Abstract
Many older adults experience a deterioration in cognitive function with aging, and this can have a negative impact on quality of life. Late-life depression has been linked to mild cognitive impairment and dementia, and treating depression with an agent that has procognitive effects could be beneficial. Vortioxetine is a novel antidepressant with a multimodal mechanism of action that works primarily via serotonin transporter inhibition, 5-HT1A receptor agonism and 5-HT3 receptor antagonism. A recent systematic review demonstrated procognitive effects of vortioxetine when indirectly compared with selective serotonin reuptake inhibitors and serotonin–norepinephrine reuptake inhibitors in adults aged 18–65 years with major depressive disorder. While this systematic review demonstrated promising procognitive effects from vortioxetine, the included studies did not enroll older adults, who are at the highest risk of cognitive impairment. Therefore, our systematic review sought to investigate the effects of vortioxetine on cognitive functioning in patients over the age of 65 years. Three studies met the prespecified search criteria and were evaluated. Overall, these preliminary data suggest that vortioxetine has promising effects in improving cognition in older adults with depressive symptoms and may have a place in therapy in older adults with depression and/or cognitive impairment, including Alzheimer’s disease. Additional long-term studies that include more diverse populations with comorbidities and direct comparisons with other antidepressants are needed to fully understand the potential cognitive benefits in older adults.
Introduction
The prevalence of major depressive disorder (MDD) in older adults is 1–5%, with rates as high as 42% in patients with varying comorbidities. 1 Depression diagnosed after age 65 years is referred to as late-life depression (LLD) and has been linked to dementia and mild cognitive impairment. 2 Some studies even suggest that LLD is a prodrome and risk factor for the development of dementia and increases the risk of progression from mild cognitive impairment to dementia. 2 Patients with LLD respond to antidepressants at a lower rate than younger adults and experience greater recurrence of depressive symptoms. 3 Age-related cognitive decline has been reported to occur in more than 40% of people over age 60 years. 4 This decline can include impairment in memory, language and judgment, and in some cases can progress to dementia. 5 Cognitive impairment can have a significant impact on quality of life for both patients and their families.
Preliminary evidence has shown certain antidepressants to potentially affect cognitive decline. Long-term use of citalopram reduced amyloid-β generation and reduced the number of plaques formed in mouse and human subjects. 6 Additionally, after analysis of patients with previous depression enrolled in the Alzheimer’s disease (AD) Neuroimaging Initiative, researchers found that use of a selective serotonin reuptake inhibitor (SSRI) for 4 years was associated with reduced rates of conversion from mild cognitive impairment to AD compared with patients receiving other antidepressants. 7 Additionally, both citalopram and duloxetine have individually demonstrated clinical improvements in cognition in older adults.8,9 However, it is unclear whether these effects were seen purely as a result of improved depressive symptoms, or whether these medications may have separate cognitive benefits. Antidepressants with additional procognitive effects could be beneficial to treat LLD and improve global cognition and executive functioning.
Vortioxetine is a novel antidepressant with potential procognitive effects. A recent systematic review demonstrated procognitive effects of vortioxetine when indirectly compared with SSRIs and serotonin–norepinephrine reuptake inhibitors (SNRIs) across studies in adults aged 18–65 years with MDD. 10 Participants treated with vortioxetine showed improvements in global cognition, executive functioning, speed of processing, and attention, which are all symptoms associated with age-related cognitive decline. According to the authors, these procognitive effects were thought to be related to the agent’s modulation of neurotransmitters, and less likely related purely to improvements in depression symptoms. 10 While this systematic review demonstrated promising procognitive effects from vortioxetine, the included studies did not enroll older adults, who are at the highest risk of cognitive decline. Therefore, our systematic review sought to investigate the effects of vortioxetine on cognitive functioning in patients over the age of 65 years.
