Abstract
Schizophrenia is a devastating mental disorder resulting in marked morbidity and mortality despite the optimal use of all currently available interventions. For this reason, the release of lumateperone (CaptylaR), also known as ITI-007, an orally administered, atypical antipsychotic provided a welcome novel tool for clinicians to utilize precision medicine to tailor an optimal treatment plan to the specific needs of each person with schizophrenia. To generate a foundation for clinicians to assess the risks and benefits of lumateperone in relation to other interventions for schizophrenia, we conducted a search of items for ‘ITI-007’ and ‘lumateperone’ on PubMed, ScienceDirect, Web of Science, Google Scholar, and www.clinicaltrials.gov. We present a critical evaluation of the limited information about lumateperone for schizophrenia, its use approved by the US Food and Drug Administration. Lumateperone merits consideration for patients with treatment-resistant schizophrenia and for patients with schizophrenia who are vulnerable to developing metabolic dysfunction and movement disorders. On the other hand, lumateperone should not be used for (a) women who are pregnant or breastfeeding, children, adolescents, and elderly patients with dementia-related psychosis, (b) patients who are at risk for cerebrovascular diseases, (c) patients who use inducers and moderate or strong inhibitors of the cytochrome P450-3A4 (CYP3A4) isozyme, and (d) patients who use alcohol and other sedating agents. Clinical trials from multiple centers without financial conflicts of interest to market lumateperone are needed to directly compare and contrast lumateperone and other antipsychotic agents to generate trustworthy evidence to be assessed objectively by clinicians treating patients with schizophrenia. Future investigations will provide the foundations to identify the evidence for comprehensive evaluations of the role of lumateperone in the treatment of people with schizophrenia and other conditions.
Keywords
Introduction
Schizophrenia
Schizophrenia is a devastating mental health disorder affecting how a person interprets reality, expresses emotions, thinks, and behaves, and involving a variety of positive, negative, and cognitive symptoms with onset in adolescence or early adulthood. 1 Positive symptoms include hallucinations (auditory, visual, gustatory, olfactory, and tactile), delusions (persecutory, referential, somatic, erotomanic, grandiose, and religious), confused thoughts, disorganized speech, trouble concentrating, catatonic behavior, 2 and movement disorders.1,3 Negative symptoms include anhedonia, apathy, asociality, alogia, flattening of affect, withdrawal, affective blunting, and inattention.4,5 Cognitive symptoms include deficits in attention, working memory, concentration, and executive functions. 1
Schizophrenia is the eighth leading cause of disability worldwide in people aged 15–44 years. 6 The prevalence rate of schizophrenia is 2%.7,8 Men are 1.4 times more likely to be diagnosed with the condition than women. 9 There are 1.5 new cases per 10,000 annually. 8 Both genetic and environmental influences may precipitate the development of schizophrenia in vulnerable individuals.
The dopamine hypothesis
The dopamine hypothesis conjectures that hyperfunctionality of dopamine D2 receptor neurotransmission in limbic and subcortical brain regions attributes to positive symptoms of schizophrenia, while hypoactivity of dopamine D1 receptor neurotransmission contributes to negative and cognitive symptoms of the disease.10–15
