Abstract
Previous research has shown that food avoidance can be learned via classical and operant conditioning. This leads to the question of whether learning deficits could contribute to the harmful food avoidance seen in individuals with anorexia nervosa. Accordingly, we tested whether healthy women with increased levels of anorexia nervosa symptoms and characteristics show learning abnormalities related to the acquisition and extinction of food avoidance behaviors. Data from a previous experiment (Spix, Schutzeichel, et al., 2023) was used and supplemented with new questionnaire data. Based on participants’ levels of anorexia nervosa symptoms and characteristics, we subtyped an analogue and a healthy group and compared their performance on a food avoidance learning task. We assessed the frequency of avoidance responses, as well as relief, frustration, eating desires, fear, and liking for the conditioned stimulus (CS) associated with food intake. The analogue group showed more unnecessary food avoidance behaviors, a greater relief about the omission of the food, and a faster and more persistent reduction in eating desires than the healthy group. There were no differences between groups in frustration, fear, and liking. Learning abnormalities might contribute to the development and maintenance of food avoidance in individuals with anorexia nervosa.
Keywords
Restrictive eating, that is, the avoidance of certain foods or calorie intake in general, presents a core symptom of anorexia nervosa (American Psychiatric Association, 2013; Schaumberg et al., 2021; Steinglass et al., 2015). Excessive food/calorie avoidance negatively impacts patients’ quality of life, reduces their opportunities for new experiences, and causes severe physical consequences, such as underweight, malnutrition, and bradycardia (American Psychiatric Association, 2013). Even though the reduction of food avoidance is one of the primary goals of many anorexia nervosa treatments (National Institute for Health and Care Excellence, 2020), behaviors often persist throughout treatment or return afterward (Walsh, 2011). Thus, a more thorough understanding of the mechanisms underlying food avoidance is direly needed to improve treatment success (Glashouwer et al., 2020; Jansen, 2016).
To understand how inherently appetitive stimuli, such as food (Simon et al., 2015), can come to trigger persistent avoidance behaviors, recent theoretical models point to an important role of classical and operant conditioning (Christian & Levinson, 2022; Garcia-Burgos et al., 2019, 2023; Melles et al., 2021; Schaumberg et al., 2021). During classical conditioning, food and eating-related stimuli (conditioned stimuli; CSs) become associated with an aversive outcome (unconditioned stimulus; US), such as (uncontrollable) weight gain, social judgment, or gastrointestinal malaise (Levinson et al., 2019; Levinson & Williams, 2020; Melles & Jansen, 2023). Consequently, food and eating-related stimuli start to trigger aversive conditioned responses (CRs), such as heightened autonomic arousal, fear and/or disgust, and avoidance tendencies (Hildebrandt et al., 2015; Melles et al., 2021; Spix, Schutzeichel, et al., 2023). During operant conditioning, individuals learn about the consequences of their avoidance behaviors (Krypotos et al., 2015; LeDoux et al., 2017): if food restriction leads to the omission of the expected aversive outcome and/or reduces anxiety and disgust, individuals are more likely to avoid food intake again in the future (negative reinforcement; De Houwer & Hughes, 2020). In other words, associations between specific actions and safety outcomes are learned, and these so-called action → safety associations guide future behavior (De Kleine et al., 2023). Additionally, food avoidance can be reinforced by its pleasant consequences, such as weight loss, compliments by others, or elevated feelings of control and self-esteem (Marzola et al., 2015; Selby & Coniglio, 2020; Walsh, 2013). In line with this model, we showed in a previous study that healthy women can acquire persistent food avoidance behaviors via classical and operant conditioning (Spix, Schutzeichel, et al., 2023).
Considering food avoidance as a learned behavior raises the question of whether individual differences in learning can facilitate the extreme food restriction seen in individuals with anorexia nervosa. Conditioning research has identified inter-individual differences in maladaptive avoidance behaviors (for an overview, see Wong et al., 2023): These may include the excessive, unnecessary, and persistent execution of avoidance behaviors, that is, deficits in the extinction of avoidance. Accordingly, individuals with or at risk of developing anorexia nervosa might (a) unnecessarily avoid stimuli not associated with aversive consequences, (b) excessively avoid food and eating-related stimuli disproportionate to the anticipated consequences, and (c) continue to avoid food and eating-related stimuli even though positive and aversive consequences of avoidance are no longer present (extinction deficits). Once in place, these maladaptive avoidance behaviors potentially exacerbate fear and prevent individuals from testing their threat beliefs (Lovibond et al., 2009; Pittig et al., 2020; Spix, Melles, et al., 2023; Spix, Schutzeichel, et al., 2023). Thereby, maladaptive food avoidance can contribute to the development and maintenance of eating disorders—making it of crucial importance to understand what mechanisms reinforce and facilitate these behaviors.
Potential contributors to maladaptive avoidance are relief, defined as the pleasant feeling after the omission of an anticipated aversive event (Leng et al., 2024) and frustration, defined as the negative emotional reaction to the omission of a rewarding event (Abler et al., 2005; Blechert et al., 2014). While pleasant feelings of relief act as a reinforcer of food avoidance (De Kleine et al., 2023; Leng et al., 2022, 2024; Papalini et al., 2023; Vervliet & Indekeu, 2015), aversive feelings of frustration reduce the likelihood that avoidance behaviors are executed again in the future (positive punishment; De Houwer & Hughes, 2020). Thereby, strong relief and little frustration after food avoidance could provide a potent motivation for further avoidance behaviors irrespective of the associated threat (De Kleine et al., 2023; Vandael et al., 2023). Moreover, impairments in action → safety learning might contribute to excessive food avoidance: if individuals only acquire a weak action → safety association and consequently, do not trust that their avoidance behavior cancels an expected aversive outcome, they will execute the behavior more rigidly to ensure that aversive outcomes are really prevented (Cobos et al., 2022; De Kleine et al., 2023; Flores et al., 2018). Accordingly, if individuals with anorexia nervosa doubt that restrictive eating really prevents weight gain, they may use additional avoidance behaviors, such as excessive exercise, to achieve certainty. Lastly, individual differences in fear learning and appetitive conditioning can contribute to maladaptive food avoidance: if individuals acquire a strong fear response to food and eating-related stimuli, avoiding food intake will have a greater anxiolytic effect, potentially reinforcing unnecessary or excessive avoidance behaviors (Murray et al., 2016; Strober, 2004); if individuals reduce more quickly in their eating desires and their liking for eating-related stimuli, they will be less motivated to test whether eating is still followed by negative consequences, possibly contributing to deficits in the extinction of food avoidance (Georgii et al., 2020; Murray et al., 2022). Thus, various learning abnormalities could contribute to unnecessary, excessive, and overly persistent food avoidance.
