Abstract
Background:
Post-traumatic stress disorder (PTSD) is a debilitating condition for which there are few efficacious treatments. Psilocybin is being studied for use in treatment-resistant depression but has not yet been investigated in PTSD.
Aims:
The trial’s primary outcome was to investigate the safety and tolerability of single-dose psilocybin in participants with PTSD.
Methods:
This was a Phase 2, nonrandomized, open-label, multicenter trial. Secondary outcomes were changes in PTSD symptoms (Clinician-Administered PTSD Scale for DSM-5 (CAPS-5); PTSD Checklist for DSM-5 (PCL-5)), functional impairment (Sheehan Disability Scale; SDS) and quality of life (EQ-5D-5L index score).
Results:
Amongst the 22 participants enrolled (63.6% female; mean (SD) age, 39.0 (7.91) years), there was a total of 117 treatment-emergent adverse events (TEAEs); 70 (59.8%) were reported on administration day, of which 64/70 (91.4%) resolved by the end of the next day. TEAEs commonly included headache (n = 11; 50.0%), nausea (n = 8; 36.4%), crying (n = 6; 27.3%) and fatigue (n = 6; 27.3%). There were no serious TEAEs or TEAEs leading to study withdrawal. Pre-post comparisons indicated a clinically meaningful change from Baseline in mean CAPS-5 total score at Week 4 (−29.9 (14.06)) and Week 12 (−29.5 (15.43)), which was associated with the intensity of psychedelic experience on Day 1. PCL-5 scores showed symptom reduction was rapid and sustained until Week 12. SDS total score and EQ-5D-5L index score showed similar improvements.
Conclusions:
Psilocybin at a dose of 25 mg, administered with psychological support, may be safe, well-tolerated and associated with symptomatic improvement in adults with PTSD. Further investigation is warranted.
Clinical trial registration:
ClinicalTrials.gov Identifier: NCT05312151
(https://clinicaltrials.gov/study/NCT05312151)
Introduction
Post-traumatic stress disorder (PTSD) is a serious psychiatric disorder that emerges following exposure to one or more traumatic events. It is characterized by four core symptom clusters: intrusive dreams, flashbacks or memories, avoidance of stimuli associated with the trauma, negative alterations in cognition and mood and marked alterations in arousal and reactivity (American Psychiatric Association, 2013). PTSD has an estimated prevalence in a given year of approximately 5% in the United States (US) and cross-nationally (Kilpatrick et al., 2013; Koenen et al., 2017). The prevalence is higher in at-risk groups such as military veterans, emergency care staff and those living in or migrated from conflict-affected regions (Berger et al., 2012; Charlson et al., 2019; Patanè et al., 2022; Schein et al., 2021). The disorder is severely debilitating with high levels of associated functional impairment (Jellestad et al., 2021) and negatively impacts quality of life (Monson et al., 2017). The patient burden of the disorder is mirrored by its societal and healthcare system impact; high excess economic costs have been described that go beyond direct healthcare costs and involve costs from disability, caregiving, unemployment, workplace absenteeism and presenteeism and premature mortality (Davis et al., 2022).
Despite the pervasive clinical and societal consequences of PTSD, there remains a substantial unmet treatment need for those with the disorder. Trauma-focused psychotherapies are recommended as first-line treatments by several treatment guidelines (Hamblen et al., 2019). While potentially efficacious (Lewis et al., 2020a), there are important barriers limiting treatment success: the approach itself is time intensive, may not be well tolerated and access is often limited (Finch et al., 2020; Watkins et al., 2018). Drop-out rates from psychological interventions for PTSD are high and significantly elevated for trauma-focused psychotherapies in particular (Lewis et al., 2020b). In consequence, for a large cohort study of US veterans with recently diagnosed PTSD, the rate of psychotherapy initiation was modest at 42% and of those who initiated, only 12.1% completed a sufficient treatment course (Valenstein-Mah et al., 2019). Emerging evidence suggests some treatment success can be achieved through complementary and integrative approaches such as mindfulness, meditation and yoga (Gallegos et al., 2017). These approaches are potentially more tolerable, incurring less dropout than trauma-focused interventions.
Pharmacologic advances in treating the disorder have been limited. Treatment guidelines recommend selective serotonin reuptake inhibitors (SSRIs), either the FDA-approved SSRIs sertraline and paroxetine, or SSRIs as a class more broadly (Hamblen et al., 2019). Such treatments have limited efficacy, with less than 30% of patients achieving remission (Kelmendi et al., 2016). High levels of polypharmacy and off-label medication use occur in clinical practice (eg other antidepressant, atypical antipsychotic, anticonvulsant and alpha-adrenergic medications), but the value of such treatment regimens has not been confirmed consistently in clinical trials (Krystal et al., 2017). Non-adherence to antidepressant medication is a well-reported issue in PTSD and is further associated with negative treatment outcomes (Salas et al., 2020). The shortage of evidence-based, efficacious treatment options for PTSD, which are both tolerable and rapidly acting, underscores the importance of developing novel treatments. Psychedelic-based treatments represent a promising research avenue for treatment innovation. As well as being mechanistically novel compared to existing pharmacotherapy for PTSD, psychedelics can induce psychological effects that have been hypothesized to aid trauma processing without being overtly trauma-focused and possibly incur less dropout than trauma-focused psychotherapies (Zaretsky et al., 2024).
Psilocybin is a tryptamine alkaloid prodrug that is dephosphorylated to psilocin, a serotonergic psychedelic, with potent agonist activity at several serotonin receptors, including 5-HT2A (Passie et al., 2002). COMP360 is a proprietary, pharmaceutical-grade, synthetic psilocybin formulation developed by Compass Pathfinder Limited. A Phase 2 trial examining single-dose COMP360 psilocybin suggested that treatment was well-tolerated, produced a rapid decrease in depressive symptoms and improved social functioning and quality of life in individuals with treatment-resistant depression (Goodwin et al., 2022; Goodwin et al., 2023). Like depression, serotonergic dysregulation has been assumed to be the target of antidepressants in PTSD (Yehuda et al., 2015). However, the effects of the serotonergic psychedelic psilocybin in the disorder are hitherto unknown. The objective of this trial was to assess the safety and tolerability of COMP360 psilocybin for the treatment of PTSD.
