Abstract
Brugada syndrome (BrS) presents with a characteristic electrocardiogram (ECG) and is associated with sudden cardiac death. Until now, prolongation of QTc interval and its association with Torsade de Pointe and possible fatal arrhythmia have been the focus of routine baseline ECGs before prescribing psychotropic medication. A semi-systematic literature review was conducted using PubMed. The terms ‘Brugada’, ‘Brugada Syndrome’ AND ‘psychotropic’ ‘antipsychotic’ ‘antidepressant’ ‘mood stabilisers’ ‘clozapine’ ‘Tricyclic Antidepressants’ ‘Lithium’ were searched. From a search that delivered over 200 articles, 82 articles were included. Those that included details around causative medication, doses of medication and where clear timeline on drug cause were included. Where clarification was needed, the manufacturer of the medication was contacted directly. Psychotropic medication can be associated with BrS, Brugada phenocopy or unmasking of BrS, in overdose or in normal doses. Our results include a table summarising a number of psychotropic overdoses that led to BrS unmasking. Routine screening for BrS in patients before prescribing psychotropic medication is a natural extension of the baseline ECG currently routinely done to rule out QTc prolongation. Psychiatrists need to invest in ensuring better skills in interpreting ECGs and work closer with cardiologists in interpreting ECGs.
Introduction
Brugada syndrome (BrS) is an inherited disease that is presented with a characteristic electrocardiogram (ECG) and can be associated with fatal complications and premature sudden unexpected cardiac death. 1 Having been introduced as a new clinical entity by Pedro and Josep Brugada 2 in 1992, the syndrome is considered responsible for nearly 20% of all sudden cardiac deaths (SCDs) in patients with structurally normal hearts. It is important to realise that any given patient with BrS can have marked day-to-day ECG changes making diagnosis challenging. In a large series of patients with Brugada who had repeated ECGs over the years, only every third ECG showed type 1 and every other third ECG was normal. 3 Some psychotropic medications can unmask BrS or precipitate Brugada-like ECG changes. Brugada pattern ECG has also been found to be more common in those with schizophrenia spectrum disorders. 4
Method
A semi-systematic literature review was conducted using PubMed. The terms ‘Brugada’, ‘Brugada Syndrome’ AND ‘psychotropic’ ‘antipsychotic’ ‘antidepressant’ ‘mood stabilisers’ ‘clozapine’ ‘Tricyclic Antidepressants’ ‘Lithium’ were searched. From a search that delivered over 200 articles, 82 articles were included. Those that included details around causative medication, doses of medication and where clear timeline on drug cause were included. Further articles were identified from the articles reviewed. Where clarification was needed, the manufacturer of the medication was contacted directly.
Definition of Brugada-type ECG
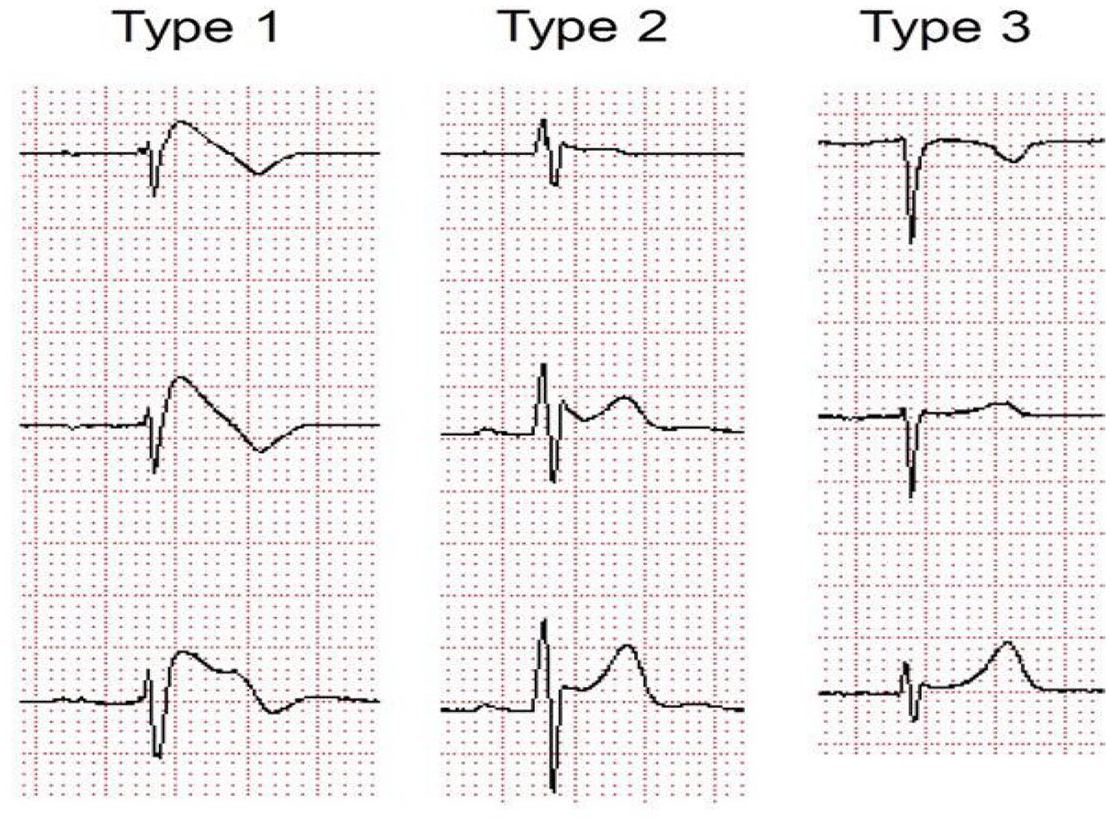
Brugada-type ECG is characterised by several different patterns, of which only type 1 is diagnostic. 5
Type 1 is a prominent cove-shaped ST-T segment with a J-wave amplitude or ST elevation ⩾2 mm (0.2 mV) at its peak followed by a negative T wave, with little or no isoelectric separation. This is the only diagnostic pattern for BrS. 1
Type 2 is a high take-off ST segment with a J-wave amplitude ⩾2 mm with gradually decreasing ST-segment elevation (remaining ⩾1 mm above baseline, followed by a positive or biphasic T wave that gives rise to saddle-back configuration). Type 3 is defined if right precordial ST-segment elevation is <1 mm of a saddle-back type, coved type, or both. Type 2 and type 3 patterns are suggestive of BrS but are not diagnostic in themselves (Figures 1 and 2).

