Abstract
Background:
Long-acting injectable antipsychotics (LAIs) have advantages over oral antipsychotics (OAPs) in preventing relapse and hospitalization in chronically ill patients with schizophrenia-spectrum disorders (SSDs), but evidence in patients with first-episode/recent-onset, that is, early-phase-SSDs is less clear.
Objectives:
To assess the relative medium- and long-term efficacy and safety of LAIs versus OAPs in the maintenance treatment of patients with early-phase SSDs.
Method:
We searched major electronic databases for head-to-head randomized controlled trials (RCTs) comparing LAIs and OAPs for the maintenance treatment of patients with early-phase-SSDs.
Design:
Pairwise, random-effects meta-analysis. Relapse/hospitalization and acceptability (all-cause discontinuation) measured at study-endpoint were co-primary outcomes, calculating risk ratios (RRs) with their 95% confidence intervals (CIs). Subgroup analyses sought to identify factors moderating differences in efficacy or acceptability between LAIs and OAPs.
Results:
Across 11 head-to-head RCTs (n = 2374, median age = 25.2 years, males = 68.4%, median illness duration = 45.8 weeks) lasting 13–104 (median = 78) weeks, no significant differences emerged between LAIs and OAPs for relapse/hospitalization prevention (RR = 0.79, 95%CI = 0.58–1.06, p = 0.13) and acceptability (RR = 0.92, 95%CI = 0.80–1.05, p = 0.20). The included trials were highly heterogeneous regarding methodology and patient populations. LAIs outperformed OAPs in preventing relapse/hospitalization in studies with stable patients (RR = 0.65, 95%CI = 0.45–0.92), pragmatic design (RR = 0.67, 95%CI = 0.54–0.82), and strict intent-to-treat approach (RR = 0.64, 95%CI = 0.52–0.80). Furthermore, LAIs were associated with better acceptability in studies with schizophrenia patients only (RR = 0.87, 95%CI = 0.79–0.95), longer illness duration (RR = 0.88, 95%CI = 0.80–0.97), unstable patients (RR = 0.89, 95%CI = 0.81–0.99) and allowed OAP supplementation of LAIs (RR = 0.90, 95%CI = 0.81–0.99).
Conclusion:
LAIs and OAPs did not differ significantly regarding relapse prevention/hospitalization and acceptability. However, in nine subgroup analyses, LAIs were superior to OAPs in patients with EP-SSDs with indicators of higher quality and/or pragmatic design regarding relapse/hospitalization prevention (four subgroup analyses) and/or reduced all-cause discontinuation (five subgroup analyses), without any instance of OAP superiority versus LAIs. More high-quality pragmatic trials comparing LAIs with OAPs in EP-SSDs are needed.
Trial registration:
CRD42023407120 (PROSPERO).
Plain language summary
Background: We explored whether antipsychotics long-acting injections (LAIs) might outperform regular antipsychotics pills for people dealing with early-stage conditions like schizophrenia. While LAIs have clear benefits for those with long-term challenges, their effectiveness for those just starting to grapple with these issues is less certain. Objective: We aimed to uncover whether LAIs or regular antipsychotic pills demonstrate better outcomes over the medium and long term for individuals in the early stages of schizophrenia. Method: We scrutinized several studies comparing LAIs to regular pills in treating early-stage schizophrenia. Employing a combined analysis, we assessed factors such as preventing relapses and hospitalizations, as well as patient treatment adherence. Design: We combined different study results in one unique analysis. We delved into whether LAIs surpassed regular pills in preventing relapses and hospitalizations and in patient treatment adherence. Results: In our study of 11 trials involving over 2000 participants, we observed that LAIs and regular antipsychotic pills were generally comparable regarding preventing relapses, hospitalizations, and treatment adherence. However, on closer inspection, LAIs appeared slightly more effective for specific groups in the early stages of schizophrenia. Conclusion: While LAIs and regular antipsychotic pills showed similar results for most individuals in the early stages of schizophrenia, our findings hint at the possibility that LAIs might have a slight edge for certain groups. Nevertheless, we emphasize the need for more high-quality studies to gain a clearer understanding. Registration: This study is registered under CRD42023407120 (PROSPERO).