Antipsychotics
Antipsychotics, dopamine D2 receptor blocking drugs, constitute a major therapeutic tool for the treatment of schizophrenia. However, Parkinsonism, akathisia, 16 and other movement disorders are common adverse effects of traditional (first-generation) antipsychotics. 17 Newer atypical (second-generation) antipsychotics modulate neurotransmission of 5-hydroxytryptamine (serotonin) and dopamine by acting as both serotonin 5-HT2A receptor antagonists and dopamine D2 receptor antagonists to treat both positive and negative symptoms of schizophrenia. 18 In contrast to first-generation antipsychotics, second-generation antipsychotics provide superior treatment for negative symptoms, cognition, mood, and aggression 19 and reduced risks of movement disorders. 20
The hallmark of currently available atypical antipsychotic medications, high levels of occupancy of striatal dopamine D2 receptors, produces Parkinsonism, akathisia, 16 and other extrapyramidal symptoms. 21 Another issue with current antipsychotic drugs is the high affinity for off-target receptors such as serotonin 5-HT2c, muscarinic, histamine H1, and adrenergic α-1A receptors, as well as excessive antagonism of dopamine receptors. Targeting of these receptors has caused serious side effects such as akathisia, 16 weight gain, dyslipidemia, hyperglycemia, type II diabetes, and cardiac conduction issues. 21
The clinical actions of antipsychotics result from their pharmacokinetic properties. First-generation antipsychotics are well absorbed orally and parentally. Peak plasma levels of first-generation antipsychotics are attained 15–30 min after parenteral administration and 1–4 h after oral administration. First-generation antipsychotics are protein-bound. Antipsychotics are highly lipophilic and are primarily metabolized by cytochrome P450 2D6 and P450 3A systems in the liver. They are excreted in urine and feces. 20 Both first- and second-generation antipsychotics provide some symptomatic improvement but also result in a variety of cardiometabolic, endocrine, and neurologic side effects. Their inability to treat effectively both positive and negative symptoms and resulting side effects requires more research into agents for schizophrenia with a risk-to-benefit ratio with more benefits and fewer risks.22,23
Methods
Data sources and searches
A search of items for ‘ITI-007’ and ‘lumateperone’ on PubMed, ScienceDirect, Web of Science, Google Scholar, and www.clinicaltrials.gov generated extant publications on lumateperone in schizophrenia. As several reviews of lumateperone have utilized the available publications from investigators who have financial conflicts of interest to present articles that emphasize the potential benefits,5,24–31 we sought to construct a selective review assessing the trustworthiness of the available evidence on the pharmacodynamics, pharmacokinetics, pharmacotherapy, and clinical trials of lumateperone in schizophrenia in order to provide expert insight for the possible role of this agent for people with schizophrenia. To enhance the trustworthiness of this article, we have limited ourselves to published articles and abstracts.5,24