Three types of Brugada ECG pattern.
Brugada-type ECG shift.
Pathophysiology
BrS was initially considered to have an autosomal dominant inheritance pattern with incomplete penetrance and primarily associated with mutations in the SCN5A gene, which encodes for the pore-forming alpha subunit of the cardiac Na+ channels. Over 300 mutations in SCN5A have been identified thus far, accounting for up to 30% of BrS probands. In recent years, however, over a dozen further pathogenic variant genes have been associated with BrS, for example, by altering the normal function of Na+, K+, Ca2+ and hyperpolarization-activated cyclic nucleotide-gated (HCN) channels. These additional gene mutations only account for around 10% of probands however. 6 Both depolarization and repolarization abnormalities have been described in BrS. 7
Although genetic mutations appear to be equally distributed between sexes, the clinical phenotype of BrS is almost 10 times more prevalent in men than in women. 8
Furthermore, women with BrS tend to present with more benign clinical characteristics and less spontaneous type 1 ECG pattern and are more likely to be asymptomatic than men. 9 Although the drivers behind this sex-related distinction remain to be identified, there is evidence suggestive of still unknown, but there is evidence to suggest a hormonal basis. Interestingly, reversion of the phenotype has been noted in two patients with prostate cancer after surgical castration suggesting a possible role for androgens. 1
Recent evidence demonstrating normalisation of the pathognomonic ECG pattern and elimination of the arrhythmic disposition in most patients following radiofrequency ablation of the right ventricular (RV) outflow tract epicardium is suggestive of the possibility that structural abnormalities, likely at a microanatomical scale, also play some role in the pathophysiology of BrS. As such, BrS and arrhythmogenic cardiomyopathy have been proposed to be part of the same disease spectrum. 10
Brugada ECG pattern is more common in those diagnosed with schizophrenia spectrum disorder, conditions which are also associated with ion channel abnormalities. A recent case control study of 338 patients recently diagnosed with a schizophrenia spectrum disorder and 844 healthy controls identified Brugada pattern ECG in 33 patients (8.5%) and 13 healthy controls (1.5%), with an adjusted odds ratio of 3.5 (p < 0.0001). 4 An earlier study found that as many as 4% of patients with schizophrenia had type 1 Brugada ECG compared with an estimated prevalence of 0.05% in the general population. 11
Brugada incidence and prognosis
The worldwide incidence and risk of death in those with a Brugada ECG pattern remains unclear despite a developing understanding of this disorder since its initial description in 1992. 12 Since the first descriptions reported high mortality, the reported incidence of SCD has been declining and there is a clearer understanding that some patients accumulate the highest risk for developing malignant ventricular arrhythmias while others follow a benign course with a long-life expectancy. 13 That being said, BrS has a significant mortality attached and accounts for 12% of all SCDs and around 20% of SCDs in patients with structurally normal hearts. 2 Symptomatic presentation with previous aborted cardiac arrest or sustained ventricular arrhythmia carries the highest risk, with a reported annual rate of repeated arrhythmic event of around 8%. In such cases, secondary prevention with implantable cardioverter-defibrillator (ICD) implantation is recommended. 10
Two meta-analyses reviews have attempted to give a world prevalence; the first gives a prevalence of Brugada ECG pattern as 0.4% with different prevalence according to region – highest in Asia (0.9%) and North America (0.2%) – and according to sex – male (0.9%) and female (0.1%). 12
