Abstract
Tardive dyskinesia (TD), a movement disorder associated with antipsychotics, most frequently affects the lower face and jaw muscles, but can also affect walking, breathing and use of the hands and limbs. Knowledge of TD among physicians may be limited, and the pathophysiology of TD is poorly understood. We conducted this review to summarise the current knowledge surrounding the pathophysiology of TD and present recommendations for prevention and treatment based on a literature search and roundtable discussion attended by psychiatrists in Japan. It has been suggested that dopamine hypersensitivity, damaged gamma-aminobutyric acidergic neurons and/or increased production of reactive oxygen species may contribute to development of TD. Symptoms can profoundly affect everyday life; patients who develop TD have poorer prognoses, worse health-related quality of life, greater social withdrawal and higher mortality than patients without TD. Traditional treatment options include dietary supplements, although evidence for their effectiveness is low. Among pharmaceutical interventions, there is moderate evidence that switching to the second-generation antipsychotic clozapine, which has a lower affinity for dopamine D2 receptors than other antipsychotics, may improve symptoms. Vesicular monoamine transporter 2 (VMAT-2) inhibitors, which oppose the increased dopaminergic activity associated with prolonged antipsychotic use by interfering with dopamine uptake and storage, have the strongest evidence for efficacy. VMAT-2 inhibitors are approved in the United States for the treatment of TD, and the first VMAT-2 inhibitor was approved in Japan for this indication in March 2022. Most guidelines recommend treating TD by first reducing the dose of antipsychotics or switching to clozapine or other second-generation antipsychotics, which have a lower association with TD than first-generation antipsychotics. We recommend focusing on prevention and monitoring for TD when prescribing antipsychotics, given that TD is often irreversible. Physicians should treat with antipsychotics only when necessary and at the lowest effective dose, and frequently monitor for TD symptoms.
Plain Language Summary

Visual Summary

Introduction
Tardive dyskinesia (TD) is a potentially serious neurological hyperkinetic movement disorder that is associated with the use of antipsychotics.1,2 The epidemiology, risk factors and recommendations for diagnosis and symptom monitoring for TD in Japan are discussed in-depth in the first article of this supplement. Symptoms of TD include spontaneous, repetitive and jerking motions that commonly affect the muscles of the lower face and jaw and occur in a choreiform fashion; patients may also have difficulty in walking, breathing and using their hands.1–4 In rare cases, TD can be life-threatening.4,5 TD carries a debilitating social and functional burden for both patients and caregivers.6,7
Knowledge of TD among Japanese physicians is limited, and they may not fully understand the importance of monitoring for this drug-induced movement disorder among patients treated with antipsychotics. An understanding of how to effectively manage patients and when to initiate treatment once diagnosed may also be lacking. There are several factors that may contribute to this. First, accurate and early diagnosis of TD may be difficult owing to its delayed presentation and similarity to other movement disorders.8–10 Second, TD is less common in Asian patients than in other races, 11 and there is a corresponding lack of published research dealing specifically with TD in Asians. Third, the social stigma associated with mental illnesses in Japanese culture and reluctance of patients and caregivers to seek treatment may also exacerbate the problem. 12
TD is difficult to treat 3 and often irreversible.13,14 In a study examining the reversibility of tardive syndromes in patients in whom the causative agent was discontinued, a complete resolution of the movement disorder was achieved in 13% of 108 patients, and none of the patient clinical characteristics (e.g. age, sex, baseline severity or duration of drug exposure) were associated with reversibility. 14 Therefore, minimising the risk of TD development is the first step for clinicians. This is done by following best practice when prescribing antipsychotics: ensuring the drug is optimal for a specific illness, limiting the duration of treatment and using the minimum effective dose. 15 Once TD is diagnosed, there are several approaches that can be used for treatment. Vesicular monoamine transporter 2 (VMAT-2) inhibitors can be quite effective in treating TD and should be considered as first-line treatment for TD in countries where they are approved.15,16 In addition, antipsychotics can be modified via discontinuation, dose reduction or switching to another antipsychotic;15–19 switching to alternative second-generation antipsychotics (SGAs) has been associated with reduced TD symptom severity. 18 Increasing the dose of antipsychotics to mask TD symptoms is not recommended as the benefits are short-lived and symptoms return for most patients. 15 Botulinum toxin can be used in patients with focal TD16,18 and alternative therapeutic modalities such as short-term treatment with vitamin B6, amantadine or deep brain stimulation can be considered if patients do not respond to more established therapies.15,19 While other treatments have been evaluated, the studies have been small, making the results difficult to interpret.15,17,19
Knowledge around the underlying pathophysiology of TD remains imperfect, which has hampered development of effective pharmacological therapies until the recent development of VMAT-2 inhibitors. 20 The purpose of the present review is to provide a synopsis of the current understanding of the mechanisms and pathophysiology of TD, and provide an up-to-date summary of existing and upcoming treatment options, with a specific focus on Japan. The implications for prognosis and unmet patient needs will also be discussed.
