Abstract
Background:
Clozapine is the most effective medication for treatment–refractory schizophrenia but is associated with significant adverse drug effects, including hypotension and dizziness, which have a negative impact on quality of life and treatment compliance. Available evidence for the management of clozapine-induced hypotension is scant.
Objectives:
Due to limited guidance on the safety and efficacy of pharmacological treatments for clozapine-induced hypotension, we set out to systematically review and assess the evidence for the management of clozapine-induced hypotension and provide guidance to clinicians, patients, and carers.
Design:
We undertook a systematic review of the safety and efficacy of interventions for clozapine-induced hypotension given the limited available evidence.
Data Sources and Methods:
PubMed, Embase, PsycINFO, CINAHL, and the Cochrane trial Registry were searched from inception to November 2021 for literature on the treatment strategies for clozapine-induced hypotension and dizziness using a PROSPERO pre-registered search strategy. For orthostatic hypotension, we developed a management framework to assist in the choice of intervention.
Results:
We identified nine case studies and four case series describing interventions in 15 patients. Hypotension interventions included temporary clozapine dose reduction, non-pharmacological treatments, and pharmacological treatments. Midodrine, fludrocortisone, moclobemide and Bovril® combination, and etilefrine were associated with improvement in symptoms or reduction in orthostatic hypotension. Angiotensin II, arginine vasopressin, and noradrenaline successfully restored and maintained mean arterial pressure in critical care situations. A paradoxical reaction of severe hypotension was reported with adrenaline use.
Conclusion:
Orthostatic hypotension is a common side effect during clozapine titration. Following an assessment of the titration schedule, salt and fluid intake, and review of hypertensive and nonselective α1-adrenergic agents, first-line treatment should be a temporary reduction in clozapine dose or non-pharmacological interventions. If orthostatic hypotension persists, fludrocortisone should be trialled with monitoring of potassium levels and sodium and fluid intake. Midodrine may be considered second-line or where fludrocortisone is contraindicated or poorly tolerated. For patients on clozapine with hypotension in critical care settings, the use of adrenaline to maintain mean arterial pressure should be avoided.
Registration:
PROSPERO (Registration No. CRD42020191530)
Introduction
Clozapine is the most effective medication for treatment–refractory schizophrenia 1 and is associated with lower rates of hospitalisation, lower mortality, and improved quality of life when compared with other antipsychotic medications.1–4 Despite this, clozapine is often underutilised, 5 due to serious haematological, 6 metabolic, 7 and cardiac 8 adverse drug effects, including autonomic dysfunction 9 and orthostatic hypotension,10–13 which affects patient adherence and prescribing practices.
Orthostatic hypotension is defined as a sustained reduction in systolic blood pressure of > 20 or of > 10 mmHg diastolic pressure within 3 min of rising from lying to a standing position and is associated with increased cardiovascular and cerebrovascular mortality, and syncope and falls.14–16 The incidence of clozapine-induced orthostatic hypotension is around 9%.17–19 However, common diagnostic methods to assess clinical orthostatic hypotension, such as sit–stand testing, have limited diagnostic accuracy. 20 Patients may experience dizziness due to changes in blood pressure that do not meet the clinical criteria for orthostatic hypotension but may still increase the risk of falls and lead to serious harm or discontinuation of clozapine. As a result, individual symptoms are more common than the full syndrome of orthostatic hypotension. In one British sample of patients newly initiated on clozapine, dizziness was the fourth most common reason for clinician-led discontinuation. 11 The incidence of ‘orthostatic faintness’ in phase 3 of the Clinical Antipsychotic Trials for Intervention Effectiveness (CATIE) trial was 24%. 21 In a 2021 cluster analysis of consumer perspectives, dizziness on standing was reported in 40% of patients, was significantly associated with lower consumer safety ratings, and was the most influential adverse event on consumer dissatisfaction with clozapine. 22
Clozapine’s α1-adrenoceptor blockade causes vasodilation by preventing α1-adrenoceptors in vascular smooth muscle from mediating peripheral resistance. 23 Clozapine, such as other α1-adrenergic agents, is associated with a ‘first-dose effect’. Patients are more likely to experience greater hypotensive effects when commencing treatment or during dose increases, especially with rapid titration. 24 Desensitisation of the α1-adrenergic receptors occurs with repeated exposure to α1-adrenergic antagonists and tolerance usually develops over several weeks of treatment. 24 A recent meta-analysis showed no relationship between orthostatic hypotension and clozapine/norclozapine plasma levels at steady state. 25 However, a dose-related relationship has been observed in other studies. Patients receiving a dose of 150–300 mg of clozapine per day in clinical trials experienced less dizziness than those taking 300–600 mg per day. 26
There is considerable interpatient variability in both sensitivity to α1-antagonists and metabolism of clozapine by cytochrome P450 enzymes, and thus for some individuals, clozapine-induced orthostatic hypotension may be challenging to manage and prove to be a dose-limiting adverse effect, particularly in older age and in those taking concurrent antihypertensive medicines.27,28 Clozapine has also been shown to cause hypotension unrelated to positional change refractory to fluids and vasopressors, and a paradoxical reaction of severe hypotension due to clozapine and adrenaline co-administration.29–33
There is limited guidance on the safety and efficacy of pharmacological treatments for clozapine-associated hypotension. We therefore conducted a systematic review to assess the evidence for the management of clozapine-induced hypotension and provide guidance to clinicians, patients, and carers.