To enhance the trustworthiness of the available evidence for lumateperone, we sought to identify the risk of bias, inconsistency, indirectness, or publication bias. 23 The current literature about lumateperone is hindered by the dearth of data from authors who are not supported by the pharmaceutical company that markets lumateperone.26,32 Thus, published studies emphasize the putative promise of lumateperone by tilting the risk-to-benefit ratio of articles in favor of the benefits. 33 The paucity of available evidence for extant clinical studies for lumateperone in schizophrenia is inadequate for a definitive systematic review 34 of the topic. However, because lumateperone offers great promise to alleviate the suffering of people with schizophrenia, we seek to share our objective assessment to aid clinicians, administrators, policy planners, and advocates to apply precision medicine to the development of optimal treatment plans for people with schizophrenia.
Introduction to the compound
Lumateperone is an orally administered, atypical antipsychotic developed by Intra-Cellular Therapies (New York, NY, USA) for the treatment of schizophrenia and other neurologic and neuropsychiatric disorders. 35 Lumateperone is the first in class selective and simultaneous modulator of dopamine, serotonin, and glutamate,5,18,22,30,32,36–41 approved by the US Food and Drug Administration for the treatment of schizophrenia. 42
Chemistry
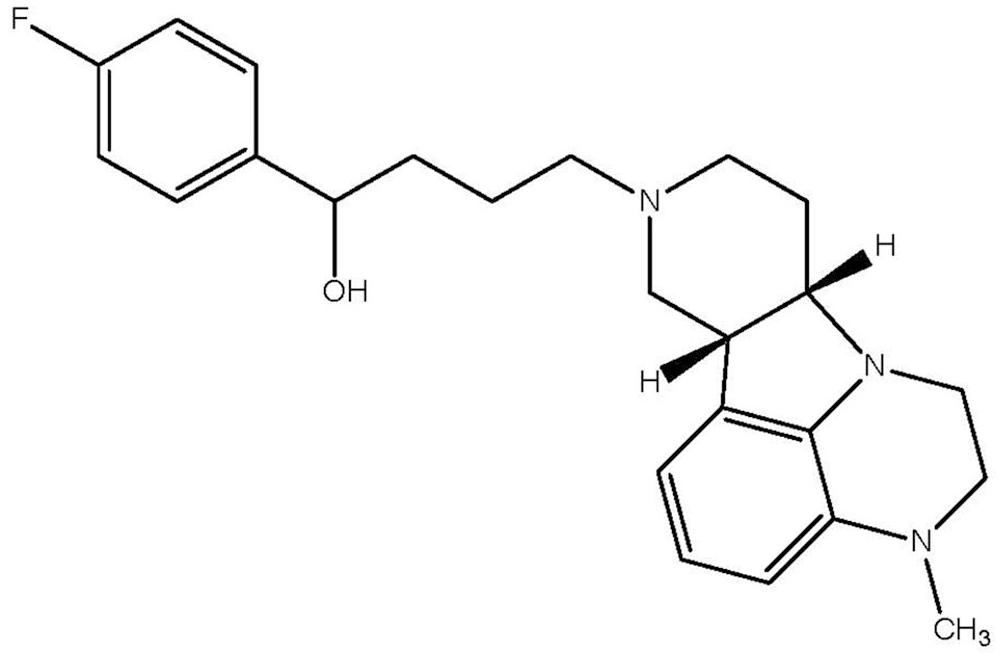
Lumateperone (4-((6bR,10aS)-3-methyl-2,3,6b,9,10,10a-hexahydro-1H,7H-pyrido[3′,4′:4,5]pyrrolol[1,2,3-de]-quinoxalin-8-yl)-1-(4-fluoro-phenyl)-butan-1-one 4-methylbenzenesulfonate), 43 also known as ITI-007 and ITI-722, exists as the tosylate salt5,20,36,37,44 (Figure 1). The principal metabolite of lumateperone, ICI200131, is the alcohol produced by the reduction of the carbonyl side-chain by ketone reductase.21,38,43,44
Mechanism of action
Lumateperone is a serotonin transport inhibitor and a potent antagonist at serotonin 5-HT2A receptors5,24,45–49 acting as a presynaptic partial agonist and a postsynaptic antagonist at dopamine D2 receptors,5,50,51 and as a dopamine receptor phosphoprotein modulator.5,22,28,45–49 It is also a dopamine D1 receptor-dependent indirect modulator of glutamatergic α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid 52 and N-methyl-D-aspartate (NMDA) GluN2B receptors.5,19,24,53,54
Routes of administration
The once-daily recommended dose is 42 mg administered orally with food.24,55 In addition, a 26-year-old man with treatment-resistant paranoid schizophrenia required escalation of the dose of lumateperone to 84 mg/day to experience amelioration of auditory and visual hallucinations without adverse effects after 6 months of sustained therapy. 56 Thus, lumateperone could be a beneficial intervention for treatment-resistant schizophrenia. 55
Preclinical development of a long-acting injectable formulation of lumateperone for schizophrenia is ongoing in the USA. 24
Lumateperone comes with a black box warning that states that elderly patients with dementia-related psychosis are at an increased risk of death. The drug is not approved for the treatment of dementia-related psychosis.24,43
Other indications for lumateperone
Clinical development of lumateperone for possible use in major depressive disorders, behavioral disorders associated with Alzheimer’s disease and dementia, bipolar disorder, and sleep maintenance is underway in many countries around the world.5,24 The use of lumateperone for conditions other than schizophrenia is beyond the scope of this review.
