Abstract
Background:
Lenalidomide is an immunomodulatory agent thought to inhibit the growth of del(5q) haematopoietic progenitors in myelodysplastic syndromes (MDS) that was approved for use in Europe for certain forms of these conditions in 2013.
Objectives:
This study aimed to identify the distribution of lenalidomide treatment and safety outcomes in patients with MDS in Europe among those treated within (on-label) and outside (off-label) of the European Commission-approved indication.
Design:
This observational, retrospective study of prospectively collected disease registry data (13 June 2013 to 1 May 2023) summarised lenalidomide treatment patterns and safety outcomes by on- and off-label treatment in 11 European countries.
Methods:
Safety outcomes included progression to acute myeloid leukaemia (AML), second primary malignancies (SPMs), adverse events (AEs) and all-cause mortality. Hazard ratios (HRs) and 95% CI were reported for progression to AML comparing on- to off-label treatment.
Results:
Among 523 qualifying patients, more were prescribed lenalidomide off-label (
Conclusion:
Analysis of real-world registry data indicated that off-label lenalidomide use in patients treated for MDS was common. Across the registries studied, the overall incidence of safety events of lenalidomide in patients with MDS was consistent in both on-label and off-label use, highlighting the importance of researching off-label use of lenalidomide in MDS.
Trial registration:
This study was registered with the EU Post-Authorisation Studies (PAS) Register: EUPAS22604.
Plain language summary
MDS are a group of cancer-causing abnormalities in the bone marrow that affect blood stem cells and can lead to AML, a cancer of the blood and bone marrow. Lenalidomide is a drug approved to treat certain types of MDS. This study investigated the use of lenalidomide in patients with MDS in the way it was approved in 2013 by the EC for treatment with lenalidomide (on-label) and in patients whose use was not approved by the EC (off-label). This study measured the proportion of patients with MDS in each group and important safety issues from treatment, such as development of AML, development of other cancers, side effects during treatment, and death.
The majority (66%) of patients with MDS in the disease registries received off-label treatment while 30% received on-label treatment. For 4% of patients, the diagnosis for lenalidomide treatment was unknown. Across the registries, the proportion of safety issues were similar in the on- and off-label groups, including similar rates of AML, other cancers, side effects, and death. Overall, lenalidomide was deemed to be acceptably safe for treating patients with MDS. Future research is needed to investigate how well lenalidomide works in off-label patients with MDS.
Keywords
Introduction
The myelodysplastic syndromes (MDS) group is comprised of haematopoietic malignancies characterised by blood cytopenia, ineffective haematopoiesis and hypercellular bone marrow. 1 Although the disease is highly heterogeneous, common clinical manifestations include anaemia, neutropenia, thrombocytopenia, infections and bleeding complications.2,3 MDS were first identified among individuals with cytogenetic deletion of the long arm of chromosome 5 (del[5q]); however, further research has shown that MDS are characterised by a high degree of genetic variation and not all patients with MDS have del(5q).4,5
MDS have an annual European standard population age-standardised incidence of 2.51 per 100,000 person-years. 6 Overall, MDS have a worldwide prevalence ranging from 0.22 to 13.2 per 100,000 individuals, primarily affecting older adults. 7 One study in the Netherlands estimated MDS to have a 5-year survival ranging from 18% to 58% depending on other clinical factors. 8 Overall prognosis is predicted by a variety of patient attributes, including karyotype, bone marrow blast count, peripheral cytopenia, serum ferritin and lactate dehydrogenase levels, among other factors.9–11
MDS exist on the same continuum of disease progression as acute myeloid leukaemia (AML) and can progress to AML through genetic clonal evolution, which occurs in 30%–40% of patients with MDS.1,2,12,13 Rates of leukemic progression for MDS vary based on karyotype and other clinical factors.1,4,5,10 Cytogenetic abnormalities and bone marrow failure are directly implicated in both the risk of progression to AML and overall prognosis of MDS with del(5q), with a complex karyotype indicating high risk of progression to AML.10,14,15 Moreover, gene mutations have been implicated in disease prognosis and progression to AML. 2
An immunomodulatory agent, lenalidomide, demonstrates multiple mechanisms of action that can restore erythropoiesis and inhibit angiogenesis. 16 Lenalidomide is thought to inhibit the growth of del(5q) haematopoietic progenitors in MDS and has previously demonstrated efficacy in the MDS indication.1,16,17 Clinical trial findings have shown that treatment with lenalidomide aids in achieving sustained blood transfusion independence, increased haemoglobin concentrations, resolution of cytologic abnormalities and reduced risk of death or progression to AML among patients with International Prognostic Scoring System (IPSS) Low- or Intermediate (Int)-1 risk MDS. Overall, the drug has been considered to have an acceptable safety profile for treatment in this indication.16,17
In 2013, the European Commission (EC) approved lenalidomide for use in patients with transfusion-dependent anaemia due to IPSS Low- or Int-1-risk MDS associated with isolated del(5q) cytogenetic abnormality, when other therapeutic options are insufficient or inadequate. 18 Since the approval of lenalidomide for the treatment of MDS in Europe, there remain important questions about its real-world use in this indication, including long-term risk of progression to AML, development of second primary malignancies (SPMs) other than AML and occurrence of adverse events (AEs). Compared to real-world patients with MDS who have been treated with other therapies, lenalidomide-treated patients with MDS have shown improved or similar survival rates, no increased risk of progression to AML and comparable rates of SPMs.14,19,20 According to one study from the United States, it is estimated that 24.2% of MDS patients treated with lenalidomide have low-risk defined non-del(5q) MDS. 21 Prior observational studies in Europe have largely evaluated outcomes in lenalidomide treatment for on-label use only, defined as usage in Low- or Int-1 risk patients with MDS with del(5q) and transfusion-dependent anaemia, without consideration of off-label lenalidomide use in MDS, defined as lenalidomide use in any MDS condition other than Low- or Int-1-risk del(5q) transfusion-dependent anaemia.19,22 Thus, it is important to understand the real-world patterns and outcomes of on- and off-label use of lenalidomide treatment in Europe. In particular, off-label use of lenalidomide warrants attention in safety monitoring, given its lack of regulatory approval.
