Abstract
Background:
Cardiovascular adverse events represent critical complications of antineoplastic therapy with profound implications for cancer survivorship and treatment outcomes. Despite the clinical significance, comprehensive pharmacovigilance data characterizing distinctive cardiotoxicity profiles across modern cancer therapeutics remain limited.
Objectives:
This investigation systematically analyzes cardiotoxicity patterns associated with antineoplastic agents using the FDA Adverse Event Reporting System (FAERS) database to inform evidence-based cardiovascular monitoring strategies.
Design:
A retrospective pharmacovigilance study utilizing disproportionality analysis and time-to-onset evaluation.
Methods:
We conducted a comprehensive analysis of FAERS data spanning 2004–2024, employing validated disproportionality metrics including reporting odds ratio (ROR) and proportional reporting ratio (PRR) to detect significant drug-event associations. Advanced time-to-onset analysis revealed temporal patterns of cardiotoxicity development across therapeutic classes. Statistical significance was defined as ROR >1 with 95% confidence intervals excluding 1.0, and PRR >2 with chi-square >4.
Results:
Analysis of 18,289,374 reports identified 51,402 cases of antineoplastic-related cardiovascular toxicity, demonstrating distinct class-specific patterns. Anthracyclines exhibited profound associations with structural cardiac damage (doxorubicin-cardiomyopathy: ROR = 20.64, 95% CI: 19.87–21.45). Immune checkpoint inhibitors demonstrated unprecedented immune-mediated cardiac inflammation (pembrolizumab-myocarditis: ROR = 245.36, 95% CI: 218.42–275.88). Fluoropyrimidines showed distinctive vasospastic effects (5-fluorouracil-Prinzmetal angina: ROR = 18.27, 95% CI: 14.72–22.69). Critical temporal patterns emerged: fluoropyrimidines caused early-onset cardiotoxicity (median: 11 days, IQR: 4–28), anthracyclines showed intermediate onset (doxorubicin median: 64 days, IQR: 21–156; epirubicin median: 72 days, IQR: 28–168), while mitoxantrone demonstrated delayed effects (median: 457 days, IQR: 182–891). Cardiogenic shock emerged as the most lethal manifestation with a 43.08% mortality rate (95% CI: 40.12–46.14).
Conclusion:
This landmark pharmacovigilance study reveals previously uncharacterized temporal and mechanistic patterns of antineoplastic cardiotoxicity, providing an essential evidence-based framework for cardiovascular monitoring strategies. The findings highlight critical intervention windows: immediate monitoring for fluoropyrimidines, intermediate surveillance for anthracyclines (2–6 months), and extended follow-up for agents like mitoxantrone (>12 months). These insights support the development of risk-stratified cardio-oncology protocols tailored to specific therapeutic classes.
Plain language summary
Cancer treatments can sometimes cause serious heart problems, but doctors need better information about which drugs cause which heart issues and when they occur. This study looked at over 18 million safety reports sent to the FDA between 2004 and 2024 to understand patterns of heart problems caused by cancer drugs.
The researchers found more than 51,000 reports of heart problems linked to cancer medications. Different types of cancer drugs caused different heart problems at different times:
• Anthracyclines (like doxorubicin) - older chemotherapy drugs that can permanently damage the heart muscle, typically causing problems after 2-5 months of treatment
• Immune checkpoint inhibitors (like pembrolizumab) - newer immunotherapy drugs that can cause dangerous heart inflammation, often within the first few weeks
• Fluoropyrimidines (like 5-fluorouracil) - drugs that can cause heart artery spasms, usually within the first 1-4 weeks
• Mitoxantrone - a drug that can cause heart problems much later, often after more than a year
The study found that cardiogenic shock (when the heart can’t pump enough blood) was the most dangerous heart problem, with nearly half of patients dying from it.
These findings help doctors know when to watch patients most carefully for heart problems. For example, patients getting fluoropyrimidines need immediate heart monitoring, while those getting anthracyclines need regular heart checks for several months, and patients on mitoxantrone need long-term follow-up for over a year.