Methods
This study was registered with PROSPERO (Registration No. CRD42020191530), an international database of prospectively registered systematic reviews. 34 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement recommendations were followed for backgrounds, search strategy, methods, results, discussion, and conclusions. 35 Ethical approval was not required as all the included studies had been previously published with ethical approval.
We searched PubMed, Embase, PsycINFO, CINAHL, and the Cochrane trial Registry from inception to November 2021 using the following search terms: Clozapine OR Clopine OR Clozaril OR Zaponex AND hypotension OR orthostasis OR orthostatic OR postural OR dizz* OR autonomic* OR sympathetic* OR presync* OR presyncope OR orthostatic intolerance OR hypotension [MeSH Terms]. Studies were not limited by language or article type. All studies identified in the electronic searches were screened at the title and abstract level by three independent researchers (T.B., S.D.P., and T.D.T.). Studies that met the inclusion criteria or could not be excluded based on information provided in the abstract were reviewed at full-text level by three independent researchers (T.B., S.D.P., and T.D.T.). For studies published in languages other than English, Google Translate was used to translate to English so that review was possible. Articles published in German were translated by one author (T.D.T.).
Studies were included if the participants prescribed clozapine demonstrated hypotension or dizziness and reported a pharmacological or non-pharmacological intervention. All study designs were included where available, including case reports, case series, observational studies, cohort studies, and randomised controlled trials (RCTs). There were no restrictions on age, sex, comorbidities, adjunct therapies, or diagnosis. Study quality was assessed using the Joanna Briggs Institute (JBI) Critical Appraisal Tool for case studies and case series 36 to assess the methodological quality and risk of bias in both the reporting and analysis of findings.
Data extraction was conducted by two researchers (T.B. and S.D.P.) and validated by a third researcher (T.D.T.). Where available, the pre-specified variables for the collection included demographics, medical and psychiatric history, concurrent medication, clozapine dose and plasma level, clozapine-associated adverse events, details of the hypotension intervention, and adverse effects of the intervention. Complete response was defined as a resolution of symptoms associated with orthostatic changes in blood pressure, resolution of measured drop in blood pressure with orthostasis, or achieving desired mean arterial pressure (MAP) measurement. Partial response was defined as an improvement in symptom severity, reduced change in blood pressure with persisting orthostasis, or improvement in MAP without achieving target measurement. Missing or non-individualised data were documented as ‘not reported’. Descriptive statistics and qualitative analysis were employed.
Results
There were 3234 studies found in the initial database search and 14 studies identified through other sources (Figure 1). After removal of duplicates, 2823 were excluded at the title and abstract level and 60 articles were assessed at the full-text level. Overall, 13 studies (9 case studies and 4 case series) published between 1982 and 2021 met the inclusion criteria and were included in the review. There were no other study types found.

PRISMA 2009 flow diagram.
The 13 studies described 15 patients with pharmacological or non-pharmacological intervention for clozapine-induced hypotension (Table 1). There were 11 males and 3 females, with 1 patient’s sex not reported. Age was reported in 12 studies and ranged from 21 to 73 years with a mean age (standard deviation) of 45.3 years (15.6). Patient diagnoses included schizophrenia (11 patients), Parkinsonian dementia and delusional disorder (1 patient), psychotic depression (1 patient), and schizoaffective disorder (1 patient), with diagnosis not reported in 1 patient. Five studies were recorded in a psychiatric inpatient setting.38–42 Six studies were recorded in a critical care setting, such as emergency department, intensive care unit, and operating theatre,29–33,43 and the remaining two studies did not report the setting.44,45 Studies took place in the United Kingdom,29,30,42 Germany, 40 the United States,33,38,41,45 France, 44 and Australia,39,43 with two studies not reporting the location.31,32
Table of included studies.
