Abstract
Background:
The purpose of this study was to analyze the prevalence of long-term benzodiazepine use in older adults treated in general and neuropsychiatric practices in Germany.
Methods:
This study included 32,182 patients over the age of 65 years who received benzodiazepine prescriptions for the first time between January 2010 and December 2014 in general and neuropsychiatric practices in Germany. Follow up lasted until July 2016. The main outcome measure was the proportion of patients treated with benzodiazepines for >6 months.
Results:
The proportion of patients with benzodiazepine therapy for >6 months increased with age (65–70 years: 12.3%; 71–80 years: 15.5%; 81–90 years: 23.7%; >90 years: 31.6%) but did not differ significantly between men (15.5%) and women (17.1%). The proportion of patients who received benzodiazepines for >6 months was higher among those with sleep disorders (21.1%), depression (20.8%) and dementia (32.1%) than among those with anxiety (15.5%). By contrast, this proportion was lower among people diagnosed with adjustment disorders (7.7%) and back pain (3.8%).
Conclusion:
Overall, long-term use of benzodiazepines is common in older people, particularly in patients over the age of 80 and in those diagnosed with dementia, sleep disorders, or depression.
Introduction
Benzodiazepines are one of the most commonly prescribed drug classes in the world [Donoghue and Lader, 2010]. They are notably used in the treatment of anxiety, panic disorders, insomnia, and seizures, as well as to treat the symptoms of alcohol withdrawal [Donoghue and Lader, 2010; Baldwin et al. 2013; Kurko et al. 2015]. Although the use of benzodiazepines is associated with significant clinical benefits in patients affected by these psychiatric and neurological conditions [Montgomery et al. 2009; Offidani et al. 2013], many professionals have had major concerns about the dependency on these medications for decades [López-Muñoz et al. 2011; Hood et al. 2014]. In fact, benzodiazepines have significant side effects, such as increased risk of fall, cognitive impairment, and confusion [Stewart, 2005]. Therefore, it was suggested that benzodiazepines should be used on a short-term basis, for a maximum of a few weeks [Lader, 2008, 2014].
Despite these recommendations, several studies have highlighted the fact that long-term use of benzodiazepines is common worldwide. In 2010, Cunningham and colleagues showed that 8.4% of British Columbians used benzodiazepines and that 3.5% of the whole population were long-term users [Cunningham et al. 2010]. They further found that individuals with low income or poor health and patients over the age of 65 were more likely to use these medications on a long-term basis. Later, in 2015, Finnish researchers discovered that approximately 3% of the general population used benzodiazepines for >6 months [Kurko et al. 2015]. Even if benzodiazepine prescriptions usually involved low doses, nonrecommended prescriptions were common in older people. In line with these findings, one review published in the same year estimated that there were between 128,000 and 1.6 million benzodiazepine-dependent people in Germany [Janhsen et al. 2015]. More recently, a 2017 study underlined the fact that benzodiazepines were the most common drugs reported to be potentially inappropriate for older people [Lucchetti and Lucchetti, 2017]. Although previous work has shown that the older people are at a particular risk of being dependent on benzodiazepines, prescriptions in older patients were often excluded from analyses [Janhsen et al. 2015]. Due to this limitation, little is known about the long-term prescription of these medications in older people in Germany.
Therefore, the goal of the present study was to analyze the prevalence of long-term benzodiazepine use in older people treated in general and neuropsychiatric practices in Germany.
