Abstract
Background:
Anxiety disorders are common, associated with significant burden of disease, and have high levels of treatment resistance. Low-dose ketamine has been extensively studied in treatment-resistant depression, with fewer reports in treatment-resistant anxiety disorders.
Aims:
This systematic review and meta-analysis collected efficacy, safety, and tolerability data for ketamine as a treatment for anxiety spectrum disorders.
Methods:
We conducted a systematic search for randomized controlled trials (RCTs) of acute ketamine treatment for patients with anxiety disorders. Open-label trials of ketamine maintenance therapy were also considered. Qualitative and, where possible, quantitative syntheses of findings were performed using Review Manager software (RevMan). Acute dose-response and maintenance treatment data were also collected.
Results:
There were six eligible acute RCTs – two in social anxiety disorder (SAD), three in post-traumatic stress disorder (PTSD), and one in obsessive-compulsive disorder (OCD). Four of the six showed significant improvement in anxiety rating scores in ketamine compared with control groups. Pooled analysis showed ketamine was associated with an increased likelihood of treatment response for SAD (odds ratio (OR): 28.94; 95% confidence interval [CI]: 3.45–242.57;
Conclusion:
These preliminary analyses suggest that acute ketamine may be broadly effective across treatment-resistant anxiety spectrum disorders. These effects can be prolonged with maintenance treatment. Future studies will be needed to provide critical knowledge gaps around off-label use, side effects, and potential risks for abuse in clinical settings.
Introduction
Anxiety disorders – including specific phobia, social anxiety disorder (SAD), generalized anxiety disorder (GAD), panic disorder, and agoraphobia 1 – are among the most prevalent psychiatric conditions, affecting an estimated 284 million people worldwide. 2 Burden of disease can be significant, and despite psychotherapy and drug treatment options, treatment resistance or relapse is a frequent problem.3,4
Over 20 years ago, ketamine, an N-methyl-D-aspartate receptor (NMDAR) antagonist with anesthetic and analgesic properties, was reported to have rapid-onset antidepressant activity in treatment-resistant (TR) major depression (TRD) 5 and subsequently in TR bipolar depression. 6 Since then, these findings have been widely replicated,7–10 with the major focus on treating depressive disorders. Until a nasal spray formulation of esketamine for TRD was approved in the United States in 2019, 11 use of ketamine for TRD was off-label.
In contrast to TRD, relatively little clinical research has been published on the use of ketamine in treatment-resistant anxiety disorders. This is not because TR anxiety is a trivial or insignificant clinical problem; remission rates in clinical trials may be as low as 25–35%, 4 and relapse rates post-remission may be 30% after 10 years. 12 Possible reasons for the relatively small volume of published research in TR anxiety could be due to the lack of consistency in defining treatment resistance for anxiety disorders, 13 lack of interest from pharmaceutical sponsors in developing new anti-anxiety drugs, and/or the absence of regulatory guidance on how to develop drugs for TR anxiety.