Patients and methods
Trial design
This trial was a 12-week, Phase 2, multicenter, nonrandomized open-label clinical trial designed to investigate the safety and tolerability of a single dose of 25 mg COMP360 psilocybin in patients with PTSD (ClinicalTrials.gov Identifier: NCT05312151). The trial involved one site in the United Kingdom (UK) (King’s College London, London) and two sites in the US (Mount Sinai Hospital, New York, New York, and Sunstone Therapies, Rockville, Maryland) between June 2022 and February 2024. The trial was approved in the UK by the Brent Research Ethics Committee and in the US by Advarra Institutional Review Board and was submitted to the UK Medicines and Healthcare products Regulatory Agency and the US Food and Drug Administration. Participants’ written informed consent was obtained prior to participation, and the trial was conducted in accordance with the International Council for Harmonization (ICH) Good Clinical Practice guideline and the ethical principles of the Declaration of Helsinki.
Participants
Participants were adult outpatients with PTSD resulting from a traumatic event experienced during adulthood, aged ⩾18 years, recruited from the community and referring healthcare providers. The Life Events Checklist for DSM-5 (LEC-5) and PTSD Checklist for DSM-5 (PCL-5) were used at Screening to identify eligible participants. Diagnosis of PTSD, according to DSM-5 criteria, was confirmed by an experienced clinician using the Clinician-Administered PTSD Scale for DSM-5 (CAPS-5) (Weathers et al., 2018), and participants were required to have at least moderate symptoms of PTSD determined by a total score of 25 or higher at Baseline. Comorbid psychiatric disorders were assessed via medical records and clinical assessment using the Mini International Neuropsychiatric Interview (MINI) version 7.0.2 (Sheehan et al., 1998). Participants taking antidepressant or antipsychotic medications at Screening completed medication down-taper and discontinuation at least two weeks before Baseline. Concomitant psychological therapies were permitted, provided they were initiated at least 3 weeks before Baseline and remained stable throughout the duration of the trial.
Exclusion criteria included a medical history of psychotic disorder, bipolar disorder, personality disorder, obsessive compulsive disorder, alcohol or substance use disorder (within last 12 months), a primary diagnosis of major depressive disorder (within last 6 months), exposure to a traumatic experience in past 3 months, significant suicidal risk (identified with the use of the Columbia Suicide Severity Rating Scale [C-SSRS] at Screening, within the past year, and Baseline, since last visit) (Posner et al., 2008), and significant physical or sexual abuse during childhood based on clinician judgement with the use of the Childhood Trauma Questionnaire. Complex PTSD, a psychiatric disorder that is similar to PTSD but with the additional presentation of disturbances in self-organization symptoms (ie affective dysregulation, negative self-concept and disturbances in relationships) (Maercker et al., 2013), was also excluded. Complex PTSD is commonly associated with greater psychiatric symptom comorbidity and with prolonged or repeated exposure to traumatic stressors, often arising from a background of interpersonal abuse (Cloitre et al., 2019). As an initial study on the safety and feasibility of psilocybin in PTSD, this patient group and participants with a primary presentation involving childhood abuse were not enrolled to limit symptom heterogeneity and exclude individuals at risk of severe personality changes that commonly result from a background of chronic abuse. Dissociative symptoms were not exclusionary.
Procedures
Eligible participants completed a run-in period of 2 to 6 weeks, during which medication was washed out if applicable. Participants met with a trial therapist three times during the run-in period to receive psychoeducation and prepare for the psychedelic experience. Therapists were licensed mental health care professionals who had relevant education, training and/or experience in mental health, counselling, or psychological therapies. The training approach for therapists has been described previously (Tai et al., 2021). The support model applied was based on the principles of the Compass Psychological Support Model designed for clinical trials, described in further detail elsewhere (Kirlić et al., 2025). The purpose of pre-administration preparation sessions was to prepare the participant to navigate the psychedelic experience safely and autonomously on the day of the administration session.
Baseline assessments were obtained on Day -1 (the visit before the administration session). On Day 1, participants received a single dose of 25 mg COMP360 psilocybin. The administration session lasted 6 to 8 h and was attended by two trial therapists. During the administration session, therapists allowed the psychedelic experience to unfold naturally, providing minimal intervention and acting as safeguards. A trial psychiatrist was available on site for consultation. Administration rooms were dimly lit and designed to provide a nonclinical, calming atmosphere. During the administration session, participants were encouraged to listen to a standardized music playlist and wear eyeshades. After at least 6 h and when the psychedelic effects of the drug had fully dissipated, participants were accompanied home by a caregiver. Participants were followed up for 12 weeks after treatment. Participants received three integration sessions with a trial therapist, occurring on the day after administration and first and second weeks of follow-up. Integration sessions were nondirective and designed to support the participant in consolidating insights from their psychedelic experience without offering interpretations or solutions, during which therapists remained vigilant to potential safety concerns. Participants were to remain off medications for PTSD until after Week 4, as clinically appropriate. In-clinic follow-up visits were conducted at Day 2, Week 1, Week 2, Week 4 and Week 12. Follow-up visits were also conducted at Week 6 and Week 9, which could be in-clinic or remote.