Introduction
Schizophrenia-spectrum disorders (SSDs) are severe psychiatric conditions, which affect about 0.9% of the population, 1 with a growing incidence and prevalence in the last three decades. 2 SSDs typically have their onset during late adolescence and early adulthood, 3 and evidence suggests that early treatment is associated with better clinical outcomes compared to delayed treatment.4–6
In addition to psychosocial interventions,7–9 pharmacological treatment is critical, and relies primarily on antipsychotic medications, which are available as oral antipsychotics (OAPs) or long-acting injectable antipsychotics (LAIs).10–13
Initiating pharmacological treatment as soon as possible is crucial, as diagnosis of a first psychotic episode (FEP) is often delayed,6,14 and there is evidence that early intervention in the first 3–5 years of illness has a considerable impact on clinical trajectory.9,15 However, treatment nonadherence is a relevant clinical issue that occurs in a significant proportion of FEP patients during their early treatment phase16–19 and is related to worse clinical outcomes, including relapse and hospitalizations, lack of symptomatic remission and functional recovery, and secondary treatment resistance.20–22
Many factors are associated with medication nonadherence, including medication formulation and dosage frequency, treatment cost, denial/minimization of illness, illness duration and phase, and adverse effects. 23 LAIs have been proposed to be particularly helpful in addressing the issue of medication nonadherence and related treatment discontinuation, relapse, and rehospitalization in patients with mental disorders.11,24,25 Moreover, a recent comprehensive set of meta-analyses of three designs [i.e. randomized controlled trials (RCTs), cohort studies and mirror image studies] reported superiority of LAIs versus OAPs in all three designs for prevention of relapse and/or hospitalization in adults with SSDs independent of illness stage. 26 Additionally, LAIs were further superior to OAPs in 16.9% of the 328 meta-analyzed outcomes, with no efficacy/effectiveness outcome in which OAPs were superior to LAIs. Furthermore, while OAPs appeared to be superior, in terms of tolerability, to LAIs in 4.9% of the 328 meta-analyzed outcomes, these results were limited to adverse events where one LAI was often compared with multiple OAPs; differences in adverse event potential may have been more important than differences in formulation.27–31 When comparing the same molecules in both formulations, 97% of the 119 meta-analyzed adverse event outcomes were not significantly different. 32 Nevertheless, despite these documented benefits of LAIs for a significant population of patients with schizophrenia-spectrum disorders and increasing endorsement by clinical practice guidelines, 33 LAIs are still poorly used in clinical practice, 34 especially in patients with early-phase(EP)-SSDs. This is particularly problematic, as such patients potentially have the most to lose and/or gain in terms of functional capacity, and because nonadherence seems to be particularly high in the early illness course. 19
A systematic review and meta-analysis comparing LAIs versus OAPs in patients with EP-SSDs 35 included seven RCTs, seven observational studies and one post-hoc analysis. Results showed superiority of LAI formulations in preventing relapse compared to OAPs, but this result was lost when restricting the analyses to the four RCTs with available data. When only RCTs were considered, the authors showed an advantage of LAIs concerning the prevention of psychiatric hospitalizations in six studies. No statistically significant differences were observed regarding medication adherence and treatment acceptability. Notably, after the publication of this review (whose search dates back to 2019) three large RCTs comparing LAIs and OAPs in patients with EP-SSDs were completed,36–38 substantially increasing the amount of available evidence on the topic.
Different from Lian et al. (2022), we focused exclusively on RCTs since observational studies have methodological limitations, such as the prescribing/selection bias, the impact of possible confounders, and the absence of randomization.39,40 However, comparing LAIs and OAPs in clinical trials also comes with several challenges. LAIs are easy to track, but oral therapy compliance is more difficult to assess 41 and often overestimated. 42 This difference poses trial design difficulties, as clinical assessment can overestimate oral antipsychotic compliance, 42 and rigorous measures may boost oral antipsychotic adherence artificially. 40 Furthermore, trial environments may introduce bias, as participants in RCTs are usually less ill, have fewer comorbidities, better insight, and are more compliant, affecting results’ real-world applicability. 43
With these conceptual considerations in mind, we aimed to conduct a systematic review and meta-analysis of RCTs comparing head-to-head the efficacy, effectiveness, and safety of LAIs versus OAPs for the maintenance treatment of patients with EP-SSDs.
Methods
The protocol for this review was registered in advance with PROSPERO (International Prospective Register of Systematic Reviews) (www.crd.york.ac.uk/PROSPERO/), registration number: CRD42023407120. This review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (see Supplemental Table 1). 44
Search methods for study identification
The following string was used to identify potential records: (schizophreni* OR schizoaffective OR psychosis OR psychotic) AND (‘first episode’ OR first-episode OR first psychotic OR FEP OR ‘early phase’ OR early-phase OR ‘recent onset’ OR recent-onset OR newly) AND (‘long acting’ OR long-acting OR LAI OR depot OR injectable OR injection) AND (antipsychotic* OR neuroleptic* OR ‘dopamine antagonist’ OR ‘dopamine blocker’ OR ‘dopamine partial agonist’) AND (random* OR head-to-head OR RCT OR placebo). Systematic electronic literature searches were performed using the following databases (last update: 24 January 2023): MEDLINE, EMBASE, PsycINFO, CENTRAL, the Cochrane Central Register of Controlled Trials. In order to include unpublished studies, within the time frame of the electronic searches, additional hand searches were performed on the following websites: regulatory agencies [Food and Drug Administration (FDA) in the United States; the Medicines and Healthcare products Regulatory Agency (MHRA) in the United Kingdom; the European Medicines Agency (EMA) in the European Union; the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan; the Therapeutic Goods Administration (TGA) in Australia]; pharmaceutical companies producing antipsychotics; and clinical trial repositories [clinicaltrials.gov in the United States; ISRCTN and National Research Register in the United Kingdom; UMIN-CTR in Japan; ANZ-CTR in Australia and New Zealand; the World Health Organization (WHO) International Clinical Trials Registry Platform]. Finally, bibliographies of included and relevant articles were reviewed.