Established pharmacotherapies for anxiety disorders include drugs that interact with monoamine and GABA-A systems. 4 Antidepressants have significant limitations, including slow onset of action, high rates of nonresponse, and they may worsen anxiety acutely. Benzodiazepines are not recommended for long-term use in some anxiety disorders, due to concerns about their potential for abuse, tolerance, and withdrawal, and they are ineffective in some anxiety spectrum disorders. 14 There are some compelling reasons why drugs interacting with the glutamate system could be therapeutically effective. Glutamate is involved in fear extinction. 15 Glutamate regulates neuropeptides involved in the stress response and may be linked to the development of anxiety disorders. 16 Glutamate also has important role in synaptic and neural plasticity linked to anxiety disorders. 17 Ketamine is a noncompetitive antagonist at NMDA glutamatergic receptors. However, it has a much more complex pharmacological properties, interacting with opioid, monoamine, and nicotinic and muscarinic cholinergic receptors, 18 and having a number of active metabolites with additional pharmacologies. 19
There have been a number of published clinical trials of ketamine in TR anxiety over the last eight years, and the purpose of this systematic review and meta-analysis was to assess the current evidence base for ketamine efficacy, safety, and tolerability in a number of anxiety disorders, based on data from randomized controlled trials (RCTs). It is important to highlight a recent narrative review on this topic,
14
which also includes data from case reports and case series, open-label clinical trials, and evaluation of anxiety ratings in patients with bipolar disorder or major depression. We also included data from studies in patients with obsessive-compulsive disorder (OCD) and post-traumatic stress disorder (PTSD) as anxiety spectrum disorders. While OCD and PTSD were reclassified from anxiety disorders to their own categories in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (
Methods
The protocol for this review was registered prospectively with PROSPERO (international database of prospectively registered systematic reviews): CRD42020190282. For the meta-analysis, we selected prospective acute treatment RCTs. The intervention of interest was ketamine therapy (oral, subcutaneous, intramuscular, or intravenous) compared to a control (psychoactive or inactive) given via the same route of administration. Case reports or case series were excluded. Participants in the reviewed trials needed to meet the following inclusion criteria: adults with
As this was a structured review and meta-analysis of studies that had previously received ethics committee approval, no additional ethics approval was required.
A narrative synthesis of the findings of the included studies was provided. Where appropriate, efficacy and safety results were pooled using a random-effects meta-analysis with odds ratios for binary outcomes, along with 95% confidence intervals. Statistical heterogeneity was assessed using chi-square and I-squared tests.
