Abstract
Background:
Post-traumatic stress disorder (PTSD) is a prevalent mental illness with a high disability rate. The neurobiological abnormalities in PTSD suggest that drug therapy may have certain therapeutic effects. According to the recommendations of clinical guidelines for PTSD, the current clinical preference is for selective serotonin reuptake inhibitors (SSRIs) or serotonin and norepinephrine reuptake inhibitors (SNRIs). Nevertheless, the efficacy of other types of drugs remains uncertain, which impacts the selection of personalized treatment for patients.
Objectives:
The aim of this meta-analysis was to assess the efficacy and acceptability of drugs with different pharmacological mechanisms in alleviating PTSD symptoms by comparing the response rates and dropout rates of different drug treatment groups in randomized clinical trials.
Design:
Systematic review and meta-analysis.
Methods:
We searched and analyzed 52 reports that described the efficacy and acceptability of medication for PTSD. Among these, 49 trials used the dropout rate as an acceptability indicator, and 52 trials used the response rate as an efficacy indicator.
Results:
In the 49 trials with the dropout rate as the indicator, the dropout rate was 29% (95% confidence interval, 0.26–0.33; n = 3870). In the 52 trials with the response rate as the indicator, the response rate was 39% (95% confidence interval, 0.33–0.45; n = 3808). After drug treatment, the core symptoms of PTSD were significantly improved. This meta-analysis indicated that there was no significant difference between antidepressants and antipsychotics in improving clinical symptoms and acceptability. However, antidepressants may have a slight advantage in efficacy, although with a higher dropout rate.
Conclusion:
Drug treatment is an effective rehabilitation method for PTSD patients, and individualized drug management should be considered.
Trial registration:
This systematic evaluation scheme has been registered with PROSPERO (protocol ID: CRD42023462662).
Plain language summary
The purpose of this study was to assess the acceptability and efficacy of all types of pharmacotherapeutic agents in reducing the symptoms of PTSD. In this systematic meta-analysis, the dropout and response rates of various pharmacotherapy groups reported by randomized clinical trials were compared. A total of 52 reports that described the acceptability and efficacy of PTSD pharmacotherapies were retrieved and analyzed. This meta-analysis supports antidepressants and antipsychotics have no significant difference in improving clinical symptoms and acceptabity, however, AAs may has a slight advantage tendency in efficacy, albeit with a higher dropout rate, so individualized drug management should be considered.
Keywords
Introduction
According to the Diagnostic and Statistical Manual of Mental Disorders, fifth edition (DSM-V), post-traumatic stress disorder (PTSD) is a severe stress disorder caused by intense mental stress such as sudden disasters or natural disasters, which can lead to patients re-experiencing symptoms, persistent avoidance, negative changes in cognition and mood, and increased alertness or reactivity. 1 On the basis of the severity of the disease, it can be divided into acute type (within 3 months after the event), chronic type (more than 3 months after the event), and delayed type (more than 6 months after the event). Lifelong PTSD sufferers may have at least one comorbid mental disorder,1–3 such as depression, anxiety, sleep disturbance, or dissociation. One survey 4 revealed that as of 2017, about 5.6% of the country’s population had experienced a traumatic event, and the lifetime prevalence of post-traumatic stress disorder was 3.9%. In contrast, the National Comorbidity Survey Replication (NCS-R) 5 estimated the lifetime prevalence of PTSD in U.S. adults in 2005 to be 6.8%. 6 The workability of PTSD patients is impaired, which also has a serious impact on the social function of family members and brings a heavy load to society.7–9