Type of studies and participants
We included published and unpublished patient-level and cluster/site-level RCTs in both acutely ill and stable patients with a FEP or a study-defined recent diagnosis of schizophrenia or SSDs (including schizophrenia, schizoaffective disorder, schizophreniform disorder, brief psychotic episode and delusional disorder) according to any validated diagnostic criteria, for example, the Diagnostic and Statistical Manual of Mental Disorders (DSM) 45 or the International Classification of Diseases (ICD) 46 criteria. We chose to include studies in which patients had a mean recency of onset of disease ⩽5 years. When patients with FEP or recent-onset SSDs represented a sub-population of a more inclusive trial, we used data from subgroup analyses when available. No restrictions were applied regarding the age or sex of participants, blinding status, or language of the publication.
Types of intervention
We included trials that randomized patients to at least one LAI antipsychotic according to the WHO ATC/DDD classification 47 against any OAP. Studies comparing a specific LAI against treatment-as-usual (which might also include other LAI formulations) were only included if all patients in the comparison group were treated with an antipsychotic, regardless of its formulation, considering that LAI use is generally low, especially in EP-SSDs. Since we focused on comparing differential efficacy, effectiveness and safety of LAIs versus OAPs, RCTs comparing LAIs or OAPs against placebo were not included.
Selection of studies and data extraction
We examined all titles and abstracts and obtained full texts of potentially relevant papers. Working independently and in duplicate, two reviewers (GV and AT) read the papers and determined whether they met the inclusion criteria. Disagreements were resolved by discussion or consensus with a third team member (CUC).
Data extraction was performed in agreement with the Cochrane Handbook for Systematic Reviews of Interventions, Chapter 7. 48 Two review authors (GV and AT) independently extracted the data on study design, participant characteristics, intervention details and outcome measures. Disagreements were resolved by discussion or consensus with a third team member (CUC). For continuous outcomes, the mean scores at endpoint or the mean change from baseline to endpoint, the standard deviation or standard error of these values, and the number of patients included in these analyses were extracted. For dichotomous outcomes, the number of patients randomized, reaching the outcome and leaving the study early were recorded. When outcome data were not reported, trial authors were contacted and asked to supply the data.
Outcome measures
The two co-primary outcomes were (1) study-defined relapse at study endpoint (>12 weeks) or hospitalization when relapse was not explicitly reported, and (2) all-cause discontinuation at study endpoint (i.e. acceptability).
The following secondary outcomes were also assessed: (1) study-defined relapse only (without hospitalization as a proxy measure of relapse); (2) psychiatric (re-)hospitalization; (3) total, positive and negative psychotic symptoms as measured through standardized clinical scales, i.e. mean scores on the Positive and Negative Syndrome Scale (PANSS), 49 Brief Psychiatric Rating Scale (BPRS), 50 or on any other psychosis rating scale with evidence of adequate validity and reliability; (4) illness severity through mean score at study endpoint at the Clinical Global impression (CGI)-Severity 51 scale or other validated scales to measure global symptomatology; (5) discontinuation due to adverse events (i.e. tolerability); (6) discontinuation due to inefficacy; (7) global functioning and quality of life through mean scores at study endpoint on validated functioning scales; (8) any adverse events with meta-analyzable data.
Assessment of included studies and quality of evidence
Two review authors (GV and AT) independently assessed the quality of the included RCTs using the Cochrane Risk Of Bias 2 (ROB2) tool 52 instruments consisting of five domains (bias arising from the randomization process; bias due to deviations from intended interventions; bias due to missing outcome data; bias in measurement of outcome; and bias in selection of the reported outcome). When there was disagreement, the final rating was made by consensus with the involvement of a third review author (CUC).
Studies were also assessed independently by two authors (GV and AT) with the A Study Pragmatic-Explanatory Characterization Tool-Rating (ASPECT-R). 53 The ASPECT-R tool is designed to rate studies along the explanatory-pragmatic spectrum and consists of six domains: (1) Participant eligibility criteria, (2) Intervention flexibility, (3) Medical practice setting/practitioner expertise, (4) Follow-up intensity and duration, (5) Outcome(s), and (6) Participant adherence. All these domains are rated from 0 to 6, where 0 is considered extremely explanatory and 6 extremely pragmatic.
For each co-primary outcome, we assessed the confidence of evidence by using the GRADEpro Guideline Development Tool (GRADE), a web-based tool for summarizing and evaluating the certainty of evidence from scientific data, including systematic reviews and meta-analyses. 54
Statistical analysis
We conducted statistical analyses using RevMan 5 55 for forest plots and subgroup analyses and the R package ‘metafor’ 56 for meta-regression analyses. For continuous outcomes, we pooled the mean differences between the treatment arms at endpoint when all trials measured the outcome using the same rating scale, otherwise we pooled standardized mean differences (SMDs). If endpoint data were unavailable, change score data were employed in order to calculate the endpoint score. A modified intention-to-treat (ITT) analysis was applied, whereby all participants with at least one post-baseline measurement were represented by their last observations carried forward. For dichotomous outcomes, risk ratios (RRs) were calculated. We calculated dichotomous data on a strict ITT basis, considering the total number of randomized participants as the denominator. Where participants had been excluded from the trial before the endpoint, we assumed that they experienced a negative outcome by the end of the trial. Continuous and dichotomous outcomes were analysed using a random-effects model, with 95% CIs.
When ⩾10 studies provided data for a co-primary outcome, we assessed publication bias by visually inspecting the funnel plot, testing for asymmetry with Egger’s regression test, 57 and investigating possible reasons for funnel plot asymmetry.