Keywords
Introduction
With global cancer incidence continuing to rise, with over 20 million new cases projected for 2024, the scope and intensity of antineoplastic drug use have expanded accordingly. 1 Modern cancer therapeutics have achieved remarkable progress, with 5-year survival rates improving by nearly 30% over the past two decades. 2 However, this success casts a growing shadow: cardiotoxicity has emerged as a primary health threat for cancer survivors.3,4 Cardiotoxicity represents a particularly formidable clinical challenge in cancer management, encompassing a spectrum of cardiovascular adverse events (AEs) ranging from asymptomatic cardiac dysfunction to life-threatening conditions such as cardiogenic shock, acute myocardial infarction, and severe heart failure.4–6 These complications not only compromise treatment efficacy by necessitating dose reductions or treatment discontinuations but also substantially increase morbidity and mortality among cancer survivors. 6
Different classes of antineoplastic agents exhibit distinctive cardiotoxicity profiles. Anthracyclines, including doxorubicin and epirubicin, have been extensively documented to cause dose-dependent cardiomyopathy and left ventricular dysfunction through multiple mechanisms, including oxidative stress and mitochondrial damage.7–9 HER2-targeted therapies such as trastuzumab demonstrate unique patterns of cardiac dysfunction that may be reversible upon treatment discontinuation.5,10 More recently, as immune checkpoint inhibitors have been incorporated into first-line treatment regimens for an increasing number of cancer types, their rare but potentially fatal immune-mediated myocarditis and pericarditis have presented novel challenges in the field of cardio-oncology.11,12
Despite the clinical significance of cardiovascular complications in cancer treatment, comprehensive characterization of cardiotoxicity across the full spectrum of modern antineoplastic agents remains incomplete. 13 Existing studies are often limited by small sample sizes, focus on specific drug classes, or lack comparative assessments between different therapeutic modalities. Real-world evidence from large pharmacovigilance databases offers a valuable opportunity to assess the comparative incidence, patterns, and clinical presentations of cardiovascular adverse events across diverse antineoplastic agents.
The FDA Adverse Event Reporting System (FAERS) represents one of the largest global repositories of post-marketing pharmacovigilance data, containing millions of adverse event reports submitted by healthcare professionals, consumers, and manufacturers.14,15 Analysis of this database can provide valuable insights into the real-world cardiotoxicity profiles of antineoplastic agents that may not be fully captured in premarketing clinical trials due to their limited sample sizes and strict eligibility criteria.
This study aims to address this knowledge gap by conducting a comprehensive pharmacovigilance analysis of cardiovascular adverse events associated with antineoplastic agents using the FAERS database from 2004 to 2024. Through signal detection methodologies including disproportionality analysis, time-to-onset assessment, and mortality analysis, we seek to characterize the incidence, patterns, clinical presentation, and severity of cardiotoxicity across different classes of anticancer drugs. Our findings will inform the development of individualized cardiac monitoring strategies and guide clinical decision-making in cardio-oncology practice.
Methods
Study design and data source
This investigation represents a retrospective pharmacovigilance analysis utilizing the FAERS database. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 16 FAERS functions as a comprehensive repository containing over 18 million spontaneous reports of adverse events, medication errors, and product quality concerns submitted worldwide by healthcare providers, pharmaceutical companies, and patients since 1969, with quarterly public data releases available through the FDA’s official website. These submissions undergo evaluation by regulatory reviewers at the FDA. Each report typically includes identification numbers, suspected medications, treatment indications, adverse reactions, seriousness classification, outcomes (including hospitalization or mortality), demographic information (gender, age, and weight), chronological data (event and receipt dates), reporting source, concomitant medications, and geographical origin, while excluding personal identifiers to maintain patient confidentiality in accordance with FDA regulations (Table 1).
Patient demographics and reporting characteristics.

Study period and case selection
Our analysis examined FAERS database reports from January 2004 through December 2024, focusing on cardiovascular adverse events associated with antineoplastic agents. Data extraction was performed using standardized FDA case processing software with systematic duplicate removal protocols based on case identification numbers, FDA_DT (FDA receipt date), and MFGDT (manufacturer control number), following established FAERS cleaning procedures. After eliminating duplicates, we identified 51,402 relevant cases from a total database of 18,289,374 reports, representing 4.62% of all antineoplastic agent adverse reactions during this period.
Inclusion criteria: (1) Reports submitted to FAERS between January 1, 2004, and December 31, 2024. (2) Antineoplastic agent listed as “primary suspect” drug. (3) At least one cardiovascular adverse event coded using Medical Dictionary for Regulatory Activities (MedDRA) Preferred Terms (PTs). (4) Sufficient data quality (non-missing drug name and adverse event term). Exclusion criteria: (1) Duplicate reports (identified by identical CASEID, FDA_DT, and MFGDT). (2) Reports with antineoplastic agents listed only as concomitant medications. (3) Reports lacking identifiable cardiovascular MedDRA terms. (4) Drugs with <50 total cardiovascular event reports (insufficient for meaningful statistical analysis).