The current treatment guidelines for PTSD typically involve a combination of psychotherapy and pharmacotherapy. The frontline treatment for post-traumatic stress disorder is psychological therapy, for instance, exposure therapy, cognitive therapy, and stress inoculation training.10–12 However, psychological treatment for this disease requires a large amount of time and effort at present. In addition, research on psychotherapy often becomes complex due to certain psychobiological dysfunctions in patients with PTSD. The characteristics of PTSD are a series of neurobiological alterations that might respond to drugs, embodying the hypothalamic-pituitary-adrenal axis, and the serotonergic and noradrenergic neurotransmitter systems.13,14 Based on open-label and controlled trials, most clinical practice guidelines recommend selective serotonin reuptake inhibitors (SSRIs) such as Sertraline and Paroxetine as first-line medication for the treatment of PTSD. 15 Sertraline has been shown to have a response rate of approximately 53% in reducing PTSD symptoms compared to placebo, although it may cause side effects such as nausea and sexual dysfunction. 16 Paroxetine has demonstrated a response rate of around 62% against placebo but can lead to weight gain and other side effects. 17 Analysis of clinical practice treatment guidelines showed significant differences in the results about the therapeutic of SSRIs. 18 Furthermore, not all PTSD patients are effective with SSRIs, which require combination therapy or switching to drugs with other mechanisms of action. Many studies have attempted to incorporate patients’ clinical characteristics to select alternative types of medication, such as serotonin-norepinephrine reuptake inhibitors (SNRIs) or atypical antipsychotic drugs, for treatment.8,19,20 SNRIs such as Venlafaxine are also effective, especially in addressing hyperarousal and mood symptoms, albeit with potential side effects including nausea and increased blood pressure. 21 Risperidone has been studied for its efficacy in PTSD treatment. Some research has indicated that it can have a moderate effect in reducing symptoms such as hyperarousal, re-experiencing, and sleep disturbances. However, its use may be limited by potential side effects such as weight gain and increased prolactin levels. For example, a meta-analysis by Ipser and Stein examined the role of various medications in PTSD, including risperidone. 22 Quetiapine has shown some benefits in treating PTSD, particularly in addressing symptoms such as insomnia, nightmares, and irritability. It can contribute to a reduction in the overall severity of PTSD symptoms, although it might not be equally effective for all aspects of the disorder. The dosage typically needs to be titrated based on the patient’s response and tolerance, as described in the study by Stein et al. 23 Olanzapine has been investigated for its potential to alleviate intrusive symptoms and hyperarousal in PTSD patients. It may also have an impact on mood stability. However, it is associated with significant weight gain and metabolic side effects, which restricts its widespread use. The dosing and its effects were explored in a study by Butterfield et al. 24 However, relevant meta-analyses have yet to yield significant results. Therefore, it is necessary to comprehensively evaluate the effect of drug therapy on PTSD.
Considering patients’ varying clinical symptoms, this study evaluated the acceptability and efficacy of different classes of medication, providing the latest evidence for making medication management decisions in adults with PTSD.
Methods
Literature search
First, use the keywords to search for electronic databases Medline, PubMed, Embase, Published International Literature on Traumatic Stress (PILOTS), and the Cochrane Collaboration Depression, Anxiety and Neurosis Controlled Trials Register (CCDAN): PTSD, post-traumatic stress disorder, treatment, pharmacotherapy, medication, randomized controlled trial (RCT), and randomized controlled trial. Narrow down the search by specifying the time frame to focus on the most recent and relevant studies. Look for RCTs as they provide the highest level of evidence. This meta-analysis evaluated randomized and placebo-controlled trials published before the end of 2022. In addition, consider searching for meta-analyses that aggregate data from multiple studies to provide a more comprehensive understanding of the efficacy and safety of different medications. In addition, explore the references of the retrieved articles to identify any other relevant studies that might not have been captured in the initial search. The reporting of this study conforms to the PRISMA statement. 25 Figure 1 summarizes the selection process of randomized controlled trials, which were retrieved from the aforementioned database and subjected to meta-analysis based on the following criteria. Tables 1 and 2 present the characteristics and results of each randomized controlled trial.