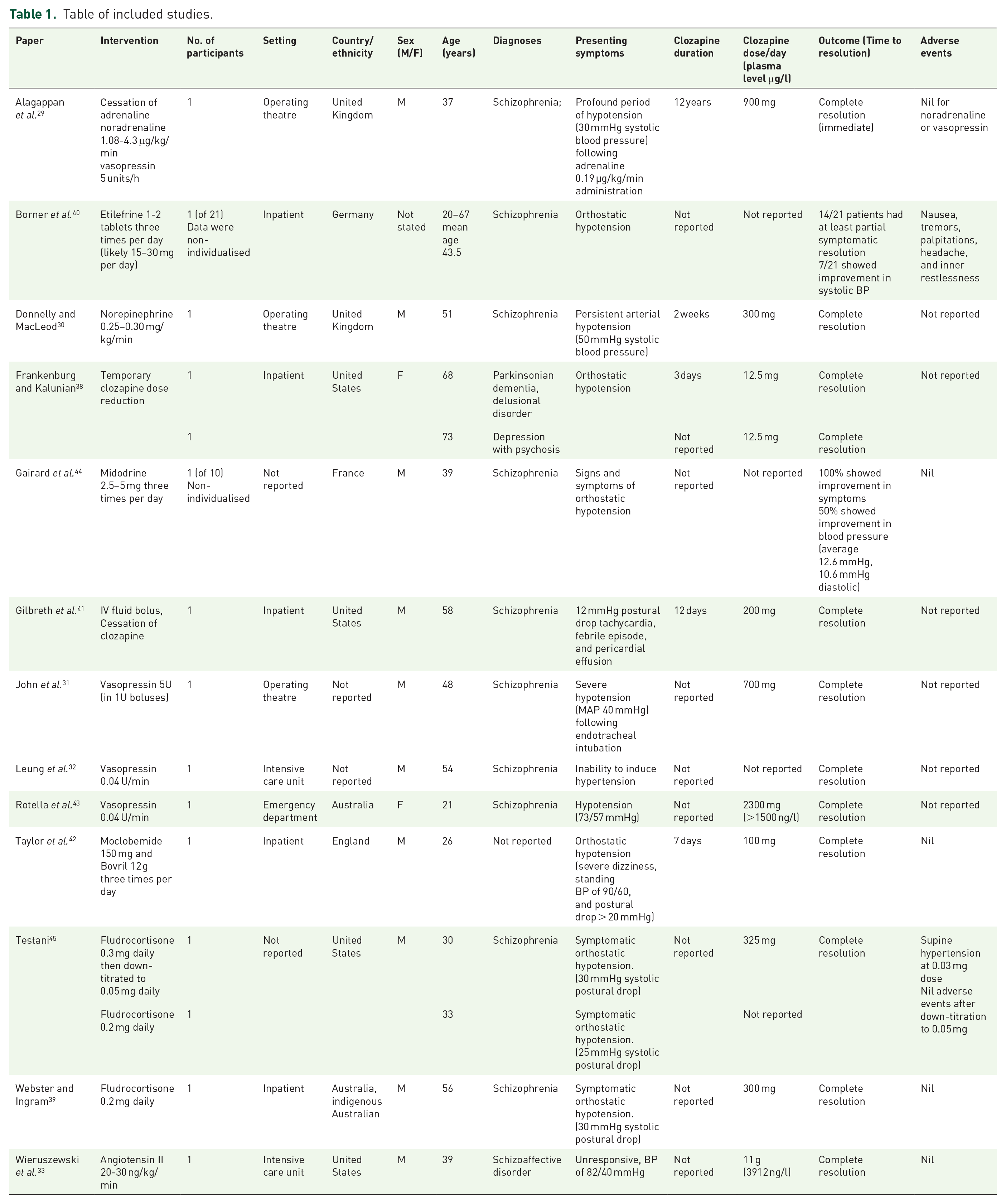
Daily clozapine dose was reported in eight studies and ranged from 12.5 to 900 mg daily. Two studies33,43 reported intentional massive ingestion of 2300 mg and 11 g of clozapine. Clozapine plasma level was only reported in these two studies (150043 and 3912 ng/l). 33 Seven studies with data for nine patients38–42,44,45 reported orthostatic hypotension on therapeutic doses of clozapine. Time of onset of orthostatic hypotension ranged from 3 to 12 days after commencement of clozapine. Other reported concurrent adverse events of clozapine included tachycardia,29,32,41,43 paralytic ileus, 29 lethargy, 38 pericardial effusion with febrile episode, 41 and weight loss. 39