Methods
Database
The Disease Analyzer database (IMS HEALTH) compiles drug prescriptions, diagnoses, and basic medical and demographic data obtained directly and in anonymous format from computer systems used in the practices of general practitioners [Becher et al. 2009]. Diagnoses (ICD-10), prescriptions (Anatomical Therapeutic Chemical Classification System), and the quality of reported data have been monitored by IMS, based on a number of criteria (e.g. completeness of documentation and linkage between diagnoses and prescriptions). In Germany, the sampling methods used for the selection of physicians’ practices were appropriate to obtain a representative database of the same. This sampling method for the Disease Analyzer database is based on summary statistics from all physicians in Germany published each year by the German Medical Association. The statistical unit of IMS uses these statistics to determine the panel design according to the following strata: specialist group, German federal state, community size category and age of physician. This panel design forms the basis for the acquisition of the practices processed in the Disease Analyzer. Cooperating software companies carry out the acquisition of and provide support for the practices using a standardized interface for IMS that enables the practices to collect the required data and send them to IMS in an anonymized form.
The validity and representativeness of the Disease Analyzer database have been previously assessed [Becher et al. 2009]. Prescription statistics for several drugs were very similar to data available from pharmaceutical prescription reports. Furthermore, the age groups for given diagnoses in the Disease Analyzer agreed well with those in corresponding disease registries [Becher et al. 2009].
Finally, the Disease Analyzer database has already been used in studies focusing on benzodiazepines and psychiatric disorders [Drosselmeyer et al. 2016; Jacob et al. 2016; Jacob and Kostev, 2016].
Study population
This study included patients 65-years old and over, who received benzodiazepine prescriptions for the first time between January 2010 and December 2014 in 1224 general and 173 neuropsychiatric practices in Germany (index date). Follow up lasted until July 2016. In order to guarantee the accuracy of the estimation of therapy duration with benzodiazepines, patients with an observation period of <365 days after the index date were excluded. Patients with a follow-up time of <365 days prior to the index date were excluded. This exclusion was necessary for the correct determination of treatment initiation. Treatment initiation was possible when there was no prescription of benzodiazepines in the year prior to index date.
Benzodiazepines included the following: brotizolam, flunitrazepam, flurazepam, loprazolam, lormetazepam, midazolam, nitrazepam, temazepam, triazolam, tetrazepam, alprazolam, bromazepam, chlordiazepoxid, clobazam, diazepam, lorazepam, medazepam, oxazepam, and prazepam.
Study outcome
The main outcome measure was the proportion of patients treated with benzodiazepines for at least 6 months without discontinuation within the year following the index date. Discontinuation of therapy was defined as a period of at least 180 days without benzodiazepines. A longitudinal dataset of medication supply was built for each individual patient, whereby the number of days of drug supply was calculated based on the quantity and dosage information associated with each prescription record. The day of discontinuation was the day when the last pill was taken (day of last prescription + days’ supply) prior to the 180-day gap.
Covariables
Demographic data included age, sex, and type of physician (general practitioner or neurologist/psychiatrist). Diagnoses associated with benzodiazepine prescriptions were: anxiety (ICD-10 codes: F41), depression (F32, F33), somatoform disorder (F45), reaction to severe stress and adjustment disorder (F43), all-cause dementia (F01, F03, G30), sleep disorders (F51, G47), epilepsy (G40, G41) and disorders related to back pain (M51–M54, M62).
Statistical analysis
Kaplan–Meier curves were used to analyze long-term benzodiazepine therapy as a function of age and diagnosis. A logistic regression model was further performed to estimate the relationship between therapy for >6 months and the predefined demographical/clinical variables previously described. The Bonferroni correction was used to reduce multicollinearity. After this correction, a p value < 0.004 was considered statistically significant. Analyses were carried out using SAS version 9.3.
Ethics
In Germany, database analyses using anonymous data do not require ethics committee consideration. Patients were not required to give informed consent to the study because the analysis used anonymous data.
Results
From a total of 6,819,472 patients available in the database (neuropsychiatric and GP practices) between January 2010 and December 2014, 364,885 patients with an initial prescription of benzodiazepines were selected. 149,317 patients were aged ⩾65 years on the index date. After excluding patients with a follow-up of <365 days prior to the index date and <365 days after the index date, 32,182 patients were available for analysis (Figure 1).

Selection of study patients.