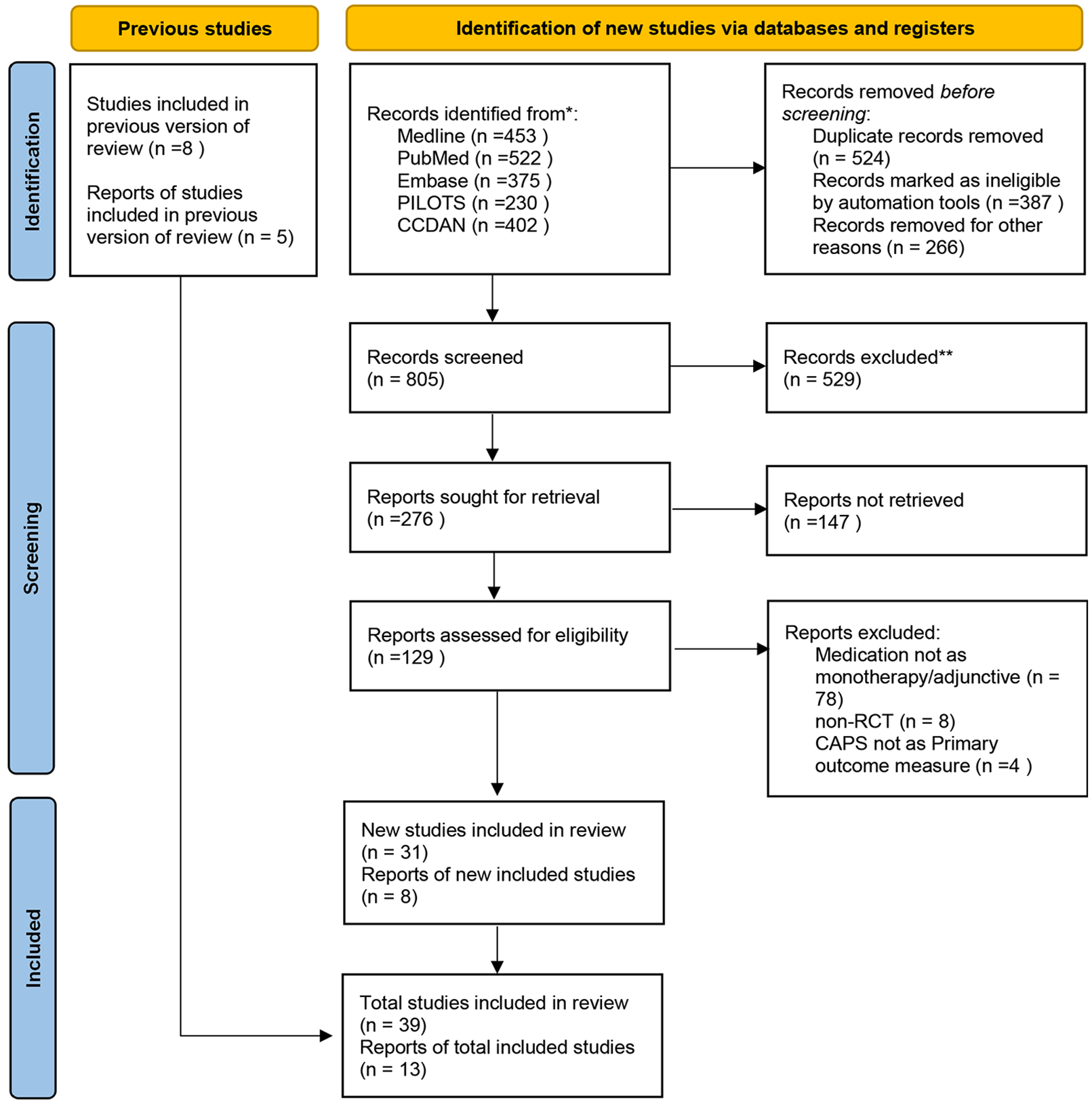
Flow diagram showing the disposition of trials identified by the systematic literature review.
Characteristics of studies reporting the dropout rates for PTSD pharmacotherapy.

Med, study medication; NA, not available; PTSD, Post-traumatic stress disorder; wk, week.
Characteristics of studies reporting the response rates for PTSD pharmacotherapy.

CAPS, Clinician-Administered PTSD Scale (I, intrusionsubscale; A, avoidancesubscale; H, hyperarousalsubscale); CGI, Clinical Global Impression; DGRP, Duke Global Rating for PTSD; PTSD, Post-traumatic stress disorder; SPRINT, Short PTSD Rating Interview scale; TOP-8, treatmentoutcome PTSD.