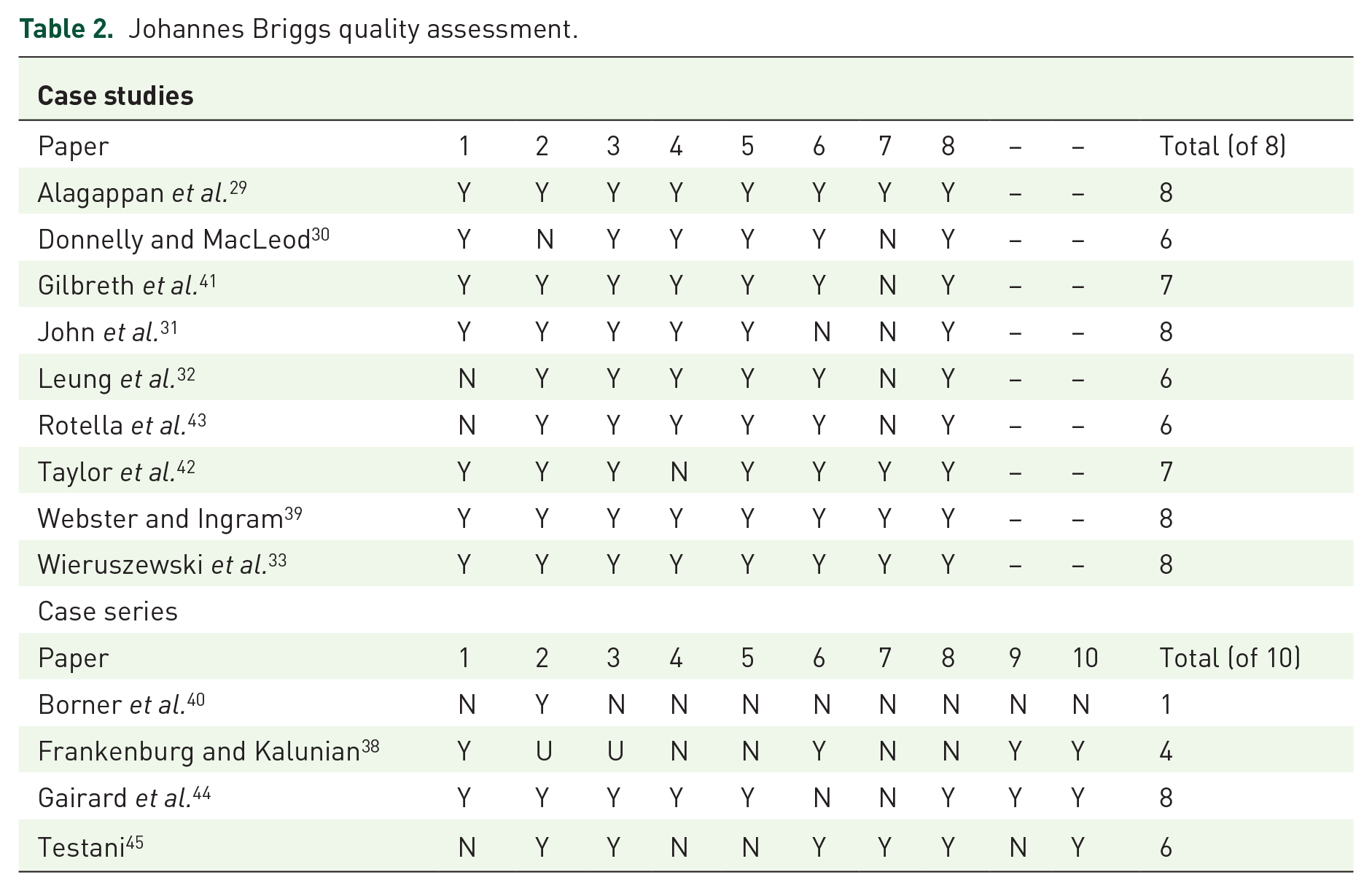
The studies were generally of high quality on the JBI clinical appraisal tool, although a failure to report adverse events of interventions was common. A summary of the study quality and a composite score has been presented in Table 2.
Johannes Briggs quality assessment.
Orthostatic hypotension and dizziness
A summary of the interventions and any safety considerations for the treatment of clozapine-induced orthostatic hypotension is presented in Table 3.
Summary of results for orthostatic hypotension interventions.
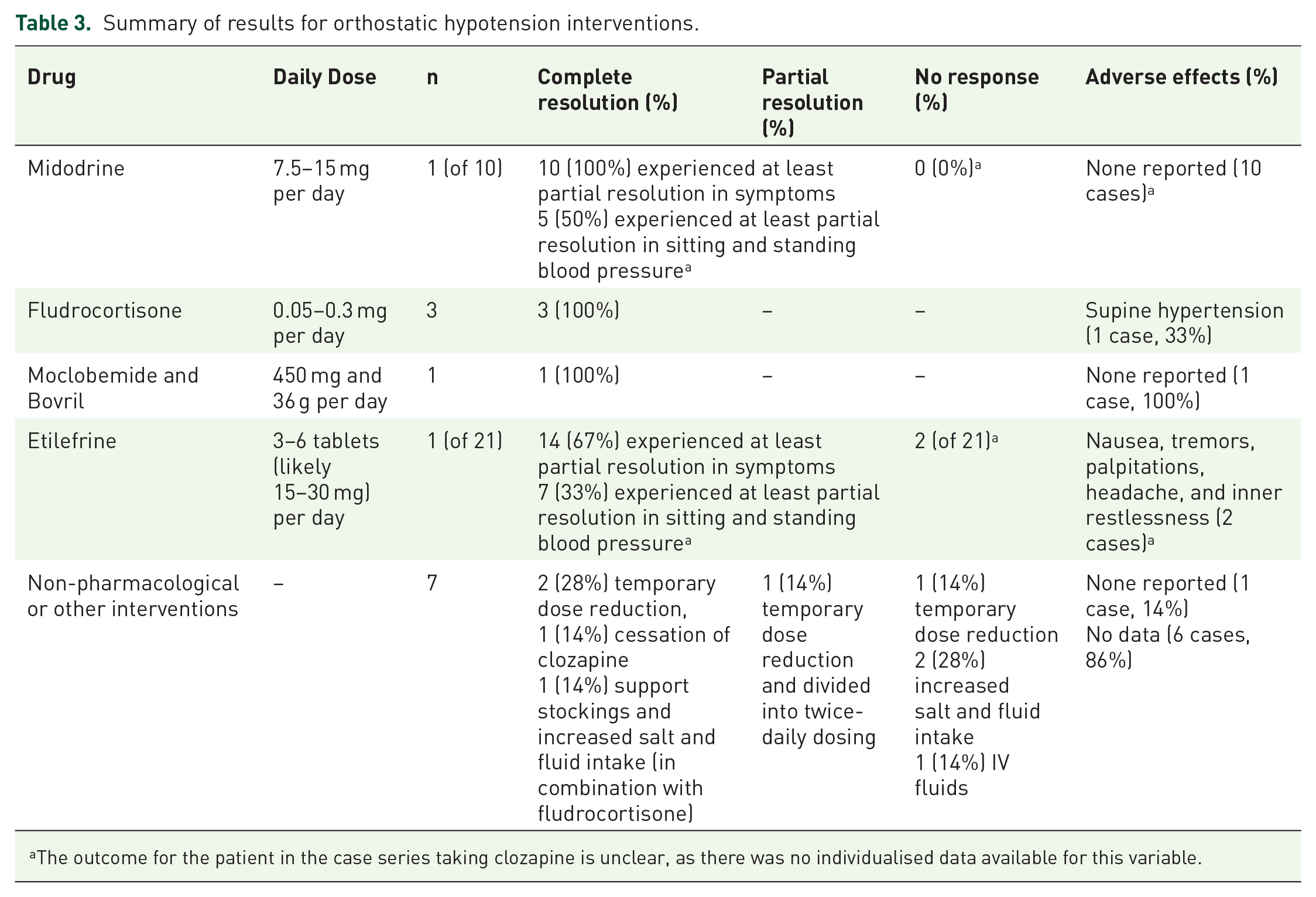
The outcome for the patient in the case series taking clozapine is unclear, as there was no individualised data available for this variable.