Background of vortioxetine
Vortioxetine is an antidepressant with a multimodal mechanism of action. Similar to SSRIs and SNRIs, vortioxetine binds to the serotonin transporter protein to inhibit serotonin reuptake in the central nervous system. However, vortioxetine is also believed to directly provide 5-HT1A receptor agonism, 5-HT1B receptor partial agonism and 5-HT3, 5-HT1D and 5-HT7 receptor antagonism.11,12 These effects on serotonergic receptors may augment the antidepressant effects resulting from serotonin transporter protein inhibition. Additionally, some of these serotonergic receptor interactions are believed to facilitate the release of other neurotransmitters. These indirect interactions may regulate the release of norepinephrine, dopamine, acetylcholine (ACh), histamine, gamma-aminobutyric acid (GABA) and glutamate. 11
Vortioxetine modulation of neurotransmitters
While vortioxetine’s precise mechanism is not completely understood, rat models suggest that it has indirect effects on several neurotransmitters. Through agonism of 5-HT1A receptors and antagonism of 5-HT3 receptors, vortioxetine likely increases norepinephrine levels.11,12 Increases in norepinephrine may help improve mood and increase brain activity. 5-HT1A agonism is also hypothesized to increase dopamine, which may lead to an improvement in symptoms such as anhedonia. Activity on 5-HT1A and 5-HT3 receptors is thought to lead to increases in ACh. Vortioxetine also increases endogenous 5-HT tone, leading to an increase in histamine. Increases in ACh and histamine are hypothesized to contribute to the procognitive effects of vortioxetine based on the importance of these neurotransmitters in neuronal functioning.
Vortioxetine is also associated with increases in glutamate and neuroplasticity in rat models. 11 This action is thought to be caused by 5-HT3 antagonism that modulates GABAergic inhibition. Neuroplasticity has been demonstrated through an increase in cell proliferation and maturation. The same model also shows an increase in dendritic branching that is more prominent with vortioxetine compared with SSRIs. These same rat models suggest that 5-HT1A receptor agonism and 5-HT3 receptor antagonism enhance memory.
Methods
Search strategy