Pharmacodynamics
Lumateperone displays a high binding affinity for serotonin 5-HT2A receptors and moderate binding affinity for dopamine D1 receptors, dopamine D2 receptors, and serotonin transporters. It also demonstrates a moderate binding affinity for dopamine D4, and α-1A and α-1B adrenergic receptors along with a low binding affinity for muscarinic and histaminergic receptors.5,24
Dopamine D2 receptor
Lumateperone has a high affinity for dopamine D2 receptors (Ki = 32 nM) selectively in mesolimbic and mesocortical regions. 22 The drug acts as a postsynaptic dopamine D2 receptor antagonist and a presynaptic partial agonist at dopamine D2 receptors. The dosage of lumateperone correlates well with D2 receptor occupancy. 21 Lumateperone rapidly penetrated the brain in healthy volunteers (n = 16) with long-lasting, dose-related, and plasma concentration-related striatal dopamine D2 receptor occupancy.21,24 A dose of 10 mg of lumateperone displayed low occupancy (≈12%) of striatal dopamine D2 receptors, while 40 mg of lumateperone displayed peak occupancy up to 39% of striatal dopamine D2 receptors. 21 In a phase II positron emission tomography (PET) study (NCT02288845), patients with schizophrenia who received 60 mg of lumateperone once daily showed peak dorsal striatal dopamine D2 receptor occupancy (mean 39%) at 1 h post-dose.21,24
Dopamine D1 receptor and glutamate GluN2B receptor
Lumateperone acts upon dopamine D1 receptors with dose-dependent high affinity (Ki = 52 nM) phosphorylating the GluN2B (NR2B) subunit of the NMDA receptor.5,36,52,57
Dopamine transporter
Inhibition of the dopamine transporter represents a special characteristic of lumateperone that may lead to beneficial effects in schizophrenia. 58
Serotonin 5-HT2A receptor
Lumateperone acts as a strong serotonin 5-HT2A receptor antagonist (Ki = 0.54 nM). 5 In a study of 16 healthy men who had a mean age of 30.7 years with an average basal metabolic index of 24.6 with a standard deviation of (±3.0), 5-HT2A receptors were fully saturated (>80%) by oral doses of lumateperone as low as 10 mg. 21 A dose of 40 mg of lumateperone demonstrated occupancy up to 33% of striatal serotonin transporters.21,31
Serotonin transporter
At medium doses, lumateperone acts as a potent serotonin transporter inhibitor (Ki = 62 nM). 5-HT2A receptor antagonism exerts a synergistic effect with serotonin transporter inhibition for greater antidepressant efficacy.5,21
Off-target actions
Lumateperone displays minimal affinity for off-target receptors, ion channels, neurotransmitter transporters, and enzymes. Those are common targets of other antipsychotic drugs that may cause adverse effects like weight gain, sedation, and orthostatic hypotension. 37 It displays a moderate binding affinity for dopamine D4 and α-1A and α-1B adrenergic receptors (Ki protected at <100 nM), with a low binding affinity (<50% inhibition at 100 nM) for muscarinic and histaminergic receptors. 24
Pharmacokinetics
Key aspects of the pharmacokinetics of lumateperone include the absorption of lumateperone by the body, the distribution of lumateperone throughout the body, the conjugation of lumateperone in the liver, and the excretion of lumateperone from the body. 59
Time course of absorption
Tmax occurs at 3–4 h after the oral administration of lumateperone. At 8 h after oral administration, plasma concentrations of 0.05–50 ng/ml of lumateperone and 0.2–100 ng/ml of metabolites were detected.5,21
Bioavailability
In ≈1–2 h, the peak plasma concentration of lumateperone is achieved. 43 Steady-state concentrations are achieved in ≈5 days. Steady-state exposure of lumateperone increases in an approximately dose-proportional manner, with multiple doses in the range 21–56 mg. The pharmacokinetics of lumateperone is variable among individuals, with a coefficient of variation for Cmax and area under the curve (AUC) ranging at steady state from 68% to 97%. This drug has an absolute bioavailability of ≈4.4%. Cmax increases by 33%, AUC increases by 9%, and the median time to Cmax is delayed by ≈1 h (from 1 h in fasting state to 2 h in presence of food) when lumateperone is given with a high-fat meal.24,43
Distribution
Following intravenous administration, the volume of distribution for lumateperone is ≈4.1 L/kg. 24 It is bound to 97.4% plasma proteins at 5 µM (≈70 times higher than therapeutic concentrations).