The primary objective of this study was to describe the patterns of lenalidomide use in routine clinical practice for patients with MDS in Europe using an observational, retrospective, registry-based drug utilisation study design. The secondary objective was to evaluate the safety outcomes of on- and off-label lenalidomide usage for the treatment of MDS in the same setting, including progression to AML, SPMs, AEs and all-cause mortality. This retrospective study, sponsored by Bristol Myers Squibb, was a condition of the European Medicines Agency (EMA) marketing authorisation for lenalidomide in the MDS indication.
Methods
Patient population and study design
This post-authorisation safety study was a retrospective, longitudinal, observational, registry-based analysis of data from patients with MDS from 11 European countries (EU Post-Authorisation Studies (PAS) Register: EUPAS22604). Patient data from 13 June 2013 to 1 May 2023 were obtained from four MDS registries, including the Düsseldorf MDS Registry, European Myelodysplastic Syndromes (EUMDS) Registry, French Registry of MDS (Groupe Francophone des Myélodysplasies (GFM)) and Spanish Registry of MDS (Registro Español de Síndromes Mielodisplásicos (RESMD)).23–26 Collectively, these registries systematically collect and maintain information about patients diagnosed with MDS from multiple countries. Data from Austria, the Czech Republic, Denmark, France, Germany, Greece, the Netherlands, Poland, Portugal, Spain and the United Kingdom were included in this analysis. The registries’ data collection methods have been previously described.6,27–31 The reporting of this study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) requirements (refer to Supplemental Material 2 for additional information). 32
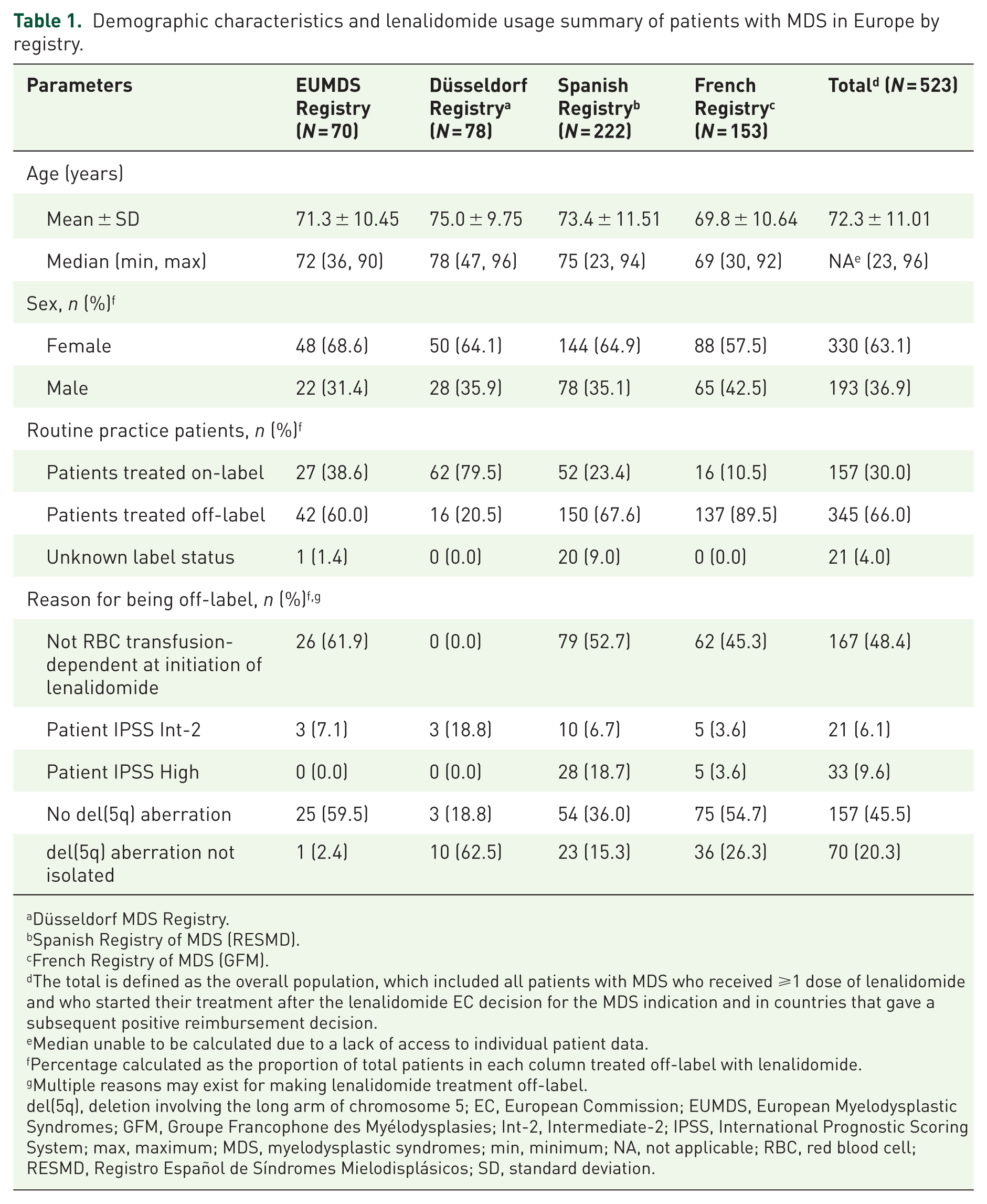
All included participants had a laboratory-confirmed MDS diagnosis (morphological and cytogenic) with defined dates of diagnosis and initiation of lenalidomide treatment (Table 1). In addition, patients must have been treated with lenalidomide in a European country that had a positive reimbursement decision for lenalidomide for the MDS indication. Finally, patients were included if they received at least one dose of lenalidomide for the treatment of prevalent or incident MDS after lenalidomide’s EC approval (13 June 2013). Prevalent MDS cases were considered to be patients diagnosed prior to the EC approval of lenalidomide, while incident cases were patients diagnosed after the decision. Patients who received lenalidomide prior to the EC’s approval date or were treated with lenalidomide in a clinical trial were excluded.
Demographic characteristics and lenalidomide usage summary of patients with MDS in Europe by registry.
Düsseldorf MDS Registry.
Spanish Registry of MDS (RESMD).
French Registry of MDS (GFM).
The total is defined as the overall population, which included all patients with MDS who received ⩾1 dose of lenalidomide and who started their treatment after the lenalidomide EC decision for the MDS indication and in countries that gave a subsequent positive reimbursement decision.
Median unable to be calculated due to a lack of access to individual patient data.
