Abstract
Objectives:
Sertindole is a nonsedating atypical antipsychotic drug with low propensity to cause extrapyramidal side effects but it has been associated with a 20 ms QTc prolongation and increased risk of cardiac events. It is uncertain whether this drug-induced increase in cardiac risk might also be revealed by dynamic measures of the QT interval such as the ratio of QT variability to heart rate variability (variability ratio [VR]). The aim of this study was to investigate the effect of sertindole on QT dynamics.
Methods:
QTc and the VR were assessed in an observational study using 24-hour Holter monitoring at baseline and after 3 weeks of treatment with sertindole 16 mg. The VR was calculated by dividing the standard deviation of QT intervals with the standard deviation of heart rates. Outcome measures were compared using paired
Results:
A total of 18 patients participated in the study, two were excluded from further analysis due to low amplitude of the T-wave. When patients were shifted to sertindole, the VR increased from 0.192 (SD 0.045) to 0.223 (SD 0.061),
Conclusion:
Sertindole was associated with 19 ms QTc prolongation and an increased ratio of QT variability to heart rate variability. Both measures may contribute to the increased cardiovascular mortality found with sertindole.
Keywords
Introduction
Treatment with antipsychotics has been associated with a two-fold risk of sudden cardiac death, partly explained by the propensity of antipsychotics to alter cardiac repolarization [Ray et al. 2009]. This effect is exerted through inhibition of the slow delayed rectifying IKr potassium channel (HERG) which presents as QT interval prolongation in the electrocardiogram (ECG) [Nielsen et al. 2011]. The QTc interval is widely used as a surrogate marker of drug-induced Torsade de Pointes arrhythmia (TdP), but the validity of QTc can vary considerably due to measurement inaccuracies, errors when correcting for heart rate and false-positive findings, i.e. not all drugs prolonging the QT interval increase the risk for TdP [Nielsen et al. 2011]. For these reasons a variety of new surrogate markers have been investigated, e.g. Tpeak−Tend interval, QT dispersion, T-wave morphology [Graff et al. 2009a, 2009b] and QT variability.
QT variability is based on the fact that cardiac repolarization is a dynamic phenomenon which varies between each beat. The small variations reflect temporal heterogeneity of repolarization, likely due to changes in intracellular calcium handling, which may lead to reentry and ventricular arrhythmias [Jensen et al. 2005]. QT variability have only sparsely been used to estimate the arrhythmogenic potential of drugs, but antiarrhythmic drugs with potassium channel blocking abilities and risk for proarrhythmias have been shown to increase QT variability. Nortriptyline, a tricyclic antidepressant was found to increase QT interval variability whereas paroxetine did not affect QT variability in patients with panic disorder [Yeragani et al. 2000]. In a recent study nortriptyline has been associated with a more than five-fold increased risk for cardiac arrest compared with the background population, whereas paroxetine did not have any increased risk [Weeke et al. 2012]. Unmedicated schizophrenia and psychosis has been associated with increased QT variability and might indicate high sympathetic activity in these patients [Bar et al. 2007; Jindal et al. 2009]. Estimating sympathetic tonus may not only be attributed to cardiac safety but has also been associated with response to antipsychotic drugs and outcome of schizophrenia [Boden et al. 2011].
The variability ratio (VR) is a recent variant of QT dynamics which has been suggested as a surrogate marker for sudden cardiac death [Jensen et al. 2005]. VR is determined from 24-hour Holter recordings and is defined as the standard deviation of all of the QT intervals divided by the standard deviations of the RR intervals.
Sertindole is an atypical antipsychotic drug introduced in 1997 and voluntarily withdrawn in 1998 due to suspected increased risk of sudden cardiac death. Several reviews have investigated the efficacy and safety of sertindole [Azorin et al. 2010; Cincotta and Rodefer, 2010; Muscatello et al. 2010; Karamatskos et al. 2012; Pae, 2013; Muscatello et al. 2014]. One of the advantages of sertindole compared with other antipsychotics seem to be the superior effect on negative symptoms [Karamatskos et al. 2012; Muscatello et al. 2014]. Besides the 20 ms QTc prolongation during treatment with sertindole, the drug has also been reported to worsen T-wave morphology [Nielsen et al. 2009]. In contrast, sertindole does not increase the heart rate variability due to its lack of affinity for cholinergic receptors [Agelink et al. 2001]. Neither has sertindole demonstrated any effect on QT dispersion or Tpeak−Tend [Nielsen et al. 2010]. Despite these contradictory findings, a large surveillance study, the Sertindole Cohort Prospective Study (SCoP) including 9858 patients, found an increased cardiac mortality of sertindole compared with risperidone but no increased overall mortality for sertindole [Thomas et al. 2010]. Sertindole remains a second-line option and QTc assessment is warranted before initiating sertindole and after 3 weeks and every 3 months [Pae, 2013].