Metabolism
Lumateperone produces more than 20 metabolites by undergoing metabolism by different enzymes. These include uridine diphosphate (UDP) glycosyltransferase (UGT)1A1, UGT1A4, UGT2B15, aldo-keto reductase (AKR)1C1, AKR1B10, AKR1C4, CYP3A4, CYP2C8, and CYP1A2.24,43
Excretion
Lumateperone and glucuronidated metabolites represent ≈2.8% and 51% of the total plasma radioactivity following a single dose of radiolabeled lumateperone. Around 58% of the radioactivity is excreted in the urine, <1% of the drug is excreted in the urine in unchanged form, and 29% is excreted in the feces. After intravenous administration, the terminal half-life is ≈18 h and the clearance is ≈17.9 L/h.24,43
Toxicokinetics
Age, sex, or race does not affect pharmacokinetics to a significant clinical extent. 43 Lumateperone should not be given with moderate or strong CYP3A4 inhibitors and UGT inhibitors. Co-administration will increase the risk of toxicity. 43 Since inducers and inhibitors of CYP34A increase the concentration of lumateperone, co-administration of these compounds is contraindicated. 60
The high plasma protein binding (97.4%) in humans suggests that this compound may be retained in the body for an extended duration. 38
Dosing routes
Currently, the dosing route of lumateperone is orally in a capsule containing lumateperone developed as a crystalline, tosylate salt. (NCT01499563). 38
Adverse effects
Common side effects of lumateperone are headache, somnolence, dizziness, 29 sedation, fatigue, and constipation (4–6.7%). 60
Contraindications
Due to interactions with the cytochrome P450-3A4 (CYP3A4) isozyme, clinicians should cautiously administer lumateperone to patients who take inducers and inhibitors of CYP3A4. In addition, to avoid sedation, clinicians should discourage the use of lumateperone with alcohol and other agents for sedation.5,25,61 Clinicians should avoid prescribing lumateperone for those at risk of cerebrovascular diseases. 62
Clinical trials
Multiple clinical trials conducted by the pharmaceutical company marketing lumateperone have reported beneficial effects without safety issues.39,45-49 Key clinical trials for lumateperone for schizophrenia are summarized in Table 1.
Clinical trials of lumateperone for schizophrenia.

Phase II trials
Stable schizophrenia
A phase II, open-label PET study (NCT02288845) was conducted to determine the relationship between lumateperone dose, plasma levels, and brain receptor occupancy in patients with stable schizophrenia. A total of 14 participants with stable schizophrenia were enrolled in the study; 10 patients aged 26–57 years received lumateperone 60 mg and completed the study. Nine (90%) patients were Black/African and one (10%) was Asian. Among these patients only one was female (10%). Patients were stable at baseline with a mean Positive and Negative Syndrome Scale (PANSS) 63 total score of 72.7 with a standard deviation of (+/−7.39) and a mean Clinical Global Impressions Severity (CGI-S) 64 score of 3.8 with a standard deviation of (+/−0.63). Before the washout period, all of the patients took at least one antipsychotic. Quetiapine (eight patients, 80%) and risperidone (three patients, 30%) were the most popular ones, while haloperidol, olanzapine, lurasidone, and paliperidone were among the others taken. During the study and washout period, none of the other antipsychotics were taken. Lorazepam (nine patients, 90%) was the only psychotic medication that was taken simultaneously. A dose of 60 mg lumateperone was administered orally open-label in the morning once daily for 2 weeks in patients with schizophrenia (n = 10) and at least after a washout period of 2 weeks. [ 11 C]raclopride was used as a radiotracer with PET to determine dopamine D2 receptor occupancy. Patients who received 60 mg of lumateperone once daily showed peak dorsal striatal dopamine D2 receptor occupancy (mean 39%) at 1 h post-dose. This study showed that lumateperone has a favorable safety profile and is well tolerated.5,46
Acute exacerbation of psychosis in schizophrenia
Another phase II (NCT01499563) randomized clinical trial was conducted in a double-blind, placebo-controlled, multi-center manner in patients with schizophrenia having an acute exacerbation of psychosis. The study was conducted to assess the efficacy and safety of two dose levels of lumateperone compared with placebo. Risperidone was included as a positive control. Patients aged 18–55 years were randomized to receive lumateperone 42 mg (n = 84), lumateperone 84 mg (n = 84), risperidone 4 mg (n = 82), or placebo (n = 85) once daily for 4 weeks. The least squares (LS) mean change from baseline in the PANSS 63 total score (primary endpoint) was −13.22, −8.3, −13.4, and −7.4 in the above groups, respectively, on day 28. After 28 days of inpatient treatment, patients were started on standard antipsychotic medication and were stabilized over 5 days before being discharged from the study clinic. Patients were assessed for final outpatient safety evaluation at the end of the study visit 2 weeks after discharge. While the difference between lumateperone 84 mg once daily and placebo was not significant, there was statistical significance between lumateperone 42 mg once daily and placebo (p = 0.017) and between risperidone and placebo (p = 0.013). Both risperidone and lumateperone 42 mg once daily significantly (p =< 0.05) improved positive symptoms and general psychopathology on the PANSS 63 subscales compared with placebo. Lumateperone 42 mg once daily improved depressive and psychotic symptoms in a subgroup of severely ill patients with comorbid depression.5,18