Percentage calculated as the proportion of total patients in each column treated off-label with lenalidomide.
Multiple reasons may exist for making lenalidomide treatment off-label.
del(5q), deletion involving the long arm of chromosome 5; EC, European Commission; EUMDS, European Myelodysplastic Syndromes; GFM, Groupe Francophone des Myélodysplasies; Int-2, Intermediate-2; IPSS, International Prognostic Scoring System; max, maximum; MDS, myelodysplastic syndromes; min, minimum; NA, not applicable; RBC, red blood cell; RESMD, Registro Español de Síndromes Mielodisplásicos; SD, standard deviation.
Data for these analyses were collected from the registries for each qualifying patient beginning at the first recorded date of lenalidomide treatment until the last follow-up date or death, whichever occurred first. The registries obtained appropriate permissions from patients to collect the data for their own purposes. For this study, the sponsor obtained permission to receive aggregated data via the service agreement with each registry. 30
Study measures
Demographic covariates recorded at baseline included age (years) and sex (male, female). The primary outcome was the proportion of lenalidomide treatment by label status (on-label, off-label and unknown label usage) to describe the real-world distribution of lenalidomide use in the MDS indication. Patients who were transfusion-dependent due to IPSS Low- or Int-1-risk MDS with isolated del(5q) were defined as being treated on-label in line with the EC’s approved use of lenalidomide in the MDS indication. Transfusion dependence was defined as receipt of ⩾2 red blood cell (RBC) units over an 8-week period prior to lenalidomide treatment initiation. Lenalidomide treatment in any other type of MDS was defined as off-label. Patients with MDS who were treated off-label were further characterised by IPSS status (Intermediate 2 risk (Int-2) or higher), presence (yes, no) and type of additional cytogenetic abnormalities according to the Revised IPSS (IPSS-R) classification and failure to meet accepted criteria for transfusion-dependent status (including lack of documentation of transfusion-dependent status) at initiation of lenalidomide treatment.
The secondary outcomes measured included the number and percentage of patients with progression to AML, the occurrence of SPMs, AEs (haematological and non-haematological) and all-cause mortality. Safety outcomes were reported as documented by the registries. These results were stratified by on- or off-label usage. Progression to AML, as well as time-to-progression from the date of first lenalidomide treatment to the date of diagnosis of progression to AML, was captured. The time-to-progression analyses were conducted by the individual registries. SPMs other than AML were categorised as haematological malignancies, non-melanoma skin cancer and solid tumours. Types of AEs evaluated included bleeding events, infections, major cardiac events, neutropenia, thrombocytopenia and venous thromboembolic events. AEs were further summarised by seriousness for the EUMDS and Spanish registries or by AE severity per the Common Terminology Criteria for Adverse Events (CTCAE) grading for the French and Düsseldorf registries.