Patient characteristics are shown in Table 1. A total of 32,182 patients with benzodiazepine prescriptions at the index date were included in this study. The mean age among them was 75.1 years [standard deviation (SD) = 6.7 years], 34.8% were men, and 9.0% were treated in neuropsychiatric practices. Only 8.7% of patients had a private health insurance coverage. In patients with statutory health insurance coverage, 14.1% of benzodiazepine prescriptions were private and had to be paid for by the patients rather than by their insurance funds.
Baseline characteristics of patients with benzodiazepine therapy in general and neuropsychiatric practices in Germany.
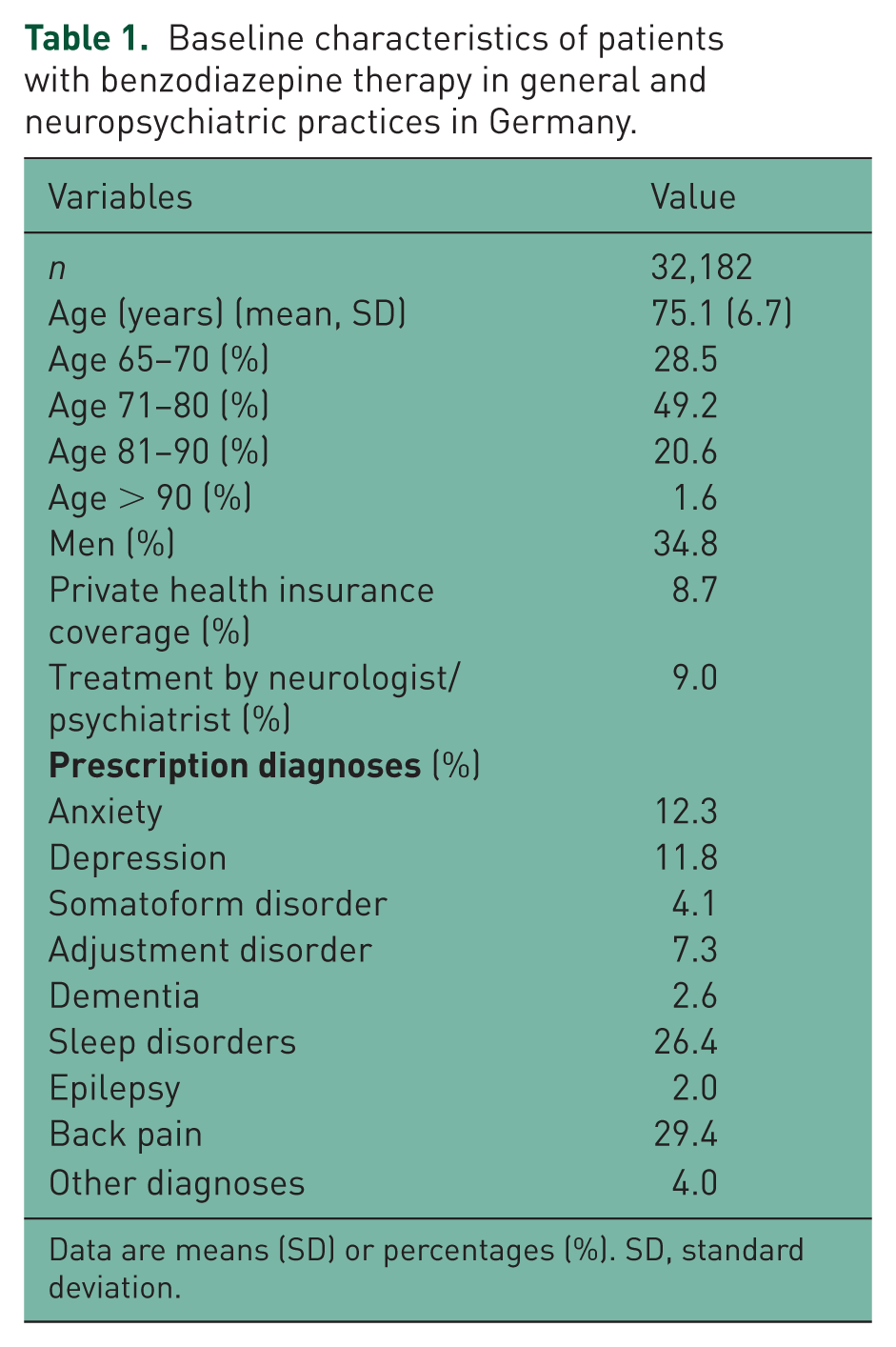
Data are means (SD) or percentages (%). SD, standard deviation.
