Abstract
Background:
The aim of this study was to describe dosing patterns and medication adherence among bipolar patients who initiated lurasidone in a real-world setting.
Methods:
Adult bipolar patients who initiated lurasidone between 1 November 2010 and 31 December 2012 (index period) with 6-month pre- and post-index continuous enrollment were identified from the IMS RWD Adjudicated Claims US database. Patients were grouped by starting lurasidone daily dose: 20 mg (7.1%), 40 mg (62.2%), 60–80 mg (28.7%), and 120–160 mg (2.1%). Patient characteristics were compared across doses using Cochran–Armitage trend tests. Multivariable ordinal logistic regression assessed the association between initial lurasidone dose and patient characteristics. Medication adherence was measured using medication possession ratio (MPR).
Results:
Of 1114 adult bipolar patients (mean age 40.6 years, 70.6% female), 90% initiated lurasidone at 40 mg or 80 mg/day (mean 51.9 mg/day). Of these, 16.2% initiated lurasidone as monotherapy. Mean lurasidone maintenance dose was 55.2 mg/day and mean MPR was 0.53 [standard deviation (SD) = 0.34] over the 6-month follow up. Substance use, hyperglycemia, obesity, and prior antipsychotic use were associated with higher initial lurasidone doses (p < 0.05). Odds of a 20 mg/day increase in initial lurasidone dose was 1.6-times higher for patients with substance use [95% confidence interval (CI): 1.16−2.24], 2.6-times higher with hyperglycemia problems (95% CI: 1.15−5.83), 1.7-times higher with obesity (95% CI: 1.05−2.60), and 1.3 (95% CI: 1.01−1.78) and 1.8-times higher (95% CI: 1.17−2.86) with prior use of second- and first-generation antipsychotics, respectively.
Conclusions:
This real-world analysis of bipolar patients indicated that 40 mg or 80 mg/day were the most common starting doses of lurasidone. A majority of patients used concomitant psychiatric medications (polypharmacy). Higher doses of lurasidone were prescribed to patients with comorbidities or prior antipsychotic use. Adherence to lurasidone was comparable to or better than antipsychotic adherence reported in bipolar disorder literature.
Introduction
Bipolar disorder is a chronic psychiatric illness characterized by episodes of mania or hypomania alternating with depressive episodes and inter-episodic subsyndromal symptoms of mania/depression [Leboyer and Kupfer, 2010]. The initial onset is often subthreshold nonspecific symptoms in adolescence or early adulthood [Perugi et al. 2000] and the lifetime prevalence of bipolar disorder is approximately 2–4% among adults in the United States (US) [Merikangas et al. 2007]. Due in part to its increased morbidity and mortality and high risk for suicidal tendencies [Tidemalm et al. 2014], bipolar disorder is ranked as one of the top 10 leading causes of disability in the US and worldwide [National Alliance on Mental Illness, 2013].
According to treatment guidelines published by the American Psychiatric Association, first-line pharmacological treatment for bipolar patients with severe manic or mixed episodes is combination therapy with a mood stabilizer and an atypical antipsychotic [American Psychiatric Association, 2000, American Psychiatric Association, 2002]. Monotherapy with either a mood stabilizer or an atypical antipsychotic is recommended for bipolar patients with less severe symptoms [American Psychiatric Association, 2000, American Psychiatric Association, 2002].
Atypical antipsychotics have varying efficacy and tolerability profiles, and may, to varying degrees, cause weight gain and changes in metabolism [Vancampfort et al. 2013], resulting in an increased risk of metabolic syndrome [Vancampfort et al. 2013] and diabetes [Crump et al. 2013]. The problems associated with metabolic syndrome may be further exacerbated by the fact that patients with bipolar disorder are significantly more likely to have hyperglycemia, diabetes, angina, dyslipidemia, hypertension, and metabolic syndrome than the general population [Fagiolini et al. 2013].
Until recently, only two second-generation antipsychotics, olanzapine (combined with fluoxetine) and quetiapine (immediate or extended release), have been approved by the US Food and Drug Administration (FDA) as monotherapy treatment for bipolar depression. On 28 June 2013, lurasidone (Latuda) was the first second-generation antipsychotic to be approved by the FDA for major depressive episodes associated with bipolar I disorder (bipolar depression), as monotherapy or adjunctive therapy with lithium or valproate [Sunovion, 2013].
Given that lurasidone was approved for the treatment of bipolar depression in 2013, only limited real-world information exists regarding baseline patient characteristics, dosing patterns, and post-initiation adherence rates associated with use of lurasidone among patients with bipolar depression. Such information is important for the clinical community to understand the extent to which these factors contribute to the prescribers’ selection of a specific atypical antipsychotic in the treatment of bipolar disorder and patients’ adherence to it. The aim of this study was to describe dosing patterns, and post-initiation adherence rates among bipolar patients who initiated lurasidone in a real-world setting.