This study analyzed all available cases in the FAERS database meeting inclusion criteria during the study period (N = 51,402), representing a complete enumeration rather than a sample. No a priori sample size calculation was performed, as this retrospective analysis utilized the entire population of reported cases. The large sample size (>50,000 cardiovascular events) provides adequate statistical power (>99%) to detect even modest associations (ROR ⩾1.5) with commonly used agents. However, statistical power is limited for rare drug-event combinations with <10 reported cases, which are flagged as exploratory findings throughout the manuscript.
Drug classification and categorization
The medications were systematically classified into five therapeutic categories based on their primary mechanism of action and FDA-approved indications: conventional chemotherapy (N = 11,218, including anthracyclines, alkylating agents, antimetabolites, and topoisomerase inhibitors), monoclonal antibody therapies (N = 8133, including HER2-targeted and VEGF-targeted agents), small molecule targeted agents (N = 5422, including tyrosine kinase inhibitors and mTOR inhibitors), proteasome inhibitors (N = 3295), and immunomodulators (N = 14,444, including lenalidomide, thalidomide, and pomalidomide), and immune checkpoint inhibitors (N = 8890, including PD-1, PD-L1, and CTLA-4 inhibitors). Drug selection criteria included: (1) FDA approval for oncological indications, (2) availability of adequate reporting data (⩾50 cardiovascular events), and (3) sufficient temporal data for time-to-onset analysis.
Cardiovascular event definition and classification
We examined cardiovascular events categorized according to MedDRA PTs, analyzing those with substantial reporting frequencies (Table S2). Cardiovascular events were classified based on clinical presentation and included major categories such as: structural heart disease (cardiomyopathy, left ventricular dysfunction), rhythm disorders (atrial fibrillation, arrhythmias), acute coronary syndromes (myocardial infarction, coronary artery disorders), heart failure syndromes (cardiac failure, cardiogenic shock), inflammatory conditions (myocarditis, pericarditis, and pericardial effusion), and specific drug-related manifestations (immune-mediated cardiotoxicity, vasospastic disorders). Event severity was classified using FDA seriousness criteria: serious events included death, life-threatening conditions, hospitalization, disability, congenital anomaly, or other medically significant events requiring intervention. The analysis focused on the most commonly reported cardiovascular events with sufficient case numbers to enable meaningful statistical analysis. A comprehensive list of all MedDRA PTs used for cardiovascular event identification is provided in Table S2, which includes detailed PT codes, names, and their classification into the aforementioned categories.
Statistical analysis
Disproportionality analysis
To assess potential associations between specific antineoplastic agents and cardiovascular toxicities, researchers conducted a comprehensive disproportionality analysis using multiple validated pharmacovigilance metrics. We employed a three-metric validation approach to ensure robust and reliable signal detection, combining complementary statistical measures that address different aspects of drug-event associations. The three-metric validation strategy was designed based on the distinct advantages and limitations of each indicator: (1) Reporting odds ratio (ROR) provides an unbiased estimate of relative risk and is particularly sensitive to rare events, but may demonstrate instability in small sample sizes; (2) Proportional reporting ratio (PRR) effectively identifies drug-event combinations with disproportionately elevated reporting frequencies and is robust for detecting strong signals, but may have reduced sensitivity for weaker associations; (3) Chi-square testing offers statistical significance assessment and helps control for random variation, though it does not provide information about effect magnitude. The integrated use of these three metrics enhances both the sensitivity for detecting true signals and the specificity for minimizing false-positive associations.
The analysis employed three primary disproportionality metrics to ensure robust signal detection. The ROR was calculated as (a/b)/(c/d), where “a” represents cases with both the drug and the event of interest, “b” represents cases with the drug but without the event, “c” represents cases with the event but without the drug, and “d” represents cases with neither the drug nor the event. The PRR was calculated as [a/(a + b)]/[c/(c + d)], representing the proportion of specific adverse events for a particular drug compared to all other drugs. In addition, chi-square tests were applied to assess the statistical significance of the associations.