Experimental research with analogue samples, consisting of healthy individuals experiencing sub-clinical symptoms or showing other disorder-related characteristics (Abramowitz et al., 2014), presents a suitable way to study learning abnormalities related to food avoidance. In contrast to patients with anorexia nervosa, healthy individuals do usually not avoid high-calorie food intake (Steinglass et al., 2015) and, therefore, can acquire food avoidance behaviors through an experimental manipulation (Glashouwer et al., 2020; Jansen, 2016). Unfortunately, clear guidelines or best practices to identify analogue samples for anorexia nervosa are missing. Solely relying on higher levels of eating, weight, or shape concerns or a stronger drive for thinness and food restriction might be insufficient to divide individuals into an analogue and a healthy group (Papalini et al., 2021). Instead, including other characteristics associated with anorexia nervosa, such as maladaptive perfectionism and behavioral inhibition (Dahlenburg et al., 2019; Fairburn et al., 2003; Frank et al., 2012, 2013, 2018; Halmi et al., 2012; Harrison et al., 2010; Hilbert et al., 2014; Jonker et al., 2020, 2022; Miles et al., 2023; Ralph-Nearman et al., 2024; Welch et al., 2020; Wilson et al., 2019), should allow a more fine-grained identification of healthy individuals with anorexia nervosa-like features and, thus, analogue samples. Statistical methods, such as cluster analysis, can aid researchers in the selection of appropriate analogue samples by grouping individuals based on their scores across multiple measures (e.g., symptoms and characteristics associated with anorexia nervosa).
So far, experimental studies investigating individual differences in maladaptive food avoidance and potential reinforcing mechanisms are largely missing (for an exception, see Hildebrandt et al., 2015; Olatunji, 2020). To address this gap, the present study tests whether an analogue sample including healthy women with increased levels of eating disorder symptoms, maladaptive perfectionism, and behavioral inhibition displays maladaptive food avoidance behaviors, as well as abnormal relief, frustration, fear learning, and appetitive conditioning in a food avoidance learning task (Spix, Schutzeichel, et al., 2023). Compared with healthy individuals, the analogue sample is expected to show the following: (1) More unnecessary avoidance behaviors and more persistent food avoidance during extinction. (2) Impairments in action → safety learning, which is reflected in higher expectancies to receive food after engaging in food avoidance behaviors. (3) Greater relief and less frustration after food omission during avoidance acquisition and extinction. (4) A faster and more extreme reduction in eating desires and liking for the CS associated with food intake during avoidance acquisition and a more persistent reduction during extinction. (5) A faster and more extreme increase in fear for the CS associated with food intake during avoidance acquisition and a more persistent increase during extinction.
Methods
Study design
In the present study, data from a previous experiment (Spix, Schutzeichel, et al., 2023) was used and supplemented with new questionnaire data. In Spix, Schutzeichel, et al. (2023), participants could avoid a small sip of milkshake by pressing a button on the computer screen. One half of the participants was rewarded for food avoidance and punished for food intake (experimental condition), while the other half never received any consequences irrespective of their avoidance response (control condition). Additionally, all participants filled in some questionnaires measuring eating disorder symptoms and characteristics associated with anorexia nervosa. This questionnaire data has not been analyzed and published before. Using cluster analyses, participants were divided into a healthy and an analogue group based on their scores on the EDE-Q, DT, BIS, and SAPS (reflecting eating disorder symptoms, behavioral inhibition, and maladaptive perfectionism, see Questionnaires). To test the current hypotheses, classical and operant conditioning in the experimental condition was compared between the analogue and healthy group.
Participants
Participant characteristics for the analogue and healthy group in the experimental condition.

Questionnaires
Eating Disorder Examination Questionnaire
The EDE-Q (Fairburn & Beglin, 2008) assesses eating disorder symptoms over the course of the past 28 days. The EDE-Q consists of 28 items and covers four subscales: restraint, shape concern, weight concern, and eating concern. Items are answered on a seven-point Likert scale ranging from 0 = no days/not at all to 6 = every day/markedly. Subscale scores are calculated by averaging across the respective items; a global score is calculated by averaging across the four subscales. Higher scores reflect higher levels of eating disorder symptoms. The EDE-Q subscale- and global scores showed an adequate internal consistency with α’s > .79.
Eating Disorder Inventory II—Drive for thinness
The DT subscale (Garner, 1991) measures individuals’ desire to be thin. The scale consists of seven items that are answered on a six-point Likert scale with 1 = never to 6 = always. A total score is calculated by averaging across all items. Higher scores indicate a greater drive for thinness. The drive for thinness subscale had a good internal consistency with α = .93.
Short Form of the Revised Almost Perfect Scale
The SAPS (Rice et al., 2014) measures two aspects of perfectionism: one subscale assesses whether individuals set high standards for themselves (SAPS Standard), while the other captures to what extent individuals experience a mismatch between their standards and actual performance (SAPS Discrepancy). The eight items of the SAPS are answered on a seven-point Likert scale ranging from 1 = strongly disagree to 7 = strongly agree. Subscale scores are calculated by summing the respective items together. Higher scores reflect a greater level of perfectionism. Both SAPS scales possessed a good internal consistency with α’s > .84.
Behavioral inhibition system/behavioral approach system scales
The BIS/BAS (Carver & White, 1994) measures sensitivity towards reward and punishment. It is based on Gray’s reinforcement sensitivity theory (Pickering et al., 1995; Pickering & Gray, 2001): human behavior is guided by a behavioral activation system, which is responsible for the motivation to follow goals and to find new rewards and a behavioral inhibition system, which regulates the responses to aversive experiences and resolves approach-avoidance conflicts. The 20-item BIS/BAS comprises four subscales: BAS – Drive, BAS – Reward Responsiveness, BAS – Fun, and BIS. Items were scored on a four-point Likert scale (1 = very false for me to 4 = very true for me). Subscale scores are calculated by summing the respective items together. Higher scores indicate a greater activity of the behavioral activation/behavioral inhibition system. The BIS (α = .71) showed a good internal consistency, and the BAS sub-scales had adequate internal consistency (α’s > .52).