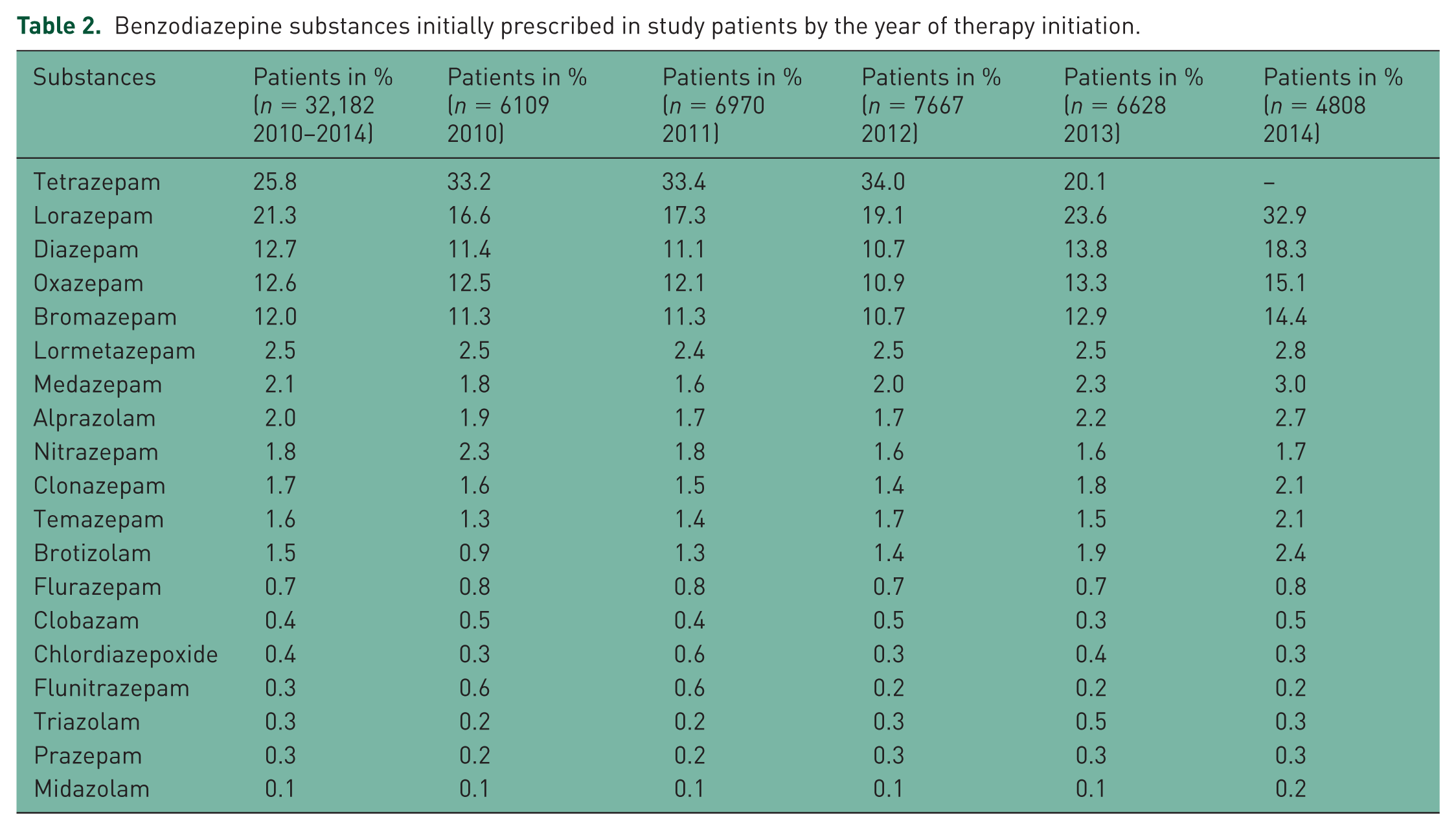
Benzodiazepines were often prescribed for back pain (29.4%), sleep disorders (26.4%), anxiety (12.3%), and depression (11.8%). The proportions of different benzodiazepines are shown in Table 2. Tetrazepam (25.8%), lorazepam (21.3%), and diazepam (12.7%) were the three most frequently prescribed drugs. However, in 2014, tetrazepam was no longer prescribed as it had been withdrawn from the market in August 2013.