For the co-primary outcomes, the following subgroup analyses were carried out: (1) type of diagnosis of participants (schizophrenia-only versus schizophrenia-spectrum patients); (2) first episode of disease within <2 years versus first episode of disease within ⩾2 years; (3) clinically stable patients versus not clinically stable patients. The classification of clinical stability depended on how patients were defined by the study authors. When not clearly stated, we used validated severity psychotic symptomatology cutoffs: BPRS: ⩽44; PANSS: ⩽78; CGI-S ⩽458,59; (4) patients switched to another antipsychotic versus patients maintaining their pre-randomization therapy; (5) first-generation LAI versus second-generation LAI; (6) compliance measured with a proxy measure (drug blood levels, pill counts, pharmacy records, caregiver involvement etc.) versus studies without a proxy measure; (7) OAPs selected according to previous treatment history versus not selected according to previous treatment history; (8) high risk of bias at ROB2 versus not high risk of bias at ROB2; (9) Pragmatic according to median ASPECT-R score of included studies (⩽21.8) versus explanatory (>21.8); (10) strict ITT analysis versus not strict ITT analysis; and (11) pharmaceutical company sponsorship/funding versus not pharmaceutical company sponsorship/funding.
Furthermore, we performed meta-regression analyses to assess if the following covariates acted as moderators of the observed treatment effect: mean age of included participants, % of female participants, total sample size, year of publication, follow-up duration, number of previous episodes, overall dropout rate, mean score of baseline psychotic symptomatology, mean age at onset, mean duration of illness, overall ASPECT-R score. Most of the secondary analyses were preplanned and reported in the review protocol registered on PROSPERO (CRD42023407120). However, certain preplanned analyses could not be executed due to insufficient data for the analyses, such as measuring treatment adherence, relapse at specific time points, or change in quality of life rating scales. The preplanned sensitivity analyses were carried out as subgroup analyses to enhance their informativeness. Conversely, other subgroup analyses were added only after a thorough extraction of essential study characteristic data, including strict ITT analysis versus not, OAPs selected according to previous treatment history versus not, compliance measured with a proxy measure versus not.
Finally, visual inspection of forest plots was used to investigate the possibility of statistical heterogeneity. This approach was supplemented by using the I-squared statistic, which provides an estimate of the percentage of variability due to heterogeneity rather than chance alone. An I2 higher than 50% indicated high heterogeneity. 48
Results
The literature search yielded 241 records. After excluding duplicates, 197 records were assessed at the abstract level, and 39 were assessed at the full-text level. Finally, 11 independent RCTs were included in the analyses (Figure 1), of which 10 were patient-level randomized RCTs36–38,60–67 and one was a cluster-randomized RCT. 37 The number of included participants ranged from 37 to 764 (median = 85). Patients had a median age of 25.2 years (interquartile range, IQR = 22.8–30.5 years, range = 21.5–32.6 years). The median illness duration was 45.8 weeks (IQR = 31.1, 134.3 weeks, range = 9.8–151 weeks), and 68.4% of the participants were male. The median follow-up duration was 78 weeks (IQR = 52–104 weeks, range = 13–104 weeks). Only one study 63 used a first-generation LAI (fluphenazine decanoate); all others used a second-generation LAI, such as LAI paliperidone,38,62,65,66 LAI risperidone,60,61,64,67 LAI aripiprazole 37 and both LAI aripiprazole and LAI paliperidone. 36 In seven RCTs, LAIs were compared against a mix of different OAPs37,38,60–62,64,66; in the other four trials, the comparators were oral risperidone 67 oral olanzapine 65 oral fluphenazine 63 and both oral aripiprazole and oral paliperidone. 36 Only three RCTs matched the antipsychotics in the LAI and OAP treatment arm.36,63,67 Five studies included only patients with schizophrenia, while six studies included patients with SSDs. Five studies included patients in their first psychotic episode. Five studies enrolled patients who were considered clinically stable,37,38,60,63,67 whereas six studies included patients who were deemed clinically unstable.36,61,62,64–66 A detailed description of included studies is available in Table 1. Altogether, 10 studies contributed to the outcome of relapse/hospitalization, while all 11 studies had available data concerning all-cause discontinuation/acceptability.

PRISMA flowchart of screened studies.
Design, patient, and treatment characteristics of included studies.