Materials and methods
Data source and study design
A retrospective database analysis was conducted using longitudinal data from 1 May 2010 to 30 June 2013 from the IMS RWD Adjudicated Claims US database (formerly known as the PharMetrics Patient-Centric database). The IMS RWD Adjudicated Claims, US database has the largest representation of the commercially-insured population in the US. It is comprised of health plan information from managed care plans and other sources, and includes fully-adjudicated medical and pharmaceutical claims for over 150 million unique patients across the US (approximately 40 million covered lives per year) [IMS Health, 2013]. In compliance with the Health Insurance Portability and Accountability Act, patient data included in the analyses were de-identified. This study was exempt from the Institutional Review Board (IRB) review.
Patient selection
Adult patients (age ⩾ 18 years) with a diagnosis of bipolar disorder [International Classification of Diseases, 9th revision, Clinical Modification (ICD-9-CM) codes of 296.0, 296.1, 296.4-296.8] were first identified between 1 May 2010, and 31 December 2012. Included patients were newly-initiated on lurasidone between 1 November 2010 and 31 December 2012. The date of the first prescription fill for lurasidone served as the patient’s index date. Eligible patients were continuously enrolled in the health insurance plan for ⩾6 months prior to the index date (pre-index period) and ⩾6 months following the index date (post-index period). They were required to have ⩾1 inpatient or outpatient claim for bipolar disorder (including those with an ICD-9-CM of 296.2 and 296.3), and no prescription fills for lurasidone during the pre-index period. Patients were excluded if they had >10% of visits for schizophrenia (ICD-9 CM code 295.X) within the pre- and post-index periods or did not receive a feasible lurasidone daily dose (i.e. 20 mg, 40 mg, 60 mg, 80 mg, 120 mg, and 160 mg, as described in the lurasidone doses section).
Baseline characteristics
Demographic characteristics (i.e. age, gender, geographic regions, insurance payer, and insurance product type), diagnosis of bipolar disorder type, and psychiatric and physical comorbidity characteristics were measured during the 6-month pre-index period (i.e. baseline period). As the course of bipolar disease is variable in nature and has the potential for misdiagnosis, bipolar patients were classified into mutually-exclusive bipolar disorder types based on diagnostic characteristics during the 6-month pre-index period [Blanco et al. 2002; Moreno et al. 2007; Chen et al. 2014]. Specifically, patients were categorized as having ‘bipolar depression’ if they had a pre-index diagnosis of bipolar depression (ICD-9-CM 296.5). Of those, bipolar patients with a concurrent major depression diagnosis (ICD-9-CM of 296.2, 296.3) during the pre-index period were re-categorized as ‘bipolar depression’ to account for potential misdiagnosis. The remaining patients were categorized as ‘bipolar mania’ (ICD-9-CM 296.0, 296.1, 296.4), ‘bipolar mixed’ (ICD-9-CM 296.6), or ‘bipolar unspecified’ (ICD-9-CM 296.7, 296.8).
Key psychiatric and physical comorbid conditions among the study patients were identified from ICD-9-CM diagnoses during the pre-index baseline period. Psychiatric comorbidities included generalized anxiety disorders, substance use, personality disorder, post-traumatic stress disorder, and dementia. Physical comorbidities included cardiovascular or metabolic disorders such as hypertension, coronary heart disease, cerebrovascular disease, diabetes, impaired glycemic control or hyperglycemia, hyperlipidemia, and obesity (see appendix for ICD-9-CM codes for each condition).
Pre-index concomitant use of psychiatric drugs was categorized as follows: (1) mood stabilizers; (2) other second-generation antipsychotics; (3) first-generation antipsychotics; and (4) antidepressants. Utilization of psychotherapy (individual, family, or group) during the 6-month pre-index period was identified using Current Procedural Terminology (CPT) codes (90785, 90802, 90804–90829, 90832–90834, 90836–90840, 90845–90847, 90849, 90853, and 90857).
Lurasidone dosing patterns
Lurasidone daily dose was measured at index (initial lurasidone daily dose) and during the 6-month post-index period (lurasidone daily maintenance dose). Initial lurasidone daily dose was calculated based on the first lurasidone prescription, with daily dose defined as prescription strength in milligrams multiplied by quantity of pills dispensed and divided by days of supply. Lurasidone daily maintenance dose was measured as an average dose during the 6-month follow-up period. As noted in the prior section on patient selection, patients were excluded if they received an initial lurasidone daily dose that was not feasible as a combination of the dosage sizes available on the market (e.g. not in 20 mg dose increments, n = 17). The remaining patients with feasible doses were grouped into categories based on the following initial lurasidone daily dose: 20 mg, 40 mg, 60 mg, 80 mg, and 120–160 mg. The 120–160 mg groups were combined due to small sample sizes. Patients with an initial lurasidone daily dose of 60 mg (n = 14) were excluded from statistical comparisons as this subgroup was too small to be analyzed using cross-dose comparisons.
