Abstract
Background:
Extant research on cognitive functioning in treatment-resistant schizophrenia (TRS) is limited and of poor quality. Cognitive impairments in patients with schizophrenia spectrum disorders (SSD) significantly influence quality of life. In patients with TRS, clozapine (CLO) is not consistently associated with improved cognitive functioning. The active metabolite n-desmethylclozapine (norclozapine (NCLO)) potentially exerts procognitive effects due to cholinergic and glutamatergic activity. Unfortunately, research on CLO/NCLO ratio and cognitive functioning is even more scarce.
Objectives:
To review the literature on the effect of the CLO/NCLO ratio on cognitive functioning in patients with SSD.
Design:
This is a systematic review.
Data sources and methods:
A search was carried out in the electronic databases Embase, PsycINFO, PubMed, Cochrane and the Cochrane Controlled Register of Trials with no restrictions in language or publication year.
Results:
We identified 15 relevant studies (longitudinal, k = 4; cross-sectional, k = 11). The study population consisted of adult clozapine users (n = 953) with varying degrees of treatment resistance. Specific cognitive domains and overall cognitive functioning were assessed using various neuropsychological tests and a composite score, respectively. Eleven studies were considered of fair quality (longitudinal: k = 2, cross-sectional: k = 9). In one longitudinal study, a negative causal relationship was found between the CLO/NCLO ratio and attention/vigilance and a negative correlation between social cognition and the composite score (n = 11). No significant correlations were found between the CLO/NCLO ratio and the cognitive domains processing speed, reasoning/problem solving, or for working memory (k = 1, n = 11), verbal learning (k = 1, n = 43) or visual learning (k = 2, n = 54). Study designs and populations were heterogeneous, and the analysis of confounding factors was limited and inconsistent.
Conclusion:
Clinical evidence is too scarce to support the hypothesis of a procognitive effect of NCLO. Personalised CLO treatment by modulating the CLO/NCLO ratio remains a distant prospect. Recommendations for future CLO research and anticipated limitations are discussed.
Trial registration:
This systematic review was preregistered with PROSPERO (CRD42023385244).
Plain language summary
Cognitive abilities determine the quality of life to a great degree in people with schizophrenia spectrum disorders. In a seriously ill subgroup who do not respond to regular antipsychotic medications, cognitive impairments are often severe. In these cases, clozapine has the best effect on psychotic symptoms. However, we do not know how or to what extent clozapine affects cognitive abilities. Perhaps instead of clozapine, its active breakdown product norclozapine has cognitive benefits. We hoped to see if we could find a direct link between the ratio of clozapine and norclozapine in the blood and cognitive abilities in these severely mentally ill people.
We searched for previous studies in people with schizophrenia spectrum disorders using clozapine to find out the impact of the ratio of clozapine and norclozapine on cognitive abilities. We determined the quality of the studies. We checked for factors like age, gender, smoking, ethnicity, and genetics that could affect this ratio. We also checked for factors that influence cognitive abilities such as years of education, duration and severity of mental problems, and duration of clozapine treatment.
We found fifteen studies, but in only two small reliable studies important cognitive areas were tested at two different points in time. We found no solid proof that the ratio of clozapine and norclozapine in the blood directly affects cognitive abilities. Factors like the clozapine dose, timing of blood tests, other medications, and individual differences in drug metabolism were often not considered.
At present, the ratio of clozapine and norclozapine is not useful for tailoring clozapine treatment to individual patients in order to improve cognitive impairments.
Keywords
Introduction
Clozapine
Approximately 30% of patients with schizophrenia are affected by treatment resistance. 1 Treatment resistance entails a confirmed diagnosis of schizophrenia based on validated criteria, showing persistence of significant symptoms to at least two different nonclozapine antipsychotics taken at therapeutic dose and duration (⩾6 weeks) with confirmed adherence (using serum antipsychotic levels). 2 Clozapine (CLO) is the only antipsychotic medication with a beneficial effect on positive symptoms in patients with treatment-resistant schizophrenia (TRS) due to its unique receptor-binding profile. 3 However, its effect on negative symptoms and cognitive deficits, most crucial for psychosocial functioning and quality of life in schizophrenia spectrum disorder (SSD), 4 is inconsistent.
Clozapine and cognitive functioning
Three meta-analyses of critically low and low methodological quality showed contradictory effects of CLO on various cognitive domains compared to other antipsychotic medications (Table 1).5–8 We used the Assessing the Methodological Quality of Systematic Reviews (AMSTAR) tool to appraise the quality of the meta-analyses and to determine the critical domains. 9 The first meta-analysis by Woodward et al. found a beneficial effect of CLO on the global cognitive index, learning, delayed recall, processing speed, verbal fluency and motor skills. 5 Nielsen et al. reported a significant beneficial effect of CLO on verbal fluency. 6 A head-to-head comparison between CLO and ziprasidone showed an inferior effect of CLO on verbal working memory. Baldez et al. found inferior effects of CLO on multiple cognitive domains (executive functioning and visuoconstruction) and the worst outcomes for CLO on working memory, verbal learning and the composite score. 7 A recent high-quality meta-analysis by Cheuk et al., comparing cognitive performances before and after CLO treatment in patients with TRS, found inconsistent effects on overall cognitive functioning due to high between-study heterogeneity. 8 However, a younger age, more years of education, improvement in positive symptoms and CLO treatment duration of at least 6 months were associated with a significant improvement in overall cognitive functioning.