Alteration of clozapine dose and non-pharmacological interventions
Three studies trialled a dose reduction of clozapine with data for four patients.38,39,42 Two patients experienced complete resolution, although the magnitude of dose reduction was not reported in this case series. 38 One patient experienced partial resolution with a reduction of clozapine dose from 125 mg once daily to 100 mg daily in combination with splitting into 50 mg twice-daily dosing. 42 One patient experienced no response with clozapine dose reduction in combination with increased salt and fluid intake before successfully trialling fludrocortisone. 39 One patient was trialled on intravenous (IV) fluids with no response before experiencing complete resolution after ceasing clozapine. 41 One patient experienced complete resolution with support stockings and increased salt and fluid intake; however in this case report, these non-pharmacological interventions were used in combination with fludrocortisone (see below). 45
Pharmacological interventions
Fludrocortisone. Two studies39,45 provided data for three patients prescribed with fludrocortisone for symptomatic orthostatic hypotension. A dose of 0.2 to 0.3 mg once daily was used. All three patients achieved full resolution of orthostatic drop in blood pressure and resolution of associated symptoms. All three studies reported on adverse effects. One patient developed supine hypertension, which subsequently resolved with dose reduction from 0.3 daily to 0.05 mg daily. 45 The other two patients experienced no adverse effects.
Midodrine. One case series 44 used midodrine, an alpha-adrenergic agonist, at a dose of 2.5 to 5 mg three times per day for the treatment of orthostatic hypotension in 10 patients for 2 weeks. The study had non-individualised data available for one patient on clozapine. An average reduction in postural drop of 12.3 mmHg systolic and 10.6 mmHg diastolic was recorded, and a mean decrease in a study specific symptom severity score of 58%. While all patients reported a subjective improvement in the severity of symptoms, there was a persisting postural drop in the systolic blood pressure of five patients and diastolic blood pressure of four patients. No patients experienced adverse events.
Moclobemide and Bovril combination
One study 42 with data for one patient used moclobemide 150 mg, a reversible inhibitor of monoamine oxidase A, and Bovril® 12 g, a tyramine-rich yeast and beef extract, three times daily. This combination was effective in achieving full resolution of symptoms associated with orthostatic hypotension. Standing systolic blood pressure also improved from 90 to 130 mmHg; however, it remains unclear if a postural drop persisted as supine measurements was not made available.
Etilefrine
One study 40 reported the use of etilefrine, an adrenergic agonist, at a dose of three tablets (dose not presented but likely 5 mg per tablet) daily for 1 week, then six tablets daily for 3 weeks for the management of orthostatic hypotension in 21 patients. Only one patient in the study was taking clozapine and there was no individualised data reported on the patients’ outcome. Overall, 14 patients showed a decreased orthostatic reaction. Two patients, despite no recorded benefit in systolic blood pressure, reported less dizziness and headaches. Adverse events were reported in two patients, including nausea, tremors, and palpitations. One patient who was taking an antidepressant medication was withdrawn from the study due to restlessness. This was reproducible on cessation and re-initiation of etilefrine.
Refractory hypotension in critical care settings
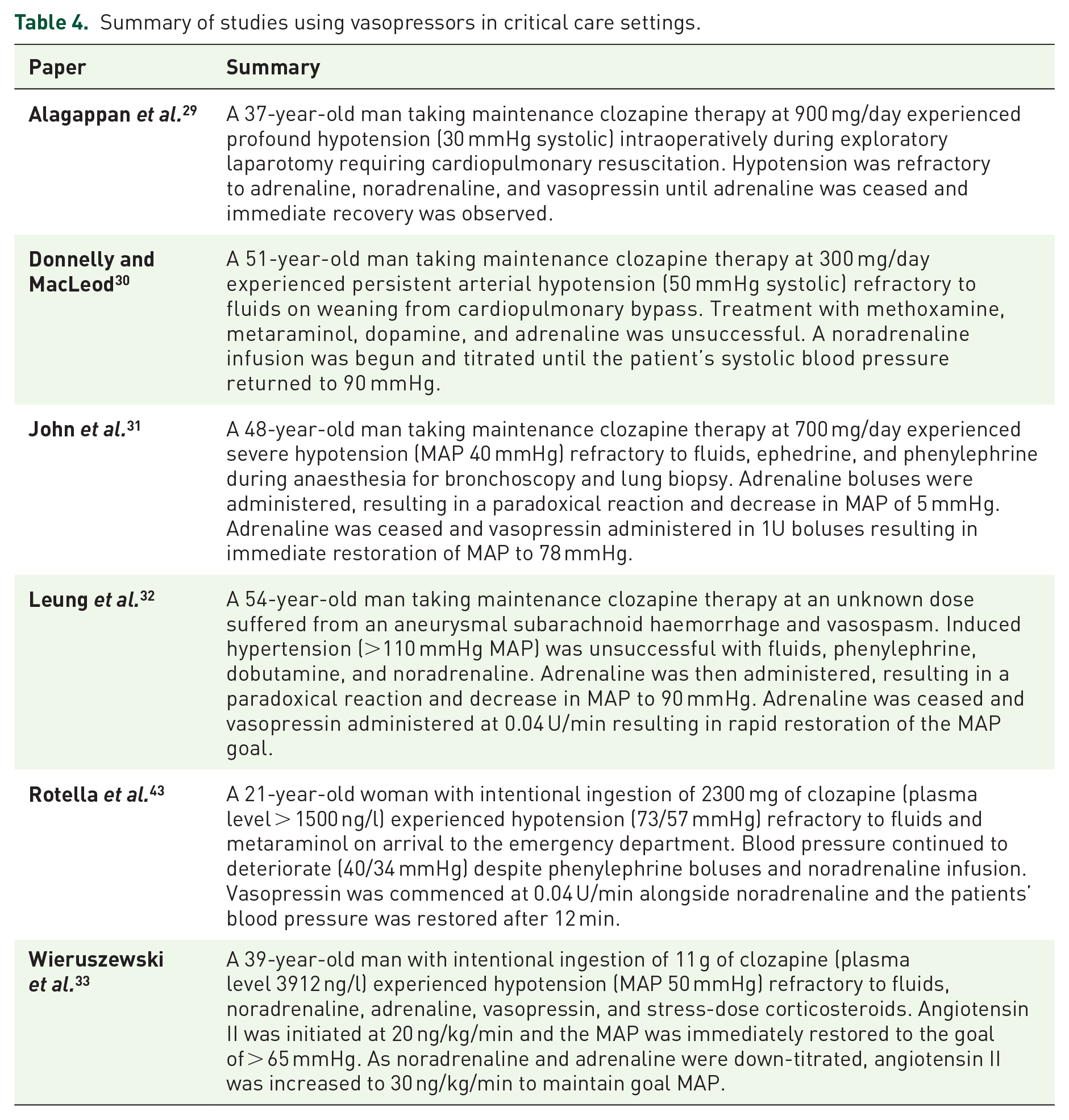
