Abstract
Background:
CDK4/6 inhibitors abemaciclib, palbociclib, and ribociclib have rapidly become an established oral treatment for patients with ER+, HER2− locally advanced or metastatic breast cancer. The use of the oral route offers convenience and flexibility to the patient; however, the co-administration of proton pump inhibitors (PPIs) to mitigate gastrointestinal adverse events induced by anticancer treatments may decrease drug solubility, bioavailability, and potentially impact treatment efficacy.
Objectives:
The present study was aimed at investigating whether PPIs may affect the progression-free survival (PFS) of patients treated with abemaciclib.
Design:
Multicenter observational cohort study on clinical data collected retrospectively.
Methods:
Patients with ER-positive/HER2-negative mBC candidates for a first-line treatment with abemaciclib as per clinical practice were enrolled. Patients were classified as “no concomitant PPIs” or “concomitant PPIs” if PPI administration covered not less than 2/3 of the treatment period with abemaciclib. All clinical interventions were made according to clinical practice.
Results:
One hundred eight patients were enrolled in this study; 66 belonged to the “no concomitant PPIs” group and 42 to the “concomitant PPIs” group. No statistically significant difference in PFS was found between the two groups (p = 0.77). Likewise, no difference in PFS was observed in endocrine-sensitive or -resistant mBC in the presence or absence of concomitant PPI treatment. No correlation with adverse events, including hematological or gastrointestinal toxicities, was found.
Conclusion:
This study demonstrates that the administration of PPIs to patients treated with abemaciclib is not associated with clinically significant drug–drug interactions on PFS.
Plain language summary
Proton pump inhibitors (PPIs) are the most commonly used anti-acid drugs worldwide, including among cancer patients. However, drug-drug interactions between PPIs and other agents may lead to decreased drug absorption with possible reduced therapeutic benefit, or even increased toxicity. Unfortunately, only scarce data exist regarding the safety of concomitant PPI use with anti-cancer agents. In this research article we describe the clinical outcome (progression-free survival, PFS) in hormone receptor-positive/HER2-negative metastatic breast cancer patients treated with the CDK4/6 inhibitor abemaciclib who were or were not taking proton pump inhibitors. This retrospective study demonstrates no statistically significant difference in PFS between the two groups (p = 0.77). In conclusion, no clinically significant drug interaction occurs between abemaciclib and PPIs.
Keywords
Introduction
CDK4/6 inhibitors (CDK4/6i) are considered the standard of care for hormone receptor (HR)-positive/HER2-negative breast cancer (BC), both in the adjuvant and the advanced settings1–10; however, they are prone to drug–drug interactions (DDIs), mainly involving CYP3A4 inhibitors/inducers. Proton pump inhibitors (PPIs), which are often prescribed to cancer patients, strongly modify the gastric pH and negatively affect the absorption and bioavailability of drugs with pH-dependent solubility.11,12 A recent review article identified over a hundred of orally administered medications involved in clinically relevant DDIs with acid-reducing agents 13 ; in about one-third of them, the mechanism was gastric pH dependent, while for the remaining drugs, the mechanisms involved were metabolic enzymes, transporters, and urine alkalization. 13 CDK4/6i are characterized by different physicochemical properties 14 ; in particular, the aqueous solubility of abemaciclib is >2 mg/ml at pH 4.5, while the solubility of palbociclib is >2 mg/ml at pH 2.0, and it is reduced at pH values above 4, indicating a significant acid-dependent solubility of the drug.15–17 Therefore, the co-administration of acid-reducing agents is likely to influence the absorption and exposure to palbociclib, while it is possible that it has no influence, but it remains to be demonstrated, on the solubility and absorption of abemaciclib. Although the pharmacokinetic profiles and DDIs of the three CDK4/6i have been described, 14 their clinical impact is still subject to debate. 18 Recently, a few studies demonstrated a significant decrease in progression-free survival (PFS) and overall survival (OS) in metastatic BC patients treated with palbociclib when co-administered with PPIs.19–21 On the contrary, it is generally agreed that the concomitant treatment with ribociclib and PPIs does not affect the PFS of patients.20,22 To the best of our knowledge, no published data exist on the effect of PPIs on clinical outcomes in cancer patients treated with abemaciclib. Therefore, this study evaluated the impact of PPI use on the PFS of BC patients treated with abemaciclib in clinical practice.