Safety endpoints
The primary outcome of the trial was the safety and tolerability of treatment. Adverse events were evaluated at every visit and coded using the Medical Dictionary for Regulatory Activities (MedDRA), version 26.1. Adverse events that emerged or worsened after treatment administration were categorized as treatment-emergent adverse events (TEAEs). ICH Good Clinical Practice criteria were used to categorize serious adverse events (ie adverse events that are life-threatening, cause or prolong hospitalization, cause significant or persistent disability, result in death or congenital disorder, or are otherwise medically significant). Suicidality was assessed using the C-SSRS at each visit with an assessment period since the last visit (Posner et al., 2008). Suicidal ideation with intent (items 4 or 5) or endorsement of any items in the suicidal-behaviour section, except non-suicidal self-injurious behaviour, were considered serious adverse events. Adverse event severity describes the level of functional impairment and hazardousness due to the intensity of a specific event and was assessed as mild, moderate, or severe for each TEAE. The Modified Discontinuation Emergent Signs and Symptoms Scale (mDESS) was used to assess the tolerability of antidepressant/antipsychotic medication withdrawal during run-in (Duffy et al., 2019). Vital signs were assessed at Baseline, on the day of administration (pre-dose) and on Day 2 for each participant. During the study, the vital sign assessment schedule was amended to additionally assess vital signs at 1 hour, 3 hours and 6 hours post-dose to describe in greater detail the on-treatment effects of the drug. Data obtained following this updated schedule are available for four participants only. Other safety assessments comprised electrocardiogram (ECG), clinical laboratory tests (including urine drug screening) and the Brief Psychiatric Rating Scale – positive symptom subscale (BPRS+) (Zanello et al., 2013).
Secondary endpoints
The secondary outcomes of the trial were treatment efficacy and the effects of treatment on functional impairment and quality of life. Treatment efficacy was assessed via change from Baseline on the clinician-rated CAPS-5 (past month) at Week 4 and Week 12, administered by trained clinicians at study sites, and the self-reported PCL-5 at each study visit. The participant’s recall period using the PCL-5 was the past day on Day 2 and the past week at all other assessment points. Treatment response was defined as a reduction of ⩾15 points in CAPS-5 total score compared to Baseline, a score considered clinically meaningful (Varker et al., 2020), and remission was defined as a CAPS-5 total score ⩽20.
Improvements in functional impairment and quality of life were assessed at both Week 4 and Week 12 using the Sheehan Disability Scale (SDS; change from Baseline) (Sheehan, 1983) and the EQ-5D 5-levels instrument (EQ-5D-5L; index score) (Herdman et al., 2011; The Euroqol Group, 1990), respectively. The SDS is a self-reported assessment of functional impairment, which assesses impairment across three domains: occupational, social and family/home responsibilities. Each domain comprises a Likert scale ranging from 0 to 10, with higher scores indicating greater functional impairment. A total score was obtained by summing the scores on each domain. The EQ-5D-5L is a self-reported instrument used to describe the health-related quality of life of an individual across five dimensions: mobility, self-care, usual activities, pain/discomfort and anxiety/depression. Participant response data is converted into an EQ-5D-5L index score, with higher index values reflecting better health-related quality of life.
The 5-Dimensional Altered States of Consciousness Rating Scale (5D-ASC) (Dittrich, 1998; Studerus et al., 2010) is a 94-item self-reported instrument that characterizes the acute psychedelic effects experienced by the participant on the day of administration. It was completed at the end of the administration session, after the acute psychedelic effects had subsided. The 5D-ASC consists of five dimensions: Oceanic Boundlessness (measuring mystical-type experiences that are often associated with positive emotional states); Anxious Ego Dissolution (measuring dysphoric experiences that are often associated with negative emotional states); Visual Restructuralization (measuring perception and visual alterations/hallucinations); Auditory Alterations (measuring changes in hearing, such as perceived sounds and auditory hallucinations); and Reduction of Vigilance (measuring a state of impaired alertness, including levels of drowsiness, awareness and functioning). Correlations between each of the 5D-ASC dimensions on Day 1 and the change from Baseline on the CAPS-5 total score (Week 4 and Week 12) were analyzed as post hoc exploratory outcomes to assess the association between psychedelic experience and treatment efficacy.
Statistical analysis
No inferential statistical analyses were performed for this open-label study. Safety analyses were performed on the Safety Analysis Set, which included all participants who received COMP360. Efficacy analyses were performed on the Full Analysis Set, which included all participants in the Safety Analysis Set. Descriptive summaries were used to evaluate the safety data and secondary efficacy endpoints. There were no imputations for missing efficacy data; only observed data were analyzed. To obtain index values using the UK value set, EQ-5D-5L data were mapped to the available EQ-5D-3L value set via the cross-walk method (NICE, 2019). Spearman’s rank correlation coefficients (rs) were calculated in post hoc analyses to assess the correlation between each 5D-ASC dimension and change from Baseline in CAPS-5 total score at Week 4 and Week 12. Effect sizes are measured using Cohen’s d and reported as absolute values throughout.
Results
Participants
Twenty-two participants (n = 14 female; 63.6%) with a mean (SD) age of 39.0 (7.91) years were enrolled and received psilocybin treatment out of a total of 39 participants screened. Thirteen were enrolled at the site in the UK, with the remainder enrolled in the US (five at Mt. Sinai Hospital, four at Sunstone Therapies). All participants had at least moderate symptoms of PTSD, with a preponderance considered severe as indicated by a mean (SD) CAPS-5 total score at Baseline of 47.5 (9.78). The mean (SD) duration of the disorder at study entry was 88.2 (68.65) months. Nine participants (40.9%) were receiving antidepressant treatment at Screening and completed medication washout. No participant commenced or re-started antidepressant or antipsychotic medications during the trial. One participant was receiving lamotrigine for depression at Screening, which remained consistent for the duration of the trial. Seven participants (31.8%) were receiving psychological therapies at trial entry, which in each case began at least 3 weeks before Baseline and remained stable throughout the trial; the minimum duration between the start of concomitant psychological therapy and Baseline was 6.3 months. Sample demographic and clinical characteristics are described in Table 1, and the participant CONSORT diagram is shown in Supplemental Figure S1.
Demographic and clinical characteristics.

BMI: body mass index; CAPS-5: Clinician-Administered PTSD Scale for DSM-5; PTSD: post-traumatic stress disorder; SDS: Sheehan Disability Scale; SD: standard deviation
Other traumatic events comprised: witnessing or learning of actual or threatened death, or serious injury, occurring to a close family member or close friend; domestic violence incident; terrorist attack; medical incident causing trauma.