We also included data from any published sources on acute dose-response and responses to maintenance treatment, for any disorder, presented as percent change-from-baseline.
Analyses are presented in three sections: (1) acute efficacy, safety, and tolerability responses in controlled studies; (2) acute dose-response data; and (3) maintenance treatment.
Results
From 3,127 records screened by title and/or abstract, 37 were assessed as potentially eligible, and eight were included in the review (Figure 1). The eight studies selected include six double-blind acute treatment RCTs (five crossover and one parallel), one acute dose-response analysis of both open-label and double-blind efficacy data, and one open-label maintenance therapy trial.
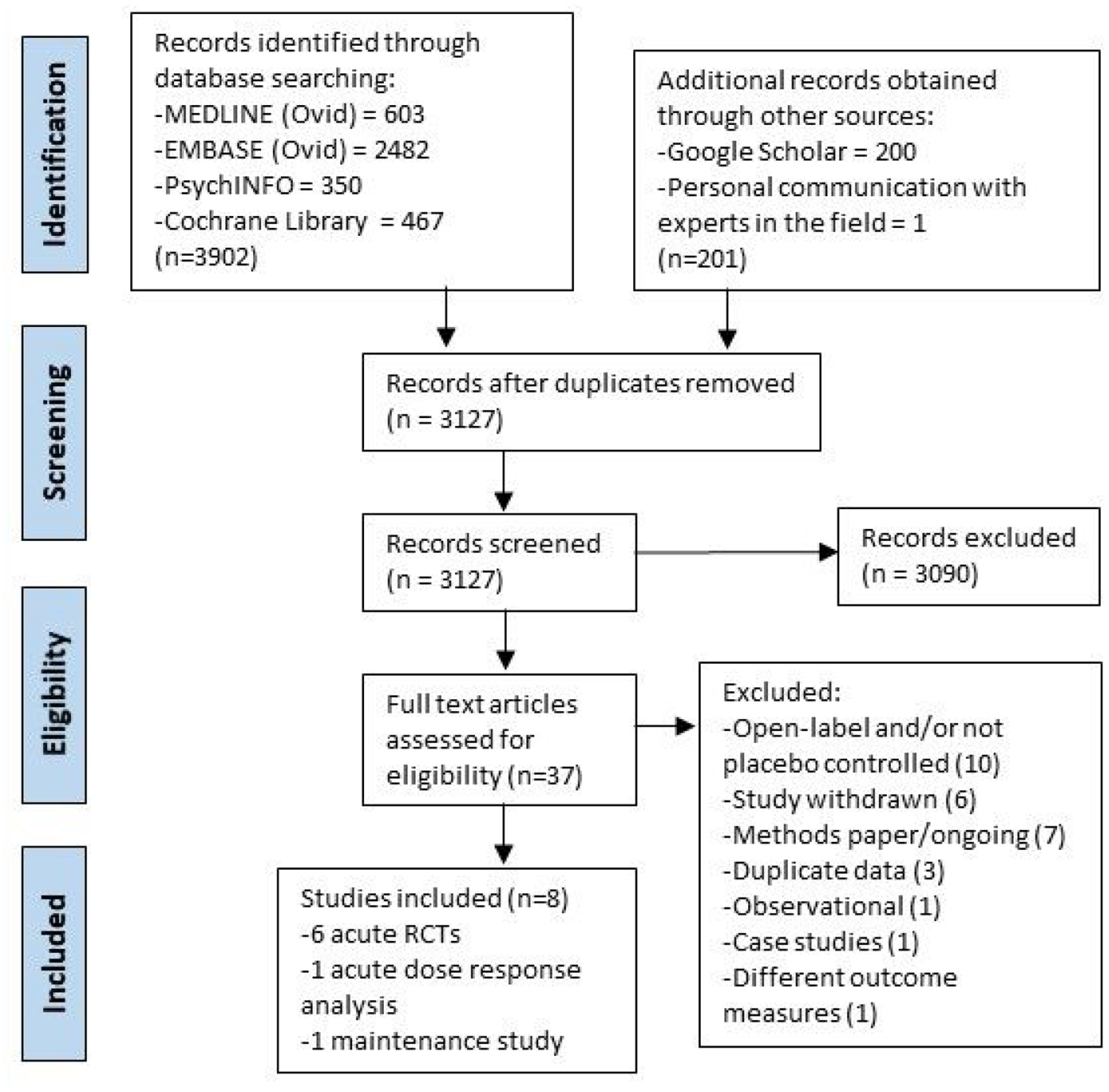
PRISMA flow diagram.
Acute treatment
Six RCTs were identified that assessed ketamine as an acute treatment for anxiety disorders. Two papers included patients with SAD and GAD;21,22 three with PTSD;23–25 and one with OCD. 26 The Cochrane risk-of-bias assessment tool for randomized trials is shown in Figure 2. Performance bias arising from functional unblinding of participants and personnel was an issue due to the dissociative effects of ketamine, particularly in contrast to non-psychoactive (e.g. saline) placebo. ‘Other bias’ included the risks of carryover and period effects, so crossover designs were categorized as high risk, and parallel studies as low risk.

Risk of bias in included acute treatment RCTs (
SAD
Glue

Responder analysis for patients with SAD and PTSD.
GAD
Both Glue
PTSD
Three studies investigated ketamine as a treatment for patients with PTSD.23–25 Feder
Dadabayev
Pradhan
Results from all three studies were pooled for responder analysis. Treatment response was defined by Feder
OCD
Rodriguez
Using published first-phase data, we calculated a statistically nonsignificant greater reduction in Y-BOCS score after ketamine treatment compared with control treatment at 7 days post-infusion: mean difference −5.79 (95% CI: −11.94 to 0.36;
Safety and tolerability
CADSS
Compared with control, Clinician-Administered Dissociative States Scale (CADSS)
35
scores tended to increase after ketamine administration, and return to baseline by 120 minutes (Figure 4). This effect was greatest at 30–40 minutes post-dose21,26 and smaller at 60 minutes.
22
Of note, patients with comorbid PTSD and CP from Dadabayev

Effects of ketamine (solid lines) and control (dashed lines) on mean Clinician-Administered Dissociative States Scale (CADSS) scores over time.
Side effects
The most common side effects reported after administration of ketamine included feeling dissociated/spaced out, nausea and vomiting, dizziness or light-headedness, blurred vision, and fatigue. Using supplementary data from Feder

Side effects (ketamine compared to controls).