Methods
Design
Multicenter observational cohort study on clinical data collected retrospectively; no additional treatments or diagnostic procedures were applied outside the validated clinical practice. Setting: HR-positive/HER2-negative mBC patients treated with first-line abemaciclib as per approved indication in the presence or absence of concomitant PPI therapy were enrolled in this study. The primary endpoint was to assess the possible differences in PFS between PPI users and non-users.
Inclusion criteria
Age ⩾18 years; HR-positive/HER2-negative mBC patients treated with first-line abemaciclib plus endocrine therapy (fulvestrant or letrozole), according to clinical practice; PPIs treatment allowed.
Exclusion criteria
Age <18 years; patients ineligible to receive abemaciclib as first line; concomitant antacid treatments other than PPIs.
Data collection
The clinical records of patients were collected at the Divisions of Medical Oncology at the University of Modena (Italy) and the University of Bari (Italy), the Units of Medical Oncology at the University of Pisa (Italy), the University of Verona and IRCCS-Istituto Romagnolo per lo Studio dei Tumori (IRST) “Dino Amadori” (Italy), the Department of Experimental and Clinical Biomedical Sciences “M. Serio” at the University of Florence (Italy), and the Radiation Oncology Unit from Azienda Ospedaliero Universitaria Careggi (Italy). Hormonal status was defined as tumors with estrogen and/or progesterone receptor expression >1% and HER2-negative (score 0 or 1+ to immuno-histochemistry). Treatment groups were defined as “no concomitant PPIs” if no PPIs were administered during abemaciclib treatment, or “concomitant PPIs” if the administration of PPIs covered not less than two-thirds of treatment with abemaciclib. According to the duration of previous endocrine response, endocrine-sensitive patients were those who relapsed ⩾12 months after the completion of adjuvant endocrine therapy or with de novo advanced BC, whereas endocrine-resistant patients were those who relapsed <12 months after ending adjuvant endocrine therapy. 23 Abemaciclib was administered orally at a dose of 150 mg twice daily, plus endocrine therapy (fulvestrant or letrozole), according to clinical practice. Abemaciclib reduction to 200 or 100 mg was made according to the toxicity profile. Patients took the dose of lansoprazole (15 mg), esomeprazole (20 mg), omeprazole (10 mg), or pantoprazole (20 mg) in the morning at breakfast. Abemaciclib was taken at lunchtime, and patients were instructed not to take strong inhibitors or inducers of cytochrome P450 3A4 (CYP3A4). The prescribing physician monitored the compliance of patients with the above-mentioned recommendations. Toxicity was graded according to Common Terminology Criteria for Adverse Events (CTCAE v5). All data were collected in a shared anonymized dataset.
Statistical analysis
Categorical variables, including Eastern Cooperative Oncology Group performance status (ECOG PS), hormone sensitivity, pre/peri-menopausal status, visceral/bone disease, and the number of tumor sites, were described by absolute and relative frequencies, while quantitative parameters were described by median values and range. Baseline characteristics of patients who received abemaciclib with or without PPIs were assessed by the chi-squared test or the Mann–Whitney U test, as appropriate. The association between PPIs and adverse events (AEs) was evaluated using the χ2 test or Fisher’s exact test, as appropriate. PFS was defined as the time from treatment start to the disease progression. Survival curves were obtained by the Kaplan–Meier method, and differences between curves were assessed using the log-rank test. The evaluation of independent risk factors for PFS was performed using the Cox hazard regression model. Differences were considered significant at p < 0.05. All statistical analyses were carried out using MedCalc Statistical Software version 14.8.1 (MedCalc Software, Ostende, Belgium). The reporting of this study conforms to the STROBE statement. 24
Results
A total of 108 patients were included in the study; 42 patients received concomitant PPI, mostly lansoprazole, during abemaciclib treatment (Table 1), while 66 patients were treated with the association of abemaciclib plus endocrine therapy (ET) without PPIs. Forty-four patients were endocrine-sensitive and received letrozole, while 64 were endocrine-resistant and received fulvestrant (groups balancing p = 0.17). Fifty-five patients received abemaciclib at a dose of 300 mg (51%), 21 patients (19.4%) reduced the dose to 200 mg, and 24 (22.2%) required a further dose reduction to 100 mg (groups balancing, p = 0.53). No significant differences between “concomitant PPIs” and “no concomitant PPIs” groups in terms of baseline characteristics have been found, including ECOG PS (p = 0.09) and visceral disease (p = 0.14), pre/post-menopause (p = 0.25). Detailed clinical characteristics of patients and the type of PPI used are reported in Table 1.