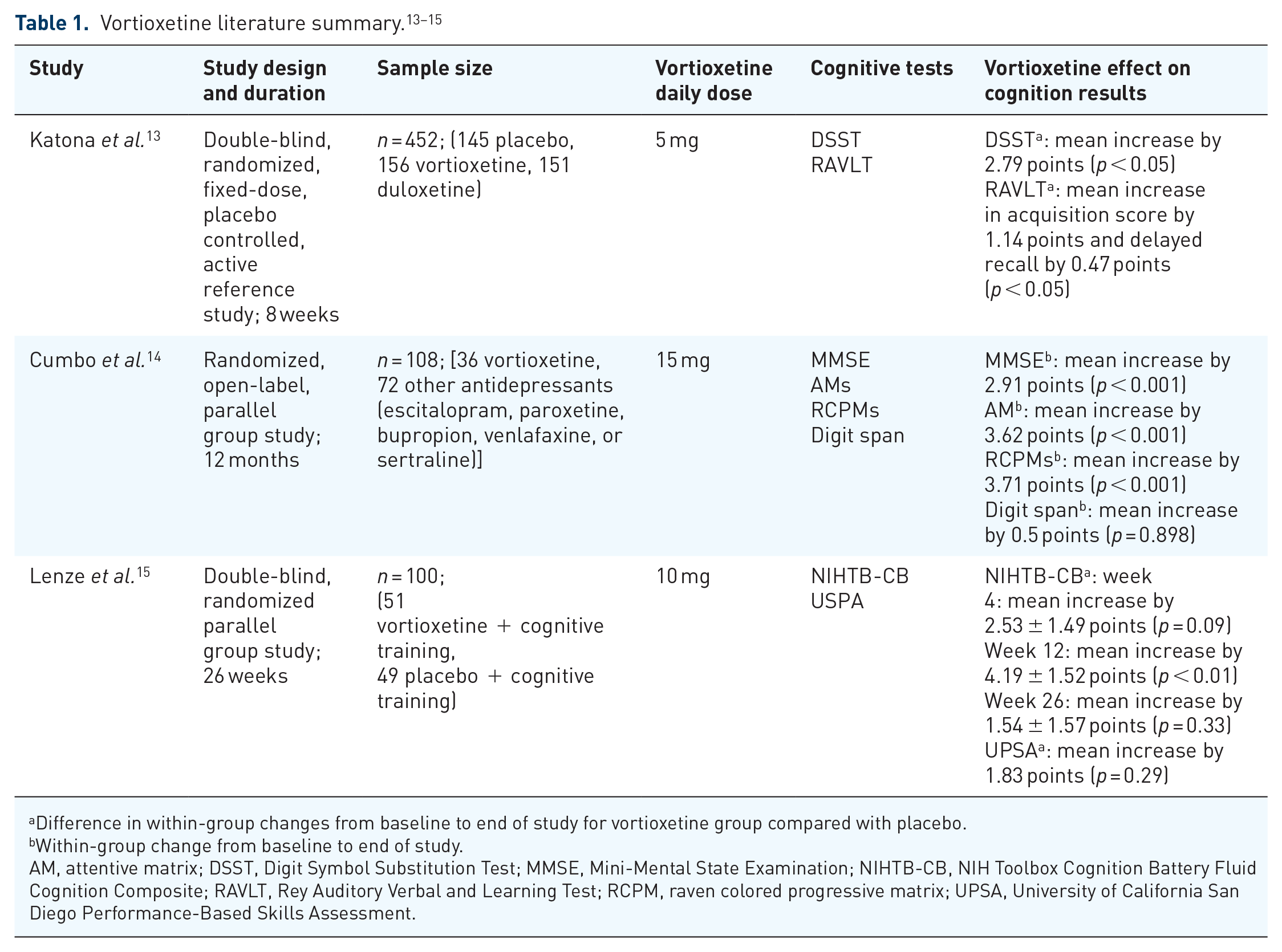
The PubMed, EMBASE, PsycINFO and Cochrane collaboration databases were searched for randomized controlled and open-label trials published to 31 August 2020 using the following Medical Subject Heading terms: vortioxetine AND cognition. Studies were included if they reported on cognitive findings and if the average age of included patients was 65 years or older. A summary of the included studies can be found in Table 1. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines were utilized in conducting this systematic review.
Difference in within-group changes from baseline to end of study for vortioxetine group compared with placebo.
Within-group change from baseline to end of study.
AM, attentive matrix; DSST, Digit Symbol Substitution Test; MMSE, Mini-Mental State Examination; NIHTB-CB, NIH Toolbox Cognition Battery Fluid Cognition Composite; RAVLT, Rey Auditory Verbal and Learning Test; RCPM, raven colored progressive matrix; UPSA, University of California San Diego Performance-Based Skills Assessment.
The initial search was conducted independently by one study author (MB), and the search was then replicated by a second study author (SP) for validation. Data extraction was then performed independently (MB) with subsequent validation by an additional author (SP). Investigators from the included studies were contacted if additional information regarding their respective study was desired.
The Grading of Recommendations, Assessment, Development and Evaluations (GRADE) checklist was used to assess the quality of papers included. 16 Selection bias and detection bias were low for all randomized and active comparator studies as well as for the open-label study owing to randomization of treatment groups and outcomes. Performance bias could not be excluded in the open-label study owing to lack of blinding of participants. All three trials met minimum 80% of participants enrolled completing the study, per recommended GRADE criteria. Reporting bias was low for all studies given reporting of significant and non-significant results. Additionally, none of these studies were ended prematurely. The GRADE criteria for the included studies can be referenced in Table 2.
GRADE criteria checklist.