Statistical analysis
Baseline characteristics, progression to AML, occurrence of SPMs other than AML, AEs and all-cause mortality were summarised as counts and proportions for categorical data and as means, standard deviations (SD), medians, minimums and maximums for continuous variables.
Progression to AML was summarised for each on- and off-label use cohort, employing Cox Proportional Hazard regression methods to estimate hazard ratios (HRs), with 95% confidence intervals (CIs) calculated separately for each registry and then by pooled HR. The number of SPMs, AEs and all-cause mortality were summarised by on- or off-label lenalidomide treatment group. Specific types of SPM, AE, AE severity and cause of death were also recorded by label status.
This study leveraged the secondary use of data. No individual-level patient data were collected, and all analyses were based on data supplied by the registries in an aggregated form. All statistical analyses were conducted in SAS® Version 9.4 or higher (Cary, North Carolina). Registries that provided the data may have used Microsoft Excel, R or other statistical computing software.
Ethics approval
Ethical approval for the study was obtained from the Independent Ethics Committees of Vall d’Hebron University Hospital’s Clinical Research Ethics Committee (approval of last protocol update on 22 September 2023) and the National Ethics Committee for Clinical Research (Reference Number: 2022_EO_23; approval of last protocol update on 12 April 2024). The need for informed consent was waived by the Institutional Review Board (IRB) due to the retrospective nature of the study. The data used in this study were de-identified, ensuring patient confidentiality and compliance with applicable data protection regulations. The final study report was submitted to the EMA and included detailed information on data sources and ethical considerations.
Results
Patient disposition and baseline characteristics
There were 30,686 patients across the four included registries, of whom 1273 received at least one dose of lenalidomide (Figure 1). Of these, 750 patients were excluded due to ineligibility for one or more of the following reasons: 35 (4.7%) received lenalidomide in a clinical trial, 687 (91.6%) were treated with lenalidomide before EC MDS approval and 30 (4.0%) due to an unknown date of treatment initiation. Ultimately, 523 patients treated with lenalidomide in routine clinical practice were included in this analysis.
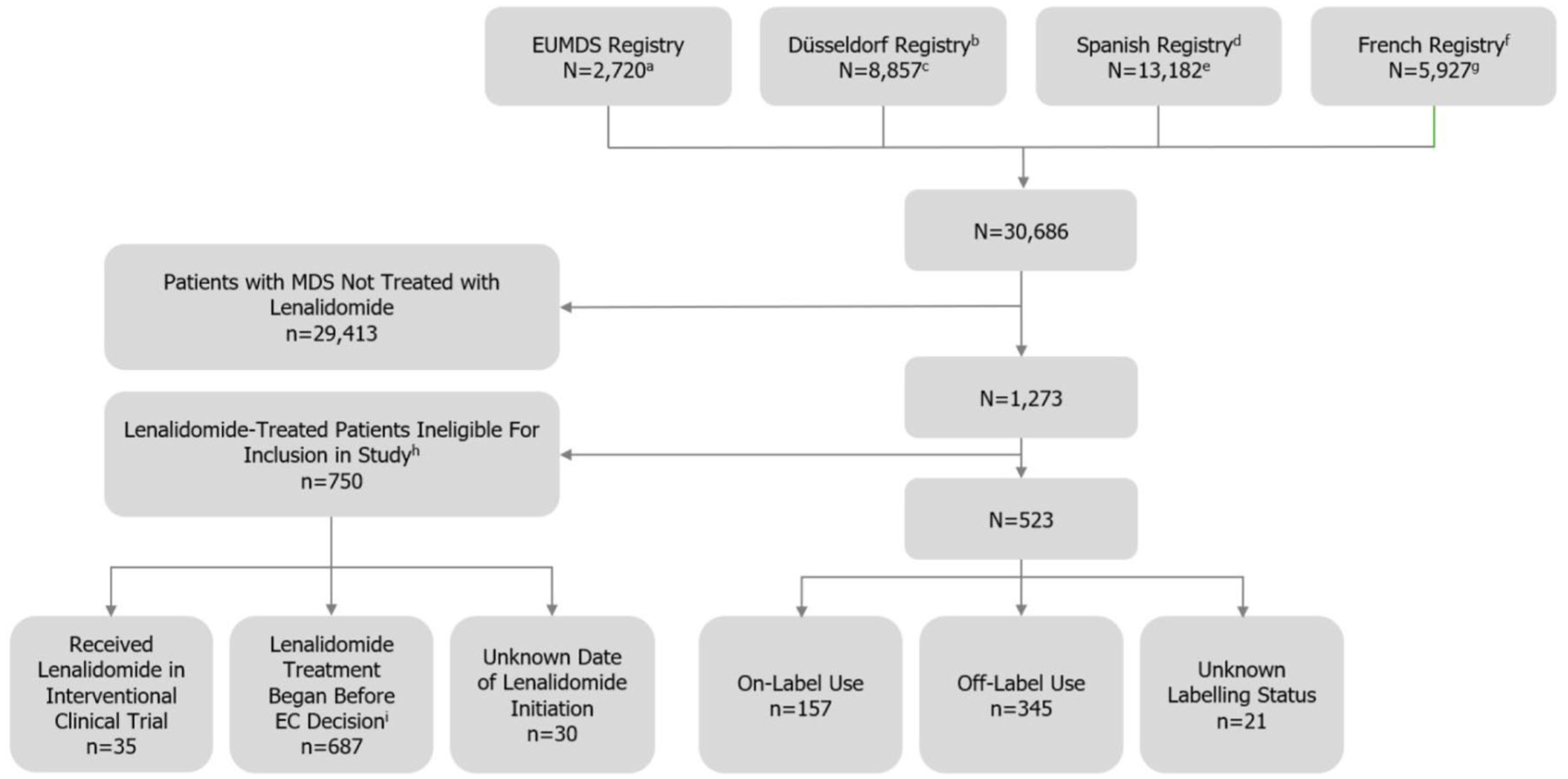
Flow diagram of MDS registries and patients included in the study.