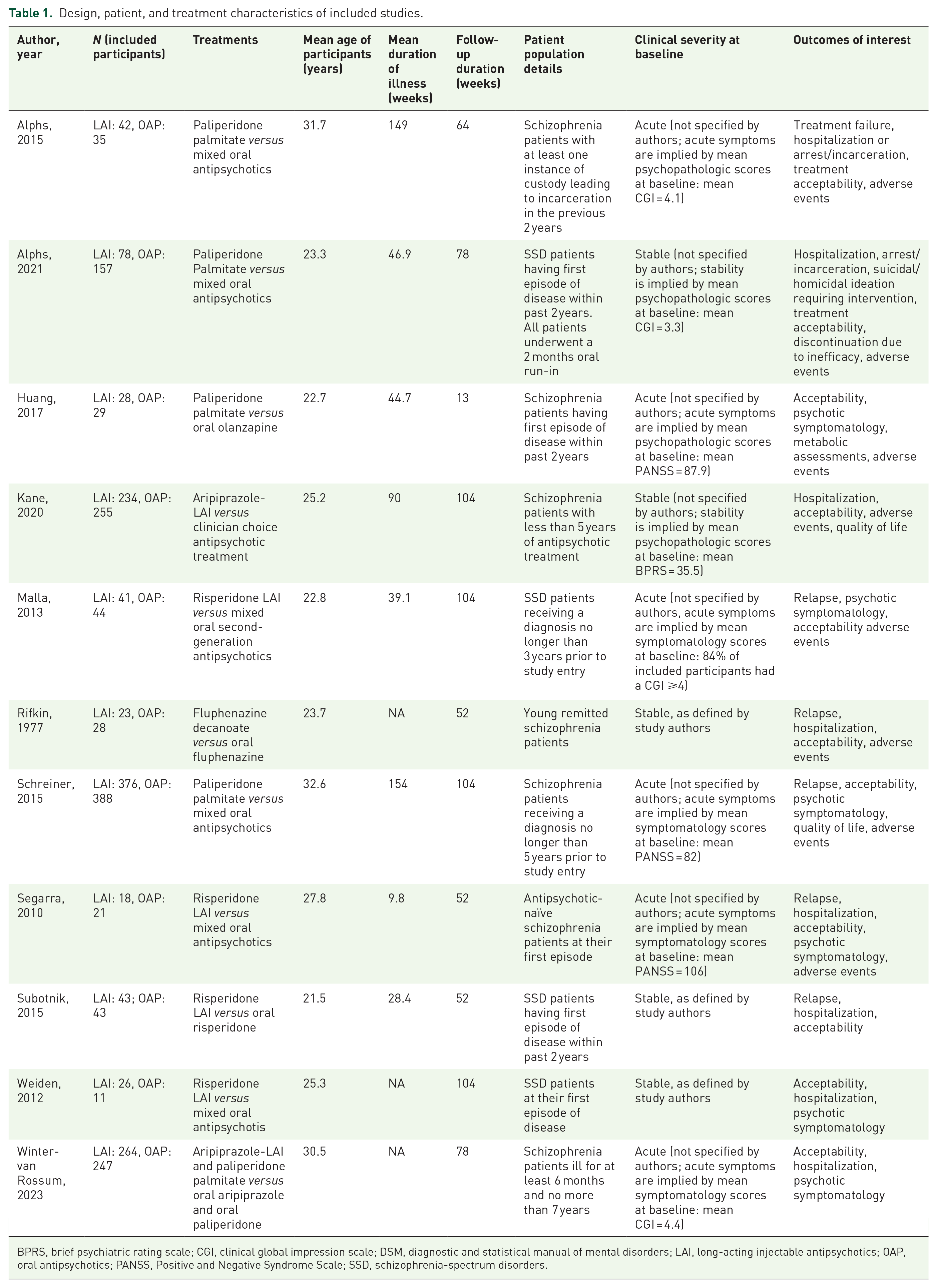
BPRS, brief psychiatric rating scale; CGI, clinical global impression scale; DSM, diagnostic and statistical manual of mental disorders; LAI, long-acting injectable antipsychotics; OAP, oral antipsychotics; PANSS, Positive and Negative Syndrome Scale; SSD, schizophrenia-spectrum disorders.
Quality of included studies
Concerning relapse/hospitalization, we found an overall poor quality of included studies, with six studies being at high risk of bias and four studies showing some concerns of bias. No studies had a low risk of bias, according to the ROB2. Critical domains were ‘Bias due to deviations from intended interventions’ (five studies scoring at high risk of bias) and ‘Bias due to missing outcome data’ (two studies scoring a high risk of bias) (see Supplemental Figure 1).
Concerning acceptability, included studies had a slightly better overall methodology, with three studies being at high risk of bias and eight studies showing some concerns of bias. No study had a low risk of bias according to the ROB2. As for relapse/hospitalization, the most critical domain was ‘Bias due to deviations from intended interventions’ (three studies scoring a high risk of bias) (see Supplemental Figure 2).
Explanatory/pragmatic rating according to ASPECT-R
Rating each study according to the ASPECT-R, the overall scores (sum of six domains rated from 0 to 6, where 0 is considered extremely explanatory and 6 extremely pragmatic, range = 0–36, median = 18) ranged from 15.5 to 28.5, with a median score of 21.8 (see Supplemental Figures 3–12).
Relapse/hospitalization rates
We did not find any significant differences between LAIs and OAPs (studies = 10, n = 2374, RR = 0.79, 95%CI = 0.58–1.06, p = 0.13, I2 = 53%) (Figure 2). The certainty in the estimate was very low according to GRADE due to high inconsistency, low internal quality and high imprecision.

Relapse/hospitalization (RR, Random effects, 95% CI).
Subgroup analyses showed that LAIs performed better than OAPs in studies including clinically stable patients (studies = 5, n = 898, RR = 0.65, 95%CI = 0.45–0.92, I2 = 31%), pragmatic studies according to ASPECT-R (studies = 5, n = 1616, RR = 0.67, 95%CI = 0.54–0.82, I2 = 0%), and studies following a strict intent-to-treat approach (studies = 6, n = 1702, RR = 0.64, 95%CI = 0.52–0.80, I2 = 3%). Results from the subgroup analyses are summarized in Table 2; forest plots of subgroup analyses are available in Supplemental Figures 13–25.