Methods
We conducted a PubMed search in August 2021 to identify suitable literature to include in the present review. The search focused on papers published between January 2000 and August 2021, although papers published prior to 2000 were also included as appropriate. The following search terms were used: ‘tardive dyskinesia’ alone and in combination with ‘mechanism’, ‘pathophysiology’, ‘prevention’, ‘treatment’, ‘management’ and ‘guideline’. A roundtable discussion was held on 9 September 2021, which formed the basis for the expert opinion presented herein. Among those who attended the advisory board meeting and contributed to the consensus recommendations were the authors of the present review, as well as Professor Koichiro Watanabe (Department of Neuropsychiatry, Faculty of Medicine, Kyorin University, Japan).
Pathophysiology of tardive dyskinesia
The pathophysiology of TD is not yet fully understood, and multiple pathways across several neurotransmitter systems have been suggested, including the dopamine hypersensitivity hypothesis, the gamma-aminobutyric acid (GABA) hypothesis and the reactive oxygen species hypothesis.21–24
The dopamine hypersensitivity hypothesis suggests that dopamine D2 receptor hypersensitivity is related to TD, and this hypothesis is the most widely accepted explanation for TD pathogenesis in the current literature.25,26 It is believed that the physiological response to dopamine receptor-blocking agents (DRBAs) is to upregulate transcription of dopamine D2 receptor genes, causing an increase of postsynaptic dopamine D2 receptors in the striatum, which plays an important role in the control of the oral musculature.27,28 The higher expression of dopamine D2 receptors results in dopamine hypersensitivity and subsequent motor impairment as shown in Figure 1. This hypothesis is supported by several clinical findings. First, increasing the dose of antipsychotics often results in initial improvement of TD symptoms, but the effect is only temporary. 23 Second, first-generation antipsychotics (FGAs), which elicit greater blockade of dopamine D2 receptors, are more likely to cause TD than SGAs. 29 Third, there is some support for this hypothesis from genetic susceptibility studies, particularly with respect to gene polymorphisms in the dopamine receptor genes DRD2 and DRD3, which have been associated with TD susceptibility in some studies.30,31 In addition, polymorphisms in the VMAT-2 gene, which regulates the release of neurotransmitters implicated in TD, are reported to be associated with TD. 32 Finally, this hypothesis is supported by the mechanism of action of VMAT-2 inhibitors, the only pharmacological treatment with strong evidence of efficacy in treating TD.33,34 Although there is support for the dopamine hypersensitivity hypothesis, it should be noted that this hypothesis cannot explain why TD is often irreversible, even when antipsychotics are stopped. 26

Dopamine hypersensitivity hypothesis. (a) In short-term administration of dopamine receptor-blocking antipsychotics, upregulation of the indirect pathway occurs: the thalamus is inhibited by excitatory projections (via glutamic acid; red arrows) from the subthalamic nucleus to the internal globus pallidus, leading to inhibition (via GABA, blue arrows) of motor output. (b) Long-term administration of antipsychotics and chronic dopamine D2 receptor blockade leads to downregulation of the indirect pathway; excitatory input to the internal globus pallidus via the subthalamic nucleus is weakened, resulting in increased inhibition of the internal globus pallidus. This in turn weakens the inhibitory projections to the thalamus, and results in overstimulation of motor outputs (i.e. tardive dyskinesia).