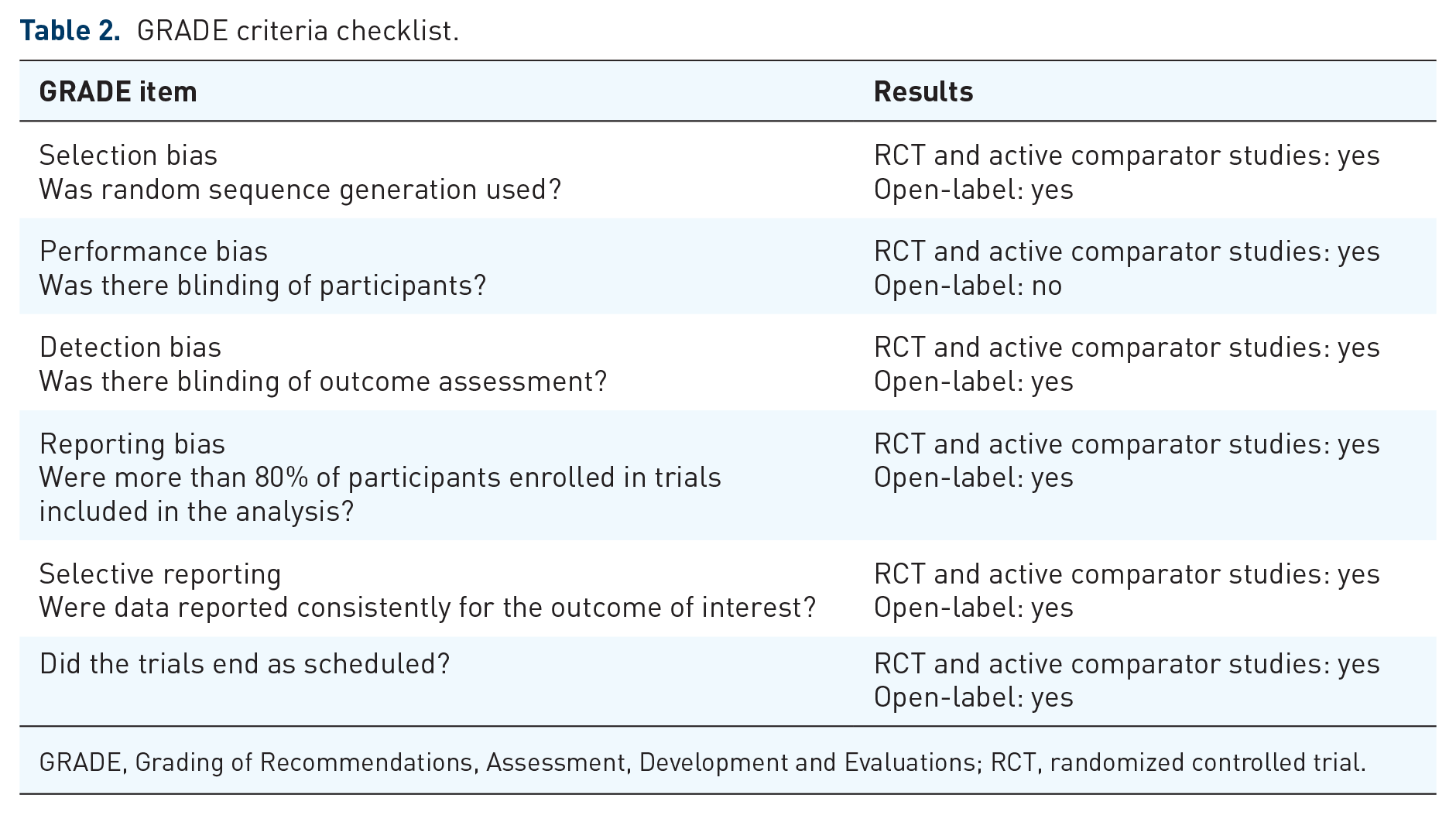
GRADE, Grading of Recommendations, Assessment, Development and Evaluations; RCT, randomized controlled trial.
Results
A total of 52 studies were identified in the literature search (Figure 1). Nineteen duplicate studies were removed and 30 were excluded. Studies were excluded for not including patients 65 years and older, having no cognitive assessment, or a combination.

Literature review findings.