Summary results from subgroup analyses.

p-value < 0.05. **p-value < 0.01. ***p-value < 0.001.
ASPECT-R, A Study Pragmatic-Explanatory Characterization Tool-Rating; CI, confidence interval; ITT, intention-to-treat, LAI, long-acting injectable antipsychotics, OAP, oral antipsychotics, ROB2, risk of bias 2, RR, risk ratio. Significant findings are reported in bold.
All-cause discontinuation (acceptability) rates
No difference between LAIs and OAPs was observed (studies = 11, n = 2431, RR = 0.92, 95%CI = 0.80–1.05, p = 0.20, I2 = 47%) (Figure 3). The certainty of the evidence was very low according to GRADE due to high inconsistency, low internal quality and high imprecision. Subgroup analyses showed that LAIs were superior to OAPs in studies including only patients with schizophrenia (studies = 5, n = 1898, RR = 0.87, 95%CI = 0.79–0.95, I2 = 0%), studies including patients with an illness duration ⩾2 years (studies = 6, n = 1977, RR = 0.88, 95%CI = 0.80–0.97, I2 = 0%), studies including clinically unstable patients (studies = 6, n = 663, RR = 0.89, 95%CI = 0.81–0.99, I2 = 0%), studies where supplementation with OAPs in the LAI arm was not a protocol violation (studies = 7, n = 1938, RR = 0.90, 95%CI = 0.81–0.99, I2 = 19%), and studies without a long OAP run-in phase (⩾2 weeks) and where patients in the OAP arm did not maintain their pre-randomization therapy (studies = 6, n = 1224, RR = 0.84, 95%CI = 0.72–0.98, I2 = 13). Results are summarized in Table 2; forest plots of subgroup analyses are available in Supplemental Figures 13–25.

Acceptability (RR, Random effects, 95% CI).
Secondary outcomes
Relapse according to study authors definition
No statistically significant differences between LAIs and OAPs were observed regarding study-defined relapse (studies = 6, n = 1254, RR = 0.71, 95% CI = 0.39–1.30, I2 = 51%).
Hospitalizations
No differences between LAIs and OAPs were observed regarding hospitalizations (studies = 9, n = 1650, RR = 0.81, 95%CI = 0.57–1.15, I2 = 46%).
Psychotic symptoms
No differences were observed between LAIs and OAPs in reducing total psychotic symptomatology (studies = 7, n = 2132, SMD = –0.04, 95% CI = −0.12 to 0.04, I2 = 0%), positive psychotic symptomatology (studies = 6, n = 1683, SMD = −0.06, 95%CI = −0.16 to 0.04, I2 = 0%), and negative psychotic symptomatology (studies = 6, n = 1683, SMD = 0.02, 95%CI = −0.20 to 0.23, I2 = 69%).
Global illness severity
No differences were observed between LAIs and OAPs in reducing global illness severity (studies = 5, n = 1192, SMD = −0.12, 95%CI = −0.32 to 0.08, I2 = 44%).
Functioning
No differences were observed between LAIs and OAPs in improving functioning (studies = 5, n = 1192, SMD = 0.16, 95%CI = −0,06 to 0,39, I2 = 55%).
Discontinuations due to adverse events
No differences were observed between LAIs and OAPs for discontinuations due to adverse events (studies = 9, n = 1903, RR = 1.01, 95%CI = 0.76–1.34, I2 = 23%).
Discontinuations due to inefficacy
No differences were observed between LAIs and OAPs for discontinuations due to inefficacy (studies = 8, n = 1854, RR = 0.73, 95%CI = 0.40–1.33, I2 = 29%).
Adverse events
No differences were observed between LAIs and OAPs concerning prolactin increase (studies = 10, n = 2380, RR = 1.11, 95%CI = 0.68–1.81, I2 = 65%), body mass index change (studies = 4, n = 948, SMD = 0.00, 95%CI = –0.24 to 0.24, I2 = 0%), weight increase ⩾7% (studies = 10, n = 2380, RR = 1.03, 95%CI = 0.87–1.23, I2 = 20%), sedation (studies = 9, n = 1834, RR = 0.77, 95%CI = 0.43–1.36, I2 = 45%), akathisia (studies = 8, n = 1849, RR = 1.50, 95%CI = 0.84–2.67, I2 = 51%) and extrapyramidal side effects (studies = 7, 1766, RR = 1.29, 95%CI = 0.77–2.17, I2 = 53%).
Details of statistical analyses of secondary outcomes are available in the Supplemental material (see Supplemental Figures 26–40).
Meta-regression and other statistical analyses
When performing meta-regression analyses for the two co-primary outcomes, % of patients dropping out in both arms (p = 0.029) and in the oral arm only (p = 0.041) was associated with a higher risk of relapse/hospitalization. Overall, ASPECT-R scores approached statistical significance without reaching it, with higher scores (more pragmatic) associated with a stronger effect of LAIs (p = 0.06). Complete meta-regression analyses are available in the Supplemental material (Supplemental Figures 41–63), together with Egger’s regression test (Supplemental Figures 64 and 65) and a summary of findings table obtained with GRADE (Supplemental Table 2).