Signal detection criteria
In the present study, an adverse event was considered a potential signal when at least one of four algorithms met the positive threshold. Signal significance was established using validated thresholds that align with established European Medicines Agency and World Health Organization (WHO) pharmacovigilance guidelines for signal detection. The primary criteria required an ROR greater than 1.0 with the lower boundary of the 95% confidence interval exceeding 1.0, along with a minimum of three reported cases for all drug-event combinations. For PRR analysis, significance was defined as PRR greater than 2.0 with a chi-square value greater than 4.0. However, the primary signal detection was based on ROR criteria, as this represents the most robust metric for pharmacovigilance analysis, ensuring the reliability and validity of detected safety signals (Table 2 and Table S1). Signals based on fewer than 10 events were flagged as exploratory findings requiring clinical confirmation due to wide confidence intervals and statistical uncertainty from limited case numbers.
Four major algorithms used for signal detection.

95% CI, 95% confidence interval; a, number of reports containing both the target drug and target adverse drug reaction; b, number of reports containing other adverse drug reaction of the target drug; c, number of reports containing the target adverse drug reaction of other drugs; d, number of reports containing other drugs and other adverse drug reactions; EBGM, empirical Bayesian geometric mean; EBGM05, the lower limit of 95% CI of EBGM; E(IC), the IC expectations; IC, information component; IC025, the lower limit of 95% CI of the IC; N, the number of reports; PRR, proportional reporting ratio; ROR, reporting odds ratio; V(IC), the variance of IC; χ2, chi-squared.
Time-to-onset analysis
Time to onset was calculated as the interval between therapy initiation date (EVENT_DT) and adverse event onset date (FDA_DT) as recorded in FAERS reports. Cases were included in the temporal analysis only when both dates were available and chronologically plausible (adverse event onset date ⩾ therapy initiation date). Reports with missing dates, negative time intervals (onset date prior to initiation date), or implausible values (>3650 days or <0 days) were excluded from time-to-onset calculations.
Stratified and subgroup analyses
To assess the robustness and generalizability of our findings, we conducted four pre-specified subgroup analyses: (1) Temporal period analysis: Cases were stratified into two periods (2004–2016 vs 2017–2024) to evaluate consistency of signals over time. (2) Geographic analysis: Reports were categorized by country of origin into high-reporting countries (top seven countries by case volume) and low-reporting countries to assess regional reporting patterns and potential geographic differences in surveillance practices. (3) Sex-stratified analysis: Cardiovascular adverse events were analyzed separately for male and female patients to identify sex-specific toxicity patterns and differential susceptibility. (4) Age-stratified analysis: Cases were divided into three age groups (0–17.9 years (pediatric), 18–64.9 years (adult), ⩾65 years (elderly)) to characterize age-related risk profiles and inform age-appropriate monitoring strategies.
Comparative analysis
The study performed stratified analyses comparing multiple dimensions of cardiovascular adverse events. The analysis included reporting patterns between serious and non-serious cardiovascular events using chi-square tests with statistical significance set at p < 0.05, demographic differences including age, sex, and geographic distribution using descriptive statistics, and mortality rates calculated as the proportion of fatal outcomes among reported cardiovascular events with 95% confidence intervals. Serious adverse events were defined according to FDA criteria as events resulting in death, life-threatening conditions, hospitalization, significant disability, congenital anomaly, or other medically important conditions requiring intervention.
Data processing and quality control
All data processing was conducted using R statistical software (version 4.3.2) with specialized pharmacovigilance packages to ensure analytical rigor and reproducibility. Quality control measures included systematic verification of drug name standardization using WHO Drug Dictionary Enhanced (WHODDE), validation of MedDRA coding consistency to ensure accurate adverse event classification, temporal data integrity checks to verify chronological accuracy, and cross-validation of statistical calculations to prevent computational errors. The FAERS database extraction was performed on February 15, 2025, ensuring the analysis incorporated the most current available safety data for a comprehensive assessment of cardiovascular risks associated with antineoplastic therapy.
Results
Cardiovascular adverse events among antineoplastic agent users in FAERS from 2004 to 2024
Using the FAERS database, we investigated the incidence of cardiovascular AEs in patients receiving antineoplastic agents between 2004 and 2024. From a total of 18,289,374 AE reports, we identified 51,402 cases of antineoplastic agent-related cardiovascular AEs after excluding duplicates. These cardiovascular AEs constituted 4.62% (51,402/1,111,579) of all adverse reactions reported for antineoplastic agents (Table 3). The number of cardiovascular AE cases showed significant growth over the study period, increasing from 748 cases in 2004 to 3239 cases in 2024, with a peak of 4157 cases in 2019. As demonstrated in Figure 1, these antineoplastic agents were categorized into five main groups: chemotherapy drugs (N = 11,218), monoclonal antibody-based targeted therapies (N = 8133), small molecule targeted drugs (N = 5422), proteasome inhibitors (N = 3295), immunomodulators (N = 14,444), and immune checkpoint inhibitors (N = 8890).