Effect of vortioxetine on depression and cognition in older adults with normal cognition
Katona et al. 13 compared vortioxetine 5 mg daily with placebo and duloxetine 60 mg daily in patients aged 65 years and older with MDD in an 8-week randomized, double-blind trial. The study was powered to assess differences between vortioxetine and placebo, but not between vortioxetine and the active comparator arm. Patients were excluded if they had cognitive impairment or any other psychological diagnosis aside from MDD. The primary endpoint of this study was change in the Hamilton Depression Rating Scale (HAM-D) score compared with placebo at 8 weeks, with cognition as a predefined exploratory endpoint. HAM-D is a common assessment of depression with the following score breakdown: 0–7 is normal, 8–16 is classified as mild depression, 17–23 is classified as moderate depression, and a score of ⩾23 is considered severe depression. 17 Cognitive assessment was a secondary outcome and focused on two aspects of cognition: processing speed and verbal learning and memory.
This study randomized 453 patients: 145 to the placebo group, 156 to the vortioxetine, and 151 to the duloxetine group (one patient did not take any study medication). At baseline, the average classification of depression based on the 24-item HAM-D score was severe depression in all three treatment groups. By the end of the 8-week trial period, the mean HAM-D score was significantly improved in both the vortioxetine (−3.3 ± 1.0; p = 0.0011) and duloxetine (−5.5 ± 1.0; p < 0.0001) groups compared with placebo, indicating efficacy in treating depression in older adults
Changes in cognition from baseline were compared between placebo and treatment groups, and were assessed by the Digit Symbol Substitution Test (DSST) and Rey Auditory Verbal and Learning Test (RAVLT). The DSST measures verbal learning and memory along with motor speed by requiring patients to follow instructions and match a symbol to a number in a given period of time.18,19 Patients with a higher level of learning memory may be better able to recall the symbol that matches to the presented number without having to refer to the key. Likewise, patients with higher levels of motor function will be able to replicate the symbol faster and replicate more symbols in the test time period compared with a patient with a slower motor speed. The multifactorial properties of the DSST allow for this test to be sensitive to different types of cognitive impairments (e.g. age-related decline versus brain damage). The DSST also shows high test–retest reliability and is frequently used in pharmacologic studies. 19 A summary of common cognitive tests used in various studies can be referenced in Table 3. The DSST correct symbol score (out of 100) was 44.6 in the placebo group, 45.2 in the vortioxetine group and 46.3 in the duloxetine group at baseline. After treatment, DSST scores in the vortioxetine group significantly improved by an average of 2.79 points more than placebo (p < 0.05). Patients receiving duloxetine showed a non-significant improvement of 0.77 points compared with placebo. Of note, DSST can also be scored on a continuous scale or by placing patients in a quartile based on score: ⩽29, 30–39, 40–48, and ⩾49. 20 This study found that while patients significantly improved their DSST score compared with placebo in this study, they remained in the same quartile with a score of 40–48 after treatment.
DSST, Digit Symbol Substitution Test; MMSE, Mini-Mental State Examination; NIHTB-CB, NIH Toolbox Cognition Battery Fluid Cognition Composite; RAVLT, Rey Auditory Verbal and Learning Test.
The RAVLT was used to measure immediate and delayed recall. Scores are determined based on the number of correct words repeated back to the evaluator and number of correct words remembered after other tests have been performed. 21 In this study, the RAVLT was broken down into acquisition scores and delayed recall scores. Mean scores at baseline were 21.8 and 6.2 for the placebo group, 22.3 and 6.6 in the vortioxetine group and 22.0 and 6.5 in the duloxetine group, respectively. Compared with placebo, mean RAVLT acquisition and delayed recall scores improved by 1.14 and 0.47 in the vortioxetine group (p < 0.05 for both) and 1.41 and 0.64 in the duloxetine group (p < 0.01 for both).
For both the DSST and the RAVLT, researchers performed a post-hoc path analysis to determine causal relationships between study drugs and changes in cognitive function tests by attempting to control for changes in depressive symptoms. This analysis showed that vortioxetine had an 83% direct effect on DSST (duloxetine 26%), 71% effect on RAVLT acquisition (duloxetine 65%) and 72% on RAVLT delayed recall (duloxetine 66%).