The GABA hypothesis proposes that long-term use of antipsychotics may cause damage to GABAergic neurons in the striatum.25,26 These damaged GABAergic neurons become hypofunctional and the balance between direct and indirect basal ganglia pathways becomes dysregulated (Figure 2). Evidence suggests an important link between GABA and dopaminergic neuronal function in specific brain regions. 35 However, human studies using GABA agonists to treat TD have shown minimal effects. 36
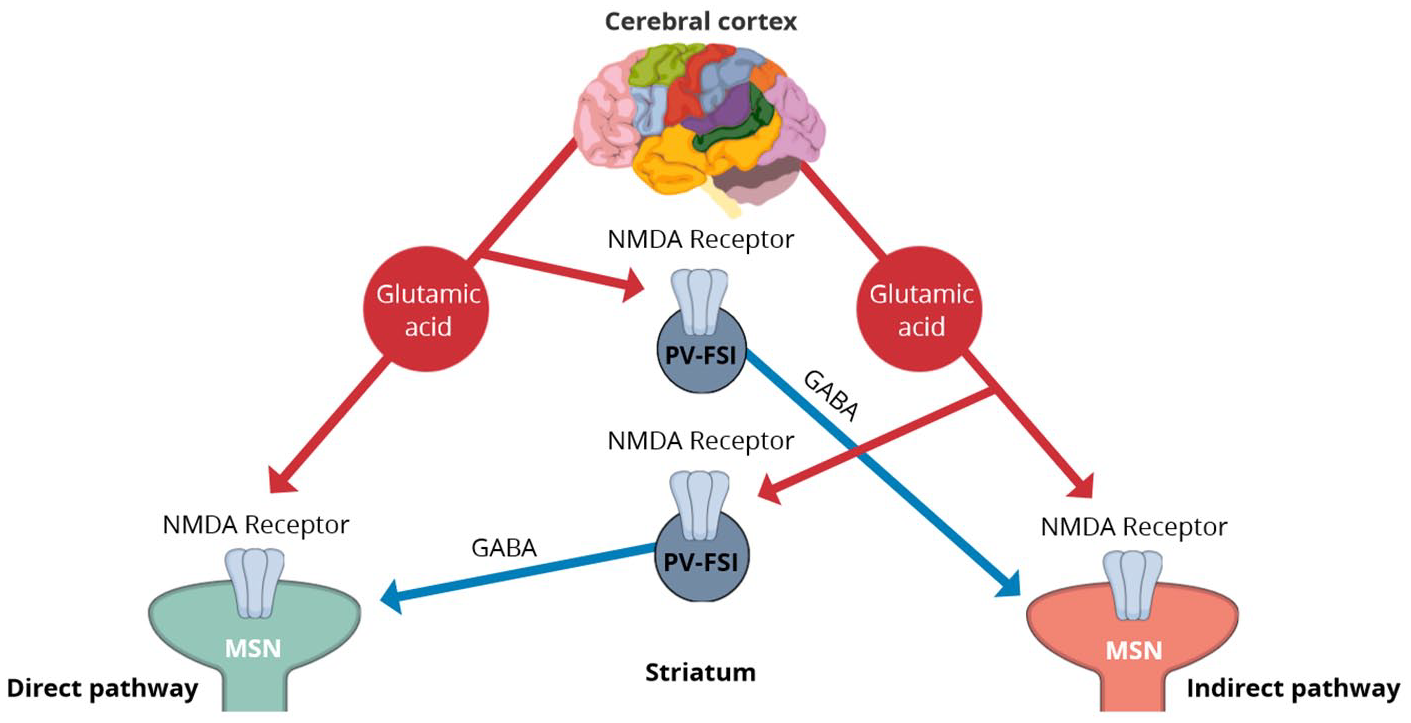
GABA hypothesis. The cerebral cortex provides excitatory input (red arrows) via glutamic acid to NMDA receptors on both MSNs and PV-FSIs in the striatum. PV-FSIs in turn regulate the balance between direct and indirect pathways by potent feedforward inhibition (blue arrows) of MSNs. Damage to these GABAergic PV-FSIs may lead to dysregulation of the pathways and subsequent motor disorders.
The reactive oxygen species hypothesis proposes that antipsychotics cause an increase in production of free radicals, which act as neurotoxins. 37 As described in the dopamine hypersensitivity hypothesis above, antipsychotics block dopamine D2 receptors, to which the brain responds by upregulating dopamine synthesis and metabolism. 21 The by-products of dopamine metabolism, which include hydrogen peroxide and oxygen free radicals, correspondingly increase. These free radicals initiate a chain of free radical production, leading to peroxidation of polyunsaturated fatty acids and subsequent damage to cell membrane lipids (Figure 3). If the production of free radicals exceeds the ability of the brain’s antioxidant systems to handle, damage to cell membrane integrity may occur, resulting in disruption of cellular communication or destruction of cells.21,37 Both preclinical and clinical studies have suggested that long-term use of antipsychotics may promote neurotoxic free radical production, particularly for FGAs.37–42 A recent meta-analysis indicated that patients with schizophrenia who received antipsychotics had higher levels of oxidative stress markers such as malondialdehyde and thiobarbituric acid reactive substances, as well as higher total oxidant status, compared with unmedicated patients. 42 Furthermore, animal studies have shown that long-term use of antipsychotics may increase the concentration of transition metals (e.g. iron, manganese and copper) in the striatum, which may also contribute to oxidative damage.43,44

Reactive oxygen species hypothesis. Long-term blockade of dopamine D2 receptors by antipsychotics results in a secondary increase in dopamine synthesis and metabolism. (a) Subsequent auto-oxidation of dopamine and oxidation by MAO and COMT results in elevated production of free radicals (hydrogen peroxide and hydroxy radicals), which are neurotoxic. HVA, as a metabolite of dopamine metabolism via MAO/COMT, is associated with dopamine levels in the brain. (b) Presence of transition metals as electron acceptors (e.g. in the form of enzymes such as superoxide dismutase) encourages formation of hydroxy radicals, which are extremely reactive and promote lipid peroxidation of polyunsaturated fatty acids, leading to cell membrane damage.
The above hypotheses are not mutually exclusive and all may contribute to the pathogenesis of TD. Some studies have associated TD with non-DRBAs such as tricyclic antidepressants, selective serotonin reuptake inhibitors or lithium, but the evidence for a causal relationship between these drugs and TD is weak, and a plausible mechanism for such a relationship is not known. 45 Many patients with psychiatric disorders who present with dyskinetic disorders following exposure to non-DRBAs also have past exposure to antipsychotics, and the latent nature of TD makes it difficult to ascribe causality in such cases.