Bold: significant; −: negative outcome compared to control; +: positive outcome compared to control; ♦: positive outcome was not significant after correction for publication bias.
Double-blind and open-label studies.
A, attention; AMSTAR, assessing the methodological quality of systematic reviews; BD, bipolar disorder; CA DS, complex attention digit symbol; CA TMT-A, complex attention trail making test-A; CoF, cognitive flexibility; CLO, clozapine; CoS, composite score; DR, delayed recall; EF, executive functioning; FGA, first-generation antipsychotics; GCI, Global Cognitive Index; k, number of studies with clozapine treatment; La, language; Le, learning; LeM, learning and memory; LTNV, long term non-verbal; LTVWM, long-term verbal working memory; MS, motor skills; n, number of individuals included in the study arm with clozapine; negative ES, more negative effect of clozapine compared to control; positive ES, more positive effect of clozapine compared to control; PS, processing speed; SGA, second-generation antipsychotics; SSD, schizophrenia spectrum disorders; TRS, treatment-resistant schizophrenia; VC, visuoconstruction; VF, verbal fluency; VL, verbal learning; VP, visuospatial processing; V/SA, vigilance and selective attention; VSS, visual spatial skills; VWM, verbal working memory; WM, working memory; Zipr, ziprasidone.
Methodological reasons for (critically) low-quality appraisals of the first three meta-analyses are enclosed in Table 1. In all four meta-analyses the quality of information was impaired due to missing values for relevant demographic and clinical variables in publications of included studies (Table 2). The treatment duration of the third meta-analysis was relatively short, and it varied widely in the other meta-analyses. In the first three meta-analyses cognitive comparisons were conducted between patients with TRS and unmedicated patients, patients who responded to regular antipsychotic treatment and patients with bipolar disorders, which are not valid. A plausible explanation for poorer response rates in TRS patients is delay of appropriate CLO treatment, often associated with trials of antipsychotic polypharmacy (APP). 10 Early intervention with CLO is vital to enhance chances of achieving favourable long-term outcomes and prevent further cognitive decline, especially in adolescents with the onset of psychotic symptoms in the critical period of prefrontal maturation with synaptic pruning. 11 The fourth meta-analysis was most informative, because the study population consisted of TRS patients before and after CLO initiation. Moreover, while study populations in all four meta-analyses were highly heterogeneous, Cheuk et al. conducted a weighted analysis to determine the heterogeneity of the study population.
−: Missing data; +: included data; ±: overall measures (mean, median, range), but no specific values mentioned in publications of included studies.
CLO, clozapine; NCLO, norclozapine.
Mechanisms of action on cognitive functioning
In vitro and in vivo preclinical findings on CLO point to multiple and possibly overlapping mechanisms of action (MOA) which might be responsible for its superior efficacy among antipsychotics. However, these MOAs contributing to its unique effect have not been fully understood. 12 While several pharmacological properties stem from the parent compound CLO, a considerable number of effects distinctly originate from the primary active metabolite NCLO (Figure 1). 13 NCLO may contribute to CLO’s clinical effects by modulating both muscarinic and glutamatergic neurotransmission.14,15 CLO and NCLO share similar receptor-binding sites. However, they act differently, even in opposite ways, at these sites (Table 3).12,16–19 More specifically, NCLO is a partial muscarinic (M)1 and M4 receptor agonist,12,16,20,21 which might explain possible procognitive effects. 12 This hypothesis is substantiated by recent positive outcomes in a phase II clinical trial involving xanomeline, an M1–M4 agonist, in conjunction with the peripheral anticholinergic agent trospium. 22
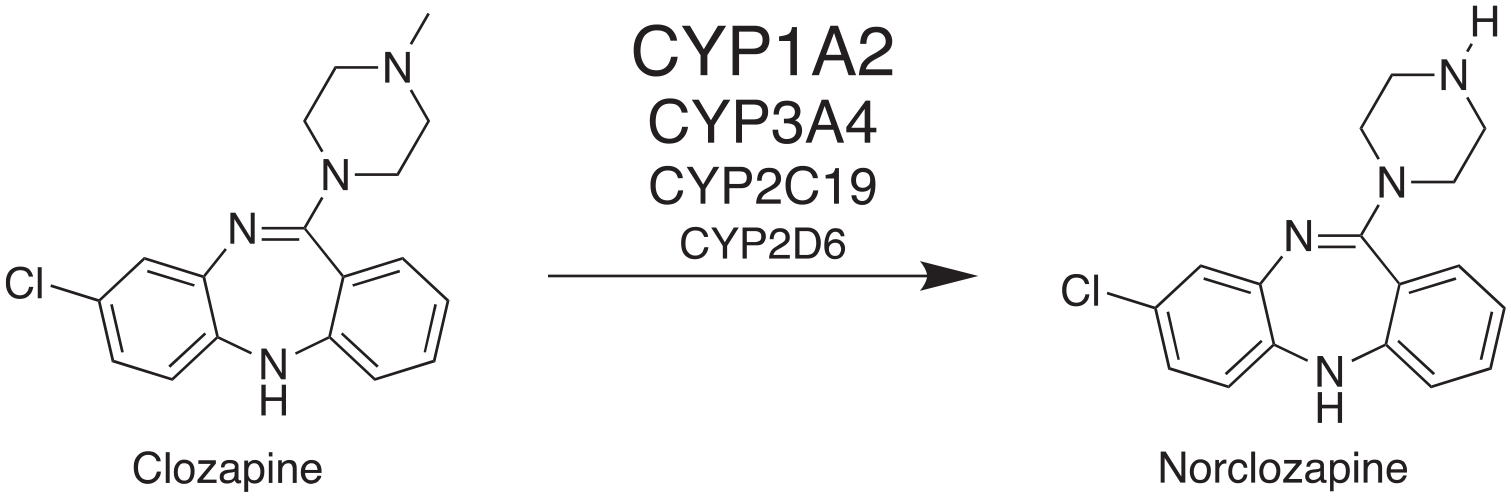
Metabolisation of clozapine through demethylation mainly by CYP1A2, CYP3A4 and to a lesser extent by CYP2C19 and CYP2D6 to the pharmacologically active metabolite norclozapine.