Acute dose response in SAD/GAD
There were two publications which reported acute ketamine dose-responses in change in anxiety rating scores: Glue
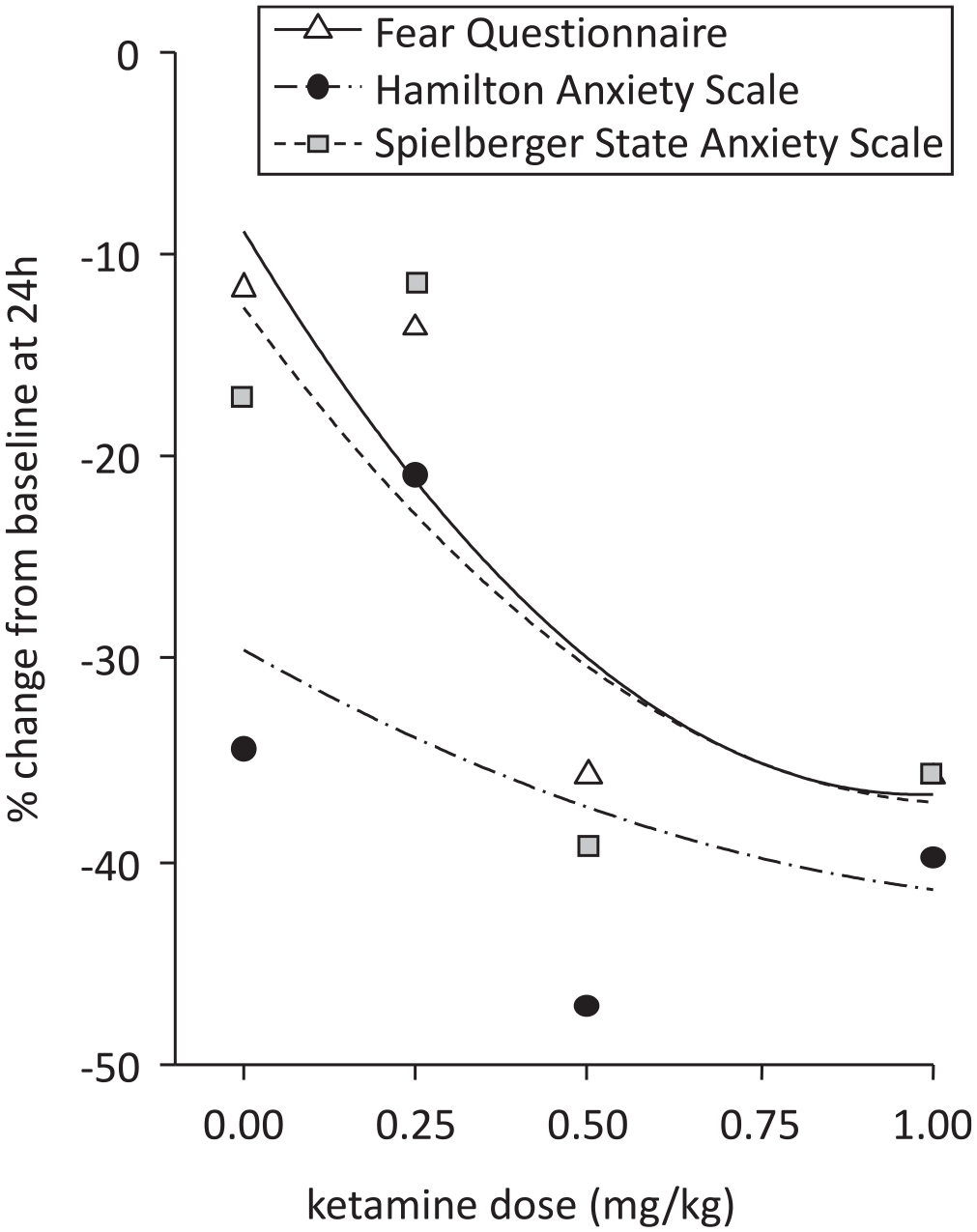
Relationship between acute ketamine dose and change in anxiety rating scores at 24 hours post-dose.
