 min, yielding generally milder early effects.46,47 Absorption after IN delivery varies with nasal physiology, administered volume, and spray technique, which can widen between-patient exposure differences. 48 Dosing also differs (weight-based for IV ketamine vs fixed 56 or 84 mg for IN esketamine), while the needle-free route confers practical advantages and avoids IV-access complications. Finally, the higher NMDA receptor affinity of the S-enantiomer may partially offset its lower systemic exposure, thereby helping preserve clinical potency of esketamine. 10
Several non-pharmacologic factors may also contribute to differences in outcomes between IV ketamine and IN esketamine, including patient expectancy, access, and side-effect profiles. The observation that IV ketamine appears to produce a faster antidepressant effect than IN esketamine may partly reflect an expectation-driven effect built into the treatment protocols: IV ketamine is typically administered as 3–6 treatments in the acute phase, whereas IN esketamine is recommended for 8. By the time patients with TRD are offered these treatments, they often have high hopes for improvement and know that continuation to maintenance treatment depends on responding during the acute phase. This anticipation may increase the likelihood of perceiving benefit by the end of the shorter IV ketamine course. Furthermore, receiving IV treatment under continuous cardiovascular monitoring for about 40 min may convey a greater sense of medical seriousness and advanced technology compared to IN delivery, which typically involves self-administration followed by observation from medical staff.
The current regulatory environment also contributes to the differences between IV ketamine and IN esketamine, reinforcing disparities in accessibility, clinical adoption, and oversight. IN esketamine, as an FDA-approved treatment for TRD, is subject to the Risk Evaluation and Mitigation Strategy (REMS) program, 11 which mandates certified clinics, witnessed dosing, and at least 2 h of post-dose observation. While both formulations require baseline cardiovascular screening, IV ketamine services follow site-specific clinical protocols; however, no federal REMS dictates post-infusion stay, and many centers discharge patients within an hour once vital signs stabilize. Economic modeling in the United States by Brendle et al. 49 found IN esketamine nasal spray unlikely to be cost-effective compared with IV ketamine from a health system perspective, although patient out-of-pocket costs may be lower due to insurance coverage or manufacturer assistance.
Differences in treatment access policies and variability in insurance coverage due to FDA approval status may invisibly select distinct patient populations, introducing a hidden socioeconomic confounder that could influence study outcomes. Socioeconomic status itself has been shown to influence depression treatment outcomes. 50 Socioeconomic factors, such as lower educational attainment and unemployment, were associated with slower and less symptom improvement across treatments, even after controlling for age, sex, and treatment type. 50 Some experts therefore caution that observed efficacy gaps between IV ketamine and IN esketamine may partly reflect selection bias rather than pharmacologic differences.51,52 Others argue that the inherent pharmacokinetic advantages of IV administration, including complete bioavailability 53 and high peak NMDA receptor occupancy 54 with potentially faster time to remission, 16 may outweigh the socioeconomic confounding.
Definitive answers will depend on well-designed trials that control for social determinants of health and directly compare these two delivery routes; notably, a large Patient-Centered Outcomes Research Institute (PCORI) funded, multisite RCT (ClinicalTrials.gov identifier NCT06713616) is now recruiting to compare IV ketamine with IN esketamine in adults with TRD. 55 Such adequately powered, methodologically rigorous head-to-head RCTs are needed to determine whether the observed trend favoring IV ketamine reflects a true efficacy advantage. A recently published small observational study of 38 adults with TRD reported longer intervals between maintenance treatments with IV ketamine than with IN esketamine (mean 18.9 vs 10.8 days; incidence rate ratio 1.8, 95% CI, 1.3–2.3; p < 0.001), which could indicate greater durability of antidepressant effect. 56 However, this single-site, nonrandomized observational cohort is vulnerable to selection bias and confounding. Building on this signal, future work should prioritize long-term comparative effectiveness and safety with standardized follow-up, durability, and relapse-prevention outcomes, and comparative tolerability; incorporate comprehensive patient-reported outcomes and economic end points; and develop and validate predictive biomarkers to guide individualized selection between IV ketamine and IN esketamine.