Effect of vortioxetine on cognitive function in patients with AD
Based on the potential procognitive benefits of vortioxetine demonstrated in the study by Katona and colleagues, and given the correlation between AD and depression, Cumbo and colleagues sought to understand the efficacy of vortioxetine 15 mg daily compared with other common antidepressants in patients with AD and depressive symptoms in a 12-month, prospective, randomized, open-label, parallel-group study. 14 To understand this relationship, researchers used change in Mini-Mental State Examination (MMSE) score as their primary efficacy endpoint to assess cognition. The MMSE is a standard cognitive assessment tool that assesses orientation, concentration, verbal memory, naming and visuospatial skills. 18 In general, MMSE scores >26 are considered “normal” and severity of cognitive dysfunction increases as the MMSE score decreases. 22 Secondary efficacy endpoints included attentive matrices (AMs), raven colored progressive matrices (RCPMs) and digit span. Patients were included in this study if they were aged 65 years and older with a probable diagnosis of AD, an MMSE score between 18 and 24 and a Geriatric Depression Scale score of 5 or greater. Patients were also required to have a caregiver help administer the medication and assist in providing information for psychometric assessments.
There were 108 patients included in this study: 36 in the vortioxetine group and 72 receiving other antidepressants [escitalopram (n = 15), paroxetine (n = 15), bupropion (n = 14), venlafaxine (n = 14), sertraline (n = 14)]. Patients had an average baseline MMSE of 20.87 in the vortioxetine group and 20.79 in the control group. At the end of the 12-month study period, patients in the vortioxetine group experienced an average improvement of 2.91 points in the MMSE, producing an average MMSE score of 23.78 ± 3.8. This change was statistically significant compared with the change seen in the control group (+2.50; p = 0.05). The secondary measures of cognition improved in the vortioxetine group; however, statistical significance compared with baseline was seen only in the RCPM and AM, but not the digit span. The degree of change from baseline for RCPM and AM scores was also significantly greater in the vortioxetine group compared with control (+2.15 and +2.94, respectively; p = 0.05 for both).
Vortioxetine plus cognitive training in patients with age-related cognitive decline
Most recently, Lenze et al. 15 sought to explore the effects of vortioxetine paired with cognitive training on cognition in adults aged 65 years and older with age-related decline. Age-related decline was defined as self-reported cognitive dysfunction attributed to aging as identified through screening questions and a score within one standard deviation of the age-matched mean score on the NIH Toolbox Cognition Battery Fluid Cognition Composite (NIHTB-CB) at baseline, as well as after 2 weeks of cognitive training. If patients were randomized into the vortioxetine group, they received 10 mg daily. All patients participated in the cognitive training program, where they performed exercises that ranged in difficulty from basic training in processing speed and attention to more complex training in working memory and executive function. The primary endpoint measure was the change in NIHTB-CB from baseline for each treatment group. Secondary endpoint measures included changes from baseline in the University of California San Diego Performance-Based Skills Assessment (UPSA).
The NIHTB-CB uses the Flanker Inhibitory Control Test, Dimensional Change Card Sort Test, List Sorting Working Memory test, Picture Sequence Memory Test and the Pattern Comparison Processing Test. The composite results of these tests measure five different aspects of cognition: attention and inhibitory control, cognitive flexibility, working memory, episodic memory, and processing speed. A meta-analysis determined that the NIHTB-CB has greater validity over the lifespan compared with other tests measuring cognition. 23 This test differs from others, such as DSST, RAVLT and MMSE, by measuring fluid cognition that is involved in problem solving as opposed to global cognition or learning and verbal memory. UPSA is a validated test that assesses aspects of everyday functioning, such as planning, comprehension, finance, transportation, and communication, by having patients participate in role playing exercises. It is scored on a scale from 0 to 100.
There were 100 patients randomized, 49 into the placebo group and 51 into the vortioxetine group. At the time of randomization, the average NIHTB-CB fluid cognition composite score in the vortioxetine group was 99.59, compared with 102.57 in the placebo group. Average UPSA scores were 80.80 in the vortioxetine group and 81.47 in the placebo group. Table 3 illustrates that a significant difference in the NIHTB-CB was found only at week 12 when comparing vortioxetine plus cognitive training with placebo plus cognitive training. No significant inter-group difference was found at weeks 4 or 26. At week 12, NIHTB-CB scores improved by an additional 4.19 points in the vortioxetine group compared with the placebo group (p = 0.0063). There was no significant difference between groups with the UPSA test, although both groups showed trends toward improvement.