Yearly distribution of cardiovascular AEs.

AE, adverse event.

Distribution of antineoplastic agents by therapeutic class associated with cardiovascular adverse events.
However, antineoplastic agents varied considerably in their association with cardiovascular toxicity. Among these, lenalidomide (10,764 cases), nivolumab (3708 cases), trastuzumab (3195 cases), and bevacizumab (3093 cases) had the highest absolute number of cardiovascular AE reports. When examined proportionally as illustrated in Figure 2, the three medications with the highest incidence of cardiovascular AEs were mitoxantrone (15.93% (280/1,758)), carfilzomib (10.01% (1342/13,406)), and doxorubicin (9.23% (2782/30,133)). The incidence of cardiovascular AEs for other commonly prescribed anticancer agents, including trastuzumab, bevacizumab, and lenalidomide, was comparatively lower at 7.43% (3195/43,007), 4.59% (3093/67,323), and 3.18% (10,764/338,814), respectively. These findings highlight the notable cardiotoxicity profile of anthracyclines and proteasome inhibitors compared to other classes of anticancer medications.

Proportional incidence of cardiovascular adverse events by antineoplastic agent.
Clinical characteristics of anticancer drug-related cardiovascular AEs
Based on a comprehensive analysis of the FAERS database, we identified 51,402 cases of cardiovascular AEs associated with anticancer drugs listed as the “primary suspect.” Demographic analysis revealed significant gender disparities, with cytotoxic agents like doxorubicin (49.7% females vs 31.1% males) and especially epirubicin (79.9% females vs 10.6% males) showing female predominance, while other drugs such as cetuximab affected males more frequently (68.4% vs 25.5%) (Table 1). Age distribution analysis demonstrated that most cardiovascular toxicities occurred in adults aged 18–64.9 and 65–85 years, exemplified by paclitaxel (43.5% and 39.9% in these age groups, respectively). Pediatric cases were uncommon, with doxorubicin being a notable exception (6.2% of cases in patients <18 years). Weight data, though frequently incomplete, showed most patients fell within the 50–100 kg range. Geographic reporting patterns varied considerably, with the United States contributing the majority of reports for several drugs, including lenalidomide (82.7%), pomalidomide (82.3%), and doxorubicin (33.4%), while Japan provided substantial data for immune checkpoint inhibitors (19.9% of nivolumab and 30.1% of pembrolizumab cardiovascular toxicity reports). Notably, targeted therapies demonstrated distinct demographic patterns, with HER2-targeted agents such as trastuzumab primarily affecting females (78.9%), consistent with their predominant use in breast cancer treatment.
Disproportionality analysis for antineoplastic agent-related cardiovascular AEs
The signal values and associations between antineoplastic agents and cardiovascular AEs are shown in Table 4. Mitoxantrone (N = 280, ROR = 4.15, 95% CI: 3.75–4.6, PRR = 3.93, χ2 = 888.45), carfilzomib (N = 1342, ROR = 2.58, 95% CI: 2.45–2.7, PRR = 2.5, χ2 = 1568.84), and doxorubicin (N = 2782, ROR = 2.31, 95% CI: 2.24–2.39, PRR = 2.26, χ2 = 2670.08) exhibited the strongest association with cardiovascular toxicity. Other agents with significant signal values included epirubicin (N = 577, ROR = 2.06, 95% CI: 1.91–2.22, PRR = 2.02, χ2 = 365.63), 5-fluorouracil (N = 1676, ROR = 2.09, 95% CI: 2–2.18, PRR = 2.05, χ2 = 1223.52), and tremelimumab (N = 3, ROR = 2.6, 95% CI: 1.15–5.9, PRR = 2.53, χ2 = 5.64) (exploratory finding based on limited case numbers). In contrast, docetaxel (N = 1440, ROR = 0.55, 95% CI: 0.52–0.58, PRR = 0.55, χ2 = 653.19), panitumumab (N = 302, ROR = 0.61, 95% CI: 0.54–0.67, PRR = 0.61, χ2 = 85.51), and lapatinib (N = 332, ROR = 0.65, 95% CI: 0.58–0.71, PRR = 0.65, χ2 = 74.2) showed lower than expected incidence of cardiovascular adverse events.
Disproportionality analysis results for antineoplastic agent-related cardiovascular adverse events.