Participants also completed the Depression Anxiety Stress Scales 21 (Lovibond et al., 1995), the Intolerance of Uncertainty Scale (Carleton et al., 2007), the Distress Tolerance Scale (Simons & Gaher, 2005), and the Disgust Propensity and Sensitivity Scale-Revised (Van Overveld et al., 2006).
Stimuli during the food avoidance learning task
A small sip of milkshake was used as the unconditioned stimulus (US). Two geometrical shapes (circle and triangle) served as the conditioned stimuli: one geometrical shape was always followed by the milkshake (CS+; 100 % reinforcement rate), while the other was never paired with the US (CS-). The allocation of geometrical shapes to CSs was counterbalanced across participants.
As a reward for avoiding the milkshake, the picture of a 50-cent coin and the text “You won 50 cents” were presented on the computer screen; as a punishment for not avoiding the milkshake, participants heard a 2-second-long aversive scream (92 dB) via headphones.
When participants could choose whether they wanted to receive the milkshake or not, the labels “Avoid” and “Not avoid” appeared in the two response boxes in the left and right corner of the screen. The location of the response labels was counterbalanced across participants.
Measures during the food avoidance learning task
Food avoidance was operationalized as (a) the actual avoidance response (i.e., clicking on the “Avoid” or “Not Avoid” button and (b) mouse movements while executing the avoidance response. Mouse movements can inform about indecisiveness and ambivalence during a choice, with straighter movements reflecting greater decisiveness and less ambivalence (Pittig & Scherbaum, 2020). On every trial without the US (CS- trials or CS+ trials with an avoidance response), we also measured how relieved and frustrated participants felt about not receiving the milkshake (How [relieved; frustrated] are you that you did not receive the milkshake?). Ratings were made on visual analogue scales (VASs) ranging from 0 = Not [relieved/frustrated] at all, and 100 = Very [relieved/frustrated]. To capture changes in appetitive and aversive conditioned responses, we assessed participants’ expectancy to receive the milkshake, fearfulness, and desire to drink the milkshake when seeing the CSs, using VASs ranging from 0 to 100. Several times throughout the conditioning task, participants also rated the pleasantness of the CS+ and CS- and their liking of the milkshake on a seven-point Likert scale. To check whether the reward and punishment were perceived as intended, participants indicated how much they liked hearing the loud scream and winning the monetary reward on a seven-point Likert scale at the end of the conditioning task. The assessment time points for each outcome measure are presented in Figure 1. Measurement time points across the avoidance and extinction phases CS+ and CS- trial. Note. T = extinction test phases; A = avoidance behavior; M = mouse trajectory data; U = US-expectancy; R = relief; F = frustration; V = CS valence; L = milkshake liking. Fearfulness and eating desires were measured in the middle of the avoidance and extinction phase. It was determined randomly whether these ratings took place on the 4th or 5th trial of the respective phase; these measurements are written in italics. The gray background indicates that measurements were obtained between trials.
Procedure
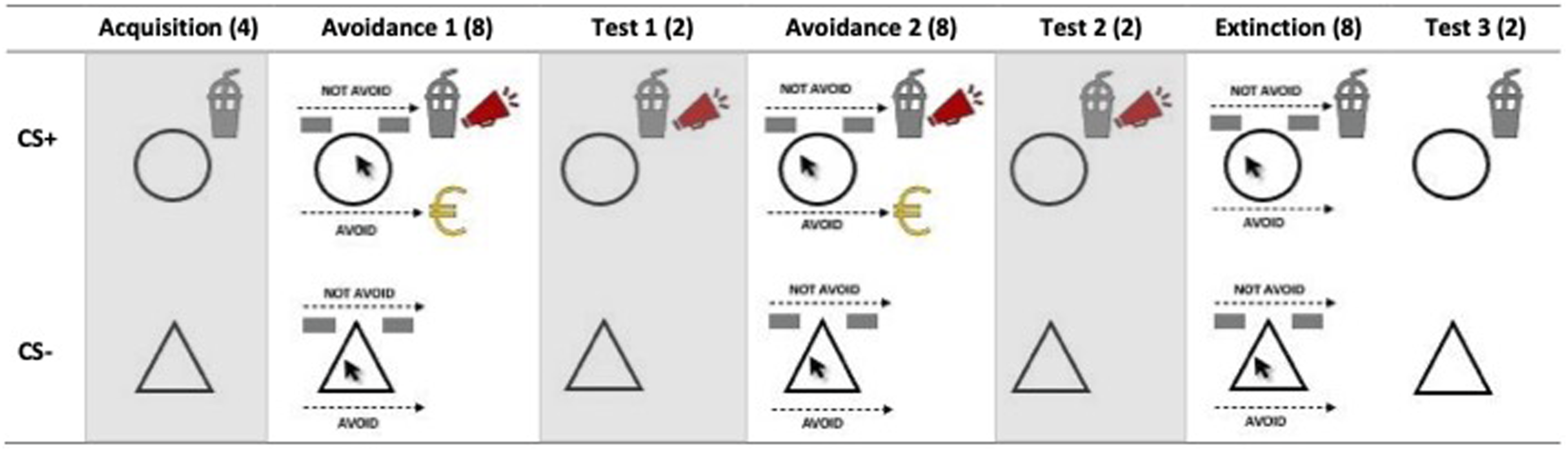
The study was approved by the Ethics Review Committee Psychology and Neuroscience at Maastricht University (approval number: ERCPN-OZL_234_27_02_2021). Participants provided written informed consent and then completed the food avoidance learning task. During the acquisition phase (4 CS+ and 4 CS- trials), participants learned about the relationship between the two geometrical shapes and the delivery of the milkshake. Then, participants entered the avoidance learning phase, which consisted of a first and second part (each with 8 CS+ and 8 CS- trials). Here, they could decide whether they wanted to receive the milkshake or not by clicking on the respective response button (“Avoid” vs. “Not avoid”) in the right or left corner of the computer screen. When avoiding the milkshake, the experimental group received a small monetary reward; when not avoiding the milkshake, they heard the aversive scream. The timeline of an avoidance trial is presented in Figure 2. At the end of both avoidance learning phases, participants entered a test phase (2 CS+ and 2 CS- trials), in which they could not avoid the milkshake and the experimental group was punished for food intake. The avoidance test phases are not of interest for the present study and, thus, are not considered in the later analyses. In the extinction phase (8 CS+ and 8 CS- trials) the consequences for (not) avoiding the milkshake stopped, while executing the avoidance response was still possible; trials followed a similar course as in the avoidance learning phases. In the following extinction test phase reward and punishment were again not delivered, but the avoidance response was not possible anymore. This phase was included to disentangle whether changes in conditioned responses over the course of the extinction phase were due to extinction learning or using the avoidance response, and thus to better identify potential extinction deficits. To answer the hypotheses of the present study, we used the avoidance learning, extinction, and extinction test phase (see Figure 3). After the food avoidance learning task, all participants filled in the questionnaires on the computer. Lastly, the experimenter measured their height and weight and handed out the reimbursements. A more detailed description of the food avoidance learning task and the general procedure can be found in Spix, Schutzeichel, et al., (2023). Timeline of trials in the avoidance learning phase. Note. On certain trials (see Figure 1), eating desires and fearfulness were measured after US-expectancy ratings. Schema of the food avoidance learning task in the experimental condition. Note. (…) = Number of trials per CS; gray background = phase was not included in the analyses of the present study.