In another meta-analysis, the worldwide pooled prevalence was predicted as 0.5 per 1000 [95% confidence interval (CI) = 0.3–0.7]. The prevalence in South East Asia was 3.7 in 1000 (95% CI = 0.7–6.7), while the prevalence was much lower in North Africa (0 in 1000). 14
A study of population >55 years old, otherwise healthy Han Chinese population (n = 5214) in Taiwan, found the overall prevalence of Brugada ECG changes to be 3.32%. 15 In a large hospital-based Taiwanese population setting where ECGs were examined as part of routine care (non-cardiology reasons for admission), just under 75,000 ECGs were examined from 20,562 patients and 26 (0.13%) were confirmed to have Brugada-type ECGs. 16 When these 26 patients were followed up, neither SCD nor hospitalised death differed between these patients and those without Brugada-type ECG. 16 Asymptomatic patients with Brugada-type ECGs were not rare in this cohort of hospitalised patients; however, their risk of SCD was not increased. 16 This differed to smaller sample size studies in hospitalised patients where participants had either Brugada-type ECGs or a family history of SCD. 17 By comparison, a Danish general population study of the prevalence and relevance of Brugada-type ECG found a prevalence of 7 in 10,000, and none of the participants presented with type 1 BrS. 18
In a study consisting of 3021 young and middle-aged participants in the Finnish population, no type 1 Brugada ECG shift was found in any participants and 18 participants had type 2 or 3 Brugada shift ECG. 19 No mortality or life-threatening ventricular arrhythmias occurred in the populations studied, and the authors concluded that type 2 or 3 Brugada ECG pattern is a normal variant rather than a specific predictor of life-threatening ventricular arrythmias especially in those without a family history of SCD. 19 In more recent studies where the long-term prognosis of 1029 patients diagnosed with BrS using patients recruited from 11 tertiary centres in four European countries (France, Italy, the Netherlands and Germany), the authors concluded that the cardiac event rate per year was 7.7% in patients with aborted SCD, 1.9% in patients with syncope and 0.5% in asymptomatic patients. 20 In this study, the presence of symptoms and a spontaneous type 1 ECG are the only independent predictors of arrhythmic events. 20 Conversely, sex, family history of SCD, inducibility of ventricular tachyarrhythmias and genetic factors had no predictive value. 20
Screening for BrS involves the following:
A baseline ECG to identify Brugada-type ECG shifts: the identification of which may require expert interpretation of ECG. However, do note the variation associated with ECG presentations previously described. 14
A challenge test: in working-age adults, this can be done using ajmaline 1 mg/kg over 5 min IV, flecainide 2 mg/kg over 10 min IV or 400 mg orally, procainamide 10 mg/kg over 10 min IV or pilsicainide 1 mg/kg over 10 min IV. Following administration, the patient’s ECG is closely monitored to determine the Brugada pattern.21,22
Genetic testing will reveal whether there is a mutation in the sodium ion channel gene SCN5A and hence whether or not there is a familial predisposition to BrS. However, an SCN5A mutation and family history do not have any impact on prognosis, so they are not used for risk stratification, and mutations are only identified in approximately 25% of people with the BrS.23,24
Fever-induced Brugada