Inclusion and exclusion criteria
The inclusion and exclusion criteria are formulated as follows:
Inclusion criteria:
(1) Adults diagnosed with PTSD based on the following diagnostic criteria (⩾18 years of age) (International Classification of Diseases-10, DSM-III, DSM-III-R, DSM-IV, DSM-IV-TR, or DSM-V);
(2) Intervention measures: Medication for PTSD in adults;
(3) Randomized control: Placebo or other pharmacological mechanism drugs;
(4) Research type: randomized controlled trials.
Exclusion criteria:
(1) A clear diagnosis of schizophrenia, schizoaffective psychosis, or bipolar disorder, unless their symptoms are secondary to post-traumatic stress disorder;
(2) Cognitive impairment;
(3) High suicide risk;
(4) Trials to prevent or prevent recurrence;
(5) Simultaneously using two or more medications, or a combination therapy of psychotherapy and medication;
(6) Data from the original study are not available or transferable;
(7) Duplicate publication.
Data extraction
Obtain the information from the retrieval: author name, publication year, sample capacity, average age, patient gender, dropout rate, response rate, medication used, dosage, follow-up time, and response criteria. Supposing that the same primary author reported several studies within 12 months, use lowercase letters to distinguish them. If different studies report the same study population, we only include one study.
Statistic analysis
All statistical analyses proceeded to apply the “meta” or “metafor” software packaging in R (version 3.5.3), and p-value < 0.05 was considered statistically significant. Using random effects models to test the acceptability and efficacy of drug therapy for adult PTSD. 26 Heterogeneity in acceptability and efficacy was determined using I 2 and forest plots. I 2 statistics were used to evaluate the heterogeneity of each analysis. 27 In case I 2 < 40%, the fixed effects model is applied. In case of I 2 ⩾ 40, statistical heterogeneity was considered, and the combined effect size was calculated using the random effects models.
First, meta-analysis was applied to ascertain the acceptability and efficacy of medication for PTSD in adults. Secondly, Egger’s funnel plots were applied to test for publication bias. Thirdly, subgroup analyses were applied to discuss potential heterogeneities and determine the potential impact factors, such as different classifications of pharmacological mechanisms.
Results
Research characteristics
According to the keywords, 1982 research reports were initially searched, and ultimately, 52 studies met the inclusion criteria. Records were removed before screening (N = 1177), records excluded (N = 619), reports not retrieved (N = 147), and ultimately 52 studies were identified after reading the full text. Among the 52 eligible research reports, 3 reflected response rates only, while the other 49 included both dropout and response rates. Tables 1 and 2 summarize the characteristics of this research.
Quality assessment in the study
We assessed the quality of all 52 studies included in our analysis, using the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Studies Reporting Prevalence Data, including nine items (1. Whether the sampling framework meets the purpose of the study? 2. Whether the study participants meet the inclusion criteria? 3. Whether the sample size is sufficient? 4. Whether the research object and method are described in detail? 5. Whether the data analysis adequately covers the identified samples? 6. Whether effective methods are used to assess the condition? 7. Were the conditions of all participants evaluated in a normalized manner? 8. Whether the method of statistical analysis is appropriate? 9. Whether the response rate is sufficient, and if not, has the low response rate data been properly processed?). 28 As all studies were quantitative data and cross-sectional design, a key evaluation tool for analyzing cross-sectional studies was used by JBI. All studies were differentiated by low, medium, or high methodological quality. The two authors (C.J.B. and Y.J.M.) evaluated each of the included studies separately and reached an agreement on the inconsistencies. In our study, with a kappa coefficient of approximately 0.7603, there is a substantial level of agreement between the two reviewers. Refer to Supplemental Figure 1 for more details. The inclusion of the study is determined by calculating the percentage of the “yes” option. If it exceeds 75%, the study will be included.28,29
Acceptability and efficacy of drug therapy for adult PTSD
Regarding the efficacy and acceptability of drug therapy for PTSD, we found that the pooled dropout rate as an acceptable indicator was 29% (95% confidence interval, 0.26–0.33; n = 3870), and the pooled response rate as an efficacy indicator was 39% (95% confidence interval, 0.33–0.45; n = 3808) based on the random effects model. Refer to Figures 2 and 3 for more details.