Clinical characteristics of patients and distribution across PPI treatment groups.

ECOG PS, Eastern Cooperative Oncology Group performance status; ET, endocrine therapy; PPIs, proton pump inhibitors.
Overall median PFS was 39.5 months; after stratification based on PFS and concomitant treatment with PPIs, the Kaplan–Meier curves were almost superimposable with no statistically significant difference in terms of PFS for patients taking PPIs versus those treated with abemaciclib plus Et alone (29.7 months vs 39.5 months in PPI non-users vs users, respectively; p = 0.77; Figure 1).
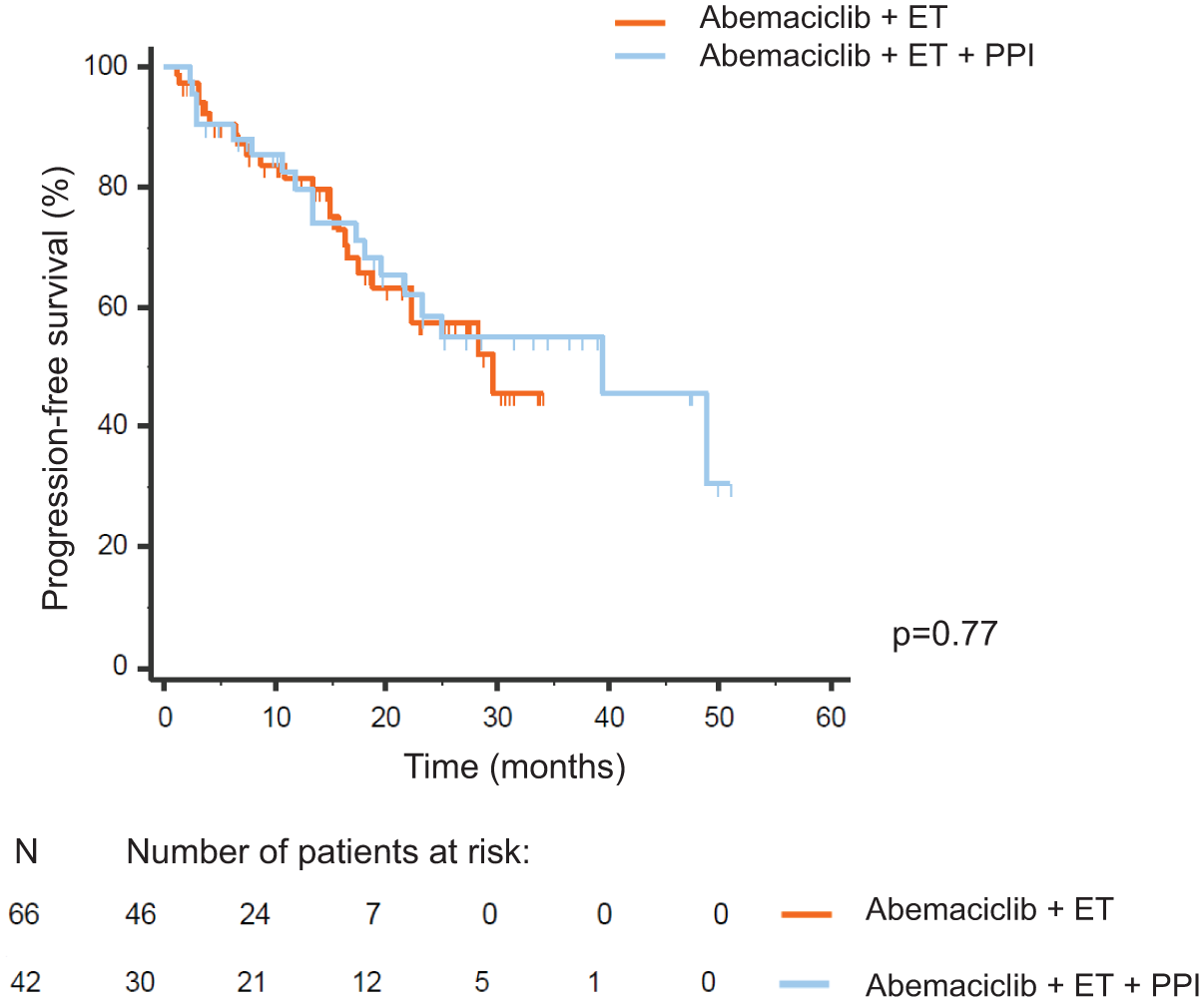
PFS of the overall population treated with abemaciclib plus endocrine therapy and stratified according to the use of concomitant PPIs.