Antidepressants withdrawn at study entry comprised: sertraline (n = 5), fluoxetine (n = 1), escitalopram (n = 1), mirtazapine (n = 1) and one participant (n = 1) withdrawn from bupropion and trazodone. One participant was using a low dose of amitriptyline for migraine at study entry, which was also discontinued. As the maximum dose used (20 mg) was not in the antidepressant therapeutic range, it is not represented in this table.
Primary outcome: Safety
TEAEs observed during the study are presented in Table 2. TEAEs occurred in all participants (n = 22) with 117 events reported in total. The most frequently reported TEAEs included headache (n = 11; 50.0%), nausea (n = 8; 36.4%), crying (n = 6; 27.3%) and fatigue (n = 6; 27.3%). Most TEAEs were reported on administration day (70/117; 59.8%), the majority of which resolved by the following day (64/70; 91.4%; 56/70 (80.0%) resolved on the day of dosing and 8/70 (11.4%) resolved the day after). Three participants (13.6%) experienced severe TEAEs, which comprised eight events as follows: euphoric mood (n = 2; 9.1%), nausea, chills, muscle twitching, visual hallucination, auditory hallucination and synesthetic hallucination (each n = 1; 4.5%). All occurred on administration day and were considered related to treatment; seven resolved on the same day, and one on the following day. There were no treatment-emergent serious adverse events or participant withdrawals from the trial.
MedDRA-coded treatment-emergent adverse events where the preferred term is observed in n > 1.
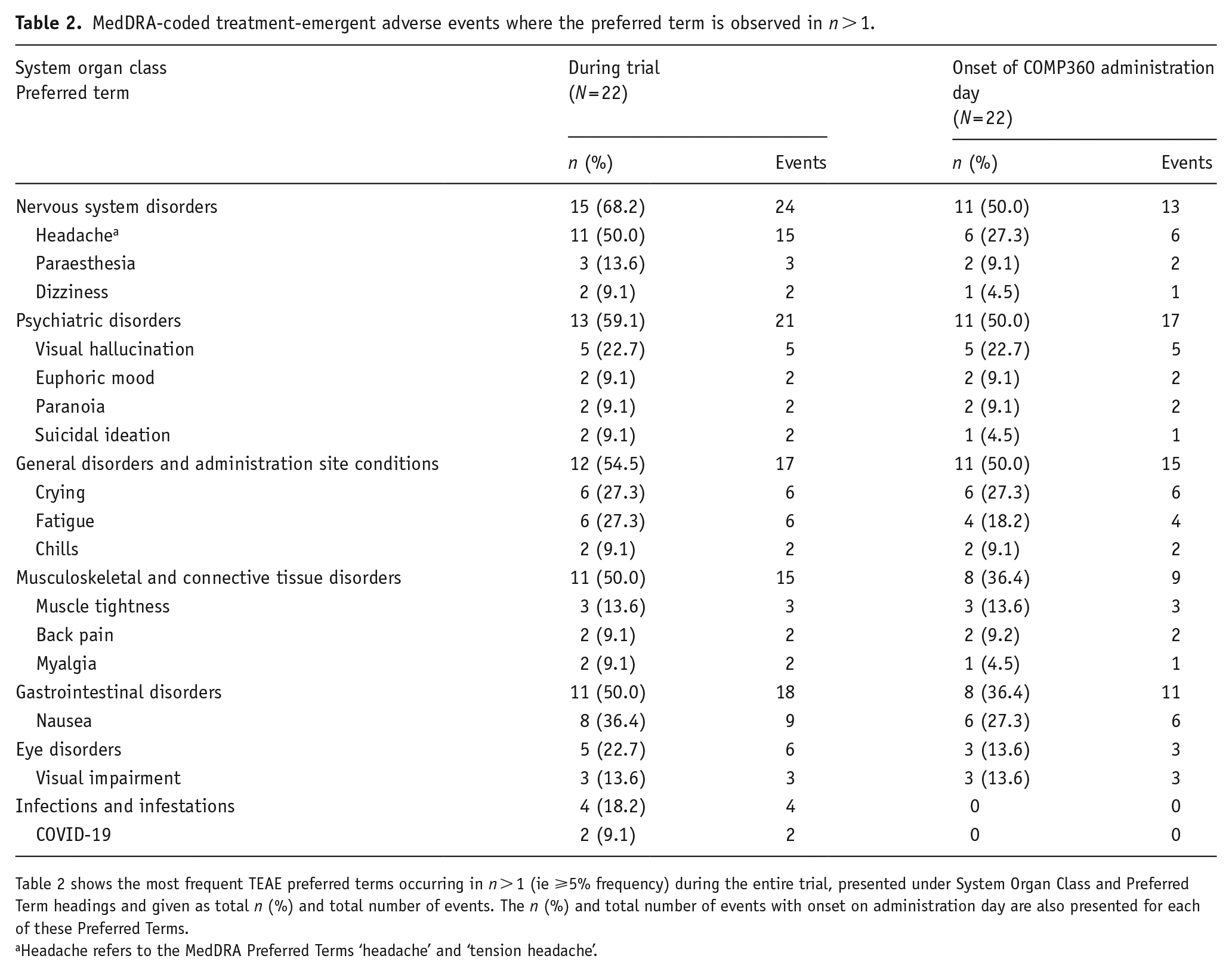
Table 2 shows the most frequent TEAE preferred terms occurring in n > 1 (ie ⩾5% frequency) during the entire trial, presented under System Organ Class and Preferred Term headings and given as total n (%) and total number of events. The n (%) and total number of events with onset on administration day are also presented for each of these Preferred Terms.
Headache refers to the MedDRA Preferred Terms ‘headache’ and ‘tension headache’.
There were two TEAEs of suicidal ideation. The first was a moderate and transient event that started and resolved on administration day in a participant who went on to be a responder and remitter at Week 4 and a responder at Week 12, and it was deemed to be related to the study drug. The second event was mild and occurred at Week 7 in a non-responder, resolved within a month and was deemed possibly related to the study drug. Both participants had a previous history of suicidality as measured by the C-SSRS. All adverse events of special interest (AESIs) that occurred during the trial are presented in Supplemental Table S1. There were no clinically concerning trends in mDESS, vital signs, ECG, clinical laboratory tests, or BPRS+ results (see Supplemental Materials).