Fever-induced Brugada was studied and ECG recordings from 402 febrile and 909 afebrile patients of a similar age were compared. 25 The authors concluded that type 1 Brugada pattern was 20 times (n = 8 in febrile versus n = 1 in afebrile) more prevalent among febrile patients (2% versus 0.1%; p = 0.0001). 25 None of the febrile-induced BrS patients were of South East Asian origin and had an electrolyte imbalance, bradycardia, prolonged PR interval or a family history of SCD. 25 Two of the febrile-induced BrS patients were taking medication associated with BrS: clothiapine and carbamazepine; their ECGs normalised, however, once the fever resolved.
Brugada phenocopy
Brugada phenocopy (BrP) is a clinical condition which leads to Brugada-like ECG but is secondary to an underlying pathological condition rather than BrS itself such as hyperkalaemia, hypokalaemia, left ventricular aneurysm, pericarditis or pulmonary embolism. 7 Once the aetiology is resolved, the BrP ECG pattern normalises. 26 Differentiating between BrP and BrS is challenging but important, not least because while the clinical implications of BrP remain uncertain, patients with BrS are at increased risk of SCD and may require an ICD. 27
Medication used to treat Brugada
Management of acute presentation in A&E
Whenever a patient presents with syncope or cardiac arrest, BrS needs to be one of the differential diagnoses and an ECG must be performed.21,22 Alongside supportive management of the patient, sodium bicarbonate IV has demonstrated efficacy when used to promote the recovery of a ‘normal’ ECG pattern.28 –31 Any fever should be reduced immediately; paracetamol can be prescribed for this. 21 Isoproterenol, a non-selective β adrenoceptor agonist, has been found to be effective and is a drug of choice for the acute treatment of Brugada, particularly in the context of electrical storm as further outlined below. 32
Ongoing management
In the longer term, the most important action is to discontinue any medicine that precipitated BrP or unmasked BrS and avoid other medicines with high risk of precipitating it. 21 The website www.brugadadrugs.org includes lists of medicines to be avoided, medicines to be avoided preferentially and medicines that are potentially anti-arrhythmic. Close collaboration between the patient, the patient’s carer(s) and all health professionals involved in the patient’s care is recommended to plan ongoing management of any co-morbid conditions, whether mental ill health or physical ill health. 33 Where BrS is clearly medicine-induced, further intervention may not be needed unless the patient also has other underlying risk factors.21,33 The patient needs to be educated to avoid excessive alcohol intake.
To determine which intervention is necessary to manage the BrS, the patient’s risk will be classified as follows:
Numerous factors have been put forward in relation to risk stratification though syncope and spontaneous type 1 ECG pattern are the only ones that have remained consistent in their predictive role across multiple studies. 34 There remains controversy around the value of electrophysiologic study (EPS) for risk stratification in patients with BrS. 35
The intervention that is most evidence-based in those deemed at higher risk of SCD with BrS, and therefore first-line, is implantation of an ICD.21,22 However, this procedure is not without risks and complications, particularly in younger, more active patients.
If a patient suffers an electrical storm, which is defined as three or more episodes of ventricular tachycardia/ventricular fibrillation (VT/VF) in 24 h, this is treated with outward potassium current blockers, for example, (hydro)quinidine, and beta-sympathomimetics, for example, isoproterenol. 21 Anything that may have precipitated this, including fever or hypokalemia, must be treated concurrently. In the case of a patient with a spontaneous type 1 Brugada ECG who is asymptomatic, (hydro)quinidine can be considered.