The mean (SD) age of included patients was 72.3 (11.01) years (range: 23–96 years). Similar mean ages were observed across the individual registries. Most patients in each registry were female, with 330 (63.1%) of all patients included in the study reporting female sex (Table 1).
Patterns of lenalidomide use
The majority (
Safety outcomes
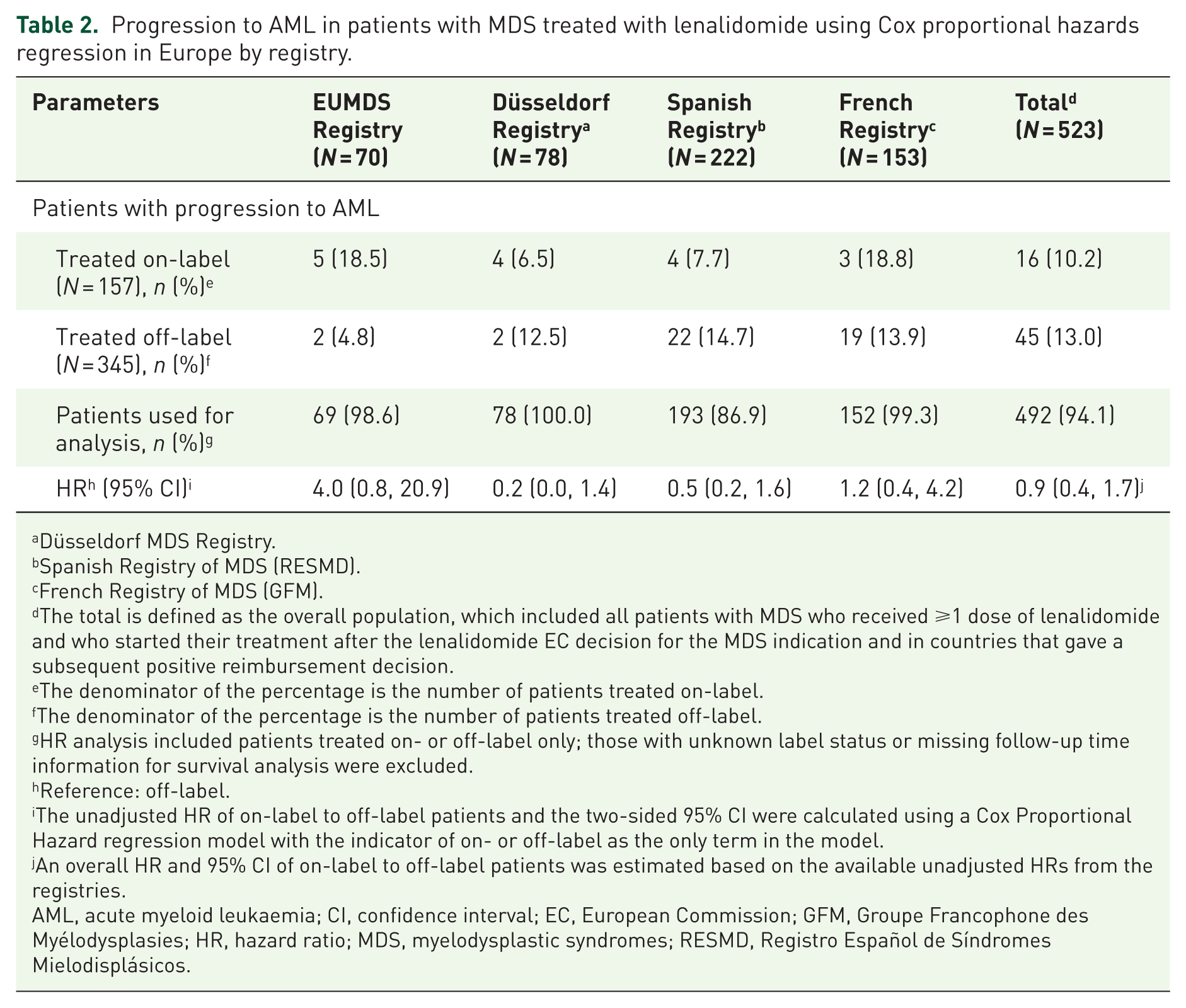
In the overall population, 10.2% (n=16) of on-label and 13.0% (n=45) of off-label patients with MDS progressed to AML over the study follow-up period (Table 2). Across all patients, there was no statistical difference in progression to AML between those prescribed lenalidomide on-label versus off-label (HR = 0.9; 95% CI: 0.4, 1.7). The HRs (95% CI) were stratified by registry, showing 4.0 (0.8, 20.9) among EUMDS registry patients, 0.2 (0.0, 1.4) among Düsseldorf registry patients, 0.5 (0.2, 1.6) among Spanish registry patients and 1.2 (0.4, 4.2) among French registry patients; all were non-significant.