Secondary outcomes: Efficacy
Treatment was associated with a clinically meaningful reduction in mean (SD) CAPS-5 total score from Baseline at Week 4 (−29.9 (14.06); 95% CI: −36.1 to −23.7; Cohen’s d = 2.13) and Week 12 (−29.5 (15.43); 95% CI: -36.4 to -22.7; Cohen’s d = 1.92) (Figure 1(a)). This translated to an 81.8% response rate and a 63.6% remission rate at Week 4 and a 77.3% response rate and 54.5% remission rate at Week 12. Inspection of CAPS-5 symptom cluster scores indicated this reduction in PTSD symptom severity was a trend observed across each symptom cluster: intrusion, avoidance, negative alterations in cognition and mood and arousal and reactivity. PTSD symptom reduction was rapid and durable indicated by a mean (SD) change from Baseline PCL-5 score (52.1 (13.68)) at Day 2 (−33.5 (14.32); 95% CI: −39.8 to −27.1; Cohen’s d = 2.34) which was sustained at each study visit through to Week 12 (−34.3 (18.13); 95% CIs: −42.4 to −26.3; Cohen’s d = 1.89) (Figure 1(b)).
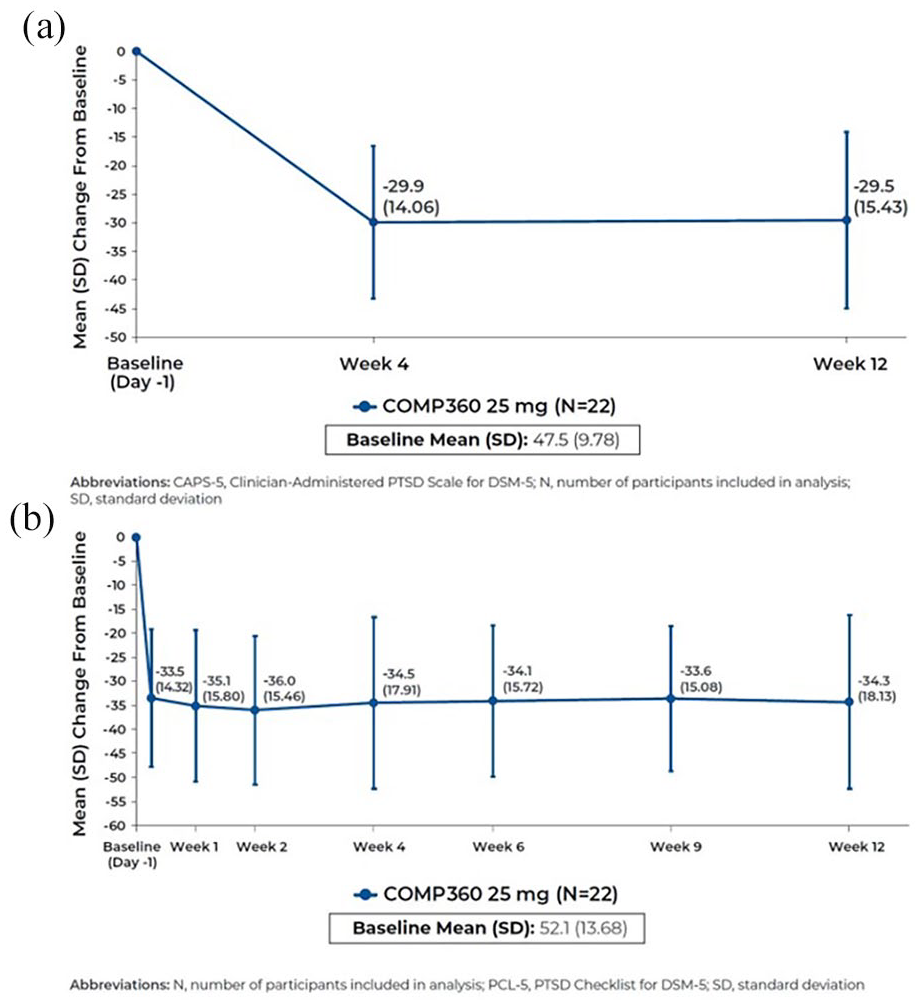
(a) Summary of Change from Baseline in CAPS-5 total score. (b) Summary of Change from Baseline in PCL-5 total score.
Participants showed an improvement in functional impairment over the 12 weeks of the study; from a mean (SD) SDS total score of 22.7 (5.38) at Baseline, there was a −11.7 ((8.41); 95% CI: −15.5 to −8.0; Cohen’s d = 1.39) point reduction at Week 4 and a −14.4 ((8.21); 95% CI: −18.1 to −10.6; Cohen’s d = 1.75) point reduction at Week 12 (Figure 2(a)). Quality of life scores improved throughout the study, indicated by an EQ-5D-5L index score of 0.51 ((0.287); 95% CI: 0.380 to 0.635) at Baseline increasing to 0.73 ((0.272); 95% CI: 0.610 to 0.851) at Week 4 and 0.78 ((0.269); 95% CI: 0.665 to 0.904) at Week 12 (Figure 2(b)). This equated to a change from Baseline in EQ-5D-5L index score of 0.22 ((0.237); 95% CI: 0.12 to 0.33; Cohen’s d = 0.94) at Week 4 and 0.28 ((0.226) 95% CI: 0.18 to 0.38; Cohen’s d = 1.22) at Week 12. All secondary outcome results, including CAPS-5 symptom cluster scores, are presented in Supplemental Table S2. CAPS-5, PCL-5 and EQ-5D-5L data were available from all 22 participants at all follow-up visits. One participant had missing SDS data at Week 12.