δ, opioid; 5-HT, serotonin; CLO, clozapine; D, dopamine; H, histamine; M, muscarinic; NCLO, norclozapine.
While the procognitive effects of CLO are possibly due to enhanced glutamatergic neurotransmission with prolonged single-channel open time of reconstituted α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) receptors, 23 the potent antagonistic activity of CLO on the M1 receptor may contribute to the anticholinergic burden that negatively impacts global cognition or specific cognitive domains such as working memory24,25 or executive functioning, and the combined domains memory/fluency and processing speed/vigilance. 26 The opposing muscarinic effects of CLO, NCLO, and their interplay may potentially explain the inconsistent cognitive effects associated with CLO.
However, differences in receptor binding affinity of psychotropic medications are generally the foundation of hypothesised variations in efficacy. Unfortunately, in clinical practice these theoretical variations are often not demonstrated in direct comparisons with significant differences in efficacy.
Objectives
This is the first systematic review of the correlation between the CLO/NCLO ratio and cognitive performance in patients with SSD. We anticipated a negative correlation between CLO/NCLO ratio and cognitive functioning given the activation of M1 and M4 muscarinic receptors by NCLO. We studied the association between the CLO/NCLO ratio and specific cognitive domains. Furthermore, we explored the predictive value of response variables such as the severity of illness, CLO treatment duration, CLO dosage regimen and blood sampling interval, the anticholinergic burden of CLO and concomitant medication and genotyping.
Methods
We conducted this systematic review according to the preferred reporting items for systematic reviews and meta-analyses guidelines 27 and we preregistered the protocol in PROSPERO (pre-registered protocol ID: CRD42023385244 (PROSPERO, 2023)). We deviated from this protocol and broadened the initial literature search to decrease the chances of missing relevant studies.
Systematic search and information sources
We systematically searched the electronic databases Embase, PsycINFO, PubMed, Cochrane and the Cochrane Controlled Register of Trials with MeSH search terms ‘clozapine’ AND ‘norclozapine’ AND ‘ratio’ AND ‘cognition / neurocognition / cognitive / neurocognitive’ (7 May 2023, updated at 12 January 2024). We broadened the initial search by omission of the MeSH term ‘ratio’ (1 August 2024). Eligibility criteria were: (1) studies published in peer-reviewed journals; (2) enrolled patients were diagnosed within the spectrum of schizophrenia disorders by The Diagnostic and Statistical Manual of Mental Disorders (3rd ed., revised.; DSM-III-R) 28 or subsequent editions, or the International Statistical Classification of Diseases and Related Health Problems (9th ed., ICD-9) 29 or subsequent editions; (3) enrolled patients were 18 years old or above; (4) enrolled patients received clozapine treatment; (5) CLO/NCLO plasma levels ratio was assessed; (6) neuropsychological assessment was conducted during CLO treatment; (7) assessment of CLO/NCLO plasma levels ratio and cognitive functioning within an interval of 1 month without any changes in CLO dosage. Exclusion criteria included unpublished articles, animal studies, case studies, case series and studies with non-quantitative outcome measures. There were no restrictions on language or publication year. Specific search queries per database are documented in Supplemental Table 1.
Study selection and data collection process
We included cross-sectional and longitudinal studies focusing on at least one domain of cognitive function in adult patients with SSD treated with CLO. The search for additional eligible studies was extended to the reference lists of included studies. The references cited in the selected studies were analysed in the same way. Abstracts were independently assessed for relevance by two researchers. Any disagreement as regards eligibility was discussed until agreement was reached. Rayyan.ai was used as a software system for recording decisions concerning the selection. 30 After the full article had been examined, relevant references relating to the effect of the CLO plasma level and CLO/NCLO plasma level ratio on cognition in patients with SSD were selected. The results were analysed independently by the same two researchers. Any disagreement as regards the results was discussed until agreement was reached. The full articles were independently assessed for certainty and risk of bias (RoB), using the Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies of the National Institutes of Health 31 and the Joanna Briggs Institute Critical Appraisal Checklist, 32 respectively. In case of disagreement, consensus was reached through discussion.