Six studies with data for six patients29–33,43 reported absolute hypotension refractory to fluids and vasopressors, with three of these studies29,31,32 reporting a paradoxical reaction of further hypotension with adrenaline co-administration. A summary of these studies, including the patient demographics and clozapine dose, is presented in Table 4.
Summary of studies using vasopressors in critical care settings.
Angiotensin II
One study 33 used angiotensin II in the setting of persistent hypotension despite escalating the use of adrenaline and vasopressin with data for one patient following intentional ingestion of 11 g of clozapine. A dose of 20 ng/kg/min of angiotensin II was successful in increasing the MAP from 56 to 66 mmHg within minutes. The dose of angiotensin II was then up-titrated to 30 ng/kg/min to maintain MAP at > 65 mmHg once vasopressin and epinephrine doses were decreased. The study reported no adverse effects.
Noradrenaline
Five studies29,30,32,33,43 reported the use of noradrenaline to restore or maintain MAP in critical care settings with data for five patients. In one study, 30 noradrenaline infusion was effective in restoring systolic blood pressure during a hypotensive episode refractory to methoxamine, metaraminol, dopamine, and adrenaline. In four studies,29,32,33,43 noradrenaline was unsuccessful in restoring systolic blood pressure or maintaining a goal MAP. One study reported no adverse effects. 30 The other four studies did not report on adverse effects.29,32,33,42
Arginine vasopressin
Five studies29,31–33,42 reported the use of IV vasopressin with data for five patients. Three studies31,32,42 reported complete resolution of hypotension refractory to fluids and other vasopressors. Of these, one study 31 used five boluses of vasopressin 1U, and two studies32,43 used a vasopressin infusion of 0.04 U/min. In two other studies,29,33 vasopressin used at 0.0833 and 0.083 U/min 29 proved unsuccessful in resolving hypotension. However, in both studies, vasopressin was co-administered with adrenaline. A clear paradoxical reaction to adrenaline was reported in one of these studies. 29 Adrenaline was ceased, and the patient remained on IV vasopressin until the MAP was stabilised. In three studies,29,31,33 patients experienced no adverse events from vasopressin. Two studies32,43 did not report on adverse events.
Discussion
Main findings
Clozapine has the highest incidence of orthostatic hypotension of all atypical antipsychotics.19,46–50 Despite evidence showing clozapine commonly causes cardiometabolic side effects, such as orthostatic hypotension and associated dizziness, leading to discontinuation, 11 limited guidance exists on the management of this adverse effect. Complications of orthostatic hypotension include cerebral hypoperfusion leading to dizziness, faintness, visual disturbances, headaches, and cognitive impairment, and autonomic compensation leading to palpitations, chest pain, and syncope. 51 Together, these mechanisms increase the risk of falls and subsequent injuries, particularly in older persons who are more susceptible to the α-adrenergic effects of clozapine. 52
This study provides the first systematic review specifically targeting studies describing the management of clozapine-induced hypotension. All 13 studies found in this review were case studies (N = 9) or case series (N = 4). Improvement in symptoms and clinical measures of orthostatic hypotension were reported in this review for fludrocortisone, midodrine, a moclobemide–Bovril combination, and etilefrine. Improvement in restoring or maintaining arterial pressure for refractory hypotension was reported for vasopressin, noradrenaline, and angiotensin II. All hypotension interventions were generally well tolerated, although 31% did not report on adverse effects.