Concomitant psychiatric treatment
Patients were also classified into monotherapy or polypharmacy groups given the frequency of polypharmacy among patients treated for bipolar disorder. Monotherapy was defined as having an active prescription for lurasidone and no other psychiatric medication during the same time period. Polypharmacy was defined as having an active prescription for psychiatric medication in addition to lurasidone on or within 14 days following the index date that was continued for at least 7 days post-index. For the purpose of this study, psychiatric treatments included any use of mood stabilizers, second-generation antipsychotics, first-generation antipsychotics, and antidepressants.
Lurasidone adherence
Adherence was measured by 6-month medication possession ratio (MPR), calculated as the total days’ supply of prescription divided by the length of follow-up during the 6-month follow-up period. MPR was described as a continuous outcome and dichotomized into the proportion of patients who met an adherence threshold of MPR ⩾ 0.8 to be defined as adherent patients. Adherence to lurasidone was reported for overall lurasidone patients, and stratified by use of concomitant treatment at lurasidone initiation.
Statistical analysis
Statistical comparisons across initial lurasidone dose categories (bivariate analyses) were conducted using analysis of variance (ANOVA) for continuous variables, and Chi-square or Fisher’s exact test (when any cell count was <5) for categorical variables. Cochran–Armitage trend tests were used to assess trends across dose categories for dichotomous patient characteristics. A two-sided alpha of 0.05 was considered statistically significant.
A multivariable ordinal logistic regression was used to determine the impact of baseline patient characteristics and prior medication use on the level of initial lurasidone dose received (with initial dose of 20 mg, 40 mg, 80 mg, or 120/160 mg as an ordinal variable).