Progression to AML in patients with MDS treated with lenalidomide using Cox proportional hazards regression in Europe by registry.
Düsseldorf MDS Registry.
Spanish Registry of MDS (RESMD).
French Registry of MDS (GFM).
The total is defined as the overall population, which included all patients with MDS who received ⩾1 dose of lenalidomide and who started their treatment after the lenalidomide EC decision for the MDS indication and in countries that gave a subsequent positive reimbursement decision.
The denominator of the percentage is the number of patients treated on-label.
The denominator of the percentage is the number of patients treated off-label.
HR analysis included patients treated on- or off-label only; those with unknown label status or missing follow-up time information for survival analysis were excluded.
Reference: off-label.
The unadjusted HR of on-label to off-label patients and the two-sided 95% CI were calculated using a Cox Proportional Hazard regression model with the indicator of on- or off-label as the only term in the model.
An overall HR and 95% CI of on-label to off-label patients was estimated based on the available unadjusted HRs from the registries.
AML, acute myeloid leukaemia; CI, confidence interval; EC, European Commission; GFM, Groupe Francophone des Myélodysplasies; HR, hazard ratio; MDS, myelodysplastic syndromes; RESMD, Registro Español de Síndromes Mielodisplásicos.
Overall, the occurrence of SPMs other than AML was somewhat more common in the on-label group. Solid tumours were the most common SPM, occurring in 11 (7.0%) on-label and 6 (1.7%) off-label patients, followed by haematological malignancies in 1 (0.6%) on-label patient and 2 (0.6%) off-label patients (Table 3). Lastly, non-melanoma skin cancers occurred in 1 (0.6%) on-label and 1 (0.3%) off-label patient.
SPMs other than AML in the overall population of patients with MDS treated with lenalidomide on- and off-label in Europe.a,b

The ‘overall population’ included all patients with MDS who received ⩾1 dose of lenalidomide either on- or off-label and who started their treatment after the lenalidomide EC decision for the MDS indication and in countries that gave a subsequent positive reimbursement decision.
SPM term is presented as originally reported by the registries.
AML, acute myeloid leukaemia; EC, European Commission; MDS, myelodysplastic syndromes; SPM, second primary malignancy.
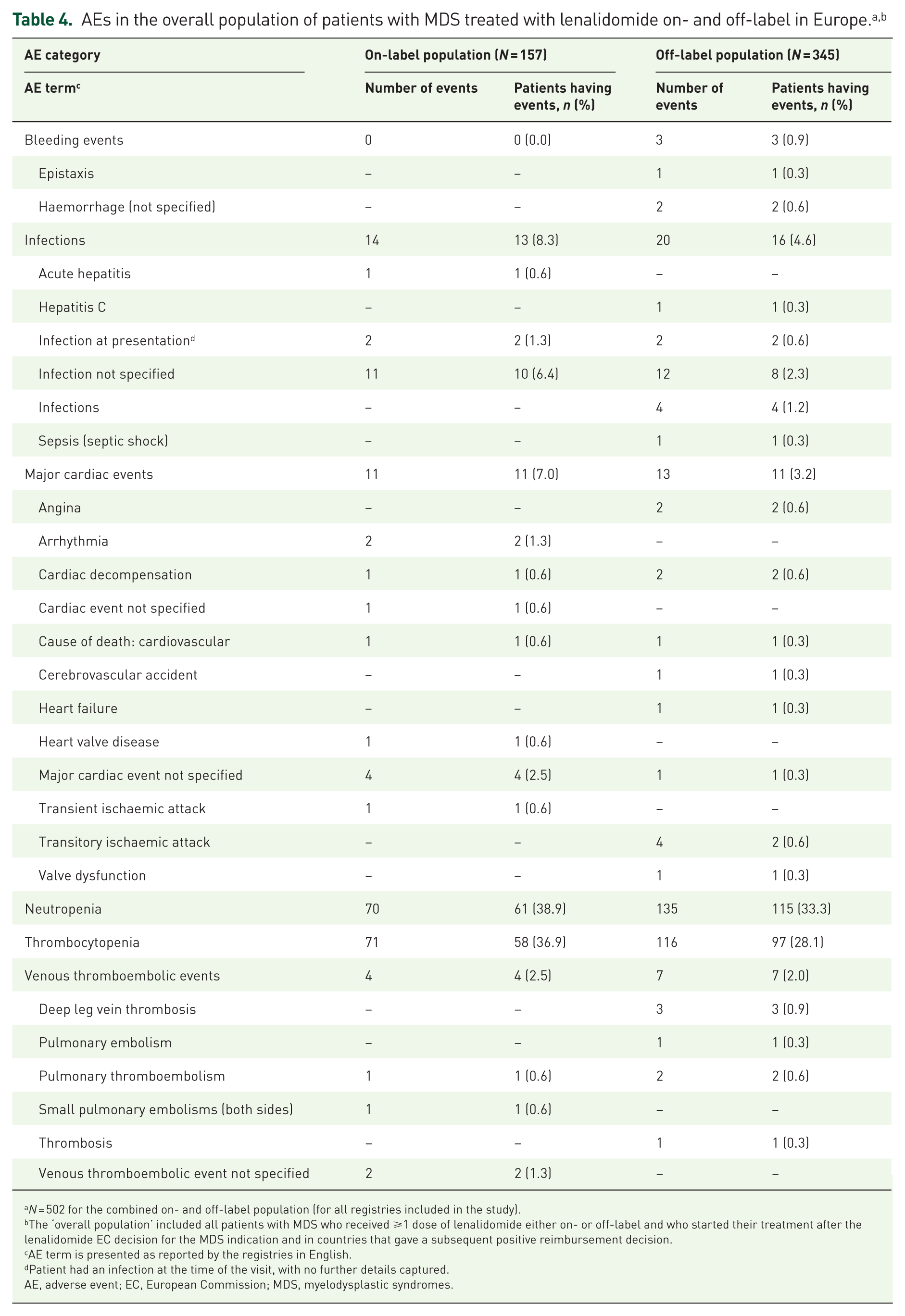
Of the specific AE categories evaluated, the most common AEs were neutropenia (61 (38.9%) on-label and 115 (33.3%) off-label patients) and thrombocytopenia (58 (36.9%) on-label and 97 (28.1%) off-label patients; Table 4). Infections (13 (8.3%) on-label and 16 (4.6%) off-label) and major cardiac events (11 (7.0%) on-label and 11 (3.2%) off-label) were the next most common AEs. Bleeding events (0 (0%) on-label and 3 (0.9%) off-label) and venous thromboembolic events (4 (2.5%) on-label and 7 (2.0%) off-label) were the least common AEs. Data regarding the AEs (by seriousness in the Spanish and EUMDS registries and by CTCAE grade in the French and Düsseldorf registries) are detailed in Supplemental Tables 1–8.
AEs in the overall population of patients with MDS treated with lenalidomide on- and off-label in Europe.a,b
The ‘overall population’ included all patients with MDS who received ⩾1 dose of lenalidomide either on- or off-label and who started their treatment after the lenalidomide EC decision for the MDS indication and in countries that gave a subsequent positive reimbursement decision.
AE term is presented as reported by the registries in English.
Patient had an infection at the time of the visit, with no further details captured.
AE, adverse event; EC, European Commission; MDS, myelodysplastic syndromes.
A total of 128 patients across the on-label (