Statistical analysis
We used (generalized) linear mixed models—based on the lme4 package (Bates et al., 2015)—to compare data from the food avoidance learning task between individuals with higher or lower anorexia nervosa-like features: For dichotomous avoidance responses (Avoid vs. Not avoid), we ran generalized linear mixed model analyses (GLMMs); for the mouse movement and self-report data (relief, frustration, US-expectancy, fear, desire, and CS valence), we computed linear mixed models (LMMs). For the analyses of the mouse movement data, we calculated the maximum absolute deviation (MAD; Kieslich et al., 2019) from the straight line between the starting position in the middle of the computer screen and the selected response button in the corner of the screen for each CS+ trial. A greater MAD reflected greater indecisiveness and ambivalence when (not) avoiding the milkshake (Kieslich et al., 2019). LMMs with US expectancy as the dependent variable violated assumptions of homogeneity and normality. To resolve this, we turned the continuous expectancy ratings into an ordinal variable with five levels (0–20, 21–40, 41–60, 61–80, and 81–100) and then analyzed the ordinal data using cumulative link mixed models (CLMMs). The phases of the conditioning task were analyzed separately. For food avoidance behaviors, and relief, frustration, and US-expectancy ratings, we looked at the avoidance learning and extinction phases; for eating desire, fear, and the CS valence ratings we additionally looked at the extinction test phase. Group (healthy vs. analogue), CS (CS- vs. CS+), Trial (depending on the dependent variable and phase of the food avoidance learning task), and their interactions were included in the model as fixed effects. Models with US-expectancy and mouse movements as dependent variables additionally included participants’ Avoidance response (Not avoid vs. Avoid) and its interactions with the other predictors as fixed effects. A random intercept per participant was included in all models, while random slopes for the predictors (Group, Trial, CS, and avoidance response) were only added when they improved model fit—reflected in a significantly lower loglikelihood in the likelihood ratio test (LRT). An overview of the estimated models per dependent variable and learning phase can be found in Supplemental material 1. The significance of fixed effects was assessed with the F-statistic for LMMs and with Wald’s z for GLMMs and CLMMs. Findings with a p-value lower than .05 were considered significant. Significant findings were followed by post hoc comparisons, and we controlled for multiple comparisons with Tukey’s HSD. The analysis of the data from the food avoidance learning task was run in R (Version 4.1.2).
Results
Manipulation check
The two groups did not differ in their liking of the milkshake (analogue: M = 2.5, SD = 0.7; healthy: M = 2.8, SD = 0.4; t(43) = −1.40, p = .170), the aversive scream (analogue: M = −2.6, SD = 1.3; healthy: M = −2.8, SD = 0.4; t(27.16) = 0.58, p = .563), and the monetary reward (analogue: M = 1.8, SD = 1.7; healthy: M = 2.2, SD = 1.2; t(43) = −0.90, p = .371).
Food avoidance learning task
The frequency of avoidance responses, and the level of US expectancy, relief, frustration, eating desires, and fear across the avoidance learning and extinction phases are depicted in Figure 4. (Standardized) regression coefficients per model are presented in Supplemental material 2. Frequency of the avoidance response (first panel) and levels of US-expectancy ratings (second panel), relief ratings (third panel), frustration ratings (fourth panel), desire ratings (fifth panel), and fear ratings (sixth panel) across the food avoidance learning task for the analogue (n = 23) and healthy (n = 22) group.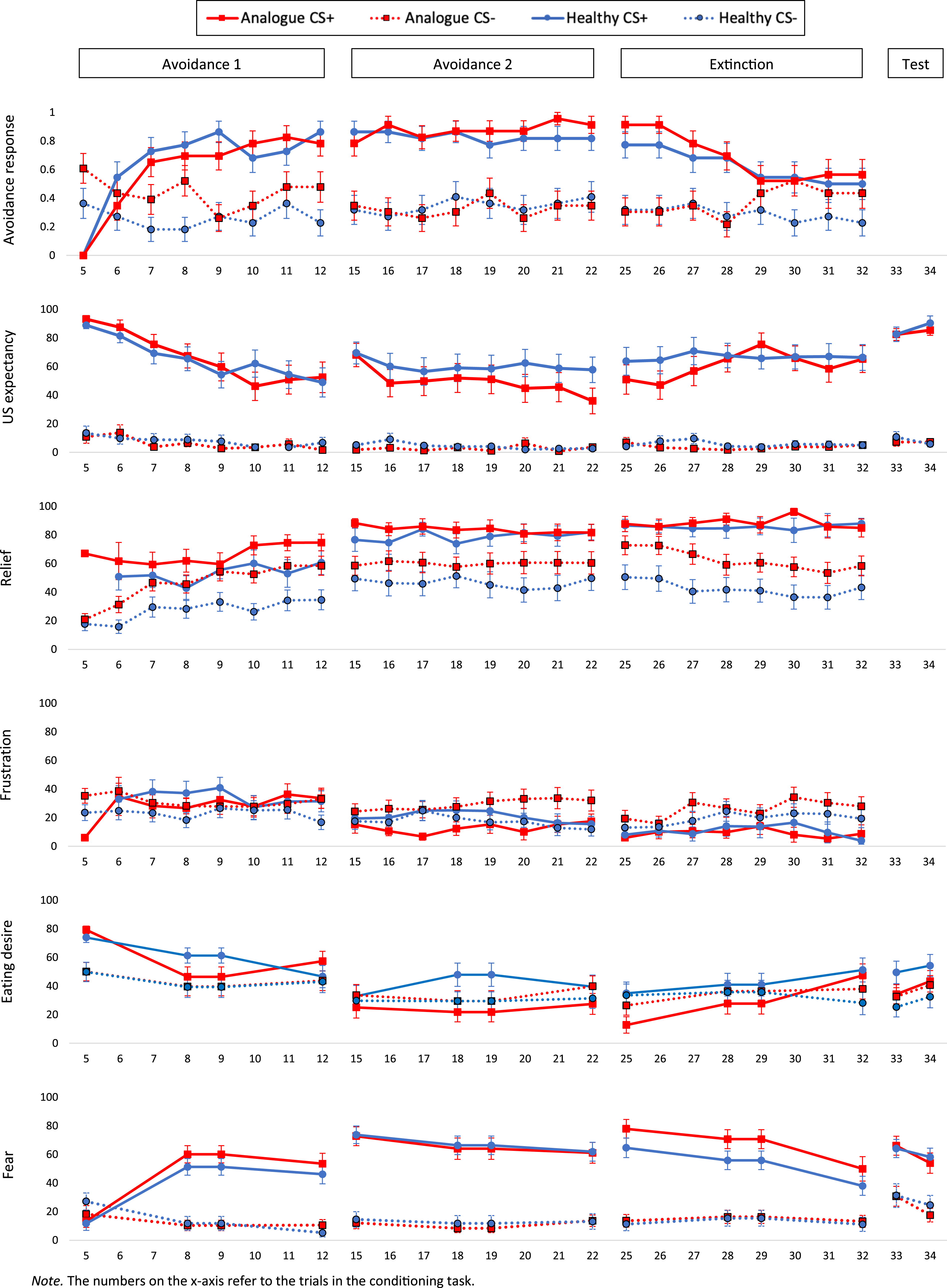
The analogue group shows more unnecessary avoidance behaviors and more persistent food avoidance during extinction than the healthy group.