Results
Study selection
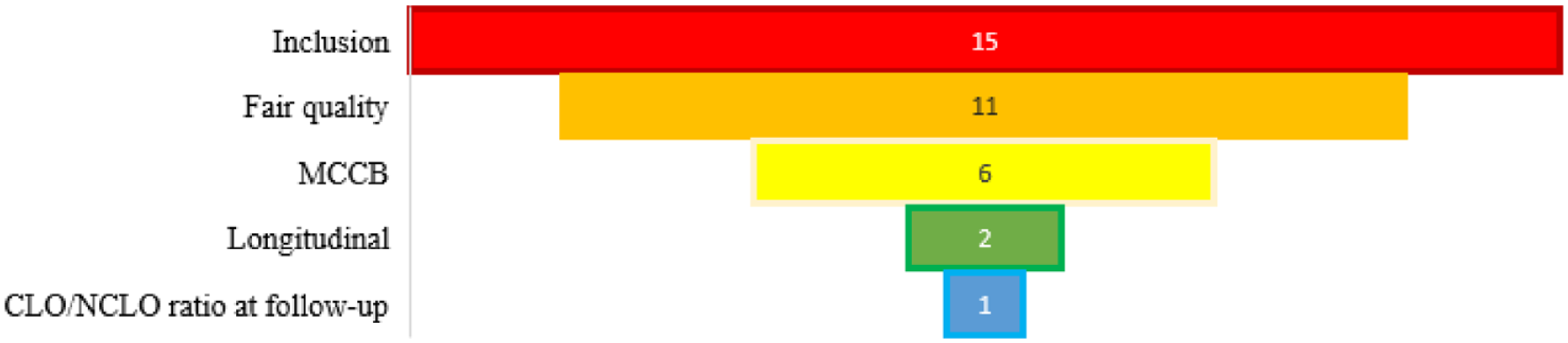
Our search strategy yielded 234 references (Figure 2). After removing 57 duplicates, 177 articles were screened based on assessment of cognitive functioning and available data for the computation of CLO/NCLO plasma level ratios. We excluded 163 references because they provided insufficient information on cognitive parameters or CLO/NCLO levels (Supplemental Table 2). One article was found after backwards citation searching. The total of 15 studies were included and analysed (Figure 3).

Flowchart according to the PRISMA guidelines. 24
Analysis of included studies on cognitive functioning and CLO/NCLO plasma level ratio.
Study characteristics
Four studies were longitudinal20,33–35 and 11 cross-sectional15,19,24,36–44 (Table 4). The study populations of the included studies (n = 593) with a sample size of 11–92 consisted of adult patients (mean age between 34.14 and 49.5 years, k = 12) with SSD being treated with CLO and with different degrees of therapy resistance. Mean severity of illness during CLO treatment was documented in 11 studies and varied from minimally ill to severely ill. The mean duration of illness was mentioned in only five studies and varied from 12 weeks to 16.1 years. Antipsychotic medication regimes consisted of CLO monotherapy (MT) in seven studies, CLO MT or APP in six studies and were not specified in two studies (Table 5). In one study fluvoxamine was specifically mentioned as additional therapy to increase the CLO/NCLO ratio. 39 In nine studies the CLO treatment duration varied from a minimum of 4 weeks on a stable CLO dose to a maximum of at least 24 weeks. The mean CLO level varied between 135.00 and 403.9 µg/L in 10 studies. The dosage regimen was once-daily in four studies and the blood sampling interval was approximately 12 h in five studies. The CLO/NCLO ratio varied from 1.28 to 2.40 in 11 studies.
I: follow-up duration ⩾ 6 months; II: follow-up duration nm; III follow-up duration ⩾30 days; IV: follow-up duration 52 weeks.
BL, baseline; BPRS, Brief Psychiatric Rating Scale; CGI-P, Clinical Global Impression for Schizophrenia positive subscale; CLO, clozapine; CRS, clozapine-resistant schizophrenia; CS, cross-sectional; DD, delusional disorder; FU, follow-up; L, longitudinal; NCLO, norclozapine; nm, not mentioned; PANSS, Positive and Negative Syndrome Scale; PD, psychotic disorder not otherwise specified; S, schizophrenia; SSD, schizophrenia spectrum disorder; SZD, schizoaffective disorder; TRS, treatment-resistant schizophrenia; TRSZD, treatment-resistant schizoaffective disorder.
ΔT0–T1 = S: difference in CLO/NCLO ratio between BL and follow-up is significant. ΔT0–T1 = NS, difference in CLO/NCLO ratio between BL and follow-up is not significant. Bold: reliable data due to non-significant difference in CLO/NCLO ratio between BL and follow-up.
NCLO/CLO = 0.75 stated in the original article.
NCLO/CLO = 0.71 stated in the original article.
NCLO/CLO = 0.416 stated in the original article.
APP, antipsychotic polypharmacy; BL, baseline; CLO, clozapine; FLUV, fluvoxamine; FU, follow-up; MT, monotherapy; NCLO, norclozapine; nm, not mentioned; ns, not specified; S, schizophrenia.