Interestingly, symptoms of hyper- and hypokinetic movements have been recorded for patients with schizophrenia prior to the introduction of antipsychotics.46–48 A meta-analysis reported that dyskinesia was strongly associated with antipsychotic-naïve patients with schizophrenia versus healthy controls (odds ratio [OR] 3.59, 95% confidence interval [CI] 1.53–8.41), and healthy first-degree relatives of those with schizophrenia were significantly more likely to have dyskinesia versus healthy controls (OR 1.38, 95% CI 1.06–1.81). 49 These findings suggest that some dyskinesias may be related to schizophrenia itself and not just exposure to antipsychotics.
Complications associated with tardive dyskinesia and patient prognosis
Patients with TD may suffer from a variety of movement impairments that can influence their everyday functioning. 50 Reduced mobility can occur because of impairments in gait speed, cadence, step length, posture/postural dynamics, arm swing, gait initiation, turning and gait efficiency. In severe cases, patients may have an increased risk of falling or hip fractures. Speech impairment often leads to a reduced ability to be understood when speaking and a loss of naturalness of speech, hindering verbal communication. 50 Respiratory irregularities, which involve the laryngeal and pharyngeal muscles, can include dysphagia or dyspnoea and may lead to choking, aspiration, pneumonia, aspiration pneumonia and asphyxia.27,51,52 A loss of fine motor skills in the upper extremities is common with TD and can have profound impacts on everyday tasks, including self-care, writing and manipulation of fine objects; this also impacts a patient’s ability to perform work duties and participate in social or recreational activities. 50 Lower limb strength and flexibility may also be affected. However, the effects of these factors have not been formally assessed in TD either independently or in relation to everyday functioning. Cognitive function, as measured by the Repeatable Battery for the Assessment of Neuropsychological Status, can also be affected. Among patients with schizophrenia, those with TD perform significantly worse on tasks involving visuospatial construction and attention and have significantly lower total index scores than those without TD. 53 Patients with psychiatric disorders and TD have significantly worse health-related quality of life and social withdrawal than those without TD. 54
In addition, TD is associated with a poorer prognosis than not having TD. In a 6-year cohort study of Asian patients with schizophrenia, a higher risk of mortality was reported for those with TD versus those without, and mortality was shown to increase with TD severity. 55 The small number of deaths during the study precluded a meaningful statistical analysis; however, the authors noted infections such as pneumonia and septicaemia were a numerically more common cause of mortality in patients with TD. As motor symptoms worsen, patients’ ability to care for themselves deteriorates, and they require increasing amounts of assistance from caregivers. 50 Patients with schizophrenia experience a high burden associated with mental health, and this is made worse for some patients by the presence of TD. 54 Many patients feel insecure or rejected by society because of their TD symptoms. 56 This social isolation is associated with a poorer quality of life among patients with schizophrenia 57 and the negative effects may be more pronounced in patients with more severe TD. 58
It is important for physicians to be proactive in interviewing their patients about the presence of TD symptoms and to carefully observe them during medical examination. Because of the poor prognosis for many patients with TD and the often irreversible nature of motor symptoms, prevention remains the best strategy, although in practice, this can be difficult when balancing the risks and benefits of antipsychotics. 15
Treatment
Physicians may consider stopping administration of antipsychotics as a first-line treatment for TD. However, this is often not possible as patients with schizophrenia generally require long-term treatment to prevent relapse. 15 First and foremost, prevention should be emphasised by using the lowest antipsychotic dose possible. In cases where TD is already established, the initial clinical decision is most likely to involve dose reduction if possible, but this must be weighed against the risk of worsening psychiatric symptoms. 59 Furthermore, patients may experience a temporary worsening of TD symptoms (withdrawal-emergent dyskinesia) immediately following dose reduction, caused by increased dopamine stimulation resulting from a decrease in dopamine blockade. 15 For patients taking FGAs, physicians should consider switching to an SGA, 15 and for those already receiving SGAs, dose reduction or switching to a different antipsychotic may be attempted with caution, but there is conflicting evidence in support of the efficacy of adjusting antipsychotics.60 In our experience, dose reduction and/or switching of antipsychotics have had modest effects for treatment of TD, but we have not had success with any other available pharmacological interventions.
A wide variety of treatment approaches have been investigated since the first reports of extrapyramidal symptoms emerged following the advent of antipsychotics in the 1950s. 61 These include dietary supplements, pharmacologic interventions and non-pharmacologic treatments. A summary of available treatments for TD is provided in Table 1, although it should be noted that the level of evidence for many of these therapies is low.15,62–71 Furthermore, efficacy of certain interventions for specific populations may be difficult to confirm in studies that did not stratify patients by TD severity; indeed, few studies have investigated the possible influences of age, sex or baseline TD severity on treatment efficacy.
Summary of treatments for tardive dyskinesia.

GABA, gamma-aminobutyric acid; VMAT-2, vesicular monoamine transporter 2.