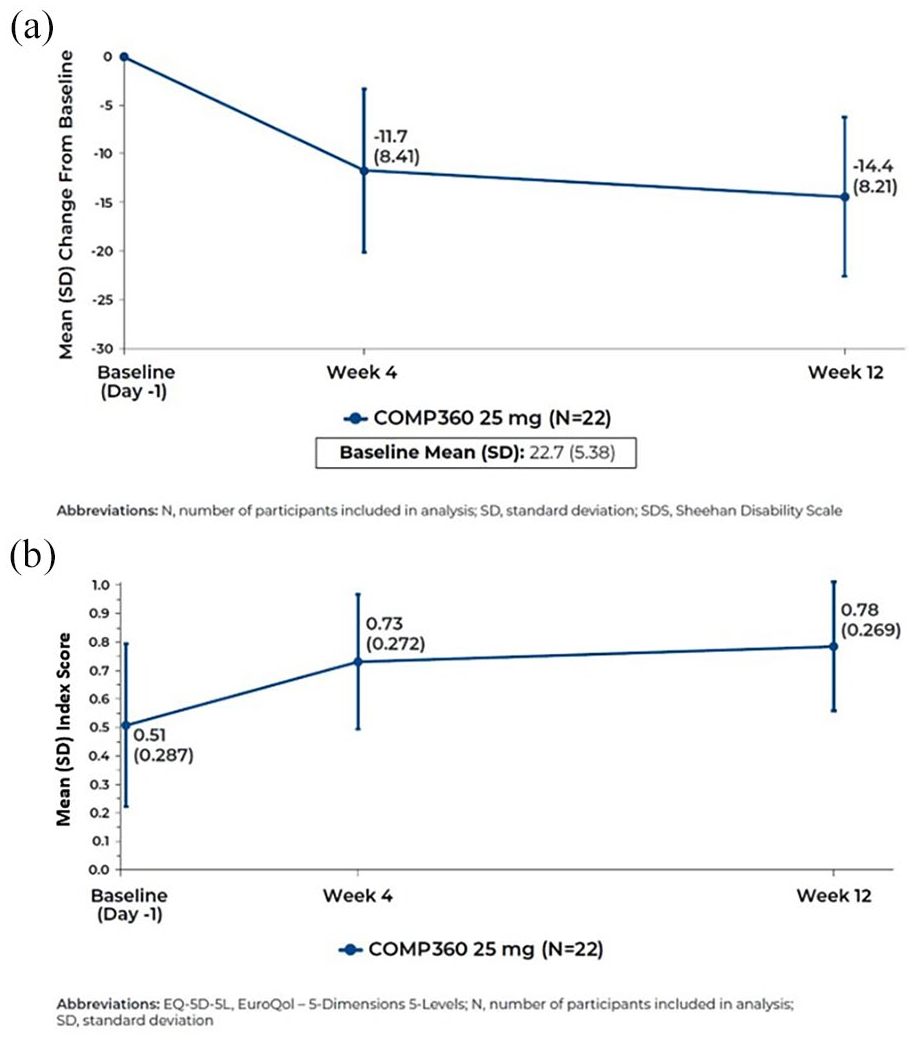
(a) Summary of Change from Baseline in SDS-5 total score. (b) Summary of Change from Baseline in EQ-5D-5L index score.
Oceanic Boundlessness (OB) was the 5D-ASC dimension most strongly correlated with CAPS-5 change from Baseline, both at Week 4 (rs = −0.442) and Week 12 (rs = −0.394); higher OB scores were associated with the greatest reductions in CAPS-5 total score (Figure 3). Contrastingly, the other 5D-ASC dimensions showed an inverse pattern, suggesting that greater intensity was associated with more modest symptom reduction. Moderate strength correlations were observed between Anxious Ego Dissolution (AED) and CAPS-5 change from Baseline at Week 4 (rs = 0.396), Reduction of Vigilance (RV) and CAPS-5 change from Baseline at Week 4 (rs = 0.425) and Visual Restructuralization and CAPS-5 change from Baseline at Week 12 (rs = 0.327). All other 5D-ASC dimension correlations were rs < 0.3 (Supplemental Table S3; Supplemental Figures S2–S5).
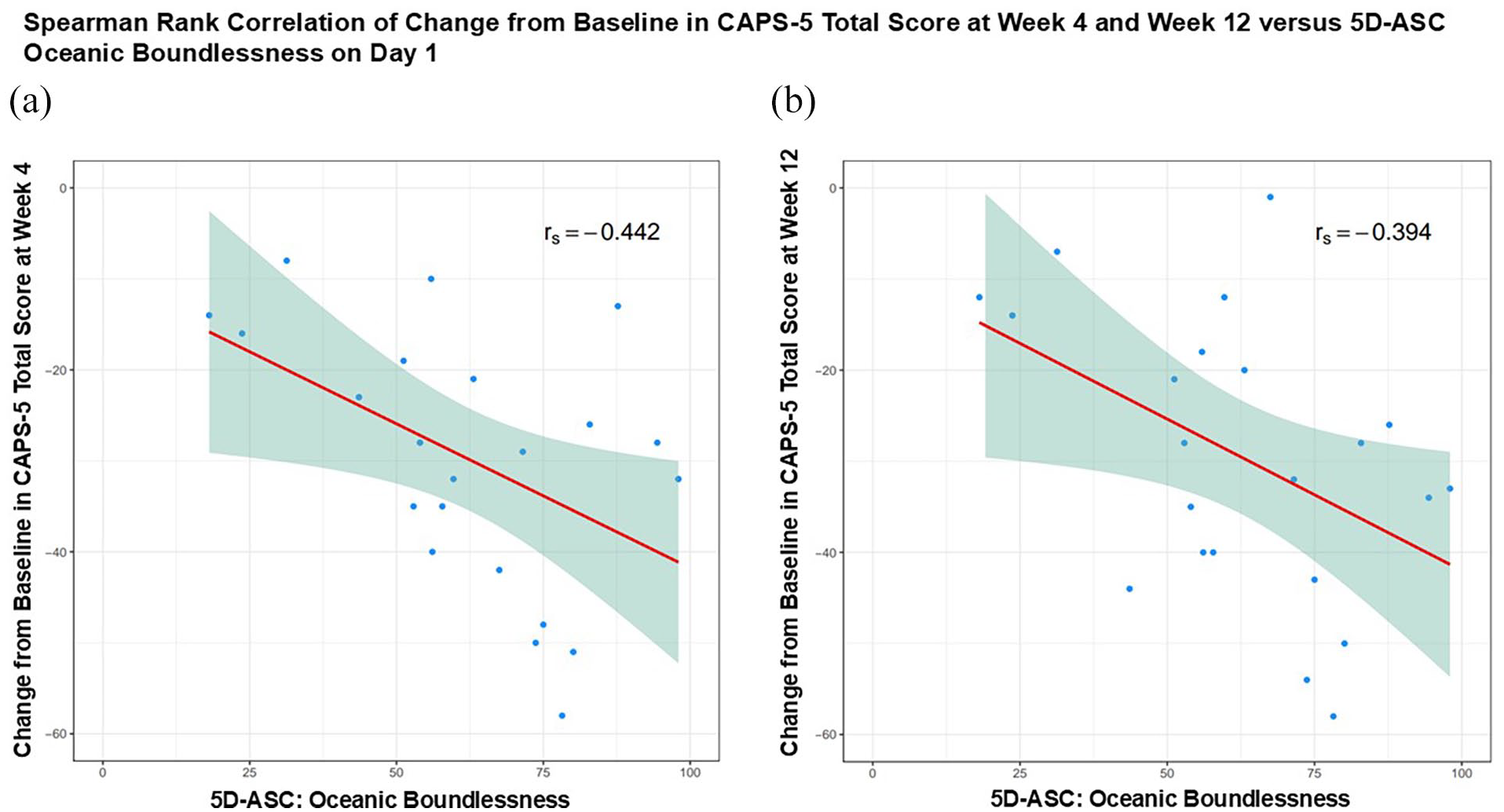
(a) Correlation between Change from Baseline CAPS-5 total score at Week 4 and 5D-ASC Oceanic Boundlessness on Day 1. (b) Correlation between Change from Baseline CAPS-5 total score at Week 12 and 5D-ASC Oceanic Boundlessness on Day 1.
Discussion
COMP360 psilocybin at a dose of 25 mg was not associated with any serious adverse events or adverse events resulting in study withdrawal in participants with PTSD. Secondary outcomes (PTSD symptom severity, functional impairment and quality of life) revealed consistent and durable pre-post treatment improvements. To our knowledge, this is the first clinical trial to report on the safety and tolerability of psilocybin in participants diagnosed with PTSD.
There were no treatment-emergent serious adverse events observed and no withdrawals over the 12-week trial period. Most TEAEs observed during the trial started on the day of dosing, resolved on the same or following day, and commonly included headache, nausea and fatigue, which is a pattern consistent with other trials examining psilocybin in healthy volunteers and in patients with treatment-resistant depression (Goodwin et al., 2022; Rucker et al., 2022). Most AESIs observed during the trial were as expected and described previously in psilocybin studies, including hallucinations, paraesthesia, mood alteration (eg crying) and euphoric mood (Goodwin et al., 2022; Rucker et al., 2022). Suicidal ideation experienced by two trial participants emphasizes the need for clinical vigilance in trials involving participants with serious mental illness. Suicidality is not uncommon amongst individuals with PTSD broadly. Indeed, for this reason, appropriate treatment access assumes importance (Cooper et al., 2020; Forehand et al., 2022).