Maintenance treatment, SAD and GAD
There were two publications reporting on mood responses to ketamine maintenance therapy. Glue
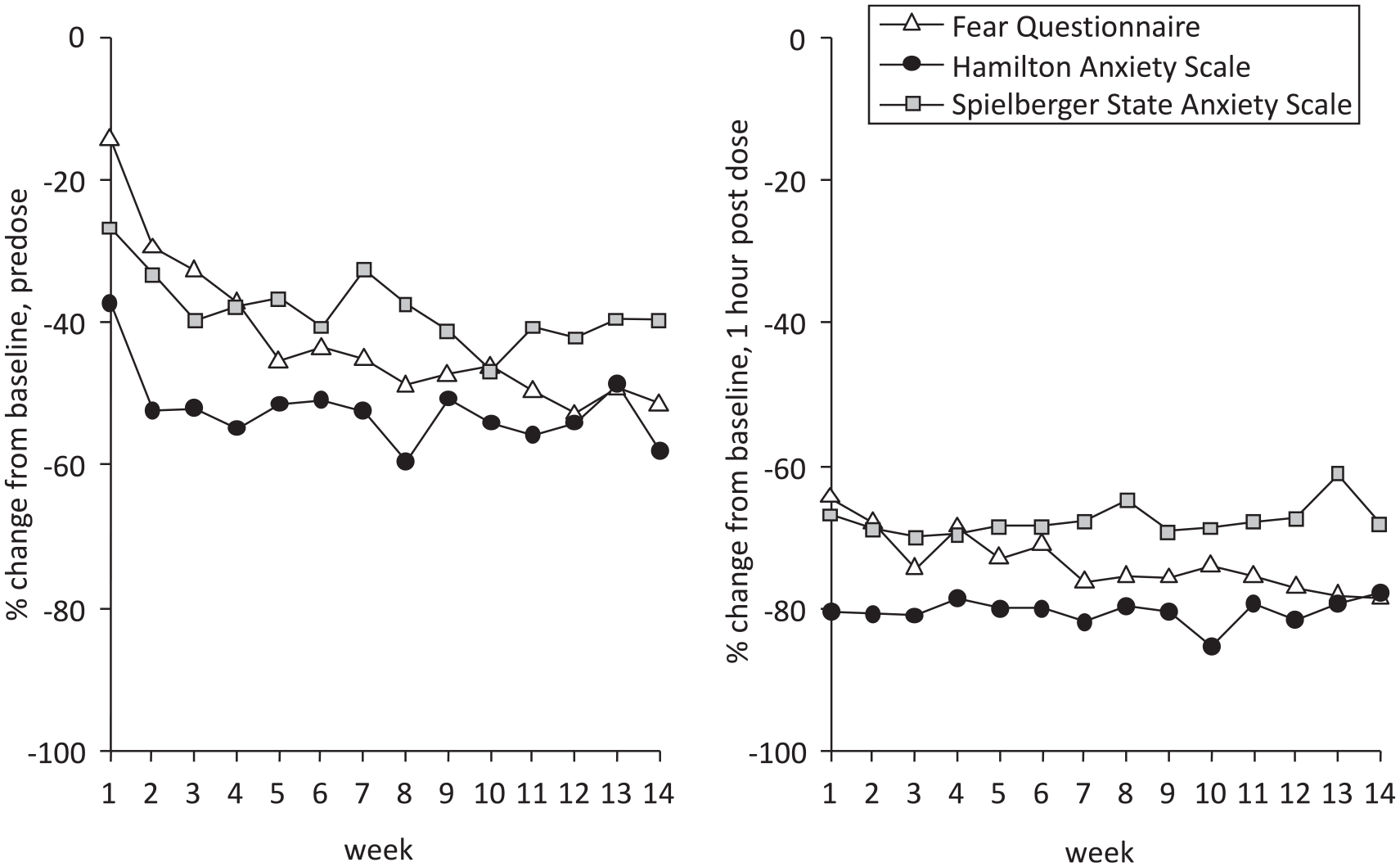
Change in anxiety rating score over time during ketamine maintenance therapy.