Avoidance response
At the beginning of the first avoidance learning phase, the analogue group pressed the “Avoid” button more frequently than the healthy group on CS- trials, Group: z = 2.08, p = .037, but not on CS+ trials, CS × Group interaction: z = −2.25, p = .024. In line with this, post hoc comparisons showed that the analogue group was more likely to avoid on CS- than CS+ trials at the beginning of the avoidance learning phase (Trial 5: β = 1.95, SE = 0.69, z = 2.84, p = .005, d = 0.42), while this was not the case for the healthy group (p = .772). Both groups became more likely to avoid on CS+ trials over the course of the avoidance learning phase, CS × Trial interaction, z = 4.38, p < .001. Over the course of the second avoidance learning phase, the analogue group increased in their avoidance frequency on CS+ trials, while there was no change in the healthy group, CS × Group × Trial interaction: z = 2.02, p = .044. However, post hoc comparisons showed no differences between the analogue and healthy group in their avoidance responses for the CS+ and the CS- on any of the trials of the second avoidance learning phase (p’s > .202). Across the extinction phase, the analogue and healthy group were more likely to avoid on CS+ than CS- trials, CS: z = 4.95, p < .001. However, this difference became smaller over the course of the extinction phase, CS × Trial interaction: z = −3.08, p = .002. Correspondingly, post hoc comparisons showed that participants became less likely to avoid on CS+ trials from trial 25 to trial 32 (β = 6.8, SE = 1.14, z = 5.95, p < .001, d = 0.63). Over the course of the extinction phase, the analogue group also increased in their likelihood to execute the avoidance response on CS- trials, Group × Trial interaction: z = 2.60, p = .009, while there was no significant change in the healthy group, Trial: z = −1.40, p = .162. Accordingly, post hoc comparisons showed that the analogue group pressed the “Avoid” button more often on CS- trials than the healthy group at the end of the extinction phase (Trial 32: β = −2.38, SE = 1.04, z = −2.28, p = .022, d = −0.68). In sum, the analogue group avoided more frequently on CS- trials than the healthy group, especially during early food avoidance learning and extinction.
Maximum absolute deviation
At the beginning of the first avoidance learning phase, the analogue group showed a larger difference in mouse movements between trials with and without an avoidance response than the healthy group, Group × Avoidance interaction: F(1, 338.84) = 6.50, p = .011. Post hoc comparisons showed that the analogue group displayed curvier mouse movements when pressing the “Not Avoid” versus the “Avoid” button (β = 244.6, SE = 87.2, t(341) = 2.81, p = .005, d = 0.15), while there was no such difference in the healthy group (β = −64, SE = 84.4, t(336) = −0.76, p = .449, d = −0.04). Thus, the analogue group was more ambivalent when choosing food intake than when choosing to avoid food intake. The MAD reduced in both groups over the course of the first avoidance learning phase, Trial: F(1, 343.41) = 10.19, p = .002. There was no significant difference in mouse movements between groups in the second avoidance learning phase (p’s > .145). Over the course of the extinction phase, the MAD changed differently in the analogue compared with the healthy group, Group × Trial interaction: F(1, 315.50) = 4.98, p = .025. Post hoc comparisons showed that in the healthy group mouse trajectories became curvier when avoiding the milkshake (Trial 25 to Trial 32: β = −89.5, SE = 46, t(322) = −1.95, p = .052, d = −0.11), while there was no change in mouse trajectories in the analogue group (Trial 25 to Trial 32: β = 28.3, SE = 42.2, t(321) = 0.67, p = .502, d = 0.04). However, post hoc comparisons showed no significant differences in the MAD between the analogue and healthy group on any of the trials of the extinction phase (p’s > .175). Changes in MAD across the food avoidance learning task are presented in Figure 5. Maximum absolute deviation (MAD) for CS+ trials with and without an avoidance response across the food avoidance learning task. Note. The numbers on the x-axis refer to the trials in the conditioning task. Positive MAD: curvature in mouse trajectories towards the unselected response option; negative MAD: curvature in mouse trajectories towards the selected response option.
Summary
Participants in the analogue group engaged in more unnecessary avoidance behaviors on CS- trials than the healthy group, while there was no consistent difference between groups in the frequency of food avoidance on CS+ trials. Participants’ mouse movements suggest that the analogue group was more ambivalent when not avoiding the milkshake during early food avoidance acquisition and continued to avoid more decisively during extinction than the healthy group.
The analogue group shows impairments in action → safety learning reflected in higher expectancies to receive food after engaging in food avoidance behaviors.