Study quality
Quality appraisal of included studies
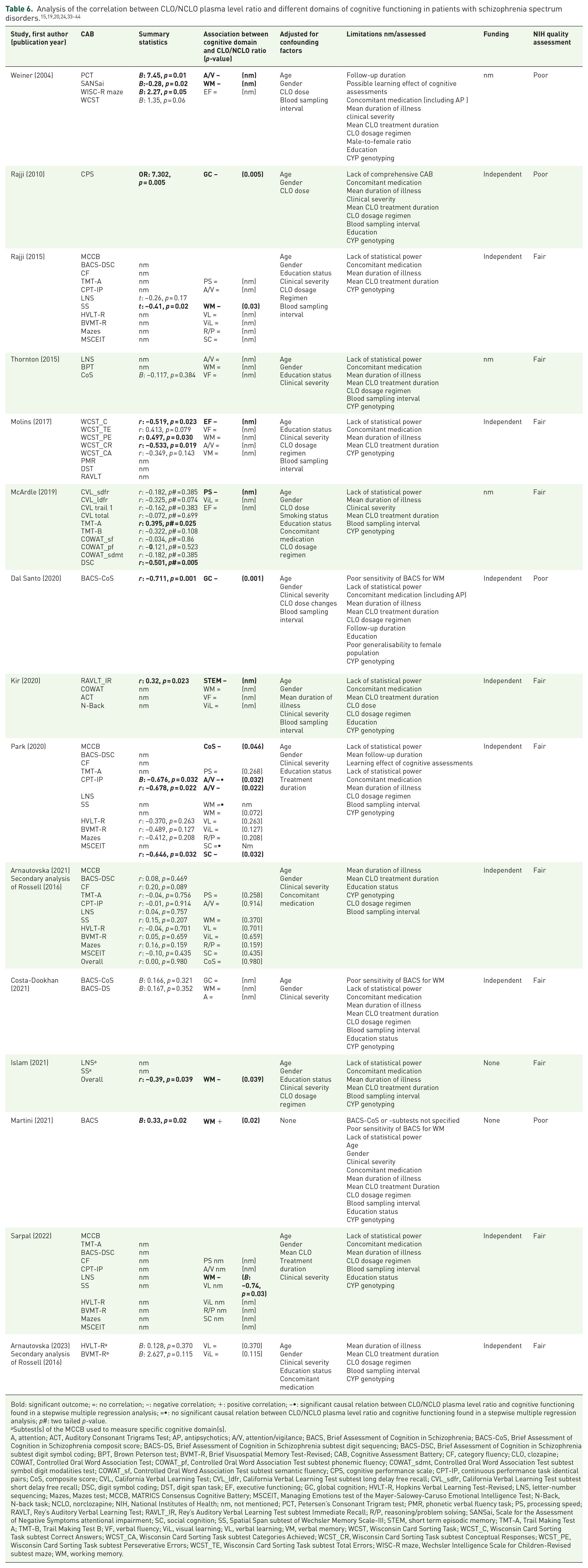
We found 11 fair-quality15,19,24,34,35,37–41,43,44 and four poor-quality studies (Table 6).20,33,36,42
Bold: significant outcome; =: no correlation; −: negative correlation; +: positive correlation; −•: significant causal relation between CLO/NCLO plasma level ratio and cognitive functioning found in a stepwise multiple regression analysis; =•: no significant causal relation between CLO/NCLO plasma level ratio and cognitive functioning found in a stepwise multiple regression analysis; p#: two tailed p-value.
Subtest(s) of the MCCB used to measure specific cognitive domain(s).
A, attention; ACT, Auditory Consonant Trigrams Test; AP, antipsychotics; A/V, attention/vigilance; BACS, Brief Assessment of Cognition in Schizophrenia; BACS-CoS, Brief Assessment of Cognition in Schizophrenia composit score; BACS-DS, Brief Assessment of Cognition in Schizophrenia subtest digit sequencing; BACS-DSC, Brief Assessment of Cognition in Schizophrenia subtest digit symbol coding; BPT, Brown Peterson test; BVMT-R, Brief Visuospatial Memory Test-Revised; CAB, Cognitive Assessment Battery; CF, category fluency; CLO, clozapine; COWAT, Controlled Oral Word Association Test; COWAT_pf, Controlled Oral Word Association Test subtest phonemic fluency; COWAT_sdmt, Controlled Oral Word Association Test subtest symbol digit modalities test; COWAT_sf, Controlled Oral Word Association Test subtest semantic fluency; CPS, cognitive performance scale; CPT-IP, continuous performance task identical pairs; CoS, composite score; CVL, California Verbal Learning Test; CVL_ldfr, California Verbal Learning Test subtest long delay free recall; CVL_sdfr, California Verbal Learning Test subtest short delay free recall; DSC, digit symbol coding; DST, digit span task; EF, executive functioning; GC, global cognition; HVLT-R, Hopkins Verbal Learning Test-Revised; LNS, letter-number sequencing; Mazes, Mazes test; MCCB, MATRICS Consensus Cognitive Battery; MSCEIT, Managing Emotions test of the Mayer-Salowey-Caruso Emotional Intelligence Test; N-Back, N-back task; NCLO, norclozapine; NIH, National Institutes of Health; nm, not mentioned; PCT, Petersen’s Consonant Trigram test; PMR, phonetic verbal fluency task; PS, processing speed; RAVLT, Rey’s Auditory Verbal Learning Test; RAVLT_IR, Rey’s Auditory Verbal Learning Test subtest Immediate Recall; R/P, reasoning/problem solving; SANSai, Scale for the Assessment of Negative Symptoms attentional impairment; SC, social cognition; SS, Spatial Span subtest of Wechsler Memory Scale-III; STEM, short term episodic memory; TMT-A, Trail Making Test A; TMT-B, Trail Making Test B; VF, verbal fluency; ViL, visual learning; VL, verbal learning; VM, verbal memory; WCST, Wisconsin Card Sorting Task; WCST_C, Wisconsin Card Sorting Task subtest Correct Answers; WCST_CA, Wisconsin Card Sorting Task subtest Categories Achieved; WCST_CR, Wisconsin Card Sorting Task subtest Conceptual Responses; WCST_PE, Wisconsin Card Sorting Task subtest Perseverative Errors; WCST_TE, Wisconsin Card Sorting Task subtest Total Errors; WISC-R maze, Wechsler Intelligence Scale for Children-Revised subtest maze; WM, working memory.