Dietary supplements
Vitamin E is a free radical scavenger and as such, may minimise damage caused by the overproduction of neurotoxic free radicals. 62 Small trials suggest that vitamin E may protect against deterioration of TD, but there is little evidence that it can improve symptoms.62,76
The pyridoxal 5′-phosphate vitamer of vitamin B6 functions as a coenzyme and is involved in the metabolic transformation of several neurotransmitters including dopamine, GABA and serotonin. 77 It is thought that supplementation of vitamin B6 may improve TD symptoms via its role in these metabolic transformations and by scavenging free radicals. Although this treatment has been shown to be clinically effective versus placebo for reducing TD symptoms, 63 the relevant studies included only a small number of patients and were of a short duration. While short-term treatment may be considered, the dose and duration of treatment have not been established and physicians should keep in mind that chronic administration of vitamin B6 > 1000 mg/day can cause neuropathy.15,78
Essential fatty acids are an important part of the neuronal membrane; variations in their levels can affect both the quantity and quality of membrane phospholipids in the brain, which can influence neuronal function. 79 Reduced membrane levels of essential fatty acids are reported in both antipsychotic-treated and antipsychotic-naïve patients with schizophrenia 79 and essential fatty acid levels in red blood cell membranes were reported to be lowest among patients with schizophrenia and severe TD. 80 An early study of essential fatty acid treatment for schizophrenia reported a reduction in abnormal movements. 81 Over the years, several studies in animals and humans have indicated a potential role for essential fatty acids in improving the symptoms of TD. 79 However, clinical studies included small patient numbers and short follow-up times, so definitive conclusions cannot be drawn, and essential fatty acids are not currently recommended for TD treatment.15,79
Branched-chain amino acids have been shown to decrease tyrosine, which is a precursor to dopamine. 82 There is some evidence from a handful of small trials that supplementation of branched-chain amino acids can improve symptoms of TD.83–85 However, given the limited data, they are not recommended in the treatment of TD.15,86
Yokukansan (yi-gan san) is a traditional Japanese medicine consisting of an herbal mixture that acts to modulate GABA and serotonin systems87,88 and has antioxidant activity. 89 A clinical trial of 22 patients with schizophrenia and TD reported that 12 weeks of treatment with Yokukansan resulted in statistically significant improvements in symptoms of both TD and psychosis. 64 However, this study was small and had no control group; thus, larger, well-controlled studies are required to fully understand if Yokukansan can provide clinically meaningful benefits for TD.
EGb 761, an extract of ginkgo biloba, is reported to have antioxidant properties. 90 A randomised, double-blind, placebo-controlled study of inpatients with schizophrenia and TD reported a significant improvement in TD symptoms for patients treated with EGb 761 versus placebo. 91 However, there is currently insufficient evidence to recommend EGb 761 for the treatment of TD. 15
Pharmacologic interventions
Given that melatonin is 6–10 times more effective than vitamin E as an antioxidant 92 and that vitamin E may protect against deterioration of TD, 62 the effects of melatonin on symptoms of TD have been evaluated. Improvements in TD symptoms have been reported for some patients 93 and a double-blind, placebo-controlled, crossover study in patients with schizophrenia reported a significant decrease in the Abnormal Involuntary Movement Scale (AIMS) score with melatonin versus placebo. 92 However, the current low level of evidence is insufficient to recommend melatonin for TD treatment.15,94
Botulinum toxin is a neurotoxin that acts to decrease synaptic transmission and focally weaken muscle; 95 injections of this toxin result in local chemodenervation to relax muscles that are contracting abnormally. Case studies and small trials have shown that botulinum toxin is effective at relieving some symptoms of TD,96–100 particularly for patients with focal TD.16,86 There is currently not enough data to support or refute the use of botulinum toxin as a treatment for TD. 65
Given the association of GABAergic neuronal dysfunction and TD, GABA agonists may increase GABA and reduce abnormal motor movements such as those observed in TD. 19 The results of clinical trials examining GABA agonists for TD have been mixed. Two meta-analyses have reported no evidence for improvements in TD symptoms with either benzodiazepine (diazepam and alprazolam) or non-benzodiazepine GABA agonists (baclofen, sodium valproate, progabide, tetrahydroisoxazolopyridinol, gamma-vinyl-GABA, gamma-acetylenic-GABA and muscimol) versus placebo.3,36 In addition, a systematic review of benzodiazepines for the treatment of TD found that the associated trials were poorly reported and provided inconclusive results. 101 It should also be noted that benzodiazepines carry risks of addiction, abuse and withdrawal symptoms. GABA agonists are not recommended for the treatment of TD given the lack of effect and risk of side effects, such as ataxia and sedation, with some GABA agonists.15,18