Single-dose psilocybin was associated with a clinically meaningful reduction in CAPS-5 total score (clinician-rated PTSD symptom severity over the past month). This reduction was observed across all four symptom clusters and sustained until the end of the trial. The patient-rated equivalent measure (PCL-5) indicated a rapid onset of this improvement, observed the day after treatment and similarly sustained throughout the trial. A reduction in PCL-5 scores was reported previously in a psilocybin feasibility study in a sample of older long-term acquired immunodeficiency syndrome survivors who had experienced demoralization and traumatic loss (Anderson et al., 2020). The current secondary outcome results suggesting efficacy for PTSD align with this previous work and are the first in a psilocybin trial to demonstrate this finding in clinically-confirmed PTSD. These data suggest COMP360 psilocybin has potential as a future treatment for PTSD.
Over 75% of participants were classified as responders, and over half were remitters at the end of the 12-week trial. These results are promising when contrasted against SSRI clinical trials in which treatment response rates scarcely exceed 60% (Kelmendi et al., 2016). Meaningful comparison with older PTSD trials is challenging, however, chiefly because those studies investigated drugs that have chronic dosing regimens and employed randomized-controlled designs. In addition, the transition to DSM-5 has since restructured how PTSD is diagnosed and assessed, introducing further divergence (Pai et al., 2017; Weathers et al., 2018). Our protocol definitions for response and remission were derived from a systematic review that highlighted the inconsistency of the criteria employed and recommended response and remission thresholds of ⩾15-point change from baseline and a total score ⩽20, respectively (Varker et al., 2020). Recent studies for 3,4-methylenedioxymethamphetamine (MDMA)-assisted psychotherapy in PTSD defined clinical response as a decrease of ⩾10 points on the CAPS-5 and remission as a loss of diagnosis and a CAPS-5 total score ⩽11 (Mitchell et al., 2021, 2023), which if applied instead, would yield a higher response rate, but lower remission rate (Supplemental Table S4). The MDMA-assisted psychotherapy studies consisted of three treatment sessions across an 18-week period and assessed symptoms at different timepoints compared with the current trial. Applying identical CAPS-5 definitions, response and remission rates at Week 4 and Week 12 were greater following single-dose COMP360 psilocybin, followed up over 12 weeks, than those achieved after two treatment sessions of MDMA-assisted psychotherapy. While the Baseline mean CAPS-5 total score in the current study indicated a similar level of severity of PTSD symptoms as was reported at baseline in the MAPP1 trial (enrolling severe PTSD) (Mitchell et al., 2021) and was even greater than in the MAPP2 trial (enrolling at least moderate PTSD) (Mitchell et al., 2023), the latter two trials did not exclude participants with childhood trauma or complex PTSD, complicating direct comparisons.