No studies with a high RoB were included (see Supplemental Table 3 for RoB scores <50% for all selected studies). 32
Quality of cognitive tests
Various cognitive assessment batteries (CABs) were used to assess different cognitive domains or a composite score (Table 6). The MATRICS Consensus Cognitive Battery (MCCB), which was specially developed and validated to assess cognitive treatment effects in clinical trials involving patients with schizophrenia, measures cognitive performance in seven domains: processing speed, attention/vigilance, working memory, verbal learning, visual learning, reasoning and problem solving and social cognition. 45 The test–retest reliability of the MCCB composite scores is considered excellent and the learning effects are small. 46 The MCCB has become the gold standard for examining cognitive performances in patients with schizophrenia.
Interpretation of cognitive functioning
In Table 7 the results of the correlations of the CLO/NCLO ratio and different cognitive domains in patients with SSD are summarised.15,19,20,24,33–44 Inter-study comparisons of the nine cross-sectional studies were not feasible due to the lack of information on dosage regimens and heterogeneity of study designs and populations. On close examination of the four longitudinal studies, only two were found to be of fair quality and had used the MCCB or subtests of CAB to assess cognitive functioning.34,35 In only one study, by Park et al., could a potential causal effect of the CLO/NCLO ratio on cognitive functioning be examined. A multiple stepwise regression analysis was restricted to attention/vigilance, social cognition and working memory, because changes between baseline and follow-up were significant in these single three cognitive domains. 34 For attention/vigilance a significant negative causal relationship was found, but for social cognition and working memory the causal relationship was not significant after a mean follow-up of 30 days. A negative correlation was found between CLO/NCLO plasma level ratio and social cognition and composite score. The second longitudinal study of fair quality, by Arnautovska et al., found no significant differences in verbal learning or visual learning after a mean follow-up of 1 year. 35 Therefore, a causal relationship could not be determined.
=: no correlation; −: a negative correlation; +: a positive correlation; −•: a significant negative causal relation, determined by a stepwise multiple regression analysis; =•: no significant causal relation, determined by a stepwise multiple regression analysis; ‘empty’: not assessed.
A, attention; CLO, clozapine; EF, executive functioning; NCLO, norclozapine; nmo, no mention of assessed outcome; STEM, short term episodic memory; VF, verbal fluency; VM, verbal memory.
Discussion
Given the limited amount of research on the correlation between CLO/NCLO plasma level ratio and cognitive functioning, we decided to analyse literature on the effects of CLO/NCLO plasma level ratio on global cognition and specific cognitive domains.
Cognitive effects of CLO/NCLO ratio
In only one study could a causal relationship be determined between CLO/NCLO plasma level ratio and cognitive functioning. 34 The most important outcomes were a negative causal relationship for attention/vigilance and no causal relationship for social cognition and working memory with initial assessments performed within 30 days and follow-up assessments performed more than 60 days after initiation of CLO.
Limitations
Study design
We found only two longitudinal studies of fair quality, with relatively small sample sizes of 11 and 43.34,35 Moreover, the minimum time to follow-up after CLO treatment initiation was short (merely 60 days) in the single study which showed a causal relationship between the CLO/NCLO ratio and cognitive domain attention/vigilance. 34 It is conceivable that the effect of CLO on all symptoms of schizophrenia, including other cognitive domains than attention/vigilance, increases after a longer CLO treatment duration than 60 days. Moreover, after a relatively short follow-up, there is a risk of cognitive bias due to improvement in positive symptoms as a result of CLO treatment.
Another major limitation was the lack of systematic analysis of the effects of the CLO/NCLO ratio on all seven cognitive domains. Unfortunately, the complete MCCB or subtests of this CAB were used in only six studies.15,24,34,35,41,43,44
In all included studies, key moderators were not documented, such as CLO treatment duration (k = 6), mean CLO dosage (k = 5), CLO dosage regimen (k = 11), blood sampling interval (k = 10), and CLO/NCLO ratio (k = 4) and concomitant medication (k = 12) (Table 6). These missing data complicate the interpretation of the results and response variables.