Discussion
This is the first systematic review and meta-analysis of controlled acute ketamine studies in anxiety spectrum disorders, and builds on the earlier thorough narrative review on this subject by Banov
Of the six included randomized acute treatment trials, four (including patients with SAD/GAD, PTSD, and OCD) reported evidence of significant benefit from ketamine compared to placebo, as measured by change in symptom rating scores at varying time points post-ketamine. Two studies found no significant difference between ketamine and controls (saline and ketorolac) in terms of reduction in PTSD scores post-infusion.23,25 Within individual studies, results also varied between different rating scales used (e.g. FQ
Based on pooled analysis, we found that compared to controls, ketamine was associated with a statistically significant increased likelihood of acute treatment response for SAD (28.94; 95% CI: 3.45–242.57;
We also report that ketamine demonstrates an acute dose-response profile, with doses of 0.5 mg/kg and above associated with greater improvement in anxiety ratings than lower doses. A similar dose-response was described for ketamine’s antidepressant effects. 40 As a maintenance treatment, ketamine was associated with sustained anxiolytic effects as well as improved social and/or work functioning. 38 A recent midazolam-controlled study of repeat ketamine dosing in patients with chronic PTSD reported reduced PTSD symptom severity after repeated ketamine infusions. 41 If ketamine is to be used clinically for treatment of other anxiety disorders, it will almost certainly require repeat dosing, and it will be important to identify optimal doses and dosing frequency for each indication.
Ketamine was reported to be generally safe and well tolerated. The dissociative and sympathomimetic effects of ketamine are well recognized. Dissociative symptoms may reduce in intensity after repeat dosing;38,42 and interestingly, patients who had comorbid CP and PTSD appeared to be more resistant to the dissociative effects of ketamine. 23 Other side effects commonly associated with ketamine dosing include dizziness/lightheadedness and nausea/vomiting. Dissociation and dizziness are of relatively brief duration and can be managed by having patients resting comfortably for the first hour after dosing. Pre-dosing patients with ondansetron may reduce the risk of nausea and vomiting.
This review has a number of limitations, including the small number of eligible studies and differences in study protocols (e.g. psychoactive vs inactive controls, different dosing regimens, and different routes of administration), which could potentially limit comparability between studies. Second, many studies had small numbers of participants (
In conclusion, this review suggests that ketamine may be a safe and broadly effective anxiolytic for patients with anxiety spectrum disorders, including treatment refractory cases. In contrast to the extensive published data for TRD, the evidence base of controlled trials of ketamine in patients with TRA is very limited. The findings from this review must be considered as tentative due to relatively small sample sizes and lack of long-term data around safety and efficacy. Of note, while ketamine may be helpful to some patients, off-label use should employ clinical decision-making that includes discussion of the limitations of the existing knowledge base. 43 Further RCTs in this area, including parallel arm studies, should be encouraged, given the significant burden of disease and high rates of treatment resistance associated with these disorders worldwide.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253211056743 – Supplemental material for Systematic review and meta-analysis of randomized controlled trials of ketamine in the treatment of refractory anxiety spectrum disorders
Supplemental material, sj-docx-1-tpp-10.1177_20451253211056743 for Systematic review and meta-analysis of randomized controlled trials of ketamine in the treatment of refractory anxiety spectrum disorders by Elizabeth Whittaker, Alisher R. Dadabayev, Sonalee A. Joshi and Paul Glue in Therapeutic Advances in Psychopharmacology
Footnotes
Acknowledgements
Drs Caroline Rodriguez and Pavithra Mukunda provided excellent feedback and advice on an earlier draft of this manuscript.
Author contributions
Drs Whittaker and Glue decided on the review topic. Dr Whittaker collected and analyzed data, and wrote the first draft of the manuscript. All authors reviewed and approved the final draft of the manuscript.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Glue has a contract with Douglas Pharmaceuticals to develop novel ketamine formulations. Within the last 3 years, Dr Glue has participated in an advisory board for Janssen Pharma. No other authors have disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