Amantadine is an antiviral agent 102 that is used to treat dyskinesia in drug-induced parkinsonism; 103 however, the underlying mechanism of action for amantadine is unclear. 86 Amantadine has been studied for over 20 years in patients with TD, but the trials have been small and the results have been inconsistent. 86 In addition, amantadine has some safety concerns, including modest anticholinergic side effects, the ability to induce psychosis in patients with Parkinson’s disease, and livedo reticularis, a condition characterised by blue/purple skin discolouration. The evidence for amantadine in TD treatment is limited, although it is sometimes administered if more established methods are ineffective. 15
Both cholinergic and anticholinergic agents have been suggested as potential treatments for TD. In general, cholinergic compounds are thought to have the potential to improve TD by enhancing acetylcholine synthesis, as it is thought that cholinergic deficiency may contribute to TD. 66 However, cholinergics were shown to have no effect on TD symptoms in a few, small trials conducted to date, and the data are currently insufficient to determine their effectiveness.15,65,66 Anticholinergics are used to treat antipsychotic-induced parkinsonism and dystonia, 67 but the rationale for using these drugs in TD is unclear. 67 For anticholinergics, no controlled studies have been performed to date, and evidence for a therapeutic benefit associated with anticholinergic usage is sparse,16,65,67 while animal studies indicate that chronic administration may increase dopamine D2 receptor sensitivity. 104 In addition, anticholinergics can cause side effects including dryness of the mouth, constipation, blurred vision and confusion. 105
Clozapine is an SGA with a lower risk of extrapyramidal side effects relative to other antipsychotics, 106 possibly because of its lower affinity for dopamine D2 receptors. 107 A meta-analysis of 16 studies found that there was a significant reduction in TD when patients were switched to clozapine from other antipsychotics; the greatest reduction was observed in patients presenting with moderate to severe TD. 68 In patients with mild TD, switching to clozapine rarely worsened TD and a trend towards reduction in symptoms was common. Furthermore, data from a systematic review also supported switching to clozapine to reduce TD risk or treat symptoms of existing TD. 69
VMAT-2 inhibitors interfere with dopamine uptake and storage in presynaptic vesicles, resulting in less dopamine available for release in the synapse, 108 thereby opposing the increased dopaminergic activity caused by prolonged antipsychotic use (Figure 4). 109 Tetrabenazine was the first VMAT-2 inhibitor identified as a potential therapeutic agent for TD, and it was subsequently optimised to improve its pharmacokinetic parameters. One optimisation used deuterisation, which resulted in deutetrabenazine. The second optimisation was a valine esterification of the most active and selective isoform of tetrabenazine, called valbenazine (NBI-98854). 110 Both deutetrabenazine and valbenazine have more stable pharmacokinetics compared with tetrabenazine. Tetrabenazine has shown efficacy for the treatment of TD in clinical trials and side effects are minimal, although depression, parkinsonism and somnolence have been reported.109,111 Deutetrabenazine and valbenazine have demonstrated strong evidence of their effectiveness in treating TD, in both acute and long-term settings, and they do not carry an increased risk of depression or suicide.109,112 However, VMAT-2 inhibitors may cause worsening of DRBA-induced parkinsonism and tremor. 113 Because VMAT-2 inhibitors reduce levels of available synaptic dopamine, TD symptoms return after withdrawal of the drug due to the resulting surplus of dopamine. However, based on AIMS scores, symptoms do not become worse than the original baseline level,71,114 suggesting that VMAT-2 inhibitors may not cause further upregulation of dopamine D2 receptors or increase dopamine hypersensitivity. Both valbenazine and deutetrabenazine were approved in the United States for treatment of adults with TD in 2017.115,116 At the time of writing, valbenazine had only just received approval in Japan for treatment of TD; 117 the approval status in Europe of the VMAT-2 inhibitors for this indication is inconsistent.

Schematic of mechanism of action of VMAT-2 inhibitors. (a) Normal release of dopamine by fusion of vesicles with the presynaptic membrane and release into the synaptic cleft. VMAT-2 facilitates incorporation of dopamine into the synaptic vesicles. (b) VMAT-2 inhibitors block transporters on synaptic vesicles, preventing uptake of dopamine. Dopamine is subsequently degraded by monoamine oxidase.