Risk of bias
The quality of data synthesis depends on uniform reporting of study parameters and outcomes. Therefore, the analysis was limited by missing and incomplete data on demographic and clinical variables (Table 6). Although no studies with high RoB were selected (Supplemental Table 3), publication bias could not be excluded. The results of this review may be biased by selective reporting of positive findings, which may lead to overestimation of the impact of the CLO/NCLO ratio on cognitive performances.
Severity and duration of illness
Unfortunately, in 10 studies the mean duration of illness was not mentioned. In the two longitudinal studies of fair quality, patients were severely ill (Positive and Negative Syndrome Scale (PANSS) 77.92 and 73.9, respectively).34,35 It is hypothesised that the severity of illness might override any procognitive effects driven by NCLO.19,37 Furthermore, a beneficial effect of CLO on other symptom domains of schizophrenia may also positively affect the cognitive CLO response. 37 However, in 14 of the studies included, CLO response was not assessed using the PANSS or Brief Psychiatric Rating Scale. Unfortunately, Arnautovska et al. did not assess symptom severity at follow-up after 1 year. 35 Park et al. was the only study in which the CLO response on all symptoms of schizophrenia was assessed using the PANSS. 34 At follow-up no difference in PANSS total score was found. CLO resistance may explain the lack of effect on other cognitive domains than attention/vigilance. Although severely ill patients showed CLO resistance, a negative causal relationship was still found between CLO/NCLO ratio and attention/vigilance.
CLO/NCLO plasma level ratio ΔT0–T1
Unfortunately, the difference in CLO/NCLO ratio was not specified in the longitudinal study by Arnautovska et al., which complicated interpretation of the absence of correlations. 35 The smaller the differences are between CLO/NCLO plasma level ratio at baseline and follow-up, the greater the risk of type II errors.
Dosage regimen and blood sampling interval
CLO has a shorter terminal half-life than NCLO and therefore NCLO plasma levels are more stable than the CLO plasma levels. 47 Hence, the dosage regimen modulates the CLO/NCLO ratio: 12 h after CLO intake the CLO plasma level reaches a higher peak relative to NCLO in once-daily dosing than in twice-daily dosing. Therefore, in a once-daily dosage regimen, the CLO/NCLO ratio measured 12 h after CLO intake is higher compared to a twice-daily dosage regimen.48,49 Because CLO dosage regimens and the interval between CLO ingestion and blood sampling (e.g. a 12-h interval or a 24-h interval) were not systematically analysed, inter-study comparisons (and possibly even within-study comparisons) are not valid. Additionally, the timing of cognitive assessments after CLO intake was not mentioned. Consequently, there may be substantial interindividual variability in the level of sedation during the cognitive tests.
Genetic variation in anticholinergic burden
Genetic variation affects the anticholinergic burden of CLO and concomitant medication. M1 receptor agonism and antagonism is hypothesised to impact certain cognitive domains (Table 3).7,12 Islam et al. studied the correlation between CLO/NCLO plasma level ratio and working memory in patients with SSD (n = 30) and found a negative association only in patients with higher transcription of the M1 receptor (non-carriers of the haplotype T-A of the cholinergic M1 gene) compared to patients with lower transcription of the M1 receptor (carriers of the haplotype T-A of the M1 gene). 15 They found that higher expression of the M1 receptor results in increased sensitivity to the CLO/NCLO plasma level ratio. Therefore, a small variation in the CLO/NCLO ratio can more easily result in a detectable change in working memory performance. A mediating effect of CYP1A2 enzyme expression was ruled out.
In the study by Park et al., the anticholinergic burden of concomitant medication was not considered. 34 Significant sedation may overshadow any procognitive effects of NCLO, potentially diminishing the impact of the CLO/NCLO ratio on cognitive functioning. This could result in type II errors for the cognitive domains problem solving, visual learning, reasoning/problem solving and working memory. Unfortunately, M1 receptor transcription was not assessed and sensitivity to changes in CLO/NCLO ratio were not determined.
Future clozapine research
Prospective longitudinal trials are needed to elucidate the value of CLO/NCLO ratio in predicting cognitive improvement in patients with SSD. Future research should entail pretreatment cognitive assessment, a validated standardised test battery (i.e. MCCB), long-term follow-up and a focus on changes in cognitive functioning at an individual level rather than at an aggregate level. Important covariates are education status, duration and severity of illness, CLO treatment duration, CLO titration scheme and experienced sedation level. Gradual titration can avoid confounding due to sedation or secondary negative symptoms. The anticholinergic burden due to concomitant medication also influences cognitive functioning.