Results
Of the 363,173 bipolar disorder patients identified during the study period, 141,353 (38.9%) used one or more second-generation antipsychotics between 1 November 2010 and 31 December 2012 (Figure 1). Of those patients, 2678 (1.9%) were prescribed lurasidone. After applying the study inclusion and exclusion criteria, a total of 1114 patients remained in the analytic cohort.
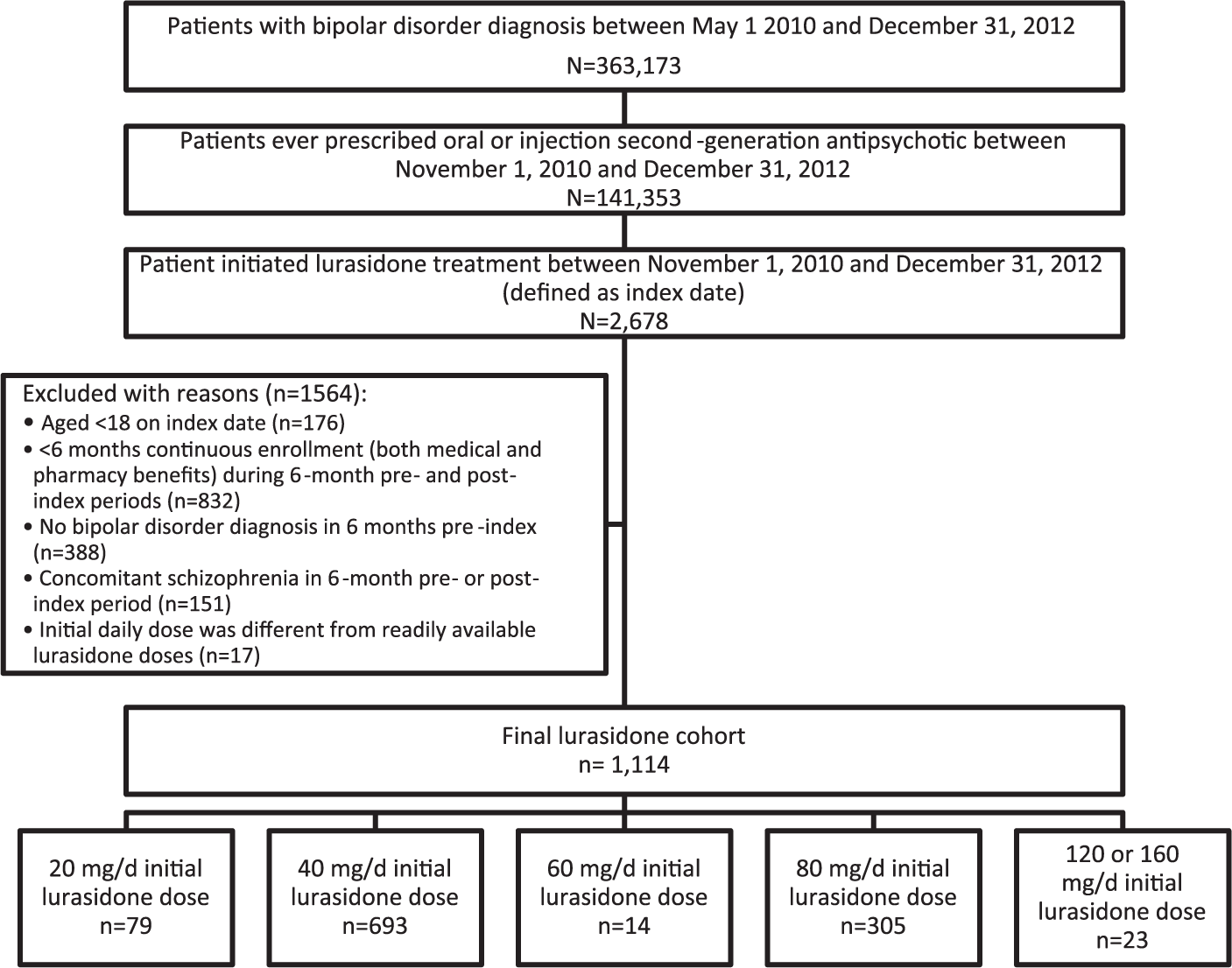
Flow diagram of patient selection.
Baseline characteristics of the analytic cohort
Among the 1114 lurasidone patients, the mean ± standard deviation (SD) age was 40.6 ± 13.1 years and 70.6% of patients were female (Table 1). About two-thirds of patients had commercial health insurance (63.6%) and were enrolled in preferred-provider organization plans (83.8%). Bipolar depression was the most common bipolar diagnosis (57.1%), followed by bipolar unspecified (21.3%), bipolar mania (11.8%), and bipolar mixed (9.9%). Substance use (17.5%) and hypertension (18.5%) were the most common mental and physical comorbidities, respectively.
Pre-index patient demographic, clinical characteristics, and medication treatment patterns, for the entire sample and by dose category.
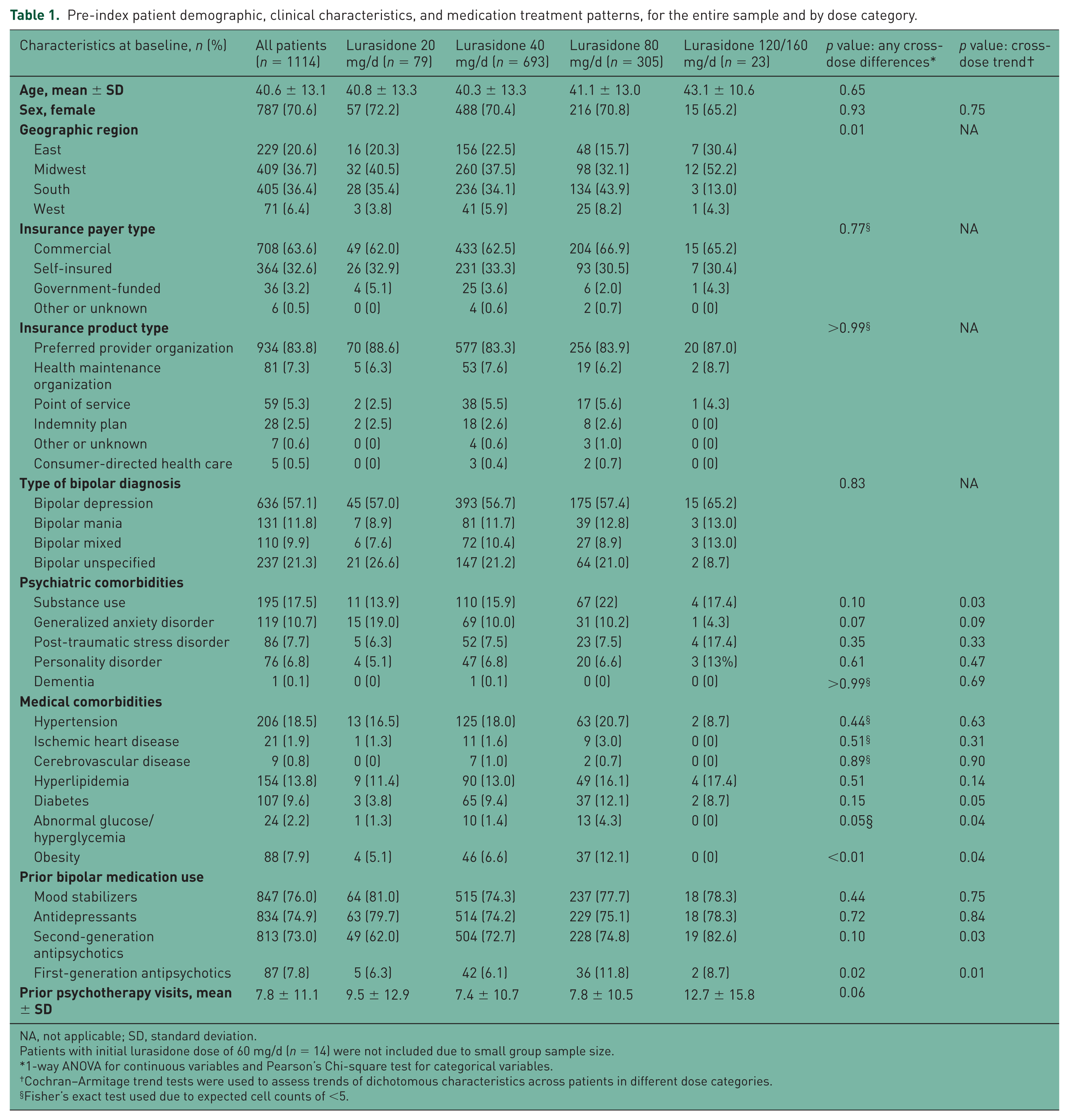
NA, not applicable; SD, standard deviation.
Patients with initial lurasidone dose of 60 mg/d (n = 14) were not included due to small group sample size.
1-way ANOVA for continuous variables and Pearson’s Chi-square test for categorical variables.
Cochran–Armitage trend tests were used to assess trends of dichotomous characteristics across patients in different dose categories.
Fisher’s exact test used due to expected cell counts of <5.