Discussion
This is, to the best of our knowledge, the largest and most comprehensive meta-analysis of RCTs comparing LAIs versus OAPs in adults with EP-SSDs. We did not identify significant differences between LAIs and OAPs regarding the two co-primary outcomes relapse/hospitalization prevention and acceptability. However, the GRADE assessment revealed very low confidence in our findings. This implies that we cannot exclude the possibility of a difference, particularly in favor of LAIs, as suggested by the lower confidence interval bound. Findings were similarly negative for all secondary outcomes, including relapse and hospitalization considered separately, positive and negative psychotic and global illness severity symptomatology, functioning, discontinuation due to adverse events, discontinuation due to inefficacy and pertaining to specific adverse events.
We did not confirm the results observed by Lian et al., as no significant differences between LAIs and OAPs were observed in terms of relapse/hospitalization prevention. However, the prior meta-analysis only identified positive results favoring LAIs when analyzing RCTs and observational studies together, while, in their analyses restricted to RCTs, the authors also did not find any significant difference between LAIs and OAPs in any outcome.
Nevertheless, our meta-analysis included not only four more studies and 1292 more patients than the meta-analysis by Lian et al. 2022, but we were also able to interrogate the database further with specific subgroup and meta-regression analyses that considered relevant design, patient population and treatment characteristics. While the overall analysis of 10 RCTs yielded an inconclusive finding, in four subgroup analyses LAIs were superior to OAPs, while none of the subgroup analyses favored the OAP arm.
Notably, in these methodology-guided subgroup analyses, we found that LAIs outperformed OAPs for preventing relapse/hospitalization in studies including more clinically stable patients (defined according to their baseline psychotic symptomatology). This finding was consistent with an independent patient data meta-analysis of relapse prevention RCTs by Rubio et al., 68 who found that treatment with LAIs was associated with lower relapse rates when remission was previously achieved.
Furthermore, LAIs were superior to OAPs regarding relapse in studies where the outcome was measured with a strict intent-to-treat approach. Given the high treatment discontinuation rates associated with EP-SSDs, data should be analyzed, avoiding the exclusion of randomized patients, 69 and ideally trying to verify the occurrence of relapse/hospitalization from other indirect sources, such as medical/hospital records.
Finally, when dividing the studies into two subgroups according to the median ASPECT-R overall score, LAIs were superior for relapse/hospitalization versus OAPs in more pragmatic studies, while no difference emerged in the explanatory study subgroup. As proposed previously,70,71 trials designed with a pragmatic and greater ‘real-world’ approach might be able to show greater benefits for LAIs versus OAPs, as explanatory studies generally select patients with greater illness insight, less illness severity and greater medication adherence compared with patients seen in clinical practice. 40 In fact, a recent French national database mirror image study found that LAIs only outperformed OAPs in the subgroup of patients with nonadherence (<80% of OAP dispensation) in the 1 year prior to LAI- initiation, except for aripiprazole-LAI. 72
In explanatory studies, ecologic aspects of treatment may be altered in relevant ways, such as randomizing patients directly to LAIs or OAPs without an oral lead-in phase as done in clinical practice, or truncating analyses at certain doses or medication augmentation strategy points,36,73 or by handing patients the study medication at regular research visits. 40 Furthermore, the way treatment adherence is measured in clinical trials represents a critical issue, as many included studies assessed adherence to OAPs with a proxy measure (drug blood levels, pill counts, pharmacy records, caregiver involvement), 23 thereby artificially influencing patients assigned to OAPs to be more adherent than in usual care settings. Nevertheless, we did not observe any differences concerning relapse/hospitalization prevention in the four studies not using a proxy measure to assess adherence.
Concerning acceptability, no differences were observed in the main analysis between LAIs and OAPs (p = 0.20). However, results for this outcome were strongly influenced by the findings of Alphs et al. 2021, 38 where patients in the LAI arm had significantly more drop-outs than in the OAP arm (RR = 1.80, 95%CI = 1.15–2.84). This result might have been biased by the 2 months run-in phase with oral paliperidone, which was completed by 84% of patients and might have helped to pre-select a subset of patients who were more prone to regularly take oral therapy. Furthermore, there was greater flexibility in allowing patients to remain in the OAP than the LAI arm in that oral paliperidone could have been switched to another oral antipsychotic according to clinical judgment, and there was greater flexibility concerning dose adjustments in the oral arm, with oral supplementation in the paliperidone-LAI arm not being allowed. Interestingly, when this study was excluded from the analysis, LAIs were associated with significantly lower all-cause discontinuation compared to OAPs (RR = 0.87, 95%CI = 0.79–0.95, I2 = 28%) and the heterogeneity of the results dropped from a relevant value of 53–28%.
Moreover, five subgroup analyses revealed that treatment with LAIs resulted in better acceptability than with OAPs, with none of the subgroup analyses favoring OAPs. LAIs were associated with significantly less all-cause discontinuation than OAPs in studies including patients with schizophrenia-only, ⩾2 years of illness, and clinical instability, as well as in studies where patients in the OAP were switched to a new antipsychotic, without a pre-randomization oral run-in phase of ⩾2 weeks and where supplementation with OAPs in the LAI arm was allowed, and those at low/medium risk of bias.
However, when measuring differences in acceptability, trial design characteristics may also play an important role. For example, some studies allowed patients randomized to the OAP arm to maintain their pre-randomization therapy, while patients randomized to the LAI arm were often switched to a different type of antipsychotic, which would favor persistence on the OAP arm.