Patients with MDS in Europe who died during on- and off-label lenalidomide treatment in the overall population.a,b

The ‘overall population’ included all patients with MDS who received ⩾1 dose of lenalidomide either on- or off-label and who started their treatment after the lenalidomide EC decision for the MDS indication and in countries that gave a subsequent positive reimbursement decision.
The denominator of the percentage is the total on-label or off-label population (
The cause of death term is presented as reported by the registries in English.
Not all causes of death were reported as AEs in the Spanish Registry of MDS (RESMD) and may have been captured during or after lenalidomide treatment.
MDS progressed to AML, and death was due to AML.
Not AML or unknown if AML.
AE, adverse event; AML, acute myeloid leukaemia; COVID-19, coronavirus disease 2019; EC, European Commission; GI, gastrointestinal; GVHD, graft-versus-host disease; MDS, myelodysplastic syndromes; MM, multiple myelomas; RESMD, Registro Español de Síndromes Mielodisplásicos; SCT, stem cell transplant.
Discussion
This study investigated patterns of lenalidomide routine clinical use in the treatment of MDS as well as safety outcomes, including progression to AML, development of SPMs other than AML, occurrence of AEs and all-cause mortality, for patients with MDS receiving lenalidomide within and outside of the EC-approved indication. As such, this study provides important insights about the real-world use and safety of lenalidomide in the treatment of MDS.
The majority of patients with MDS who were prescribed lenalidomide received it off-label, defined largely by a lack of transfusion dependence or lack of isolated del(5q) aberration. This was true for patients across all registries included in the study, apart from the Düsseldorf Registry, where the majority of patients were treated on-label. The results of this study corroborate previous findings that lenalidomide is commonly used outside of transfusion-dependent Low- or Int-1-risk del(5q) MDS in real-world practice. 33
Such findings may be explained in part by the use of data from registries at specialty or tertiary care centres for haematologic diagnoses. Physicians at these centres may either be more comfortable using lenalidomide off-label due to the volume of patients treated or have patients with the most difficult-to-treat disease presenting at these sites, which necessitates less traditional treatment approaches. As a result, the findings of this study may represent an upper bound of the true proportion of off-label use in real-world practice. Additionally, in this study, transfusion dependence was defined as requiring ⩾2 RBC units over an 8-week period prior to lenalidomide initiation. Prescribers may have considered their patients transfusion-dependent based on other definitions, such as the World Health Organization’s classification-based Prognostic Scoring System (WPSS) or the International Working Group’s definition, but these were considered off-label for this study.34,35
Regarding lenalidomide’s safety profile, the incidence of progression to AML was similar to that observed in previous studies. Prior research has suggested that lenalidomide does not increase the risk of progression to AML in its approved indication above what would be considered the natural progression of the disease.20,22 The observed incidence of progression to AML (10.2%–13.0%) in this study did not exceed previously reported estimates (30%–40%).1,2,12,13 Furthermore, the overall incidence of progression to AML did not notably differ between patients treated on- or off-label. Although the proportion of each treatment group progressing to AML varied between registries in this study, the HR and 95% CI across all registries pointed to null differences between on- and off-label treatment.