Limitations
There are limitations that need to be considered with these results. We were unable to provide any quantitative analysis of studies due to the absence of any RCTs or cohort studies. All studies were case reports or small case series and may be subject to selection, reporting, and publication bias. The recommendation of fludrocortisone is based on one case study and one case series only and is supplemented with clinical experience and pharmacological reasoning. Poor reporting on clozapine plasma level, duration of treatment, and short duration of following-up may limit the reliability of the results. There remains a need for RCTs of pharmacological interventions for clozapine-induced orthostatic hypotension that use validated rating scales, and inclusion of symptomatic patients only to accurately assess the safety and efficacy of treatments.
Framework for the management of clozapine-induced orthostatic hypotension
Given the limited available evidence, we have created a management framework designed to assist clinicians, patients, and carers on the management of clozapine-induced orthostatic hypotension, faintness, and dizziness (Figure 2). We have balanced this based on the risks and benefits of each intervention rather than solely the number of cases available.

Management framework for treatment of clozapine-induced posutral hypotension.
Before initiating clozapine, antipsychotics with potent α1-antagonism should be cross-tapered, and a review of concomitant medicines which may contribute to clozapine’s strong α1-adrenergic effects should be considered. Medications that commonly cause or contribute to orthostatic hypotension include anti-hypertensives, anti-anginal agents, diuretics, tricyclic antidepressants, antipsychotics with α1-adrenergic effects, and nonselective α1 antagonists-for benign prostatic hyperplasia or urinary symptoms.14,15,53
Careful up-titration of clozapine is recommended during the initiation of therapy. Doses should be given twice daily and any increase limited to between 12.5 and 25 mg per day, in association with frequent monitoring of sitting and standing blood pressure. 54 As symptoms of orthostasis may occur without meeting clinical criteria for orthostatic hypotension, direct questioning on the frequency and severity of symptoms is recommended. Patients should be reminded that dizziness is a common side effect of clozapine during initiation and be encouraged to stand slowly and in stages from lying to sitting and sitting to standing.14,15,53,55 Patients should also be encouraged to report the first signs of dizziness to allow time for dose adjustments of clozapine or other medications and to prevent serious adverse events and discontinuation. Grading tools are available for both clinicians and patients to assist in assessing the severity of orthostatic hypotension.56,57
Elderly patients, non-smokers, those with a history of autonomic dysfunction, hepatic disease, or patients taking medications that inhibit the hepatic CYP450 metabolism of clozapine (1A2, 2C19, 3A4, and 2D6)58,59 or medications with hypotensive effects may require a slower dose titration schedule or more frequent monitoring of orthostatic vital signs during initiation phase.60,61
If symptomatic orthostatic hypotension occurs, a temporary reduction of clozapine to the most recent tolerable dose or a slower titration schedule may be required as tolerance to its α-adrenergic activity is established. Dose reduction and cross-tapering of interacting medicines or spacing medicines with hypotensive effects 12 h away from the clozapine dose may also be helpful. For some patients, clozapine-induced orthostatic hypotension may become a persistent clinical issue resulting in a dose-limiting effect requiring behavioural or pharmacological intervention. Although only one case study describing behavioural intervention was found in this systematic review, general treatment guidelines for orthostatic hypotension recommend them first-line and reserving pharmacological therapy for refractory cases.51,52,56,57
Medical causes should be excluded including volume depletion, neurodegenerative conditions and autonomic neuropathies, cardiac conditions, and adrenal insufficiency. 51 Adequate hydration (1.5–3 litres/day) and dietary sodium intake (6-10 g/day) should be encouraged.14,15,53,55,62 If the patient is experiencing post-prandial orthostatic hypotension, encourage small, frequent meals, drinking water during meals, and limiting consumption of alcohol, hot drinks, and high-glycaemic index foods. Pre-emptive consumption of fluid prior to standing may help alleviate symptoms.14,62,63 Other interventions include decreasing nocturia by raising the head of the bed approximately 15 cm and decreasing venous pooling with physical manoeuvres or compression garments.14,15,53,62–64 Compliance with compression garments, such as abdominal binders and elastic tights, is likely to be poor in warmer climates. 65
Should symptomatic orthostatic hypotension be refractory to clozapine dose adjustments and non-pharmacological management, fludrocortisone may be effective as an acute agent (refer Figure 2 for dosing and precautions). Fludrocortisone, a synthetic mineralocorticoid, is contraindicated for patients with congestive heart failure, chronic renal failure, and hypertension. 66 Hypokalaemia occurs in up to 50% of patients and potassium levels should be monitored. 67 Fludrocortisone is associated with peripheral oedema and weight gain, exacerbation of supine hypertension, 66 end-organ damage, including left ventricular hypertrophy, 68 congestive cardiac failure and renal failure, 69 and an increased risk of hospitalisation. 66 Given clozapine’s association with cardiomyopathy 8 and metabolic syndrome, 70 long-term use of fludrocortisone should be avoided. 71