Approximately three-quarters of patients used mood stabilizers (76.0%) and antidepressants (74.9%) during the 6-month period before initiating lurasidone. Approximately 73% patients used other second-generation antipsychotics prior to initiating lurasidone; quetiapine (26.8%), aripiprazole (21.1%), and ziprasidone (14.5%) were the most frequently prescribed second-generation antipsychotics.
Lurasidone doses
Most patients initiated a daily dose of lurasidone of 40 mg (n = 693; 62.2%), followed by 80 mg (n = 305; 27.4%), 20 mg (n = 79; 7.1%), 120–160 mg (n = 23; 2.1%), and 60 mg (n = 14; 1.3%) (Figure 1). The initial mean SD lurasidone daily dose in the analytic cohort was 51.9 mg (23.4 mg), while the daily maintenance dose during 6-month follow-up period was 55.2 mg (22.9 mg) (Figure 2).
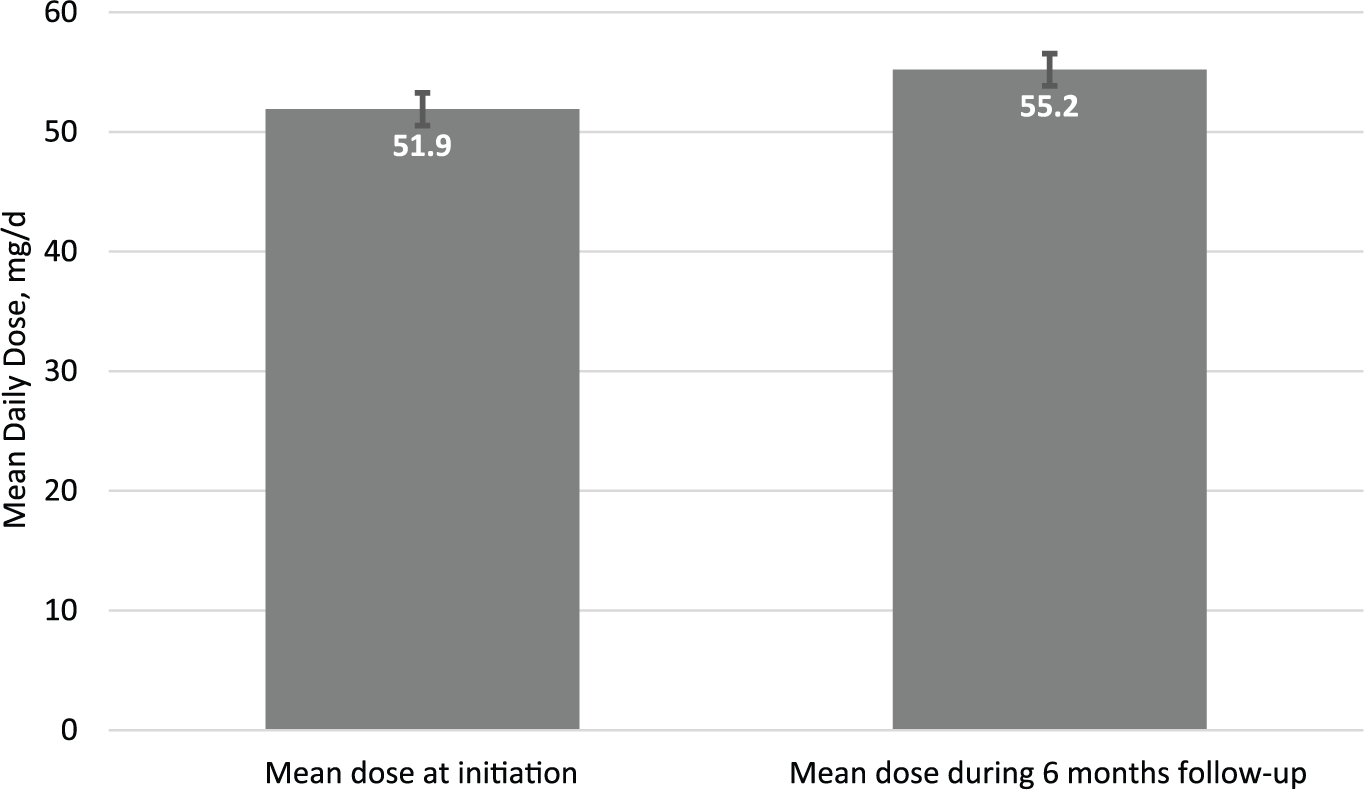
Mean (95% confidence interval) lurasidone dose at initiation and during follow up.
For the comparisons across lurasidone dose groups, patients with an initial lurasidone daily dose of 60 mg were excluded due to the small sample size of the group (n = 14 patients). Bivariate analyses across the remaining initial lurasidone dose groups showed that there were significant differences in geographic region, proportion of patients with obesity, and proportion of patients with prior use of first-generation antipsychotics (p < 0.05) (Table 1). A higher initial lurasidone daily dose was associated with higher likelihood of substance use, abnormal glucose/hyperglycemia, obesity, and prior use of second-generation antipsychotics and first-generation antipsychotics during the pre-index baseline period (p < 0.05). Baseline bipolar disorder diagnosis was not significantly associated with being prescribed a higher initial lurasidone dose.