Psychotropic medications associated with Brugada
As already described, a significant number of factors can influence the electrocardiographic and arrhythmic manifestations of BrS. With regard to psychotropic medication, the principal mode of action is blockade of ventricular sodium and calcium channels. 36 This differs from the most common mechanism underlying QT prolongation in psychotropic drugs, namely blockade of potassium channels in the myocardium. 37
In some of the cases reviewed in this article, a particular psychotropic medicine unmasked BrS while in others may have precipitated BrP.28 –31,38 –40 For example, intoxication with drugs that inhibit cardiac sodium channels, such as tricyclic antidepressants, can trigger BrP in otherwise normal individuals. 31 Differentiating between the two is challenging but important in informing the patient’s future care plan.
Although women are considered to be at greater risk of arrhythmia associated with prolonged QT interval, interestingly, in BrS, men are at higher risk of both developing the clinical phenotype for the disorder and also of suffering cardiac events following exposure to psychotropic medication that can unmask the disorder. 41
Although there is little in the literature on the topic, age is potentially a further complicating factor in relation to the presentation of Brugada-type ECG, in the context of psychotropic drug use and indeed more generally. For example, there is an increased likelihood of experiencing comorbidity as we grow older, including cardiac comorbidity. There is also likely to be additional medication burden that could include drugs that impact sodium and calcium channels.
Tricyclic antidepressants
The cardiovascular adverse effects of tricyclic antidepressants, both in therapeutic doses and in overdose, are well documented. This section will review case reports that specifically mention the BrS.28 –31,38 –40 Please see Table 2 for further details of the cases.
Risk stratification and treatment of Brugada Syndrome. 21 .

ECG, electrocardiogram; ICD, implantable cardioverter-defibrillator; VF, ventricular fibrillation; VT, ventricular tachycardia.
Demographically, the cases include both men and women aged between 33 years and 66 years. In some of the cases, there was no evidence of pre-existing BrS.28,30,31 However, with others, it was difficult to tell whether the antidepressant precipitated the ECG pattern or unmasked an existing BrS. 39 A baseline ECG may have clarified this.
It is noteworthy that in two of the cases, the patient presented as collapsed and unconscious.38,39 In two other cases, clinical signs were normal on first presentation, though subsequently deteriorated. 39 In a further four cases, heart rate, temperature, blood pressure and respiratory rate were found to be deranged while all other results were normal.28,30 It was only on performing an ECG that BrS was identified. Therefore, whenever a patient, who is prescribed a tricyclic antidepressant, presents with syncope, an ECG is recommended to screen for BrS whether or not any other cardiovascular signs are present.
Minoura et al. 42 investigated the possible underlying ionic and cellular mechanisms by examining the effects of amitriptyline on the experimental model of a coronary-perfused canine RV wedge preparation. Their results suggested that because of its potent blockade of the fast sodium current with little inhibition of the transient outward potassium channel current, amitriptyline can cause Brugada in patients with a genetic predisposition. In a subsequent study, Chen et al. 43 explored both the acute and long-term effects of some tricyclic antidepressants using neonatal rat ventricular myocytes. The acute effects of amitriptyline on the gating properties of the Nav1.5 cardiac sodium channel resulted in decreased peak current and delayed recovery from activation. In the longer term, there was a significant reduction in the expression of the Nav1.5 protein on the cell membrane, suggesting that amitriptyline caused the protein to be retained within the cell. Furthermore, the interaction between Nav1.5 and the Ankyrin-G or dystrophin antibodies was disrupted. Thus, with both amitriptyline and clomipramine, long-term treatment produces greater decreases in peak sodium current than acute treatment. With nortriptyline, both acute treatment and long-term treatment resulted in similar decreases in peak sodium current. There were no significant changes, either acute or long-term, with desipramine.