Benzodiazepine substances initially prescribed in study patients by the year of therapy initiation.
Figures 2 and 3 display Kaplan–Meyer curves for the proportion of patients receiving benzodiazepines within the first year after the index date by age and diagnosis, respectively. Moreover, Table 3 shows the number of benzodiazepine prescriptions per patient during the study period. A total of 72.0% of patients treated by GPs and 60.9% of patients treated by neuropsychiatrists received only one prescription, 11.1% and 12.8% received two prescriptions, and 3.6% and 4.8% received three prescriptions.

Proportion of patients receiving benzodiazepine therapy by age within the first year of follow up after the index date in general and neuropsychiatric practices in Germany.

Proportion of patients receiving benzodiazepine therapy by diagnosis within the first year of follow up after the index date in general and neuropsychiatric practices in Germany.
Number of benzodiazepine prescriptions per patient in general and neuropsychiatric practices in Germany.

The results of the logistic regression model are shown in Table 4. The proportion of patients receiving benzodiazepine therapy for >6 months increased with age, from 12.3% in individuals aged 65–70 to 15.5% in individuals aged 71–80 [hazard ratio (HR) = 1.30], 23.7% in individuals aged 81–90 (HR = 2.19) and 31.6% in individuals aged over 90 (HR = 3.23). The prevalence of long-term therapy did not differ significantly between men (15.5%) and women (17.1%). Interestingly, long-term use of benzodiazepines was more frequent in neuropsychiatric practices (25.6% and HR = 1.47) than in general practices (15.6%). Finally, the proportion of patients receiving benzodiazepine therapy for >6 months was higher among those with sleep disorders (21.1% and HR = 1.49), depression (20.8% and HR = 1.37), and dementia (32.1% and HR = 1.96) than among those with anxiety (15.5%). By contrast, this proportion was lower among people diagnosed with adjustment disorders (7.7% and HR = 0.48) and back pain (3.8% and HR = 0.24).
Association between benzodiazepine therapy for >6 months and demographical/clinical variables.