Phase III trials
Acute exacerbation of psychosis in schizophrenia
A phase III (NCT02282761) randomized, double-blind, parallel-group, placebo-controlled, multi-center study was conducted in patients diagnosed with schizophrenia having an acute exacerbation of psychosis. The goal of the study was to determine the efficacy and safety of lumateperone for the short-term treatment of schizophrenia. A total of 450 patients aged 18–60 years were enrolled in the study from 12 clinical sites in the USA. A total of 150 patients were randomized in each group to receive lumateperone tosylate 60 mg or 40 mg (corresponding to 42 mg or 28 mg of the active moiety) or placebo once daily for 4 weeks. The change from baseline to day 28 in the PANSS 63 score versus placebo was the main endpoint. The LS mean change from baseline in the PANSS 63 total score at day 28 was −14.5 in the lumateperone 42 mg once daily group compared with −10.3 in the placebo group (LS mean difference −4.2; p = 0.02) in the prespecified modified intention-to-treat analysis (n = 435). The treatment effect of lumateperone 28 mg once daily versus placebo was not statistically noteworthy (LS mean difference −2.6). Significant improvements (p < 0.05) with lumateperone 42 mg versus placebo were also presented on the PANSS 63 positive symptom and general psychopathology subscales, and in psychosocial function, as measured by the PANSS-derived prosocial factor score and personal and social performance (PSP) 65 scale at day 28. 61 Both doses of lumateperone were well tolerated without changes in cardiometabolic or endocrine factors or significant treatment-emergent motor adverse effects versus placebo. The drug demonstrated a favorable safety profile and efficacy for improving symptoms of schizophrenia.5,61
Acute exacerbation of psychosis in schizophrenia
A phase III (NCT02469155) clinical trial was conducted to evaluate the antipsychotic efficacy of lumateperone in a randomized, double-blind, parallel-group, placebo- and active-controlled, multi-center study in patients with schizophrenia having an acute exacerbation of psychosis. A total of 696 patients were randomized to receive lumateperone 42 mg, lumateperone 14 mg, risperidone 4 mg, or placebo once daily for 6 weeks. 36 Concerning the change in baseline score on PANSS, 63 there was no significant difference among doses. A high placebo response was noticed.5,45
Stable schizophrenia
A phase III (NCT03817528) 12-month open-label safety study of lumateperone was conducted in patients with stable symptoms of schizophrenia. In part one of this study, 303 patients with schizophrenia were switched from standard of care (SOC) antipsychotic therapy to 6 weeks of 42 mg lumateperone once daily. The patients were then switched back to SOC for 2 weeks. Part one of the study showed that lumateperone was associated with improvements from baseline in the PANSS total score, 63 Marder et al. 66 negative factor scale scores, positive symptom and general psychopathology subscale scores, and on the PSP scale.65,67 Greater improvements were visible in patients with comorbid symptoms of depression and prominent negative symptoms at baseline. 67
In the second part of this study, 602 patients with schizophrenia were moved from SOC to 42 mg lumateperone once daily for up to 1 year.48,49,68 The mean change in the Calgary Depression Scale for Schizophrenia score 69 from the baseline was −0.6 (p = 0.01) and the mean change from SOC baseline in the PANSS total score 63 was −4.0 (p < 0.001) at day 368. In patients with moderate to severe depression, improvements were more noticeable at baseline.68,70–72
Phase II and phase III pooled trials
By combining results from multiple studies, the sponsors of lumateperone argued that overall lumateperone was effective for patients with schizophrenia. Since the authors have a financial interest in the marketing of lumateperone, there may be biases in the publications. 23 A pooled analysis (n = 520) showed that 42 mg of lumateperone once daily reduced the PANSS total score 63 (LS mean difference versus placebo −4.76; p < 0.001) and established comparable efficacy to risperidone (LS mean difference versus placebo −4.97; p = 0.014). 70 Lumateperone was associated with significant improvements on PANSS 63 subscales and the CGI-S 64 scale and significantly higher PANSS 63 response rates. On combining all three studies together, the ability of lumateperone 42 mg once daily to demonstrate statistical dominance over placebo was not impacted by the negative results of one study. 73
Relevance to clinical practice
Key questions need answers. Does lumateperone have greater or equivalent efficacy when compared with other antipsychotics? Is lumateperone more metabolically friendly? Further research is needed to determine its comparative ranking. Due to the dearth of trustworthy evidence 23 to assess the role of lumateperone in the treatment of people with schizophrenia, we utilize the available indirect assessment to provide guidance to providers for patients with schizophrenia.