In the multivariable ordinal logistic regression analyses (Table 2), the odds of having a 20 mg/day increase in initial lurasidone dose was 1.6-times higher for patients with substance use disorder [adjusted odds ratio (AOR) 1.62, 95% confidence interval (CI): 1.16–2.24]. The odds of having a 20 mg/day increase in initial lurasidone dose was 2.6-times higher for patients who had abnormal glucose or hyperglycemia problems (AOR 2.58, 95% CI: 1.15–5.83) and 1.7-times higher for patients with comorbid obesity (AOR 1.65, 95% CI: 1.05–2.60). Additionally, the odds of having a 20 mg/day increase in initial lurasidone dose was 1.3-times higher (AOR 1.34, 95% CI: 1.01–1.78) and 1.8-times higher (AOR 1.83, 95% CI: 1.17–2.86) for patients with prior use of second- and first-generation antipsychotics, respectively. In contrast, age, gender, insurance type, type of bipolar diagnosis, other psychiatric and physical comorbidities, and prior use of mood stabilizers, antidepressants, and psychotherapy were not significantly associated with initial lurasidone dose.
Multivariable ordinal logistic regression of patient characteristics associated with initial lurasidone dose.

CI, confidence interval; HMO, health maintenance organization; OR, odds ratio; POS, point of service; PPO, preferred provider organization.
Note: The ordinal logistic regression included patients with initial daily lurasidone doses of 20, 40, 80, and 120/160 mg. The 14 patients with initial daily lurasidone dose of 60 mg were not included as a dose category due to the limited sample size.
Adjusted for all other variables listed in the table. ORs indicate the odds associated with a one-unit increase in the predictor (e.g. from 20 to 40 or 80 to 120/160), when all other covariates are held constant.
Concomitant psychiatric treatment and adherence
Only 16.2% of patients initiated lurasidone as monotherapy (Table 3). Nearly one-fifth of patients combined lurasidone with mood stabilizers (18.2%), antidepressants (18.6%), or mood stabilizers and antidepressants (19.7%). Approximately 1 in 4 patients used other second-generation antipsychotics in conjunction with lurasidone.