The analogue and healthy group did not differ in their US expectancy ratings during the first avoidance learning phase (p’s > .163). During the second avoidance learning phase, the analogue group reported lower US expectancies on CS- trials with an avoidance response than the healthy group, Group × Avoidance interaction: z = −2.51, p = .012. Post hoc comparisons showed that the healthy group expected the milkshake more after CS- trials with than without an avoidance response (β = −8.95, SE = 2.87, z = −3.12, p = .002, d = −0.23), while there was no such difference in expectancy ratings in the analogue group (β = 1.04, SE = 1.35, z = 0.77, p = .443, d = 0.06). In the extinction phase, the analogue group again reported a lower expectancy for the milkshake after CS- trials with an avoidance response than the healthy group, Group × Avoidance interaction: z = −2.32, p = .021. However, post hoc comparisons showed no significant differences between CS- trials with and without an avoidance response in the analogue (β = 1.99, SE = 1.08, z = 1.84, p = .065, d = 0.14) and the healthy group across the extinction phase (β = −1.01, SE = 0.71, z = −1.42, p = .155, d = −0.10). There were no differences in US-expectancy ratings for CS+ trials across the food avoidance learning task (p’s > .077). In sum, both groups successfully learned that the avoidance response on CS+ trials canceled the delivery of the milkshake. The groups differed in their expectancy ratings on CS- trials with an avoidance response. Whether these differences are due to abnormal learning in the analogue group, however, cannot be deducted based on the present findings.
The analogue group shows greater relief and less frustration after food omission during avoidance acquisition and extinction than the healthy group.
Relief
Throughout the food avoidance learning task participants in both groups reported greater relief about the omission of the milkshake after CS+ than CS- trials, CS: first avoidance learning phase: F(1 195.53) = 18.65, p < .001; second avoidance learning phase: F(1, 55.52) = 21.64, p < .001: extinction phase: F(1, 47.22) = 25.47, p < .001. Over the course of the first avoidance learning phase, relief increased in the analogue and healthy group, Trial: F(1, 504.96) = 79.20, p < .001, and this raise was stronger in the analogue group, Group × Trial interaction: F(1, 504.96) = 5.98, p = .015. Correspondingly, post hoc comparisons showed that at the end of the first avoidance phase, the analogue group reported greater relief about US omission than the healthy group (Trial 12: β = −23.10, SE = 7.59, t(52.7) = −3.04, p = .004, d = −0.84). There were no differences in relief ratings between groups in the second avoidance learning phase (p’s > .275). Throughout the extinction phase, all participants reduced in their relief about US omission after CS- trials, Trial: F(1, 42.26) = 8.86, p = .005, while relief ratings after CS+ trials remained stable, CS × Trial interaction: F(1, 499.87) = 5.38, p = .021. Moreover, there was a marginally significant difference between the analogue and healthy group in their relief ratings, Group: F(1, 41.56) = 3.84, p = .057. The corresponding regression coefficient (see Table 16, Supplemental material 2) showed that relief ratings were higher in the analogue group than in the healthy group (Analogue group: β = 24.10, SE = 10.36, t(43.78) = 2.33, p = .025).
Frustration
In the first avoidance learning phase, the analogue group showed a smaller difference in frustration ratings between CS+ and CS- trials than the healthy group, CS × Group interaction: F(1, 140.89) = 6.57, p = .011. Post hoc comparisons showed that the analogue group indicated comparable levels of frustration after CS+ and CS- trials (β = 0.99, SE = 4.69, t(40.1) = 0.21, p = .834, d = 0.03), while the healthy group reported greater frustration after CS+ compared to CS- trials (β = −12, SE = 4.67, t(39.2) = −2.57, p = .014, d = −0.41). However, post hoc comparisons showed no significant difference in frustration levels between the analogue and healthy group for CS+ and CS- trials in the first avoidance learning phase (p’s > .226). In the second avoidance learning and extinction phase, the analogue and healthy groups reported comparable levels of frustration (p’s > .083), and in both groups, differences in frustration between CS+ and CS- trials were not present anymore (p’s > .065).
Summary
The analogue group displayed more relief than the healthy group during food avoidance acquisition and extinction. Participants in the analogue and healthy groups showed no consistent differences in frustration after US omission.
The analogue group shows a faster and more extreme reduction in eating desires and liking for predictors of food intake during avoidance acquisition and a more persistent reduction during extinction than the healthy group.
Eating desires
In the first avoidance learning phase, eating desires on CS+ trials reduced faster in the analogue group than in the healthy group, CS x Trial × Group interaction, F(2, 129.52) = 3.11, p = .048. Post hoc comparisons showed a significant reduction in eating desires from trial 5 to 9 in the analogue group (β = 32.78, SE = 5.40, t(100.2) = 6.07, p < .001, d = 0.61) while eating desires reduced significantly from trial 9 to 12 in the healthy group (β = 26.80, SE = 6.41, t(84.1) = 4.18, p < .001, d = 0.46). There were no significant differences between groups on the last trial of the avoidance learning phase (Trial 12: β = −10.24, SE = 9.35, t(52.4) = −1.10, p = .279, d = −0.30). Thus, the analogue group showed an earlier reduction in eating desires than the healthy group. In the second avoidance learning phase, the analogue group reported lower eating desires on CS+ trials than the healthy group, CS × Group interaction: F(1, 43.04) = 4.36, p = .043. Thus, the reduction in eating desires was more persistent in the analogue than in the healthy group. Over the course of the extinction phase, eating desires on CS+ and CS- trials increased more strongly in the analogue than the healthy group, Trial × Group interaction: F(2, 170.32) = 5.66, p = .004. Post hoc comparisons showed that initial differences in eating desires on CS+ trials between the groups (Trial 25: β = 22.17, SE = 10.3, t(61) = 2.16, p = .035, d = 0.55) disappeared in the middle of the extinction phase (Trial 29: β = 13.21, SE = 10.3, t(61) = 1.29, p = .204, d = 0.33). While the healthy group reported higher eating desires on CS+ than CS- trials at the end of the extinction phase (Trial 32: β = −22.38, SE = 7.44, t(91.2) = −3.01, p = .003, d = −0.32), there was no difference between CSs in the analogue group (Trial 32: β = −9.48, SE = 7.22, t(89) = −1.31, p = .193, d = −0.14). Thus, the analogue group showed no return of conditioned eating desires at the end of the extinction phase. Participants changed in their eating desires from the extinction phase to the extinction test phase, Trial: F(2, 170.40) = 6.82, p = .001. Post hoc comparisons showed that eating desires reduced from trial 32 to trial 33 (β = 5.82, SE = 2.04, t(170) = 2.85, p = .014, d = 0.22) and increased again from trial 33 to trial 34 (β = −7.06, SE = 2.04, t(170) = −3.45, p = .002, d = −0.26). The healthy group again reported greater eating desires on CS+ compared with CS- trials, CS: F(1, 43.10) = 10.95, p = .002, while the analogue group reported comparable eating desires for both CSs, CS × Group interaction: F(1, 43.1) = 4.92, p = .032. In sum, conditioned eating desires did not normalize in the analogue group although executing the avoidance response was not possible anymore.