Antipsychotics such as brexpiprazole, cariprazine, and lurasidone, which are known better for their tolerability than efficacy, have comparable effect sizes as lumateperone. The higher efficacy antipsychotics (i.e. olanzapine and clozapine) come with increased metabolic side-effect profiles. 74 Lumateperone’s strongest selling points are its novel mechanism of action and its tolerability.74–77 Lumateperone is well tolerated with a low risk of extrapyramidal and cardiometabolic side effects and improves or maintains schizophrenia symptoms in outpatients with stable schizophrenia. 71 For patients who are not tolerant of current antipsychotic medications either because of extrapyramidal side effects or metabolic adverse effects, lumateperone may be proven as a good choice of medication as it is not significantly associated with metabolic and extrapyramidal symptoms. 29
However, the high cost of lumateperone should also be considered. Some healthcare systems with financial limitations might not be able to consider lumateperone as the first choice because of the financial costs. 76 In a multi-country comparison study, lifetime costs associated with anti-psychotics (cost related to adverse effects, relapse, drug cost, schizophrenia management, and treatment discontinuation) were compared. Costs of 10 antipsychotics including aripiprazole, brexpiprazole, cariprazine, lumateperone, lurasidone, olanzapine, paliperidone, quetiapine, risperidone, and ziprasidone were assessed. Costs from seven countries (France, Hungary, Italy, Slovenia, Spain, Sweden, and the UK) were obtained. The results showed that the lifetime costs are greatly driven by diabetes and the cardiovascular side effects of antipsychotics. These effects were lowest for lurasidone, second lowest for ziprasidone, and third lowest for lumateperone. 76 To make an informed treatment choice, costs must be considered before prescribing antipsychotics to patients.
Expert insight
Although the literature contains recommendations that may reflect the marketing strategies of the authors and lacks multi-center comparisons between lumateperone and other atypical antipsychotics, 51 we seek to provide clinicians guidelines to apply the principles of precision medicine to structure treatment plans tailored to the needs of individuals with schizophrenia. A frank discussion with patients of the risks and benefits of available antipsychotic agents can then result in obtaining informed consent based on the extant evidence. Lumateperone merits consideration for patients who are vulnerable to developing metabolic adverse effects 72 and movement disorders.27,70,71,78
There are several issues that lead clinicians to avoid prescribing lumateperone to patients with schizophrenia. Due to the increased risk of death, lumateperone must be avoided in the treatment of elderly patients with dementia-related psychosis. 43 To avoid unwanted drug interactions, lumateperone should not be used for patients who are taking inducers of the CYP3A4 isozyme or moderate or strong inhibitors of the CYP3A4 isozyme. 43 Due to the sedative property of lumateperone, it should be avoided in the treatment of patients who take alcohol and other soporific agents.5,25 To avoid the increased risk of stroke and transient ischemic attacks, clinicians should avoid administering lumateperone to those at risk of cerebrovascular diseases. 62 Due to the unknown effects on developing organs and children, lumateperone should be avoided in women who are or may become pregnant or breastfeeding, and in children and adolescents.