Concomitant prescription bipolar disorder medications at used at lurasidone initiation.

FGA, first-generation antipsychotics; SGA, second-generation antipsychotic.
Lurasidone + 2 or more medication categories were collapsed into ‘other combination’ if their prevalence was <5% of the sample. All lurasidone + 1 category (mood stabilizer, other SGA, FGA, antidepressant) were listed regardless of prevalence.
The mean MPR of lurasidone treatment for the overall sample was 0.53 (SD = 0.34) during the 6-month follow-up period, and 33% of patients had MPR greater than or equal to 0.8 (Table 4). The mean lurasidone MPR varied across the nine subgroups of patients with different concomitant psychiatric treatments. Patients treated with a combination of lurasidone, mood stabilizer, and other second-generation antipsychotics had the highest mean MPR of 0.6, while patients treated with lurasidone and first-generation antipsychotics had the lowest mean MPR of 0.35. The remaining treatment subgroups (i.e. lurasidone and mood stabilizer, lurasidone and antidepressant, lurasidone and mood stabilizer and antidepressant, lurasidone and other second-generation antipsychotics, lurasidone and antidepressant and other second-generation antipsychotics, lurasidone and mood stabilizer and antidepressant and other second-generation antipsychotics, lurasidone and other combinations) had a similar mean MPR, with a range of 0.49–0.58.
MPR of lurasidone treatment for the entire sample and by polypharmacy category.

FGA, first-generation antipsychotics; MPR, medication possession ratio; SD, standard deviation; SGA, second-generation antipsychotic.
Lurasidone + 2 or more medication categories were collapsed into ‘other combination’ if their prevalence was <5% of the sample. All lurasidone + 1 category (mood stabilizer, other SGA, FGA, antidepressant) were listed regardless of prevalence.
Discussion
To the best of our knowledge, this is the first study to describe the real-world clinical use of lurasidone and to examine the association between patient characteristics and lurasidone dosing patterns in patients with bipolar disorder. Overall, nine out of ten patients in this study initiated lurasidone at a daily dosage of 40 mg or 80 mg, which is consistent with lurasidone prescribing information. Mean initial lurasidone daily dose and mean daily maintenance dose were 51.9 mg/day and 55.2 mg/day, respectively.
Lurasidone was approved by the US FDA for the treatment of schizophrenia in 2010, with a recommended dose of 40–80 mg/day. Higher doses of 120 mg and 160 mg/day were later approved in 2012 [Sunovion, 2013]. In 2013, the US FDA approved lurasidone for the treatment of bipolar depression in the dose range of 20–120 mg/day. Close to two-thirds of the bipolar patients in this study initiated lurasidone at 40 mg/day, which may be explained in part by the influence of the initial 40 mg/day recommended starting dose for schizophrenia.
There were no significant differences in demographic and clinical characteristics between patients initiating higher lurasidone doses (120 mg or 160 mg/day) versus patients initiating lower doses (20 mg, 40 mg or 80 mg/day). However, the small sample sizes in the higher dose subgroups may have obscured the detection of any real differences in patient demographic and clinical characteristics.
More than 80% of patients with bipolar disorder were polypharmacy users (defined as having an active prescription for psychiatric medication in addition to lurasidone), which is consistent with prior research [Baek et al. 2014]. Given the complex nature of the disease and the comorbidities present in patients with bipolar disorder, combination treatment regimens are often used. Mood stabilizers were the most commonly used concomitant treatment in our study population.
Patients with abnormal glucose or hyperglycemia, obesity, prior antipsychotic use, and comorbid substance use issues were more frequently prescribed higher initial lurasidone doses in our study. Close to three-quarters of patients with bipolar disorder used other second-generation antipsychotics prior to the initiation of lurasidone. Clinicians may have prescribed lurasidone to patients who had more medical complexities, had failed previous treatments, or had multiple prior pharmacological treatments.
Patients with bipolar disorder treated with lurasidone were found to have high baseline rates of certain comorbid conditions, including heart diseases, diabetes, obesity, thyroid disease, and migraines [Krishnan, 2005; Kupfer, 2005]. A study from Kemp found an association between medical comorbidity and reduced medication treatment responsiveness in patients with bipolar disorder [Kemp, 2014]. Another study by Kemp and colleagues [Kemp et al. 2010] found that for every 1-unit increase in body mass index in patients with bipolar disorder, the likelihood of medication treatment response decreased by 7.5% and that of remission decreased by 7.3%. In the current analysis, patients with abnormal glucose/hyperlipidemia and obesity had significantly greater odds of using a higher initial dose of lurasidone. Polypharmacy, a proxy indicator of disease severity, is associated with more symptomatic bipolar disorder [Goldberg et al. 2009; Weinstock et al. 2014] and high-dose antipsychotic use [Biancosino et al. 2005]. Higher initial lurasidone doses were prescribed to patients with prior use of antipsychotics or substance abuse in the current study, suggesting that clinicians may have prescribed this newly available agent to patients with greater disease complexity.