AE, adverse event; EBGM, empirical Bayesian geometric mean; EBGM05, the lower limit of 95% CI of EBGM; IC, information component; IC025, the lower limit of 95% CI of the IC; PRR, proportional reporting ratio; ROR, reporting odds ratio.
A total of 1290 positive signals at the PT level were identified (Table S3). Doxorubicin showed particularly strong signals for cardiomyopathy (ROR = 20.64, 95% CI: 18.8–22.66), left ventricular dysfunction (ROR = 20.32, 95% CI: 17.63–23.43), and systolic dysfunction (ROR = 17.61, 95% CI: 12.01–25.83). Epirubicin exhibited notable signals for cardiac dysfunction (ROR = 53.11, 95% CI: 40.45–69.71) and cardiac perfusion defect (ROR = 216.17, 95% CI: 66.8–699.59). For immune checkpoint inhibitors, the strongest signals were observed for immune-mediated myocarditis with pembrolizumab (ROR = 245.36, 95% CI: 213.87–281.49), immune-mediated pericarditis with cemiplimab (ROR = 364.12, 95% CI: 49.64–2670.69), and autoimmune pericarditis with avelumab (ROR = 275.54, 95% CI: 102.23–742.66). The most commonly reported cardiovascular AEs were atrial fibrillation (N = 7453, ROR = 1.61, 95% CI: 1.57–1.65), cardiac failure (N = 6570, ROR = 1.74, 95% CI: 1.69–1.78), and congestive cardiac failure (N = 4560, ROR = 1.08, 95% CI: 1.05–1.12).
Detailed assessment of drug-specific cardiovascular toxicity profiles revealed distinctive patterns among antineoplastic classes (Figure 3), with mitoxantrone associated with restrictive cardiomyopathy (ROR = 35.59) and ventricular flutter (ROR = 156.4); anthracyclines linked to acute cardiotoxicity, including doxorubicin with acute cardiomyopathy (ROR = 63.86) and epirubicin with immune-mediated pericarditis (ROR = 301.1); fluoropyrimidines demonstrating vasospastic effects with 5-fluorouracil strongly associated with Prinzmetal angina (ROR = 18.27) and coronary artery dissection (ROR = 11.72); targeted therapies showing unique profiles with carfilzomib linked to cardiac failure (ROR = 6.23) and cardiac amyloidosis (ROR = 26.1), while anti-HER2 agents trastuzumab and pertuzumab primarily affected ventricular structure and function through ventricular dysfunction (ROR = 17.2), dyssynchrony (ROR = 27.65), and remodeling (ROR = 34.75); immunotherapies predominantly causing immune-mediated cardiac phenomena with exceptionally high signals for pembrolizumab with immune-mediated myocarditis (ROR = 245.36) and pericarditis (ROR = 469.54), and similar patterns for nivolumab and cemiplimab; multiple myeloma therapies revealing distinctive patterns with bortezomib associated with cardiac amyloidosis (ROR = 37.05) and hypersensitivity myocarditis (ROR = 56.61), while thalidomide linked to Adams-Stokes syndrome (ROR = 25.06); and several unexpected associations including sunitinib with cardiopulmonary failure (ROR = 17.04), all highlighting the importance of drug-specific cardiovascular monitoring strategies and suggesting potential mechanisms, risk factors, and areas for future cardio-oncology research.

Drug-specific cardiovascular toxicity profiles by antineoplastic class.
Based on the data from Table 5 and Figure 4, the 10 most commonly reported cardiac adverse events are atrial fibrillation (N = 7453, ROR = 1.61, 95% CI: 1.57–1.65, PRR = 1.61, χ2 = 1571.69), cardiac failure (N = 6570, ROR = 1.74, 95% CI: 1.69–1.78, PRR = 1.74, χ2 = 1861.35), cardiac failure congestive (N = 4560, ROR = 1.08, 95% CI: 1.05–1.12, PRR = 1.08, χ2 = 26.8), pericardial effusion (N = 2564, ROR = 2.53, 95% CI: 2.42–2.63, PRR = 2.52, χ2 = 2057.99), myocarditis (N = 2122, ROR = 5.31, 95% CI: 5.06–5.57, PRR = 5.31, χ2 = 5668.36), cardiomyopathy (N = 2022, ROR = 3.04, 95% CI: 2.9–3.19, PRR = 3.04, χ2 = 2353.32), acute myocardial infarction (N = 1819, ROR = 1.22, 95% CI: 1.16–1.28, PRR = 1.22, χ2 = 67.01), left ventricular dysfunction (N = 1070, ROR = 3.85, 95% CI: 3.6–4.12, PRR = 3.85, χ2 = 1844.79), cardiogenic shock (N = 882, ROR = 1.35, 95% CI: 1.26–1.45, PRR = 1.35, χ2 = 74.39), and acute coronary syndrome (N = 814, ROR = 2.01, 95% CI: 1.87–2.16, PRR = 2.01, χ2 = 370.96).