For a valid interpretation of the CLO/NCLO ratio, the CLO dosage regimen and blood sampling interval are important. Sources of variability in pharmacokinetics of CLO that affect CYP 1A2 metabolism are smoking behaviour (polycyclic aromatic hydrocarbons in cigarette smoke induce CYP1A2), 50 caffeine consumption (CYP1A2 substrate), 50 and oestrogen and progesterone levels (inhibit CYP1A2). 51 Excessive smoking is correlated with reduced plasma levels of both CLO and NCLO. 52 Coffee consumption is correlated with increased CLO and NCLO plasma levels and negatively correlated with the CLO/NCLO plasma level ratio. In premenopausal women CLO levels are higher compared to postmenopausal women. 51
Pharmacokinetic drug-drug interactions are caused by the inhibition or induction of CYPs. 53 Fluvoxamine increases the CLO/NCLO ratio through inhibition of the CYP1A2 enzyme. The demethylation of CLO to NCLO is reduced, resulting in an increase of the CLO plasma level and a decrease of the NCLO plasma level. 54
Pro-inflammatory cytokines interleukin (IL)-6 and IL-1 inhibit CYP1A2, which can be detected indirectly using the biomarker C-reactive protein (CRP). 55 Furthermore, CLO is highly bound to the acute phase protein alpha-1-acid glycoprotein (AGP) and less to serum albumin (95% of CLO binds to AGP). 56 During inflammation, concentrations of AGP increase, resulting in a decreased CLO unbound fraction. Pharmacogenetics also affects pharmacokinetics through CYP genotyping (Asian ancestry is associated with lower CYP1A2 activity compared to other ethnic groups), and transcriptional activity of the M1 receptor gene, which is correlated with increased sensitivity to the CLO/NCLO ratio. 15
Finally, CLO clearance is affected by age, sex, ethnicity and smoking status. 57 Higher age results in a reduction in CLO clearance for both male and female smokers en non-smokers of all ethnicities. Females have decreased CLO clearance compared to males. Patients of Afro-Caribbean ethnicity have increased CLO clearance, whereas patients of Asian ethnicity have decreased CLO clearance compared to their White counterparts. Additionally, smokers have increased CLO clearance compared to non-smokers.
Experts in clozapine-resistant schizophrenia hypothesise that the CLO/NCLO ratio is modulated by renal function. Gemfibrozil is used in the treatment of hypercholesterolemia. In one published case study the CLO/NCLO plasma level ratio was inverted due to gemfibrozil addition. 58 It is hypothesised that gemfibrozil increases NCLO plasma levels by inhibiting the renal transporter that excretes NCLO, while leaving the demethylation process of CLO untouched, and therefore the CLO plasma levels unaltered.
Personalised clozapine treatment
Hypothetically, the CLO/NCLO plasma level ratio is a target for personalised medicine to improve symptoms of SSD and limit side effects. 59 However, currently there is insufficient evidence to add fluvoxamine as an indirect cognitive enhancer in CLO users. In this systematic review, we focused on the limited amount of literature on cognitive effects. In a small low-quality meta-analysis (n = 120) a positive correlation was found between CLO levels and elevated triglycerides, heart rate and overall combined adverse drug reaction. 60 Furthermore, a positive correlation was found between NCLO plasma levels and triglycerides, cholesterol and weight gain. In short, at present there is too little evidence to identify the role of the CLO/NCLO plasma level ratio in the minimisation of CLO side effects.
Conclusion
While a negative correlation between CLO/NCLO plasma level ratio and cognitive functioning was hypothesised, there was a lack of robust evidence for a procognitive effect of a relatively low CLO/NCLO ratio. The ratio for the optimal balance between cognitive efficacy and tolerance is yet to be established. Ideally, future studies should be prospective and longitudinal, and should include pretreatment cognitive assessment using the MCCB, with a follow-up duration of at least 6 months after CLO initiation, a description of the calculation of the CLO/NCLO plasma level ratio and an analysis of confounding factors. However, in clinical practice, research on CLO users is complicated due to the severity of illness and noncompliance, in particular before CLO initiation. This might explain the absence of pretreatment cognitive assessment in most longitudinal studies. Large longitudinal cohort studies in patients with SSD are only feasible if clinicians focus more on neuropsychological assessments. After all, cognitive functioning is one of the most important factors for the quality of life.
Supplemental Material
sj-doc-1-tpp-10.1177_20451253241302603 – Supplemental material for Clozapine/norclozapine plasma level ratio and cognitive functioning in patients with schizophrenia spectrum disorders: a systematic review
Supplemental material, sj-doc-1-tpp-10.1177_20451253241302603 for Clozapine/norclozapine plasma level ratio and cognitive functioning in patients with schizophrenia spectrum disorders: a systematic review by Timo T. Moscou and Selene R. T. Veerman in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253241302603 – Supplemental material for Clozapine/norclozapine plasma level ratio and cognitive functioning in patients with schizophrenia spectrum disorders: a systematic review
Supplemental material, sj-docx-2-tpp-10.1177_20451253241302603 for Clozapine/norclozapine plasma level ratio and cognitive functioning in patients with schizophrenia spectrum disorders: a systematic review by Timo T. Moscou and Selene R. T. Veerman in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-3-tpp-10.1177_20451253241302603 – Supplemental material for Clozapine/norclozapine plasma level ratio and cognitive functioning in patients with schizophrenia spectrum disorders: a systematic review
Supplemental material, sj-docx-3-tpp-10.1177_20451253241302603 for Clozapine/norclozapine plasma level ratio and cognitive functioning in patients with schizophrenia spectrum disorders: a systematic review by Timo T. Moscou and Selene R. T. Veerman in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-4-tpp-10.1177_20451253241302603 – Supplemental material for Clozapine/norclozapine plasma level ratio and cognitive functioning in patients with schizophrenia spectrum disorders: a systematic review
Supplemental material, sj-docx-4-tpp-10.1177_20451253241302603 for Clozapine/norclozapine plasma level ratio and cognitive functioning in patients with schizophrenia spectrum disorders: a systematic review by Timo T. Moscou and Selene R. T. Veerman in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.