Medication adherence is a well-known challenge in patients with bipolar disorder [Berger et al. 2012]. Rates of adherence for patients newly initiating lurasidone in this study were comparable to or better than the MPR rates of adherence for patients with bipolar disorder treated with antipsychotics reported in the literature. In a claims database analysis of patients with bipolar I disorder who newly initiated an oral atypical antipsychotic treatment, the mean MPR was 0.21 [Chen et al. 2013]. In other claims analyses among bipolar patients receiving atypical antipsychotic treatment, mean MPRs ranged between 0.42 [Lage and Hassan, 2009] and 0.56 [Stephenson et al. 2012]. Among patients with bipolar disorder who received atypical antipsychotics at hospital discharge, mean MPRs of 0.37 [Berger et al. 2012] and 0.46 [Hassan and Lage, 2009] were reported. Variations of MPR reported in the literature are expected due to differences in study design, sample selection criteria, and sample size. Poor adherence to and high discontinuation rates of bipolar disorder medications are associated with worsened outcomes, such as increased risk of relapse and hospitalization [Svarstad et al. 2001; Weiden et al. 2004; Colom et al. 2005]. To better understand the underlying reasons accounting for the observed MPR and to identify ways to improve adherence in patients with bipolar disorder, future research is needed.
This study has several limitations. The study analysis period was prior to the US FDA approval of lurasidone for the treatment of bipolar depression in the US (June 2013). It is possible that studies examining real-world prescribing of lurasidone in patients with bipolar depression conducted after the approval for the indication may yield different results. However, recent market utilization estimates suggest that the lurasidone dose per day remains the same, with a significant proportion of patients starting on 40 mg per day [IMS Health, 2015]. As with other retrospective analyses using health claims databases, data generated primarily for administrative purposes lack detailed clinical information and may have insufficient validity and accuracy. Information about the prescribers’ clinical rationale for prescribing and dosing lurasidone were not available in the administrative claims data. Pharmacy claims document the medications that have been prescribed and filled, but cannot validate whether prescriptions are used as prescribed by patients. The database may not capture all medications that patients may take for bipolar depression. For instance, the database omits medications prescribed during hospitalization, thus underestimating the medications prescribed to hospitalized patients with bipolar disorder. Coding errors and omissions in the database also may cause misclassification. In spite of these limitations, these analyses provide potentially useful insight into how a newly-available medication may be used by clinicians treating patients with bipolar disorder in standard clinical settings.
Conclusion
In this analysis of dosing patterns among patients with bipolar disorder receiving lurasidone, a majority of patients started lurasidone at 40 mg or 80 mg/day. Higher dosages of lurasidone may have been prescribed to bipolar patients with more complex illness, greater disease severity, and concurrent comorbidities. About 85% of lurasidone patients with bipolar disorder in this sample used concomitant medications defined here as mood stabilizers, antidepressants, and antipsychotics. Medication adherence among the patients treated with lurasidone was observed to be comparable to or better than other oral antipsychotics as reported in the published literature. Further studies are needed to understand the decision making process in clinicians’ choice of treatment and dose selection for newly available pharmacotherapies.
Footnotes
Appendix: key psychiatric and physical comorbid conditions (ICD-9-cm diagnoses),and pre-index and concomitant psychiatric drugs
Psychiatric comorbidities measured included the following:
Physical comorbidities included the following:
Pre-index and concomitant use of psychiatric drugs was categorized as follows:
Acknowledgements
The authors would like to acknowledge the support of Xin Gao (Pharmerit International, Bethesda, MD, USA) in the design and conceptualization of the study and Sonam Mehta (Pharmerit International) for help in the design, analysis, and interpretation of the study.
Funding
This work was supported by Sunovion Pharmaceuticals Inc (Marlborough, MA, USA).
Conflict of interest statement
This study was funded by Sunovion Pharmaceuticals Inc. Martha Sajatovic is an employee of Case Western Reserve University (Cleveland, OH, USA) and of University Hospitals Cleveland Medical Center (Cleveland, OH, USA). Daisy Ng-Mak, Krithika Rajagopalan, and Antony Loebel are employees of Sunovion Pharmaceuticals Inc (Marlborough, MA, USA). Caitlyn Solem and Fang-Ju Lin are employees of Pharmerit International (Bethesda, MD, USA) and were paid consultants to Sunovion (Marlborough, MA, USA) in the development and execution of this study.