The randomised, double-blind, placebo-controlled phase III KINECT study enrolled patients diagnosed with moderate to severe TD (mean ± SD baseline AIMS total score 10.0 ± 4.0). 70 Although patients were not stratified by baseline TD severity, the baseline AIMS total score was included as a covariate in the mixed-effects model for repeated measures used to assess treatment efficacy. Patients with TD receiving 80 mg/day valbenazine had significantly reduced AIMS total scores compared with those receiving placebo after 6 weeks of treatment. In the valbenazine (40 mg/day or 80 mg/day) group, 6.6% of patients experienced a serious treatment-emergent adverse event (TEAE) versus 3.9% in the placebo group. Furthermore, 6.0% of patients in the valbenazine group experienced a TEAE that led to discontinuation versus 5.3% in the placebo group. 70 The most commonly reported TEAEs in the valbenazine group were somnolence, akathisia, and dry mouth (5.3%, 3.3% and 3.3%, respectively). Suicidal ideation was also reported, but at a lower rate than that of the placebo group (2.6% versus 5.3%). Safety and efficacy were further evaluated in the 1-year KINECT 3 extension study, which reported that 69.2% of patients experienced at least one TEAE, the most common of which included headache (7.1%), urinary tract infection (6.6%), and diarrhoea and dizziness (5.6% each). Serious TEAEs were reported in 14.6% of patients and 15.7% discontinued because of a TEAE. 118 The phase III open-label KINECT 4 trial evaluated valbenazine over a 52-week period in patients with moderate to severe TD (mean ± SD baseline AIMS total score 10.0 ± 3.8). 71 AIMS scores were improved by ⩾ 50% from baseline at week 48 in approximately 90% of patients in both dosing groups (40 mg/day or 80 mg/day) and safety results were similar to previous studies, with the most common TEAEs including urinary tract infection (8.5%) and headache (5.2%). 71 A post-hoc analysis that pooled results from patients with mood disorders who participated in either KINECT 2 (a 6-week, double-blind, placebo-controlled trial), KINECT 3 or the long-term KINECT 3 extension study found that valbenazine significantly improved the AIMS total score, and a ⩾ 50% AIMS total score improvement versus placebo at week 6 was shown in these patients. 72 All patients included in the KINECT studies were diagnosed with moderate to severe TD, and the pooled analysis incorporated baseline AIMS total score as a covariate in the efficacy assessment.
Two randomised, double-blind, placebo-controlled, phase III trials of deutetrabenazine in patients with TD (ARM-TD 73 and AIM-TD, 74 both enrolling patients with a minimum baseline AIMS total score of 6) reported significant improvement in AIMS scores at 12 weeks versus placebo. Serious AEs were reported in 4–8% of patients treated with deutetrabenazine versus 6–8% with placebo. The most commonly reported TEAEs among patients treated with deutetrabenazine included somnolence, fatigue, insomnia, headache, diarrhoea and akathisia in ARM-TD, and headache, anxiety, diarrhoea, nasopharyngitis and fatigue in AIM-TD. Patients from the ARM-TD and AIM-TD trials were eligible to enter into an open-label single-arm study to evaluate the long-term safety and efficacy of deutetrabenazine. 114 The mean change in the AIMS score from baseline was −4.9 at week 54, −6.3 at week 80 and −5.1 at week 106, demonstrating long-term efficacy. The incidence rates of serious AEs were similar to the short-term phase III studies.
A randomised, double-blind, placebo-controlled phase III trial evaluating the efficacy of valbenazine in Japanese patients with moderate to severe TD (J-KINECT) has recently been completed (NCT03176771) and publication of the results from that trial are expected in the near future. The primary endpoint was change from baseline in the AIMS total score after 6 weeks of treatment, and long-term safety and efficacy (up to 48 weeks of treatment) were also assessed.
Valbenazine may have several advantages over other VMAT-2 inhibitors. First, the half-life of valbenazine is 15–22 h, which is longer than that of deutetrabenazine (9–10 h) and tetrabenazine (5–7 h). This allows for once-daily administration rather than multiple daily doses as needed for deutetrabenazine (2 doses/day) and tetrabenazine (3 doses/day). 119 In addition, in contrast to other VMAT-2 inhibitors that have multiple metabolites, valbenazine has only a single metabolite, and is expected to elicit fewer off-target effects and thus fewer side effects than either deutetrabenazine or tetrabenazine.115,116,120,121 Finally, deutetrabenazine and tetrabenazine are contraindicated in patients with hepatic impairment and those taking monoamine oxidase inhibitors or reserpine, whereas valbenazine can be used safely in patients with mild hepatic impairment or mild/moderate/severe renal impairment. In Japan, the only VMAT-2 inhibitor approved for treatment of TD is valbenazine, which received approval very recently (March 2022).
Non-pharmacologic interventions
Deep brain stimulation acts via several mechanisms to treat motor symptoms. 122 There are several case reports that have demonstrated improvements in TD symptoms with deep brain stimulation;123–125 however, evidence for this treatment is limited. 65
Electroconvulsive therapy has also been used for treatment of TD, but the evidence supporting its use is limited to case series and case reports, and no controlled trials have been reported to date.75,126 Although favourable results have been observed in case reports, the lack of high-quality data preclude recommending electroconvulsive therapy as an isolated treatment for TD.
Consensus recommendations for prevention and treatment of TD
A summary of the recommendations for treating TD from recent clinical guidelines is provided in Table 2. Most treatment guidelines for schizophrenia recommend either dose reduction of antipsychotics or switching to clozapine or other SGAs to lower the risk of TD. 127 It is also recommended that anticholinergic drugs be either reduced or discontinued.
Summary of guidelines for treatment of schizophrenia with associated considerations for tardive dyskinesia in Japan and overseas.

CINP, International College of Neuropsychopharmacology; DBS, deep brain stimulation; FGA, first-generation antipsychotic; SGA, second-generation antipsychotic; TD, tardive dyskinesia; VMAT-2, vesicular monoamine transporter-2.