Treatment was also associated with sustained improvements in functional impairment and quality of life. This is important because the burden of PTSD on psychosocial function and quality of life is so substantial (Jellestad et al., 2021; Monson et al., 2017), and can linger even after remission of symptoms (Bryant et al., 2023; Larsen et al., 2019). Addressing such outcomes, therefore, assumes importance for comprehensively treating PTSD.
As in other psilocybin studies (Aaronson et al., 2023; Roseman et al., 2018; von Rotz et al., 2023), 5D-ASC OB, a dimension associated with desynchronization of brain networks (Smigielski et al., 2019) and positive, more connected emotional states, was correlated with better therapeutic outcomes. Assessed after administration, this dimension was robustly associated with CAPS-5 change from Baseline until the end of the study. Psychedelic experience intensity is correlated with plasma psilocin and fMRI-assessed cortical network connectivity (Madsen et al., 2021) and may therefore represent a proximal pharmacodynamic predictor of later clinical response. Such a correlation may predict a dose-effect relationship as seen for psilocybin in treatment-resistant depression (Goodwin et al., 2022, 2025). This OB effect was well differentiated from the correlations of response with other dimensions of the 5D-ASC, which were all of opposite direction or negligible strength. That greater AED and RV were negatively correlated with efficacy is dissimilar to the patterns seen in treatment-resistant depression, in which no such associations were observed (Goodwin et al., 2025). In PTSD, a condition with marked somatic and emotional reactivity, it may be intuitive to suspect that negative experiences, such as those captured by the AED, undermine the likelihood of therapeutic response. The link between efficacy and RV is less straightforward. This dimension captures diminished awareness of the external environment and altered attention. While these qualities are not necessarily indicative of treatment-emergent dissociation, they may resemble cognitive and perceptual shifts familiar to those with PTSD (particularly those with dissociative symptomatology) and could be perceived as disorienting, potentially moderating the therapeutic impact of the psychedelic experience. Further exploration of how the quality of psychedelic experience shapes treatment response in PTSD may be informative for next-generation psychedelic treatment for this patient group.
This was a feasibility study with significant limitations. First, it was a small, open-label trial without a comparator arm; larger, controlled studies are required to understand fully the safety and efficacy of COMP360 psilocybin for PTSD. Second, participants with complex PTSD or at clinically significant risk of suicide were excluded from the study. Finally, there were limited vital sign data available during the administration session: more frequent on-treatment assessment is needed to characterize the acute effects of psilocybin on pulse and blood pressure.
The landscape of PTSD pharmacotherapy and drug development has been described previously as in crisis, requiring urgent redress (Krystal et al., 2017). In this trial, COMP360 psilocybin administered in a single session with psychological support was found to be generally well-tolerated and without serious risk to safety. Trial secondary endpoints indicated that treatment was associated with a rapid and sustained reduction in PTSD symptoms and improved functioning and quality of life over a 12-week follow-up period. Future studies are needed to ascertain the safety and efficacy of psilocybin as a novel treatment for this regrettably underserved patient group.
Supplemental Material
sj-docx-1-jop-10.1177_02698811251362390 – Supplemental material for Investigating the safety and tolerability of single-dose psilocybin for post-traumatic stress disorder: A nonrandomized open-label clinical trial
Supplemental material, sj-docx-1-jop-10.1177_02698811251362390 for Investigating the safety and tolerability of single-dose psilocybin for post-traumatic stress disorder: A nonrandomized open-label clinical trial by Niall M. McGowan, James J. Rucker, Rachel Yehuda, Manish Agrawal, Nadav Liam Modlin, Hollie Simmons, Agata Tofil-Kaluza, Shriya Das and Guy M. Goodwin in Journal of Psychopharmacology
Footnotes
Acknowledgements
We sincerely thank the participants—without them, this research would not have been possible. We also thank the staff at all trial sites, including therapists, study coordinators, nurses, physicians and researchers, for their help with the recruitment of participants, data collection and study procedures. We thank Claudia Sisa, PhD, of Compass Pathways plc for her work supporting the initial design of this trial.
Author contributions
Conception or design of the work (NMM, JJR, RY, NLM, GMG); finalization of analysis plans (NMM, HS, ATK); the acquisition of the data (NMM, JJR, RY, MA, NLM, SD); the analysis of the data (HS); the interpretation of data (NMM, NLM, HS, ATK, GMG). Drafting of the initial manuscript (NMM). All authors reviewed and revised the manuscript critically for important intellectual content, approved it before submission and vouch for the adherence of the trial to the protocol, the completeness and accuracy of the data and analyses, and the reporting of adverse events. Authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy of integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: NMM, HS, ATK, SD and GMG are current or past employees of and own shares, share options and/or restricted share units in Compass Pathways Plc. GMG has also consulted for Signant Health, Sun Pharma and Takeda in the last 3 years. JJR has received payments for advisory boards for Clerkenwell Health (past), Beckley Psytech Ltd (past) and Delica Therapeutics (past) and articles for Janssen. He has been awarded grant funding (received and managed by King’s College London) from Compass Pathfinder Ltd, Beckley Psytech Ltd, Multidisciplinary Association for Psychedelic Studies, National Institute for Health Research, Wellcome Trust and Biomedical Research Centre at the South London and Maudsley NHS Foundation Trust. RY is Director of the Center for Psychedelic Psychotherapy and Trauma Research, which conducts or has conducted clinical trials sponsored by the Multidisciplinary Association for Psychedelic Studies and Compass Pathfinder Ltd. MA has no competing interests to disclose. NLM provides services for the psychoactive trials group at King’s College London. The psychoactive trials group at King’s College London receives grant funding (received and managed by King’s College London) from Compass Pathways Plc, Supporting Wounded Veterans, Multidisciplinary Association for Psychedelic Studies, Transcend Therapeutics, Beckley PsyTech and National Institute for Health Research. NLM has received consulting fees from Compass Pathways Plc, Small Pharma Ltd, Beckley Psytech Ltd and Sunstone Therapies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded and sponsored by Compass Pathfinder Ltd, London, UK. A contract research organization (Worldwide Clinical Trials), paid by the sponsor, supervised the conduct of the trial under the direction of the sponsor.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