Safety of vortioxetine
All three referenced studies also evaluated the safety of vortioxetine in older adults. Katona and colleagues found the only adverse effect significantly more common with vortioxetine than placebo was nausea, experienced by 21.8% of patients receiving vortioxetine versus 8.3% with placebo (p < 0.001). The only other notable adverse effect (>10%) identified in this study included headache, though there was no statistical significance between groups. Cumbo and colleagues also described the most common adverse effects as nausea (8.6%) and headache (8.6%), but rates were not significantly different from in the placebo group (p = 0.13 and p = 0.45, respectively). Nausea was also the only adverse effect significantly more common with vortioxetine (29.4%) than placebo (4.1%) in the study by Lenze and colleagues (p < 0.001).
Discussion
The three studies identified in this search assessing cognition in older adults all showed statistically significant improvements in cognitive function with the use of vortioxetine. However, it is important to consider the clinical significance of these individual findings. Katona and colleagues demonstrated that vortioxetine significantly improved DSST scores compared with placebo, yet the change in score was not enough to change the scoring quartile the patient was in, potentially limiting the clinical significance of this result. The vortioxetine group also showed significant improvement in both RAVLT scores in this study. A benefit of this test is that it is multifactorial and can measure different aspects of memory impairment. 21 However, there are many different forms of RAVLT, which means generalizability may be difficult to determine. While acquisition and delayed recall scores did improve in patients, it is difficult to assess the clinical impact of this improvement. Similarly, Cumbo and colleagues showed significant improvement in MMSE, another test assessing global cognition, with a moderate dose of vortioxetine compared with placebo in patients with AD and depressive symptoms. While the average improvement in MMSE scores seen with vortioxetine did not change the average classification of patients (e.g. moderate to mild severity), a mean change in score by 2.91 points indicates that some patients did likely improve in classification from mild cognitive impairment to normal cognition, offering some potential clinical benefit from vortioxetine.
In contrast, Lenze and colleagues utilized tests that measured fluid cognition, whereas the other studies focused on global cognition. Fluid cognition is involved more in day-to-day tasks and problem solving, which may be more important to some older adults as opposed to learning and verbal memory. This study showed that vortioxetine treatment significantly improved the composite of the NIHTB-CB compared with placebo, but individually only one test of cognition that measured cognitive flexibility was significantly improved. Significance was also seen only at 12 weeks, with differences at week 26 being non-significant. However, there were trends toward improvement in NIHTB-CB favoring vortioxetine at weeks 4 and 26, and therefore the lack of statistical significance at these time points may have been a result of the study being underpowered to detect these differences. While improvement was shown, all patients received cognitive training, making it difficult to assess what changes were due to vortioxetine, cognitive training or a combination of both. In the three studies referenced, patients received therapy for 8, 26 and 52 weeks, respectively. These benefits were recognized as early as 12 weeks after the initiation of therapy, indicating a fairly early response, but it is not clear how long cognitive benefits are maintained, or whether there is further improvement over time.
Each study investigated different doses of vortioxetine: Katona and colleagues studying 5 mg daily, Cumbo and colleagues studying 15 mg daily and Lenze and colleagues studying 10 mg daily. Doses of 5–20 mg daily are approved in Europe and the United States.24,25 All three studies showed improvements in cognition, suggesting any dose of vortioxetine may have procognitive effects, although additional dose-ranging studies may be beneficial to further quantify the effects of dose on cognition, particularly in specific populations (e.g. patients with AD). Sample sizes varied across these studies. Katona and colleagues had the largest population with 453 patients studied and 156 receiving vortioxetine. This study was powered to detect differences between vortioxetine or duloxetine and placebo, however, it was not powered to detect differences between the vortioxetine and duloxetine groups. Cumbo and colleagues and Lenze and colleagues included a smaller number of subjects, with 108 (36 randomized to vortioxetine) and 100 patients (51 randomized to vortioxetine), respectively. The vortioxetine group in the study by Cumbo and colleagues included only 36 patients. Because Cumbo and colleagues and Lenze and colleagues had smaller study sizes, these studies may not have been powered to detect significant differences between groups. Generalizability of all three studies is also limited by the characteristics of patients included. Participants of all three studies were predominantly Caucasian women, limiting the generalizability of these results to broader populations.