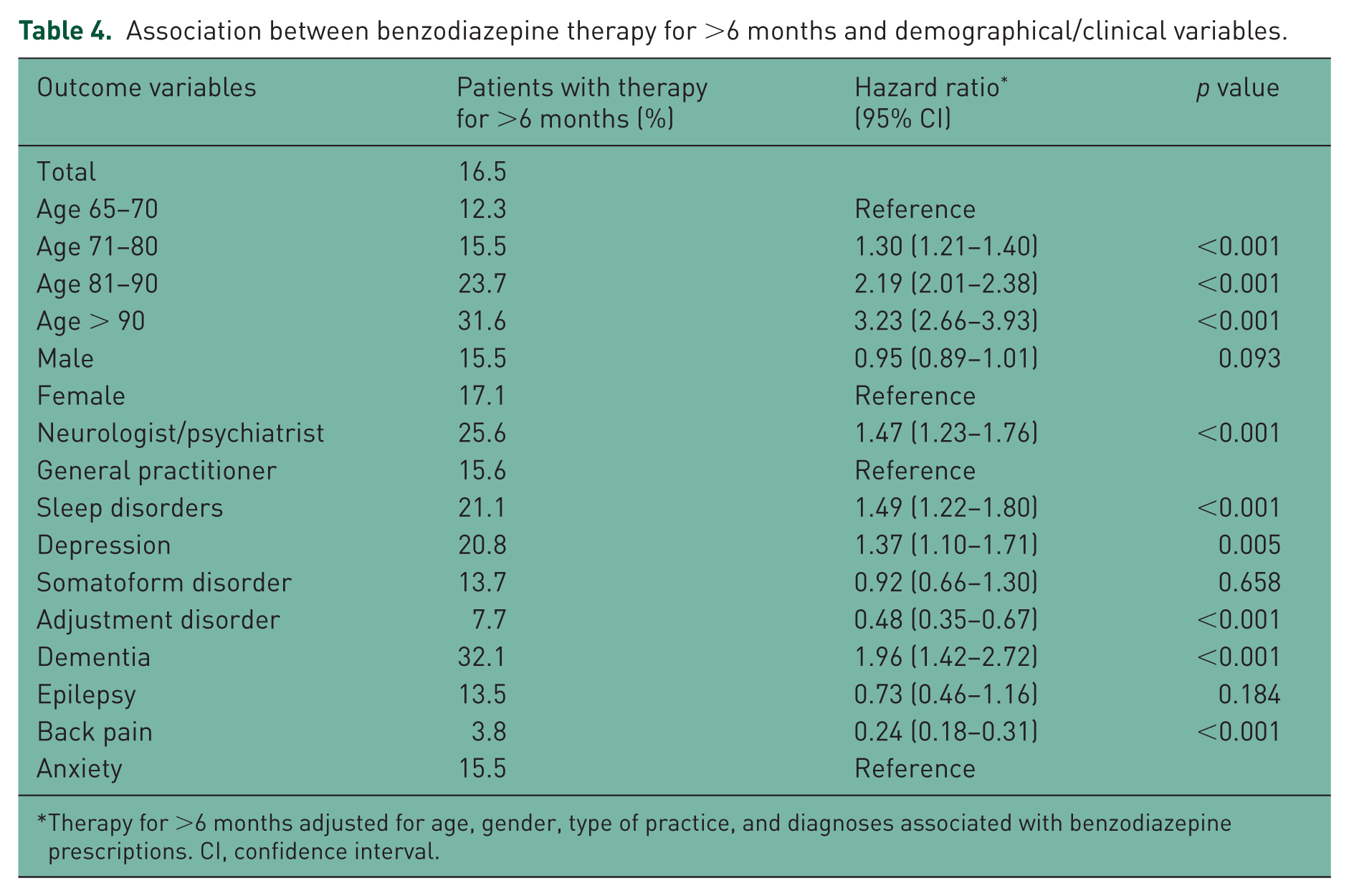
Therapy for >6 months adjusted for age, gender, type of practice, and diagnoses associated with benzodiazepine prescriptions. CI, confidence interval.
Discussion
This German retrospective study of >32,000 older people showed that the proportion of patients receiving benzodiazepine therapy for >6 months was approximatively 15–20%. The long-term use of benzodiazepines was more frequent in older people, patients treated in neuropsychiatric practices, and individuals diagnosed with dementia, sleep disorders, or depression. However, no difference in this proportion was found between men and women.