Top 10 most frequently reported cardiovascular adverse events and their signal strengths.
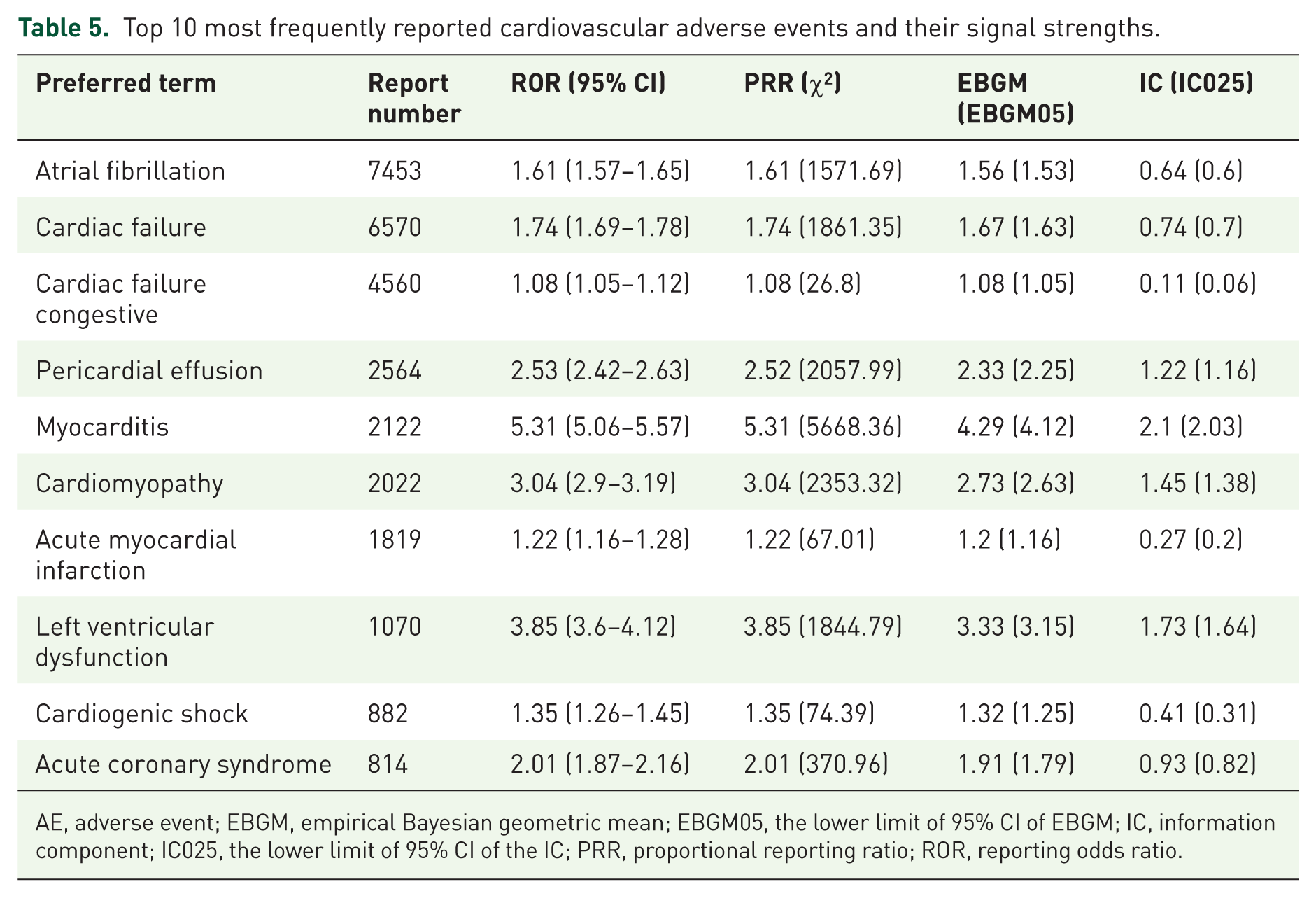
AE, adverse event; EBGM, empirical Bayesian geometric mean; EBGM05, the lower limit of 95% CI of EBGM; IC, information component; IC025, the lower limit of 95% CI of the IC; PRR, proportional reporting ratio; ROR, reporting odds ratio.