Additionally, there are several limitations with regard to the individual study designs. Katona and colleagues compared vortioxetine 5 mg daily to duloxetine 60 mg daily, which is not an equivalent dose and may have impacted interpretation of comparisons between these two treatments. Similar to Cumbo and colleagues, Lenze and colleagues did not include a true placebo group, as patients in the comparator group received cognitive training. The results of this study may have been more impactful if researchers included a reference group of vortioxetine alone to assess how the agent compared against cognitive training. In the study by Cumbo and colleagues, the comparator group included 72 patients, 15 of whom were receiving paroxetine. This agent has the greatest anticholinergic properties of the SSRIs and, because of this, could theoretically worsen cognition in patients with dementia. 26 This could have potentially allowed for a greater cognitive benefit to have been realized with vortioxetine treatment, depending on how many patients received paroxetine. Finally, the study by Cumbo and colleagues was open-label, potentially biasing patients and researchers toward finding improvement in the vortioxetine group. Due to the limited body of evidence assessing vortioxetine use in older adults, this study was still included in the review despite these limitations.
While vortioxetine has shown improvement in cognitive function in older adults, several other antidepressants have shown these effects as well. Patients receiving duloxetine showed improvement in their composite test of cognition. 8 However, since their composite included four different tests, it is difficult to compare these results with those of vortioxetine. Similarly, participants treated with citalopram showed improvement in components of their comprehensive neuropsychological test battery. As with duloxetine, these results are difficult to directly compare against vortioxetine. Citalopram treatment was associated with improvement in global performance, language and attention, which are cognitive areas vortioxetine has shown improvement in as well. 9 With cognitive improvements seen among other antidepressants and with long-term use of SSRIs showing reduced conversion of mild cognitive impairment to AD, it is possible that antidepressants as a class may improve cognitive function in older adults, or treating depression may be the primary reason cognition improved. 7 However, these positive cognitive effects have not been found in all studies of antidepressants. 27 Additionally, the path analysis referenced in the study by Katona and colleagues supports the hypothesis that cognitive benefits with vortioxetine are independent of its effects on depression, which is also in line with findings reported with vortioxetine in patients under 65 years of age. 28 It is unclear whether this is the case for citalopram and/or duloxetine. Therefore, it is possible that vortioxetine may be a preferred antidepressant in older adults, although additional head-to-head studies directly comparing cognitive effects of vortioxetine with other antidepressants are needed. Additionally, newer drug entities such as vortioxetine have higher associated costs than generic medications, and this may need to be taken into consideration as well when making treatment decisions.
Taken with these findings, given the different populations included in the three trials discussed but the similar improvements in cognitive function after receiving vortioxetine, vortioxetine’s procognitive effects may extend beyond its effects on depression. The fact that these studies all focused on different primary endpoints and patient populations but still showed improvement in cognition supports the idea that procognitive effects of vortioxetine are to some degree attributed to the agent’s multimodal mechanism of action rather than solely from improvement in depressive symptoms. However, replication of these findings, ideally in healthy control patients, would be necessary to validate these conclusions. The cognitive benefits seen in patients with AD also raise the question of whether this medication could provide procognitive effects in patients who are not experiencing depressive symptoms. This may prove to be an additional area of future study.
Conclusion
Overall, preliminary data suggest vortioxetine has promising effects in improving cognition in older adults with depressive symptoms and may have a place in therapy for older adults with depression and/or cognitive impairment, including AD. Additional long-term studies that include more diverse populations with comorbidities and direct comparisons with other antidepressants are needed to fully understand vortioxetine’s potential cognitive benefits in older adults.
Footnotes
Acknowledgements
All authors gave consent for publication.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.