Forest plot of the dropout rate in patients receiving the test drug as an index of the acceptability of the study drug in the treatment of PTSD.

Forest plot of the response rate in patients receiving the study drug as an index of the efficacy of the study drug in the treatment of PTSD.
Subgroup analysis
Perform subgroup analysis based on different drug treatment groups (type 1 = Atypical Antipsychotics(AAs), type 2 = Antidepressant, and type 3 = Other drugs(e.g., anticonvulsants, hypnotics, and benzodiazepines). Out of the 3870 patients reviewed in 52 studies, 259 were distributed to the AAs group, 3362 to the antidepressant group, and 249 to the other drugs group to assess dropout rates in the different drug treatment groups. Out of the 3808 patients reviewed in 52 studies, 273 were distributed to the AAs group, 3286 to the antidepressant group, and 249 to the other drugs group to assess response rates in the different drug treatment groups. For more detailed information, please refer to Figures 4 and 5. The results showed no difference in the dropout rate or response rate between different drug treatment groups. While the distinctions observed in subgroup analysis lack statistical significance, there are discernible trends suggesting that AAs may exhibit superior efficacy, albeit with a higher dropout rate. Conversely, the efficacy of antidepressants appears to be somewhat inferior, albeit with a slightly lower dropout rate.

Forest plot of the dropout rate in patients receiving different pharmacotherapy groups.

Forest plot of the response rate in patients receiving different pharmacotherapy groups. Type 1 = Atypical Antipsychotics (AAs); type 2 = Antidepressant; type 3 = Other drugs (e.g., anticonvulsants, hypnotics, and benzodiazepines).
Publication bias
Publication bias was determined by Egger’s funnel chart analysis. We found neither the dropout rate for publication bias (p = 0.71) nor the response rate for publication bias (p = 0.28) was found. For more detailed information, please refer to Figures 6 and 7.

Funnel plots for the dropout rate.

Funnel plots for the response rate.
Discussion
Because people with PTSD have a low response rate to medication and a high recurrence rate, finding more effective treatments is a serious and urgent issue. Previous studies on the treatment of PTSD have mainly focused on antidepressants, and comparisons have also been made between different antidepressants. Among them, the efficacy of SSRIs has been confirmed in previous meta-analyses.10,12,30–32 Some studies have shown that AAs are effective in either monotherapy or adjuvant therapy for treatment-resistant PTSD or PTSD with accompanying psychiatric symptoms.33–35 On the one hand, AAs can affect certain neurotransmitter receptors (such as serotonin, dopaminergic, and α1-adrenergic) involved in the underlying mechanisms of PTSD.36,37 On the other hand, individuals with PTSD are at an increased risk of developing psychiatric symptoms. The total symptom burden and the psychiatric symptoms, as assessed by the Positive and Negative Symptom Scale (PANSS), in veterans with PTSD, are comparable to those observed in patients with schizophrenia.38,39 However, there have been few randomized controlled trials using AAs or other drugs to treat PTSD, and these trials have not yielded a consensus. Therefore, we performed a meta-analysis, which is a quantitative statistic technique that summarizes and elucidates the therapeutic effects of drugs with different pharmacological mechanisms on PTSD by summarizing and separating data. Our meta-analysis exclusively encompasses randomized controlled trials published in peer-reviewed journals, while excluding all open-label trials and other non-randomized controlled trials to mitigate potential biases. The outcomes of our meta-analysis indicated that there were no statistically significant disparities in the acceptability and efficacy of AAs, antidepressants, and other medications for the treatment of PTSD. However, there are not yet sufficiently well-established studies to improve upon. Randomized controlled trials that include different patient populations, use more consistent outcome measures, or assess long-term efficacy could be conducted to evaluate the efficacy of medication for PTSD.