The univariate analysis on age, number of metastatic sites at abemaciclib baseline, endocrine sensitivity or resistance, ECOG, menopausal status, visceral disease, and abemaciclib dose reduction showed that only age, endocrine sensitivity, and number of metastatic sites were significantly associated with PFS (Table 2). The multivariate analysis showed age, endocrine sensitivity, and number of metastatic sites as independent predictive biomarkers for shorter PFS (Table 2).
Univariate and multivariate analyses for PFS.
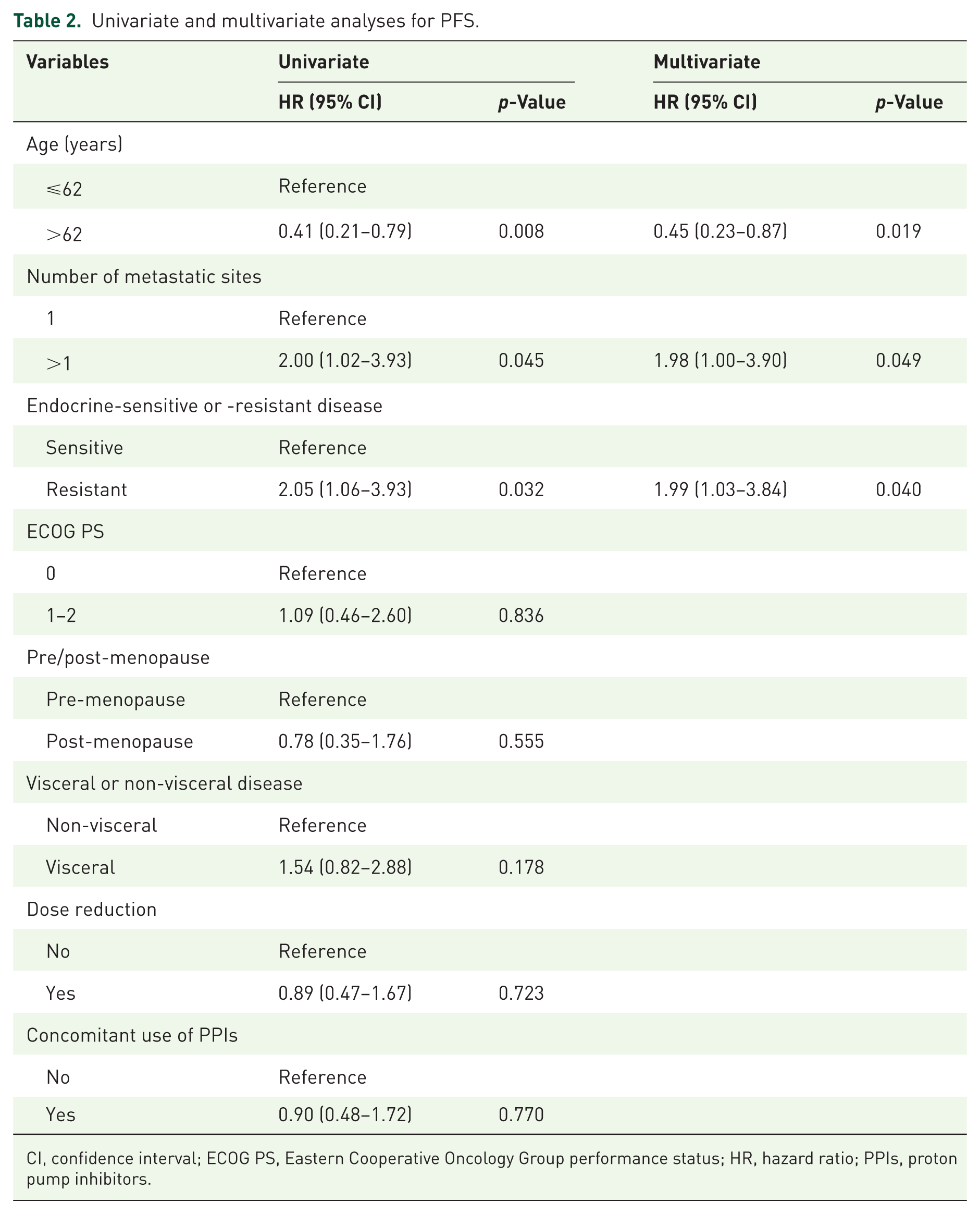
CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; PPIs, proton pump inhibitors.
To assess the impact of PPIs on endocrine sensitivity in determining PFS, patients were classified into two groups: endocrine-sensitive and endocrine-resistant (with or without concomitant PPIs in both settings). No statistically significant differences in terms of PFS between the two groups were found, both in the presence and in the absence of concomitant PPIs (Figure 2). The AEs showed no correlation with PPI administration, including hematological and gastrointestinal toxicities (p = 0.24 and p = 0.82, respectively). Among patients who experienced hematological or gastrointestinal toxicity, the distribution of severity (⩽Grade 2 vs ⩾Grade 3) did not differ according to PPI use (Table 3).

Effect of concomitant PPIs on progression-free survival over endocrine sensitivity in patients treated with abemaciclib. Patients were stratified into four groups: (a) endocrine-sensitive patients and no concomitant PPIs versus endocrine-sensitive patients and concomitant PPIs; (b) endocrine-resistant patients and no concomitant PPIs versus endocrine-resistant patients and concomitant PPIs.