The current recommendations for prevention of TD in Japan emphasise the use of SGAs over FGAs. 128 For the treatment of TD, the guideline prioritises dose reduction of the antipsychotic (if possible), and for serious cases of TD, switching to another SGA. 128
The American Psychiatric Association practice guideline for the treatment of patients with schizophrenia recommends VMAT-2 inhibitor treatment for patients with moderate to severe or disabling TD. 130 This recommendation for the use of VMAT-2 inhibitors differs from the recommendations in Japan, as the only VMAT-2 inhibitor approved for the treatment of TD, valbenazine, received approval in March 2022. Therefore, the recommendation in Japan is expected to change in the near future. In light of the recent approval of valbenazine in Japan, patients with moderate to severe or disabling TD should be considered as candidates for VMAT-2 inhibitor therapy. VMAT-2 inhibitors do not cure TD and symptoms eventually return to baseline after treatment cessation; thus, prevention remains the optimum strategy.
To reduce the risk of TD, we advise physicians in Japan to carefully consider the benefits and risks of prescribing antipsychotics in individual patients. If antipsychotics are considered necessary, physicians should preferentially prescribe SGAs rather than FGAs and prescribe the lowest effective dose.
We have the following recommendations for how physicians can achieve better outcomes for their patients. First, physicians should focus on prevention. The decision to prescribe antipsychotics should be considered carefully, antipsychotic use should be limited to specific indications, and the lowest effective dose should be prescribed. Although the evidence for the benefits of antipsychotic dose reduction to treat TD remains insufficient, 59 we recommend dose reduction if possible in stable patients because TD has, to an extent, been shown to be dependent on the dose of antipsychotics. 137 In addition, reducing the dose of antipsychotics for patients with schizophrenia on a maintenance dose has been shown to reduce extrapyramidal symptoms. 138 Both international treatment guidelines/algorithms and Japanese pharmacological treatment algorithms for schizophrenia recommend that dose reduction should be attempted with the goal of using the minimum effective dose after acute symptoms are resolved.139,140 However, it should be emphasised that the potential benefit of dose reduction should be weighed against the risk of worsening of the underlying psychiatric condition, and patients should be monitored appropriately. Second, physicians in Japan need to be aware that although TD is less commonly seen in patients administered SGAs than FGAs, patients who receive antipsychotics are still at risk of this potentially serious side effect. The promise shown by VMAT-2 inhibitors and the recent approval of valbenazine for use in Japan represents a beacon of hope for patients; thus, physicians should be vigilant in both monitoring for TD symptoms in their patients and consulting with caregivers as much as possible.
Concluding summary
With the advent of VMAT-2 inhibitors, we anticipate that TD awareness in Japan will increase and that both physicians and patients will be motivated not only to be cognizant of prevention, but also to proactively monitor for symptoms in an effort to detect TD early and initiate treatment as early as possible.
Supplemental Material
sj-tif-2-tpp-10.1177_20451253221117313 – Supplemental material for Pathophysiology, prognosis and treatment of tardive dyskinesia
Supplemental material, sj-tif-2-tpp-10.1177_20451253221117313 for Pathophysiology, prognosis and treatment of tardive dyskinesia by Hiroyoshi Takeuchi, Yasuhiro Mori and Yuichiro Tsutsumi in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-tif-3-tpp-10.1177_20451253221117313 – Supplemental material for Pathophysiology, prognosis and treatment of tardive dyskinesia
Supplemental material, sj-tif-3-tpp-10.1177_20451253221117313 for Pathophysiology, prognosis and treatment of tardive dyskinesia by Hiroyoshi Takeuchi, Yasuhiro Mori and Yuichiro Tsutsumi in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-tif-4-tpp-10.1177_20451253221117313 – Supplemental material for Pathophysiology, prognosis and treatment of tardive dyskinesia
Supplemental material, sj-tif-4-tpp-10.1177_20451253221117313 for Pathophysiology, prognosis and treatment of tardive dyskinesia by Hiroyoshi Takeuchi, Yasuhiro Mori and Yuichiro Tsutsumi in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-tiff-1-tpp-10.1177_20451253221117313 – Supplemental material for Pathophysiology, prognosis and treatment of tardive dyskinesia
Supplemental material, sj-tiff-1-tpp-10.1177_20451253221117313 for Pathophysiology, prognosis and treatment of tardive dyskinesia by Hiroyoshi Takeuchi, Yasuhiro Mori and Yuichiro Tsutsumi in Therapeutic Advances in Psychopharmacology
Footnotes
Acknowledgements
The authors thank Sarah Bubeck, PhD, of Edanz, Japan, for medical writing assistance and publication support, which was funded by Mitsubishi Tanabe Pharma Corporation. The authors have authorised the submission of this manuscript via Edanz and have approved the funding and conflict of interest statements.
Declarations
Correction (January 2023):
Article updated online to correct Figure 1. In the previous version of this figure, the ‘external globus pallidus’ was incorrectly labeled as the ‘internal globus pallidus’ in the indirect pathway (panels (a) and (b)).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