Since treatment with sertindole has a confirmed increased risk of cardiac mortality, we decided to investigate its effect on VR to explore more about the usefulness of this new surrogate marker for drug-induced repolarization alterations.
Methods
Patients switching to sertindole as part of their clinical routine treatment were asked to participate in this observational study estimating sertindole’s effect on arrhythmic surrogate markers from 24-hour Holter monitoring. Written informed consent was obtained and the study was approved by the Regional North Jutland Ethics Boards and the Danish Data Protection Agency.
Patients were recruited from Aalborg, Brønderslev and Aarhus Psychiatric hospitals between August 2008 and April 2010. Patients were maintained on baseline medications during treatment with sertindole.
Three-channel 24-hour Holter monitoring was performed at baseline and after 3 weeks of treatment with 16 mg sertindole using a digital Reynold Lifecard CF recorder (Delmar Avionics, Irvine, CA, USA). Data was transferred to a DelMar Impressario Workstation (Delmar Avionics Irvine, CA, USA), and the recording was analyzed semi-automatically by an experienced cardiologist ensuring correct beat labeling and identification of artifacts. The cardiologist used on-screen calipers to mark the end of the T-wave, in cases where it was necessary to manually adjust automatic QT intervals to insure proper measurements. All measurements of QT and RR intervals were transferred to a PC, and ectopic beats of supraventricular and ventricular origin were removed. As described earlier [Jensen et al. 2004] every normal beat was compared with the mean of the two previous and the two succeeding beats and if the normal beat varied by more than 25% of this mean, the normal beat was considered a premature beat and was discarded.
The VR, which can be considered a measure of autonomic disconcordance between the sinus node and the ventricular myocardium, was calculated as
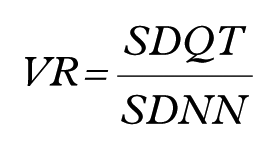
where SDNN was calculated as the standard deviation of the RR intervals of all normal sinus beats and SDQT was calculated as the standard deviation of the QT intervals of all normal sinus beats.
QTc was calculated as the average of all QT intervals corrected by Bazett’s formula (QTcB) [Bazett, 1920]. Outcome parameters were normally distributed and compared using a paired
Results
A total of 18 patients were switched to sertindole and underwent Holter monitoring. Two of them were excluded from further data analysis due to poor quality of the recordings or low amplitude of the T-wave. A total of 16 subjects, 9 females (56.3%) and 7 males (43.8%) were included in the analyses. Mean age was 39.1 years (SD 10.9). Mean body weight was 85.3 kg (SD 18.3) and body mass index was 29.1 kg/m2 (SD 6.7). Baseline medication was: olanzapine 4, risperidone 5, paliperidone 1, aripiprazole 2, quetiapine 1, and perphenazine 1. Concomitant medications were: selective serotonin reuptake inhibitors (SSRI) 6, selective serotonin and norepinephrine reuptake inhibitors (SNRI) 6, benzodiazepines 3 and anticholinergics 5.
None of the patients experienced dizziness, palpitations or syncopes during treatment with sertindole. A QTc prolongation above 60 ms was not observed in any patient. Neither was a QTcB or QTcF interval exceeding 500 ms. As shown in Table 1, the QTc interval and VR increased, whereas heart rate variability (SDNN), TpTe interval and QT variability (SDQT) remained unchanged. A T-wave amplitude reduction were seen, which did not reach statistical significance
Outcome measures.