Given that SPMs are a notable non-MDS-related cause of death in patients with MDS, it was important to investigate their occurrence in lenalidomide-treated patients in the real world.36–38 Overall, the incidence of SPMs other than AML in our study was low (⩽7%). Although there was a higher incidence of solid tumours among on-label patients compared with off-label patients, minimal differences were found between label status groups in regard to other SPMs, suggesting no additional risk of SPMs from off-label versus on-label lenalidomide use.
The occurrence of haematologic and non-haematologic AEs was also similar between on- and off-label patients. In line with prior studies, the most common AEs were neutropenia and thrombocytopenia.16,17 The incidence of other types of AEs was low in both cohorts overall, providing further evidence of the tolerable safety profile of lenalidomide use both on- and off-label.
Despite the low survival rates generally expected among patients with MDS, the majority of both on- and off-label patients in this study survived to the end of follow-up. Among those who died, the most common causes of death were AML, infection or unknown cause, which correspond to previous findings for patients with MDS.36–38
This study benefited from several strengths. It offers a perspective on the long-term safety of lenalidomide for patients with MDS based on patient data spanning nearly a decade across 11 European countries. Patient data from multiple registries across these countries provided insight into the patterns of use of lenalidomide in the real world. These data offer valuable insights about the number and characteristics of patients receiving treatment off-label, as well as the safety profile of lenalidomide within this population.
However, several limitations must also be considered. The registries that provided patient data for this analysis may differ in their primary purposes, patient characteristics and availability of specific information. Therefore, the potential inter-registry heterogeneity may limit the interpretability of the results. This may be particularly pertinent regarding AEs, as not all of the registries collect data on all types of AEs, potentially leaving some events unreported within the study population. Additionally, the regression analysis for progression to AML could not be adjusted for relevant confounders because of an insufficient number of AML cases for the analysis. Lastly, due to the secondary nature of the data analysed, there were varying degrees of missing or incomplete data.
Conclusion
The observed real-world patterns of lenalidomide treatment in this study demonstrated highly prevalent use outside of the EC-approved indication, underscoring the importance of including patients with MDS outside of transfusion-dependent del(5q) Low- or Int-1 MDS in research on lenalidomide. Furthermore, there was no evidence to suggest any new safety concerns for lenalidomide use in MDS, and there remains no change to the existing benefit–risk profile. Across the registries studied, the similar incidence of safety outcomes between on- and off-label groups suggests that the benefits of lenalidomide already documented in the transfusion-dependent IPSS Low- or Int-1-risk MDS with del(5q) indication may exist in other groups of patients with MDS. These results may have important clinical implications for the treatment of other types of MDS as well as for future research directions to further explore this area.
Supplemental Material
sj-doc-2-taw-10.1177_20420986251407181 – Supplemental material for Evaluating the patterns of use and safety of lenalidomide treatment in myelodysplastic syndromes: a European, observational multi-registry study
Supplemental material, sj-doc-2-taw-10.1177_20420986251407181 for Evaluating the patterns of use and safety of lenalidomide treatment in myelodysplastic syndromes: a European, observational multi-registry study by Iris Kim, Aidan Makwana, Victoria Ezenma, Stefania Navarcikova, Muskan Mittal, Quinn Ho, Hannah Borda and Stefan Kaehler in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-1-taw-10.1177_20420986251407181 – Supplemental material for Evaluating the patterns of use and safety of lenalidomide treatment in myelodysplastic syndromes: a European, observational multi-registry study
Supplemental material, sj-docx-1-taw-10.1177_20420986251407181 for Evaluating the patterns of use and safety of lenalidomide treatment in myelodysplastic syndromes: a European, observational multi-registry study by Iris Kim, Aidan Makwana, Victoria Ezenma, Stefania Navarcikova, Muskan Mittal, Quinn Ho, Hannah Borda and Stefan Kaehler in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The authors would like to thank all of the investigators, their clinical staff, the registries that provided the data for this analysis, and the patients and their caregivers who contributed to this study. The authors also acknowledge Zivile Zakaite, MPH, of Bristol Myers Squibb, Boudry, Switzerland, for operational support as well as Lee Hughes, MS, and Steph Beaver, MChem, of Costello Medical Inc., Boston, MA, USA, for medical writing and editorial assistance based on the authors’ input and direction. Support for third-party medical writing was funded by Bristol Myers Squibb.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