Discussion
The role of lumateperone in the treatment of people with schizophrenia can be estimated utilizing limited available evidence 23 and indirect comparisons with other available agents. To personalize prescription, efficacy and side-effect profiles of antipsychotics should be considered. 79 A good overall safety profile is demonstrated by the newer antipsychotics.60,79 Lumateperone has the advantageous properties of relatively low risks of common antipsychotic adverse effects, including weight gain, Parkinsonism, and akathisia.78,80
New antipsychotics should be compared with older ones because the newer antipsychotics are not always the most efficacious. Older drugs may have a higher efficacy but newer ones are important when it comes to the side-effect profile.79–82 The available literature is limited by the use of pooled studies and statistical arguments to explain the negative results of some clinical trials.81–82 In addition, the current evaluation of some clinical trials is limited to examination of abstracts and posters because the findings have not been published as original research articles.
Limitations
The trustworthiness of the available evidence for lumateperone is hindered by the risk of bias, inconsistency, indirectness, or publication bias. 23 The conduct of studies by the pharmaceutical company with a financial interest to market lumateperone confirms the risk of bias, inconsistency, indirectness, or publication bias 23 in the extant literature. The current literature about lumateperone is hindered by the dearth of data from authors who are not supported by the pharmaceutical company that markets lumateperone. Low to very low confidence in published articles limits the ability to assess the value of lumateperone and other antipsychotics in comprehensive reviews.34,83,84
Future directions
Future clinical trials to detect relapse will benefit from the careful design, checking cognitive 85 and other adverse effects,78,86,87 cautious discontinuation, and measurement of environmental influences to minimize the confounding effects of withdrawal. 88
When data from multiple centers without financial interest in lumateperone for schizophrenia are available, well-conducted systematic reviews34,83,84 can be conducted. Future reviews will be enhanced by the inclusion of postmarketing surveillance for adverse effects. 5
Future investigations may be enhanced by the identification of subtypes of schizophrenia incorporating genetic and environmental influences,19,41 as well as specific indications and optimal doses, 32 utilizing valid, reliable, and responsive single-item global impression 89 and other rating scales in order to apply the principles of precision medicine to tailor an optimal treatment plan to the specific needs of individual patients. Head-to-head comparisons of newer and older agents for efficacy and adverse effects will enhance objective assessments of pharmaceuticals for schizophrenia. 32
Future investigations will be enhanced by the use of biomarkers to provide quantitative measurements of receptor expression before, during, and after treatment with lumateperone. PET provides an invaluable tool to determine precisely the occupancy of each receptor affected by lumateperone.21,46 The utilization of PET along with behavioral, cognitive, physiological, 90 and environmental measurements14,89,91 provide a foundation for optimal clinical trials of schizophrenia,10,15 and other neuropsychiatric disorders92–100 and medical disorders.101–103 PET will provide a tool to identify the effects of lumateperone on neurotransmitter systems already studied21,46 and other possible pathways.92,100 Comprehensive multi-modal investigations by teams of experts around the world will facilitate the identification of biomarkers for clinical phenotypes of schizophrenia 104 to provide clinicians with the foundations to apply precision medicine to individuals with schizophrenia.
Conclusion
Lumateperone, a first in class selective and simultaneous modulator of dopamine, serotonin, and glutamate, 22 received its first global approval in the USA on 20 December 2019 for the treatment of schizophrenia in adults. 30 Lumateperone has demonstrated efficacy against both positive and negative symptoms of schizophrenia and has a good tolerability and safety profile. 80 Lumateperone has proven to be a promising drug in clinical trials for bipolar depression, dementia, and sleep maintenance insomnia. 31 It is currently being used in clinical trials around the world. While lumateperone has been approved for the treatment of schizophrenia in adults, more research is needed for its use in bipolar depression, dementia, 105 and sleep maintenance insomnia. Clinicians must balance the optimistic outcomes of the trials funded by the sponsor of lumateperone with a frank assessment of limited data to utilize precision medicine to generate the best treatment plan for individuals with schizophrenia. 106 A comprehensive evaluation of its efficacy in long-term human studies will be required to demonstrate its efficacy in the above mentioned conditions.
Footnotes
Acknowledgements
The authors thank the Welch Medical Library, Johns Hopkins University, Baltimore, MD, USA for assistance in obtaining the references.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