CS valence
There were no significant differences in CS valence ratings between groups (p’s > .072). Changes in CS valence across the food avoidance learning task are presented in Figure 6. CS valence ratings per group and CS across the food avoidance learning task. Note. Begin 1 and End 1 = beginning and end first avoidance learning phase; Begin 2 and End 2 = beginning and end second avoidance learning phase; Begin Ext and End Ext = beginning and end extinction phase; End test = extinction test phase.
Summary
While both groups gave comparable CS valence ratings throughout the food avoidance learning task, the analogue group showed a faster and more persistent reduction in conditioned eating desires during the acquisition of food avoidance behaviors. While eating desires increased in both groups during the extinction phase, the analogue group did not reacquire conditioned eating desires.
The analogue group shows a faster and more extreme increase in fear for predictors of food intake during avoidance acquisition and a more persistent increase during extinction than the healthy group.
There were no significant differences between the analogue and healthy group in fear ratings in any phase of the food avoidance learning task (p’s > .149). Thus, the analogue group showed no abnormalities in fear learning during food avoidance acquisition and extinction compared with the healthy group.
Discussion
In the present study, we investigated whether healthy women with higher and lower levels of anorexia nervosa symptoms and anorexia nervosa-related characteristics differed in the acquisition and extinction of food avoidance behaviors. Moreover, we tested whether women with more symptoms would show learning abnormalities potentially related to maladaptive food avoidance. In line with our hypotheses, we found that individuals in the analogue group engaged in more unnecessary avoidance behaviors, reported greater relief after the omission of the milkshake, and reduced faster and more persistently in conditioned eating desires than the healthy group. Contrary to our expectations, the analogue group showed no deficits in action → safety learning—meaning they successfully learned that using the avoidance response on CS+ trials canceled the milkshake—and did not differ from the healthy group in fear learning, frustration levels after US omission, and their liking of the CSs throughout the food avoidance learning task.
Irrespective of their level of anorexia nervosa-related symptoms and characteristics, participants persistently avoided the milkshake during the avoidance learning phases and reduced in food avoidance behaviors during extinction. Thus, they did not show excessive food avoidance or extinction deficits. Instead, individuals in the analogue group executed the avoidance response more frequently on CS- trials than the healthy group. As the CS- was never paired with the milkshake (or followed by rewards and punishments), avoidance responses on CS- trials were not necessary and held no costs for the individual. Thus, individuals with higher levels of anorexia nervosa symptoms and anorexia nervosa-related characteristics displayed more unnecessary low-cost avoidance behaviors (for a definition of low-cost avoidance, see Vervliet & Indekeu, 2015; Wong & Pittig, 2022). When considering the various safety behaviors used by patients with anorexia nervosa (Schaumberg et al., 2021), body checking after eating very small portions or low-calorie foods could be an example of unnecessary low-cost avoidance. While this type of maladaptive avoidance might not appear as harmful as excessive food restriction, it could still have a detrimental impact on the development and maintenance of anorexia nervosa symptoms: engaging in eating disorder-related avoidance behaviors in the absence of danger can aggravate threat beliefs (Spix, Melles, et al., 2023) and might hinder individuals to test these incorrect assumptions (Lovibond et al., 2009; Spix, Schutzeichel, et al., 2023). Specifying the type of maladaptive avoidance underlying certain eating disorder behaviors might help to target them more successfully during treatment.
Research in the context of anxiety disorders suggests that unnecessary avoidance behaviors are caused by reduced fear inhibition or increased fear generalization (Beckers et al., 2023; De Kleine et al., 2023; Duits et al., 2015). However, deficits in safety learning can only inadequately explain why individuals in the analogue group engaged in more unnecessary avoidance responses: participants did not report elevated US-expectancy and fear ratings, indicating no general problem with the inhibition of fear responses to the CS-. Thus, other factors may have motivated the use of unnecessary avoidance behaviors. For instance, participants in the analogue group may have been more inclined to follow a “Why not” strategy (i.e., “Using the avoidance responses on CS- trials costs me nothing, so why not avoid?”; Wong & Pittig, 2022) than the healthy group. Another possible driver for the higher frequency of unnecessary avoidance in the analogue group is an elevated relief after US omission: In line with their more frequent avoidance on CS- trials, the analogue group reported greater relief about not receiving the milkshake after CS- trials than the healthy group. Pleasant relief can serve as a positive reinforcement for avoidance actions and, thus, increases the likelihood that the behavior will occur again in the future (Cobos et al., 2023; San Martín et al., 2020; Vervliet et al., 2017). Accordingly, elevated feelings of relief might have motivated participants with increased anorexia nervosa symptoms to also frequently avoid on CS- trials. As patients with anorexia nervosa often show overall reduced levels of positive affect (Selby & Coniglio, 2020), relief might act as an especially potent reinforcer for avoidance in this group. For example, individuals with anorexia nervosa might experience stronger or more pleasant feelings of relief after body checking and noticing that they did not gain weight—even if that was unrealistic in the first place. Thereby, aberrant relief could reinforce unnecessary eating disorder-related avoidance behaviors.
While all participants reduced in eating desires after receiving rewards and punishments for (not) avoiding the milkshake, we found a faster and more persistent reduction in conditioned eating desires in the analogue than in the healthy group. Moreover, conditioned eating desires did not fully normalize in the analogue group—with comparable ratings on CS+ and CS- trials—after rewards and punishments stopped. When experiencing negative consequences after eating high-calorie foods (e.g., weight-based teasing), individuals with or at risk for developing anorexia nervosa might reduce more strongly in their eating desires for these foods than healthy individuals. Thus, while many women feel pressure from media, peers, or parents to be thin and to lose weight (see tripartite influence model; Shroff & Thompson, 2006), individuals with anorexia nervosa might be especially vulnerable to negative social feedback related to food intake (Diedrichs, 2017; Striegel-Moore & Bulik, 2007). This might facilitate and maintain food avoidance behaviors: if individuals experience less desire upon seeing predictors of food intake, it might be easier to abstain from eating and they might be less motivated to test whether eating high-calorie foods still results in aversive consequences. As we did not find differences between the analogue and healthy groups in their avoidance of the milkshake, future research should explicitly test how eating desires are related to excessive food avoidance and deficits in extinction.