Discussion
To the best of the authors’ knowledge, this is the first study investigating QT dynamics during treatment with an antipsychotic drug. The increased VR supports that sertindole has proarrhythmic ability in accordance with the findings from the ScOP study where the arrhythmogenic potentials of sertindole was confirmed [Thomas et al. 2010]. The Tpeak−Tend interval reflects regional differences in cardiac repolarization [Antzelevitch, 2001] and has been related to arrhythmic risk and mortality [Haarmark et al. 2009]. In our study the Tpeak−Tend interval did not change with sertindole in accordance with another study [Nielsen et al. 2010]. Sertindole is a known HERG potassium channel blocker of the IKr current, but in patients with inherited dysfunction of the HERG channel (Long QT syndrome) not only the QT interval but also the Tpeak−Tend interval are prolonged [Kanters et al. 2008]. It has recently been shown that other genetic factors are responsible for the Tpeak−Tend interval than the QT interval [Haarmark et al. 2011], which may explain why the HERG blocker sertindole mainly prolonged the QT interval before the peak of the T-wave. However, in patients with documented drug-induced TdP ventricular tachycardia, the Tpeak−Tend interval is prolonged, but sertindole is generally believed to have a low proarrhythmic risk. Since all of our patients only had mild QT prolongation and did not experience any syncopes or arrhythmias, the Tpeak−Tend interval may identify a subset of patients at higher risk.
Treatment with sertindole did not affect heart rate variability (SDNN) which is in accordance with previous findings [Agelink et al. 2001]. Although, SDNN did not change during the study, baseline values were lower than results from a study including healthy volunteers which was expected because of the psychiatric disease itself and the concomitant medications [Jensen et al. 2004]. Reduced heart rate variability has been associated with several antipsychotic drugs and is considered to be due to reduced parasympathetic tone by inhibiting cholinergic receptors [Mueck-Weymann et al. 2002].
QT dynamics in general has only been used sparsely for antipsychotic drugs. QT interval variability has only been measured with olanzapine treatment where no effect was found [Bar et al. 2008]. This results is in accordance with the Ziprasidone Observational Study of Cardiac Outcomes (ZODIAC) where both olanzapine and ziprasidone was associated with a low 1-year mortality rate [Strom et al. 2011].
An important point to remember is the use of the right correction formula as illustrated by the 20 ms difference between QTcB and QTcF. This is especially important in patients treated with clozapine since sinus tachycardia is frequent but already at a heart rate of 80 the difference between the two correction formulas is of clinical importance [Nielsen et al. 2011; Nielsen et al. 2012; Nielsen, 2012].
The present study should be interpreted within its limitations. Patients were already on antipsychotic drug at baseline, baseline values of VR and QTc may be overestimated, leading to an underestimation of the effects of sertindole on QTc and VR. All other medications were kept stable during the study. Improvement of mental state during treatment with sertindole may have influenced SDNN but unfortunately psychopathology rating was not performed. Plasma levels of sertindole were not available. Neither was electrolytes but all patients were considered cardiovascular healthy before commencing to sertindole. Power analysis was not done because this study was considered an explorative pilot study.
The clinical implication of VR is currently limited because of the small sample size of this study and the fact that many patients may not be eligible for 24-hour Holter recording. Finally the cost is also a limiting factor. However, the VR may play a role in drug development, because the current thorough QT study (TQT) used to detect cardiac arrhythmic potential of a drug is both expensive and its validity questionable [Bouvy et al. 2012]. VR may have a role as a surrogate marker for drug-induced arrhythmias but further large-scale cardiac safety studies are warranted. Ideally, these studies should have TdP and sudden cardiac death as outcomes but this is complicated by the low incidence of these events and consequently the extremely large sample size warranted to achieve substantial statistical power.
Although sertindole was associated with QTc prolongation and increased QT variability, the overall mortality may not be increased in this group of patients because of important factors such as rate of suicide or metabolic complications [Correll and Nielsen, 2010; Nielsen, 2011] as the SCoP study revealed [Thomas et al. 2010]. Clinicians should still focus on the QTc interval and especially in the presence of additional risk factors, such as hypokalemia [Girardin et al. 2013].
Conclusion
Sertindole prolonged the QTc interval and increased the ratio of QT variability to heart rate variability thus supporting the use of VR as a new marker of cardiac repolarization effects. Large-scale studies comparing newer markers of cardiac effects are warranted to determine the usefulness of such markers.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
J. Nielsen has received research grants from H. Lundbeck, Pfizer and Chempaq for clinical trials and received speaking fees from Bristol-Myers Squibb, Astra Zeneca, Hemocue and Janssen & Cilag. Other authors report no conflict of interest.