AEs and distribution across treatment groups.

Values are reported as n (%). p-Values refer to comparisons of Grade ⩽2 versus ⩾3 between PPI users and non-users (χ2 test or Fisher’s exact test, as appropriate). Three patients had missing data for hematological toxicity (n = 105), and one patient had missing data for gastrointestinal toxicity (n = 107).
AE, adverse event.
Discussion
The present study demonstrates that the concomitant use of PPIs and abemaciclib in metastatic BC patients does not affect the PFS of patients treated as first line.
The acid-solubility properties of CDK4/6i may influence their absorption, oral bioavailability, and systemic exposure in the presence of a pharmacologic reduction of gastric acid secretion. Abemaciclib is soluble in solutions up to pH 6.8 17 ; therefore, acid-suppressant agents should not affect the pharmacokinetic profile of this drug. On the contrary, the solubility of another CDK4/6i, palbociclib, declines as pH increases, being >2 mg/ml at pH 2.0 and less than 0.5 mg/ml at pH >4.5, whereas that of ribociclib is greater than 2.4 mg/ml at the same pH level,16,25 thus providing important information to understand the effect of PPIs on drug pharmacokinetics, particularly bioavailability, with potential implications on clinical efficacy.19,22 Unfortunately, no data on the pharmacokinetics of abemaciclib in patients given the drug alone or in combination with PPIs are available. This is a limitation to the interpretation of the present results, although the characteristics of drug solubility and the absence of significant effects of PFS, as demonstrated in the present study, suggest that the bioavailability of abemaciclib is not impaired by the increase in gastric pH upon administration of PPIs.