The analogue group did not show faster, stronger, or more persistent fear learning than the healthy group: all participants acquired fear for the CS+ after receiving rewards and punishments and decreased in fear during extinction. Together, with previous research that did not find a difference in fear learning between individuals with anorexia nervosa and healthy controls (Fyer et al., 2020; Murray et al., 2024), these findings suggest that abnormal fear learning might not underlie the extreme fears and avoidance behaviors seen in many individuals with anorexia nervosa (Levinson & Byrne, 2015; Melles & Jansen, 2023). Instead, excessive eating disorder fears might be caused by an interplay between maladaptive avoidance behaviors and individual differences in fear-unrelated learning. Accordingly, future research could test how aberrant relief and eating desires are related to the development and maintenance of fear of food and eating. At the same time, the absence of differences in fear learning between individuals with higher and lower levels of anorexia nervosa symptoms and character traits could be due to our strong manipulation (100% reinforcement rate and highly aversive punishment). In line with the notion that individual differences are more likely to manifest under high ambiguity (Lissek et al., 2006), the analogue and healthy groups primarily differed during the first avoidance learning and the extinction phase, and on CS- trials. Consequently, future studies should test the stability of the present findings with a food avoidance learning task that constitutes a “weaker situation” by using a lower reinforcement rate, costly avoidance behaviors, and more ambivalent rewards and punishments.
More research is needed to confirm the clinical relevance of the present findings: Future studies should investigate whether and how individual differences in learning predict food avoidance behaviors later in time and whether comparable learning patterns are present in patients with anorexia nervosa. Additionally, experimental research could help to identify ways to “unlearn” food avoidance. Given that individuals with anorexia nervosa-like features readily acquired maladaptive avoidance behaviors and rapidly reduced in eating desires once food intake was paired with punishment, behavioral techniques, such as exposure therapy, might be a useful tool for the reduction of anorectic food avoidance. During exposure therapy, clinicians help patients to identify aversive stimuli contributing to food avoidance, to confront feared stimuli without avoidance behaviors, and to correct dysfunctional threat beliefs (Cardi et al., 2019; Melles et al., 2023; Steinglass et al., 2011). For example, patients, who are afraid of negative social feedback after food intake, could eat in front of others and observe their reactions (do they respond as expected) or eat after a (perceived) negative comment, like “Are you really going to eat that?” or “You are eating well!” (for more clinical examples, see Melles et al., 2023).
We want to emphasize that the present study constitutes a first step towards understanding maladaptive food avoidance and potentially related abnormalities in learning and that findings should be considered in light of the study’s limitations. While we based our choice of anorexia nervosa-related characteristics on the existing literature and clinical experience, the inclusion of variables in the cluster analysis was primarily based on correlations between the variables in the present data instead of a priori hypotheses. Interestingly, maladaptive perfectionism and behavioral inhibition significantly correlated with eating disorder symptoms, fitting previous research that identified those characteristics as important factors for the development and maintenance of anorexia nervosa (Dahlenburg et al., 2019; Fairburn et al., 2003; Frank et al., 2012, 2013, 2018; Halmi et al., 2012; Harrison et al., 2010; Jonker et al., 2020, 2022; Miles et al., 2023; Ralph-Nearman et al., 2024). As perfectionism and behavioral inhibition also play a role in the etiology of other eating disorders (Claes et al., 2021; Dahlenburg et al., 2019; Fairburn et al., 2003; Harrison et al., 2010; Puttevils et al., 2019; Wilson et al., 2019), the identified learning processes might not be specific to food avoidance in anorexia nervosa but could also contribute to these behaviors in other diagnoses, such as bulimia nervosa. Therefore, future research should disentangle whether similar learning mechanisms underpin food avoidance behaviors across eating disorders, for example, by considering characteristics that differentiate more strongly between individuals with restrictive and binge/purge symptoms (e.g., impulsivity or negative urgency; Culbert et al., 2015; Harrison et al., 2010; Schaumberg et al., 2020). Furthermore, the present study possibly lacked power to identify small differences in learning between the analogue and healthy group, given the relatively small sample size (n = 45). Consequently, findings need to be interpreted with caution. To draw more definite conclusions, the present findings should be replicated (a) with similar subtypes (based on eating disorder symptoms, maladaptive perfectionism, and behavioral inhibition) in another sample, (b) with more participants, and (c) with pre-registered, specific hypotheses based on the present results. Thereby, replication studies could profit from using Bayesian statistics that allow researchers to consider evidence across studies. Lastly, the setup of the conditioning task did not allow us to evaluate extinction learning independent of participants’ avoidance responses: if a participant persistently avoided during the extinction phase, they could not notice that food intake was not punished anymore. This “protection from extinction” effect may have slowed down or stopped extinction learning in both groups and, thereby, overshadowed potential extinction learning deficits. Future studies interested in extinction learning in relation to maladaptive food avoidance should include trials without the possibility of avoiding in the extinction phase.
To conclude, the present study shows that healthy individuals with higher levels of anorexia nervosa symptoms and anorexia nervosa-related characteristics present a combination of unnecessary avoidance, heightened relief, and aberrant conditioned eating desires. These findings support the idea that individual differences in learning may contribute to the development and maintenance of harmful avoidance behaviors in anorexia nervosa.
Supplemental Material
Supplemental Material - Linking maladaptive food avoidance and anorexia nervosa symptoms: An analogue study
Supplemental Material for Linking maladaptive food avoidance and anorexia nervosa symptoms: An analogue study by Michelle Spix and Anita Jansen in Journal of Experimental Psychopathology.
Supplemental Material
Supplemental Material - Linking maladaptive food avoidance and anorexia nervosa symptoms: An analogue study
Supplemental Material for Linking maladaptive food avoidance and anorexia nervosa symptoms: An analogue study by Michelle Spix and Anita Jansen in Journal of Experimental Psychopathology.
Footnotes
Acknowledgments
This study is part of an ongoing project that is financed by the Netherlands Organization for Scientific Research (NWO): Open Competition Grant 406.18.GO.069, awarded to Anita Jansen.
Author Contributions
Michelle Spix: Conceptualization, methodology, investigation, formal analysis, and writing—original draft. Anita Jansen: Conceptualization, methodology, and Writing—review and editing. All authors approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Sociale en Geesteswetenschappen, NWO (Open Competition Grant 406.18.GO.069).
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