In the absence of definitive head-to-head evidence, treatment choice should be individualized. IV ketamine may be selected when rapid symptom relief is needed or when a short-term bridge is planned and infusion monitoring is available and affordable. IN esketamine may be preferred when care within an FDA-approved, REMS-certified pathway and favorable insurance coverage are priorities, or when avoiding IV cannulation aligns with patient preference and clinic workflow. Both options remain reasonable after failure of at least two adequate antidepressant trials, and decisions should account for urgency, access, comorbidities, and patient goals.
Strengths and limitations
This meta-analysis included only studies that reported outcomes for both IV ketamine and IN esketamine groups, which minimizes confounding from cross-study heterogeneity. The consistent direction of effect for remission across cohorts, along with an overall trend favoring IV ketamine for response despite some individual study variability in effect direction, coupled with negligible statistical heterogeneity for both outcomes (I2 = 0), strengthens its internal validity. However, a primary limitation is the predominance of observational studies in our analysis; only one included study was an RCT, 29 and it compared IV esketamine to IV ketamine, not the IN formulation used elsewhere. This RCT was excluded from the meta-analysis portion. In addition, our analysis incorporated data from two abstracts with very limited methodological information and unclear risk of bias. Outcome measurement instruments varied across studies, including both clinician-rated and self-reported scales, and in one study, different instruments were used across arms; this heterogeneity precluded pooling continuous change scores and may introduce residual measurement bias despite synthesizing aligned dichotomous response and remission definitions and observing negligible statistical heterogeneity. Observational studies are susceptible to selection bias, unmeasured confounding (e.g., variability in concomitant medications, illness severity, prior treatments, which are intrinsic challenges when studying these interventions in real-world settings), and potentially incomplete adverse event reporting. The current evidence base, therefore, may lack sufficient collective statistical power to definitively detect modest but clinically relevant differences. Furthermore, follow-up was rarely extended beyond the acute phase, and the small number of studies precluded formal evaluation of publication bias.
Conclusion
Existing comparative evidence, which is almost entirely observational, suggests no significant difference between IV ketamine and IN esketamine for depression in short-term efficacy, though there is a slight numerical trend favoring IV ketamine in remission and response. Both formulations appear beneficial for adults with TRD in the acute/induction phase. Bioavailability, regulatory burden, and invasiveness give each treatment a distinct profile of feasibility, cost, and patient acceptability. Given the risk of selection bias and unmeasured confounding inherent to observational cohorts, these estimates should be interpreted as associations and viewed as hypothesis-generating. Until well-powered randomized trials report definitive evidence, clinicians should weigh IV ketamine’s potential for a faster onset against the convenience and structured safeguards offered by the esketamine REMS program, tailoring therapy to patient preference, clinical profile, and available resources.
Supplemental Material
sj-pdf-1-tpp-10.1177_20451253251394127 – Supplemental material for Intravenous ketamine versus esketamine for depression: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-tpp-10.1177_20451253251394127 for Intravenous ketamine versus esketamine for depression: a systematic review and meta-analysis by Ahmed Elmosalamy, Idil Tarikogullari, Liliana Patarroyo-Rodriguez, Gwen Wilson, Jennifer L. Vande Voort, Simon Kung, Mark A. Frye and Balwinder Singh in Therapeutic Advances in Psychopharmacology
Footnotes
Acknowledgements
None.
Authors’ note
Part of the data was presented as a poster at the Annual Meeting of the American Society of Clinical Psychopharmacology, Scottsdale, Arizona (28 May 2025).
Declarations
Supplemental material
Supplemental material for this article is available online.
AI tools disclosure
During the preparation of this manuscript, the authors used OpenAI ChatGPT to assist with text editing (grammar, punctuation, and formatting). The authors subsequently reviewed and edited the content as needed and take full responsibility for the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