For decades, benzodiazepine therapy has been associated with major side effects and an important risk of dependence, which can be more common when these drugs are misused or prescribed on a long-term basis [Gorenstein et al. 1995; Voyer et al. 2010; Windfuhr et al. 2011; Manthey et al. 2012]. In 2010, Voyer and colleagues showed that almost 10% of community-dwelling older people receiving benzodiazepines were dependent on these medications based on DSM-IV-TR criteria [Voyer et al. 2010]. Even more concerning, 43% of users believed that they were dependent on these medications, suggesting that the real share of dependency was potentially much higher. Furthermore, a Dutch study found that insomnia, antidepressant use, and alcohol dependence increased the risk of benzodiazepine dependence [Manthey et al. 2012]. Furthermore, benzodiazepines cause major side effects, such as increased risk of fall, cognitive impairment, and confusion [Stewart, 2005]. In 2007, Hartikainen and colleagues found in 28 observational studies that benzodiazepines were associated with an increased risk of fall [Hartikainen et al. 2007].
Although it was suggested that benzodiazepines should only be prescribed for several weeks and for specific indications [Lader, 2008, 2014], these drugs are often prescribed on a long-term basis and for conditions for which better treatments are available (e.g. insomnia or anxiety syndromes) [Kapil et al. 2014]. A 2010 Canadian study including almost four million people estimated that 4.9% of patients were short-term users and 3.5% were long-term users [Cunningham et al. 2010]. Women, older patients, and individuals with a low income or poor health status were more likely to be prescribed benzodiazepines. The same analysis further revealed that being in the lowest income quintile, having very poor health, and being over the age of 65 were risk factors for the long-term use of these drugs. Interestingly, the long-term prescription of benzodiazepines had decreased in patients aged 70 years and over between 1996 and 2006, but had also increased in middle-aged individuals. In 2014, a UK analysis showed that approximately 8% of people had misused benzodiazepines for sleep disorders and anxiety symptoms [Kapil et al. 2014].
More recently, Kurko and colleagues found in a systematic review of 41 studies that the proportion of long-term benzodiazepine therapy was 3% in the general population [Kurko et al. 2015]. Although the studies varied significantly, the authors discovered that the relative proportion of long-term users of benzodiazepines was higher among older people (47%) than among younger patients (24%), and that such prescriptions usually involved low doses. Finally, Kurko and colleagues investigated the key characteristics related to the misuse of benzodiazepines. Despite there being no clear association between gender, socioeconomic status, and benzodiazepine prescriptions, the long-term use of these drugs was very frequent among older patients and individuals with comorbidities, particularly psychiatric conditions. Furthermore, according to several studies included in this systematic review, the risk of long-term benzodiazepine use was increased in people receiving high-potency molecules (i.e. alprazolam, lorazepam or oxazepam), multiple drugs, and high doses. In contrast, there was no clear finding regarding the potential association between long-term use and prescribers’ characteristics.
Also in 2015, Janhsen and colleagues estimated that the number of benzodiazepine-dependent people in Germany ranged from 128,000 to 1.6 million [Janhsen et al. 2015]. Although these findings are of particular importance, most of the analyses included in this work did not take into account private prescriptions or prescriptions for older people. Therefore, the number of individuals receiving long-term benzodiazepine treatment remains underestimated. In line with previous work performed in other countries [Cheng et al. 2008; Luijendijk et al. 2008; Andersen and Frydenberg, 2011], this study found that the proportion of long-term use was around 15–20% in older German people. Furthermore, there was a positive association between age and benzodiazepine therapy lasting >6 months, with a twofold increase in the risk of using benzodiazepines on a long-term basis in patients aged 81–90 compared with patients aged 65–70. Corroborating the results of the Finnish systematic review [Kurko et al. 2015], no significant impact of gender on the prescriptions of these drugs was found. Furthermore, long-term use of benzodiazepines was more common in neuropsychiatric than in general practices. One must bear in mind that neuropsychiatrists in Germany are often responsible for initiating treatment in patients affected by neurological or psychiatric disorders. Such prescriptions may be renewed by neuropsychiatrists themselves or by general practitioners. It is also possible that general practitioners and psychiatrists have different prescription attitudes towards benzodiazepines. In 2006, Siriwardena and colleagues found that the beliefs of nonspecialists about the effectiveness and safety of these drugs were not determined by scientific evidence of national guidelines [Siriwardena et al. 2006]. Later, a review of seven studies published between 1993 and 2010 estimated that general practitioners were ambivalent in their attitude towards prescribing benzodiazepines and inconsistently applied management strategies for their use [Sirdifield et al. 2013].