Mood stabilisers
Unmasking of BrS has been reported with lithium treatment.43 –45 Similarly, Brugada-type ECG changes have been reported with long-term lithium treatment. 46 In one reported case, lithium was being taken together with lamotrigine and ziprasidone. The patient’s electrolytes were normal, and his lithium level was 0.7 mEq/L. The patient’s ECG normalised when his lithium was withheld. It is thought that lithium exerts this effect via its ability to block sodium-ion channels.
In another case, a 52-year-old woman with depression presented with atypical precordial pain having taken lamotrigine for roughly 5 months. 47 Her ECG suggested type 1 Brugada pattern, although subsequent ECGs were less typical. Examination revealed normal coronary arteries. Her flecainide test was positive. Stopping the lamotrigine led to reversal of the Brugada-pattern ECG.
A 61-year-old man with schizophrenia who was treated with carbamazepine was hospitalised and diagnosed with pneumonia. On the following day, electrocardiography showed Brugada-pattern ST elevation in the right precordial leads and a blood examination revealed that the patient’s carbamazepine concentration was at the upper limit of the standard range. The patient’s ECG normalised after the withdrawal of carbamazepine. 48
Antipsychotics
A 62-year-old man with diabetes, chronic ischemic cerebrovascular disease and bipolar disorder, who was taking pregabalin and quetiapine, presented with dyspnoea and chest discomfort. 49 His temperature on admission was 36.6°C, his pH was 7.2 and his potassium levels were 3.4 mEq/L. His ECG on admission showed Brugada-like pattern. Initially, acute coronary syndrome was suspected. However, coronary angiography showed only mild disease, echocardiography did not reveal abnormalities and troponin levels were normal. Pneumonia was identified via lung opacities and consolidation found by chest radiography. He received positive airways pressure ventilation and antibiotics and recovered after 10 days. The mechanisms by which quetiapine and pregabalin may have exacerbated this presentation are not clear. Possibly, quetiapine metabolites may impair noradrenaline recycling via their affinities for the noradrenaline reuptake transporter, the serotonin receptors and the alpha (1B)-adrenergic receptor. A proposed mechanism for pregabalin is binding the alpha2-delta protein, which is an auxiliary subunit of the voltage-gated calcium channels.
Cases of Brugada have also been noted with clozapine.11,50 In the detailed, published report, the patient was a 44-year-old man with a long history of schizophrenia. He had risk factors of smoking, a sedentary lifestyle and poor diet. However, he did not have abnormal plasma lipids, hypertension, diabetes, high bodyweight or a family history of cardiac disease. His antipsychotic history included chlorpromazine, zuclopenthixol, olanzapine and risperidone. During a previous trial of clozapine, his ECGs were unremarkable and there were no changes indicative of arrhythmogenic RV cardiomyopathy. Retitration of clozapine was started and his ECG on day 7 was normal. On day 13, he presented with retrosternal chest pain episodes and his ECG was consistent with Brugada type 1 pattern. He was afebrile and his blood tests including serial troponins were normal. Nothing abnormal was found on his echocardiogram or on chest X-ray. Clozapine was stopped and olanzapine was substituted. ECG was performed daily and had normalised 4 days after clozapine was stopped. The flecainide challenge test was positive. The mechanism whereby clozapine was associated with Brugada is not clear. The authors acknowledge the possibility of co-incidence because of the increased ECG monitoring when clozapine is initiated (Table 2).
Summary of Brugada case studies involving tricyclic antidepressants. .

AV, atrioventricular; BP, blood pressure; COPD, chronic obstructive pulmonary disease; CPAP, continuous positive airway pressure; CPR, cardiopulmonary resuscitation; CT, computed tomography; ECG, electrocardiogram; EEG, electroencephalogram; GCS, Glasgow Coma Scale; HR, heart rate; ICD, implantable cardioverter-defibrillator; JVP, jugular venous pressure; MRI, magnetic resonance imaging; QRS, is the name of the waves; RBBB, right bundle branch block; RR, respiratory rate; SPECT, Single Photon Emmision Computered Tomography; TCA, tricyclic antidepressant; TSH, thyroid stimulating hormone; VF, ventricular fibrillation; VT, ventricular tachycardia.
In conclusion, should we routinely screen for BrS before prescribing psychotropics?