An additional factor to consider is the role of metabolic DDIs due to the effect of PPIs on CYP450-dependent drug metabolism in the liver. The main isoenzymes involved in the metabolism of PPIs are CYP2C19 and CYP3A4. 26 Apart from being metabolized by CYP isozymes, PPIs are inhibitors of CYPs marginally involved in the metabolism of CDK4/6i. In particular, omeprazole inhibits CYP2C19, 27 while lansoprazole inhibits CYP2D6, 28 pantoprazole is an inhibitor of CYP2C9 29 , while esomeprazole causes strong inhibition of CYP2C19, but only weakly inhibits CYP3A4. 30 Therefore, the impact of CYP450-dependent metabolic DDIs between PPIs and CDK4/6i, in particular abemaciclib, can be considered as clinically irrelevant. Taken together, the absence of DDIs at the metabolic level and the lack of effect of pH variation on abemaciclib solubility, at least within the pH range from baseline and after PPI administration, fully supports the results of the present study. Moreover, in addition to the potential effect of PPIs, the interindividual genetic variability of drug metabolizing enzymes and transporters involved in drug absorption, distribution, metabolism, and elimination may also play an independent role on DDIs. 31 Published data demonstrated an association between polymorphisms in CYP3A4, CYP3A5, ABCB1, and ABCG2 genes and AEs in patients treated with CDK4/6 inhibitors. 32
The strengths of the study are as follows: first, it included patients from several medical institutions. Therefore, the present data may be applicable to similar populations in the same clinical settings and will presumably play a crucial role in shared decision-making between patients and a multidisciplinary care team. In particular, the data from this study provide reassurance about the risks of interactions that may have a negative impact on the clinical outcome of patients who are candidates for treatment with abemabicib and ET. Second, the study focused on a homogeneous population of patients with endocrine-resistant mBC who had never undergone chemotherapy. Third, the follow-up period was reasonably long to efficiently assess PFS. Finally, both groups were well-balanced, and the univariate and multivariate analyses returned results that are in line with what is expected clinically. These findings increase the robustness of the results and are a positive feature of the present clinical investigation.
The 10-month difference displayed by the Kaplan–Meier PFS curves reflects the effect of the curve tails, which do not separate before 29 months. However, the tail of a Kaplan–Meier curve is generally considered less reliable than the earlier portions of the curve, especially when, as in this case, the number of patients at risk is low (six total patients). This is because the outcome of a single patient can have a large impact on the estimate. Even if the tail of a Kaplan–Meier PFS curve provides insights into the long-term effectiveness of a treatment, its interpretation should be done with caution, considering the number of patients at risk and the potential impact of censoring.
Limitations
This cohort study has some limitations. First, this was a retrospective, observational evaluation; therefore, information bias exists. Second, the cohort size was relatively small, and no pharmacokinetic data were collected. The lack of data on plasma concentrations may be a shortcoming, but we must consider the difficulty of performing pharmacokinetic investigations in subjects undergoing treatment as outpatients, and the value of inevitably scattered and limited sampling times for this category of subjects would not have added much value and reliability to the data presented in this study. Moreover, an accurate predictive pharmacokinetic analysis should include long-term data collection, considering that DDIs may have a delayed effect in terms of adverse drug reactions, 33 which could not appear immediately. Third, no exact data on adherence to CDK4/6 inhibitors or PPIs were obtained, as we used prescription history. In addition, we were unable to perform quantitative or qualitative assessments to establish the patterns of drug timing.
These present findings need to be validated in future trials, and, whenever possible, prospective pharmacokinetic studies should be conducted to fully validate the present findings. We aim to evaluate the OS in these patients and increase the size of the population.
Conclusion
The relevance of DDIs is underestimated in clinical practice and, for this reason, we aim to increase awareness of the risks of DDIs for the safety of patients and clinical efficacy of treatments. The widespread use of PPIs may lead to serious problems for those orally administered drugs that have a solubility influenced by gastric pH, with unpredictable consequences in their bioavailability, drug exposure, and finally clinical efficacy; therefore, studies on this topic are highly desirable. This study suggests that the effectiveness of the CDK4/6i abemaciclib is unlikely to be affected by concomitant PPI use in patients with hormone receptor-positive and HER2-negative endocrine-resistant mBC.
This evidence could hold significance for clinical decision-making regarding the concurrent use of PPIs in real-world settings for patients undergoing abemaciclib treatment.