For patients who do not respond or for whom fludrocortisone is not appropriate, midodrine may be trialled. Midodrine is a selective peripheral α1-adrenoreceptor agonist which increases blood pressure by inducing vasoconstriction. Although our review only identified one study for midodrine, it is one of the few pharmacological agents for general orthostatic hypotension tested with placebo-controlled trials and is well tolerated with few serious adverse events.72,73 Compared with fludrocortisone, midodrine has lower rates of heart failure and all-cause hospitalisation rates.67,74,75 However, it has a short duration of action (2–4 h) and thus the recommended dosing schedule of three times daily may not be practical for patients with severe mental illness or with a history of poor medication compliance. Common adverse events are supine hypertension, piloerection, and scalp pruritus. 76 Screening for hypovolaemia is important as midodrine is not effective when plasma volume is reduced. Caution should be exercised in patients with renal failure or urinary retention.14,15,53,55,63
Moclobemide, a reversible inhibitor of monoamine oxidase A, was an alternative agent found in this review. 77 Moclobemide prevents the breakdown of tyramine, a substrate of monoamine oxidase, 78 and acts as an indirect peripheral sympathomimetic. 79 When moclobemide is co-administered with tyramine-rich foods, such as Bovril, there is potential for the inhibition of breakdown of adrenaline, noradrenaline, dopamine, serotonin, and tyramine. Moclobemide is well tolerated and is devoid of significant interactions with clozapine. 80 Hypertensive crisis may occur, 79 although, in doses of less than 900 mg per day, the risk is considered negligible compared with older, non-reversible Monoamine oxidase inhibitors. 77 It should be noted that aside from the study presented in this review, and another for a patient not taking clozapine, 81 very scant literature exists to support the use of this combination.
Although etilefrine, a direct α1-adrenergic agonist with strong vasoconstriction effects, 82 showed benefit for the management of psychotropic-induced orthostatic hypotension, we have not recommended its use. In 2014, the French Health Products Agency concluded etilefrine had an unfavourable risk-benefit profile and recommended it to be withdrawn from the market 83 due to the serious risk of cardiovascular disorders. 83
Other agents that have not been reported for patients taking clozapine but are commonly used for orthostatic hypotension include droxidopa, 84 pyridostigmine, 85 and desmopressin. 86 The use of ephedrine and pseudoephedrine has also been reported; however, evidence is weak, and the danger of misuse, diversion, and psychosis means that these agents are less suitable for patients with treatment-resistant schizophrenia. 69
Refractory hypotension in critical care
We also found that clozapine-induced absolute hypotension that was refractory to IV fluid resuscitation and vasopressors both intraoperatively following massive ingestion.29–33 Catecholamines, such as vasopressin, noradrenaline, and adrenaline, are commonly used to maintain MAP in critical care settings. However, clozapine may attenuate these effects due to potent α1-adrenergic antagonism leading to vasodilation and an α-reflexive sympathetic response. 87 Furthermore, a paradoxical reaction of severe hypotension following co-administration of adrenaline has been observed.29,31,32 There is limited guidance available on the choice of catecholamine for fluid-resistant shock, and choice of vasopressor is often determined by the preference of the treating clinician.88,89 Noradrenaline and adrenaline have similar action on α- and β-adrenergic receptors; however, adrenaline has much greater β2-adrenergic activity which is responsible for the relaxation of smooth muscle. Clozapine-induced α-blockade, especially in overdose, may result in unopposed β2-agonism on administration of adrenaline leading to severe vasodilation and hypotension refractory to IV fluid and vasopressors.29,31–33 This paradoxical reaction has also been observed following quetiapine overdose, another antipsychotic with potent α-adrenergic action. 90 Noradrenaline is the catecholamine of choice in patients with tricyclic antidepressant-induced refractory hypotension, and the use of adrenaline is not recommended. 91
Despite this, surgeons and anaesthetists should not withhold clozapine prior to surgery, due to the risk of psychotic relapse and serious withdrawal effects.92–96 Clinicians should avoid the use of adrenaline in patients taking clozapine, tricyclic antidepressants, or other antipsychotics with potent α-adrenergic antagonism. The vasopressors, angiotensin II, arginine vasopressin, and noradrenaline appear effective in maintaining and rapidly restoring MAP in patients taking clozapine.
Conclusion
Clozapine-induced hypotension is a common adverse event that is associated with significant harm, reduced quality of life, and treatment non-compliance. In addition to causing orthostatic hypotension and dizziness, clozapine attenuates the effects of several common vasopressors and may cause severe refractory hypotension in a paradoxical reaction with adrenaline co-administration. Due to the lack of quality evidence available for the use of the pharmacological and non-pharmacological interventions for orthostatic hypotension, we have developed a management framework to assist patients, clinicians, and carers. Any intervention should follow an assessment of fluid and salt intake, a temporary reduction in clozapine dose or speed of titration schedule, and a review of potent α1-adrenergic or hypotensive agents. Despite the risk of intraoperative hypotension, clozapine should not be routinely withheld prior to surgery. Should intraoperative hypotension occur, clinicians should avoid the use of adrenaline in patients taking clozapine.
Footnotes
Author contribution(s)
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: D.S. is supported in part by a National Health and Medical Research Council (NHMRC) Early Career Fellowship (GNT1111136). N.W. receives speaker fees from Otsuka, Lundbeck and Janssen.