Another important finding of this study is that individuals diagnosed with dementia, sleep disorders, or depression were more likely to be prescribed benzodiazepines for >6 months than individuals diagnosed with anxiety. It is important to keep in mind that dementia guidelines recommend only the short-term use of benzodiazepines, as their use in people with dementia is problematic because of the negative effects on cognition, the increased fall risk, possible paradoxical reactions, and the potential for dependence [S3 guideline ‘Dementia’]. Moreover, the use of nonpharmacologic intervention is recommended for most sleep disorders [Mayer, 2011]. The present findings highlight the fact that benzodiazepines are frequently used to treat these conditions in Europe, particularly in Germany [Schröder, 2013; Booker et al. 2016; Wiegand et al. 2016]. In 2011, Bourgeois and colleagues showed in Belgian nursing homes that the first medical indication for the prescription of these molecules was insomnia, with 59% of benzodiazepine users being affected by this sleep disorder [Bourgeois et al. 2011]. Daily doses in the treatment of insomnia were further estimated to exceed the geriatric upper limit for most of the prescriptions of lormetazepam, zolpidem, and zopiclone. A more recent analysis of a German health insurance fund’s routine data that included 236,843 patients diagnosed with depression discovered that 8.3% of the population received long-term benzodiazepine prescriptions [Wiegand et al. 2016]. In 2016, these findings were corroborated in a case-control study, as the proportion of benzodiazepine prescriptions was 4.2% in depressed patients without cancer and 7% in depressed patients with cancer [Jacob et al. 2016]. Finally, Majic and colleagues found that the proportion of home residents with dementia treated with benzodiazepines was around 7% [Majic et al. 2010]. In line with these results, the present study discovered that the proportion of long-term benzodiazepine use was high in individuals with sleep disorders, depression, or dementia. Although these various studies should be compared with caution, it is interesting to note that the prevalence of benzodiazepine prescriptions was higher in analyses including older people than in analyses based on the general population.
In general, retrospective primary-care database analyses are limited by the degree of completeness of the data on which they are based. The major limitation was related to medical diagnoses performed by general practitioners and neuropsychiatrists, since these diagnoses could not be fully documented. Data on socioeconomic status and lifestyle-related risk factors were also unavailable, although socioeconomic factors may be important predictors of long-term benzodiazepine use. Moreover, no central system registered the drugs prescribed to a patient by different doctors, so the possibility that some patients might have received their follow-up prescriptions from another doctor cannot be excluded. The strengths of this work were the number of patients and the various types of psychiatric and neurological disorders included in the analysis.
Overall, long-term use of benzodiazepines is high among older people in Germany, particularly in patients over the age of 80 and in those diagnosed with dementia, sleep disorders, or depression. Further studies are needed to gain a better understanding of the impact of the characteristics of benzodiazepines and prescribers on the long-term use of these medications.
Footnotes
Individual contribution to the manuscript
Karel Kostev: Study concept and design, statistical analysis, final approval, agreement to be accountable for all aspects of the work. Louis Jacob: Drafting the manuscript, critically revising the work for important intellectual content, final approval, agreement to be accountable for all aspects of the work. Jens Bohlken: Critically revising the work for important intellectual content, final approval, agreement to be accountable for all aspects of the work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Louis Jacob reports no disclosures relevant to the manuscript. Jens Bohlken reports no disclosures relevant to the manuscript. Karel Kostev is an employee of IMS HEALTH, a company that focuses on analyses of pharmacy records and runs the database used in this analysis. However, this study was not part of any business project.