Acceptability
There is a certain heterogeneity between receiving medication treatment and dropout rates. Dropout rate is an effective indicator for evaluating drug tolerance, efficacy, and safety. There are many factors affecting the dropout rate, such as adverse reactions, experimental conditions, and dosage. The high dropout rate of drugs in certain clinical research may affect the evaluation of their efficacy and adverse effects, which indicates low drug acceptability and limits the generalization of trial data. Accordingly, caution ought to be exercised when using this medication in clinical practice.
Efficacy
Although our meta-analysis results indicate that the overall efficacy of PTSD drug trials did not exceed the limit of 0.5, which is a clinical efficacy indicator consistent with a recent guideline, 40 we would like to remind that there is no direct conversion between efficacy in statistical data and clinical efficacy evaluation. Compare the response rates of different drug groups to evaluate the efficacy of drugs in the treatment of PTSD. Although AAs exhibited greater efficacy than antidepressants and other drugs, their related outcomes must be carefully considered, as the number of randomized controlled trials included in individual analysis is limited. Since different methods were used in each trial to evaluate efficacy, the accuracy of the outcomes of this meta-analysis may be subjected to affected. Accordingly, the overall conclusion largely depends on the quality of the report.
Limitations
The study has several limitations. First, due to the complexity of pharmacological mechanisms, it is best to further subdivide the types of drugs. Second, it is necessary to further compare the efficacy of drug therapy in different types of trauma, pediatric and elderly patients, comorbid substance abuse patients, and refractory patients. Third, given the substantial variability in the symptoms of PTSD, it remains imperative to identify predictive factors for drug response and to ascertain whether particular medications demonstrate greater efficacy for distinct symptom clusters, including psychosis, 41 dissociation,42,43 and stress vulnerability. 44 Future randomized controlled trials and meta-analyses should endeavor to examine these issues with greater specificity and depth. In addition, a number of other factors, such as treatment resistance, comorbidities, digitization, and adjuvant therapy, can be incorporated into studies that hopefully can provide stronger conclusions and roadmaps for the treatment of PTSD.
Conclusion
In terms of the number or scale of research on drug therapy for PTSD, clinical studies on SSRI account for the majority. However, our finding is contrary to the expert consensus40,45–47 that SSRIs are the first-line drug option for treating PTSD, supports antidepressants and antipsychotics have no significant difference in improving clinical symptoms and acceptability, however, antipsychotics may have a slight advantage tendency in efficacy, albeit with a higher dropout rate, so individualized drug management should be considered.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253251342628 – Supplemental material for Pharmacotherapy for post-traumatic stress disorder: systematic review and meta-analysis
Supplemental material, sj-docx-1-tpp-10.1177_20451253251342628 for Pharmacotherapy for post-traumatic stress disorder: systematic review and meta-analysis by Yanyan Jia, Zifeng Ye, Fude Yang, Jiabao Chai, Haiting Xu, Jingming Yang, Weiye Liang and Lili Wu in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253251342628 – Supplemental material for Pharmacotherapy for post-traumatic stress disorder: systematic review and meta-analysis
Supplemental material, sj-docx-2-tpp-10.1177_20451253251342628 for Pharmacotherapy for post-traumatic stress disorder: systematic review and meta-analysis by Yanyan Jia, Zifeng Ye, Fude Yang, Jiabao Chai, Haiting Xu, Jingming Yang, Weiye Liang and Lili Wu in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-3-tpp-10.1177_20451253251342628 – Supplemental material for Pharmacotherapy for post-traumatic stress disorder: systematic review and meta-analysis
Supplemental material, sj-docx-3-tpp-10.1177_20451253251342628 for Pharmacotherapy for post-traumatic stress disorder: systematic review and meta-analysis by Yanyan Jia, Zifeng Ye, Fude Yang, Jiabao Chai, Haiting Xu, Jingming Yang, Weiye Liang and Lili Wu in Therapeutic Advances in Psychopharmacology
Footnotes
Acknowledgements
We acknowledge Leilei Wang, Hu Deng, and Wenxuan Zhao for their contributions to this work. We are also grateful to Hong Cui for her careful oversight of the study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