Finally, Second-Generation Antipsychotics (SGA)-LAI versus First-Generation Antipsychotics (FGA)-LAI use (although only one RCT studied a FGA-LAI), using a proxy measure for adherence and pharmaceutical company sponsorship did not affect either of the two co-primary outcomes of this meta-analysis.
The results of this study need to be interpreted within several limitations. First, the number of included studies is quite low, reducing the power and generalizability of the main analyses and, especially, the subgroup analyses. Thus, although overall, these analyses yielded a null finding, we cannot exclude a type II error as GRADE revealed very low confidence in the estimate, meaning that the estimate is very uncertain and a difference between the two treatments might exist, but we were not able to observe it. Second, very few RCTs have been completed comparing LAIs versus OAPs in patients with EP-SSDs. This imposed the choice to pool studies differing greatly regarding the type of included patients and relevant study methods. In particular, the choice to include both studies of clinically stable and of not clinically stable patients affected the results in that LAIs were superior to OAPs in patients previously stabilized on OAPs but not in those with clinically not stable patients. However, large recently published meta-analyses conducted in the general population of patients with SSDs (both acutely ill and stable) have indicated that both LAIs and OAPs emerge as highly effective first-line treatment options, irrespective of the initial symptom severity.29–31 Third, while being mainly interested in patients with first-episode psychosis, we focused on the broader population of patients with EP-SSDs (mean recency of onset of disease <5 years), partly due to the fact that only five trials focused exclusively on patients with FE psychosis. However, the question of LAI versus OAP utility in the early illness phase beyond the first episode is clinically relevant, and there were no differences in relapse prevention and acceptability in studies of patients within or beyond 2 years of illness. Fourth, we included in the analyses data from the subset of patients with early-phase disease by Alphs et al. 66 Including study subpopulations might introduce a bias since the randomization was not stratified based on illness duration. However, the authors did not report significant differences between the chronic and early-phase subgroups (except for illness duration). Fifth, we included the cluster-randomized study by Kane et al., 37 which compared aripiprazole-LAI against treatment-as-usual (TAU). While cluster-randomized studies may have some imbalance regarding certain patient characteristics, they are more likely to include more generalizable patient populations in the RCT. Moreover the TAU arm allowed patients to be assigned to LAIs, potentially introducing a conservative bias against the LAI intervention group as 27% of patients in the TAU group were assigned to a LAI treatment at baseline and more 51% of these received LAI treatment throughout the study. Finally, in most of the RCTs one or two specific LAIs were tested against a mixed group of different OAPs or a different OAP than LAI, with only three trials matching the LAI and OAP treatments. These differences could have influenced the results, although likely more for acceptability than effectiveness, as differences in efficacy and effectiveness among non-clozapine antipsychotics are much smaller than adverse effect differences, possibly even more so in EP-SSDs patients.29–31
Nevertheless, despite these limitations, results from this study update the current literature on a clinically very relevant topic, that is, the relative merits of LAI versus OAP treatment in the early illness phases of patients with SSDs, and which patient and study design features may explain either lack of differences between LAIs and OAPs or support the superiority of LAIs versus OAPs. We interpret results from subgroup analyses carefully, as included studies were few and multiple analyses can lead to false positive conclusions. 74 However, in nine subgroup analyses (four for the outcome relapse/hospitalization and five for the outcome acceptability) LAIs were superior to OAPs, while none favored OAPs over LAIs, which is consistent with results in patients with schizophrenia unselected for illness duration. 26 Effect sizes of significant subgroup analyses were homogenously larger for the effectiveness outcome relapse (RR range for relapse/hospitalization: 0.64–0.71) than for the acceptability outcome (RR range fior all-cause discontinuation: 0.84–0.90).
In conclusion, the negative results of this review may be due to the small number of trials and the heterogeneous way in which current trials are designed (in terms of both included patients and methodological features), as reflected by the relevant I2 values observed in both co-primary outcomes. In other words, LAIs and OAPs might not differ consistently in their overall efficacy, as they share the pharmacodynamic neurobiological mechanism of action of dopamine receptor modulation 75 while differing most consistently in their pharmacokinetic profile. 76 However, LAIs and OAPs might have different effectiveness when tested in generalizable, real-world clinical settings where degrees of nonadherence vary across LAIs and OAPs. Subgroup analyses seemed to suggest that pragmatic studies with a strict ITT approach show a bigger advantage of LAIs compared to OAPs. Additional, high-quality trials performed with a pragmatic approach are needed to further test this hypothesis and confirm findings from observational evidence, yet with more methodological rigor.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253241257062 – Supplemental material for Efficacy and safety of long-acting injectable versus oral antipsychotics in the treatment of patients with early-phase schizophrenia-spectrum disorders: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tpp-10.1177_20451253241257062 for Efficacy and safety of long-acting injectable versus oral antipsychotics in the treatment of patients with early-phase schizophrenia-spectrum disorders: a systematic review and meta-analysis by Giovanni Vita, Angelantonio Tavella, Giovanni Ostuzzi, Federico Tedeschi, Michele De Prisco, Rafael Segarra, Marco Solmi, Corrado Barbui and Christoph U. Correll in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