Risk of hospitalisation due to cardiac arrhythmia is increased with the use of antispsychotics. 51 Case control studies suggest that the risk of SCD is increased with most antipsychotics.52 –54
In first episode psychosis, the risk of prolongation of QTc interval is associated with antipsychotic use in the first 2–4 weeks of therapy. 55 While there remains controversy surrounding the relationship between prolongation of QTc and chances of arrhythmia, clear guidance exists for a range of QTc intervals that are likely to be safe and some consensus that QTc intervals longer than 500 ms increase the arrhythmogenic potential and a QTc interval longer than 650 ms is associated with Torsade de Pointes. 56 Despite the uncertainties, QTc interval remains an important measure of arrhythmogenic potential, and in the United Kingdom, it is generally assumed that all antipsychotics are associated with the potential to increase risk of SCD. There is accepted guidance to monitor baseline ECG and yearly thereafter, before prescribing antipsychotics and some antidepressants, in particular to monitor for QTc prolongation. 56
Since its initial description by Pedro and Josep Brugada in 1992, there have been significant developments in the understanding of the pathophysiology of BrS and the associated risk of SCD.1,57 Despite the fact that BrS is responsible for around 20% of SCDs in patients with structurally normal hearts, and there is an established link between certain psychotropic medications and the unmasking of BrS or the precipitation of BrP, there is no equivalent framework in place for routine assessment of Brugada pattern. Given the known risks associated with BrS and psychotropic medication’s potential impact on cardiac ion channels, should we now include BrS ECG pattern screening as well?
There is certainly a gap in our understanding of the implications of asymptomatic Brugada ECG pattern and BrP on mortality; however, as noted above, BrS is the leading cause of SCD in those with apparently structurally normal hearts and there is a developing understanding of the risk factors which are associated with a higher incidence of arrythmia. Given the numerous psychotropic drugs that can unmask BrS or precipitate a Brugada pattern ECG as a result of their effect on ion channels, prescribers should be aware of this disorder and the associated risks. This is particularly the case given the increased prevalence of Brugada pattern ECG among people with schizophrenia.
We would propose that, alongside with the existing recommendations for monitoring QT interval, when prescribing antipsychotic medications and other higher risk psychotropic medications, baseline and annual ECGs should be screened for Brugada pattern as well as QT interval. Consideration should also, of course, be given to other abnormalities present on the ECG.
We would recommend a move to more structured guidance in relation to screening for Brugada pattern ECGs in commonly referenced texts and prescribing guidelines to raise awareness and provide a framework for clinicians to follow. We would further recommend that in addition to a baseline ECG being carried out to identify pre-existing abnormalities, further ECGs should be carried out once steady state of an initiated drug which may unmask BrS or precipitate BrP has been reached and when further dose increments are made.
This does of course raise the question of psychiatrists’ confidence in interpretation of ECGs and the relative lack of awareness of BrS among prescribers in mental health settings. 58 There has long been a call for more training in the interpretation of ECG among psychiatric trainees, non-trainee grades and consultants. 59 This would be of benefit in the general identification of ECG abnormalities which increase the risk of SCD and, with regard to this article, BrS specifically. 60
Abnormalities found on screening ECGs including, for example, ST elevation in leads V1–V3 with a right bundle branch block (RBBB) appearance should prompt the search for cardiology advice to ascertain whether Brugada pattern is likely. Where a medication is known to ‘unmask’ ECG Brugada pattern or Brugada pattern is suspected on an ECG, it is important that a multidisciplinary approach is taken, involving cardiology and mental health services, working together to coordinate more detailed assessment and consider further interventions in relation to both mental health disorder and the potential BrS.
Assessment of the risks and benefits of specific treatments will have to be made with the likely discontinuation of any medicine that precipitated BrP or unmasked BrS. Close collaboration between the patient, the patient’s carer(s) and health professionals involved in the patient’s care is recommended with a view to identifying the least risky pharmacological interventions and considering mitigation strategies or enhanced monitoring, an area which itself is likely to expand considerably in future with the ongoing, rapid development of wearable devices to monitor cardiac rhythm.
Footnotes
Author contribution
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