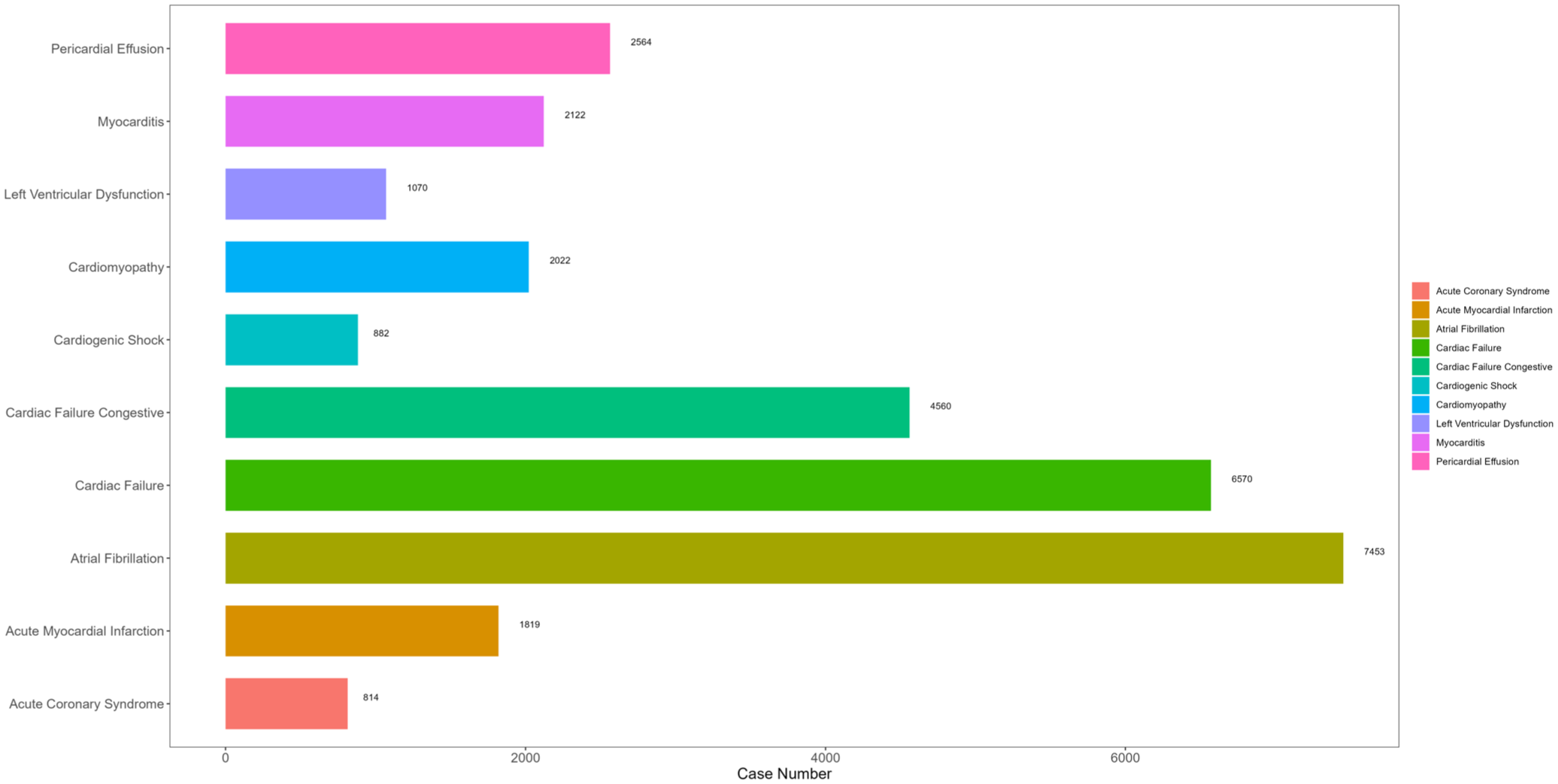
Ten most commonly reported cardiovascular adverse events in cancer patients.
To help clinicians detect potentially severe cardiovascular adverse events, we further calculated the mortality rates for each cardiovascular AE (number of deaths reported/number of AEs reported) after antineoplastic agent therapy (Figure 5). The results showed that cardiogenic shock had the highest mortality rate at 43.08%, followed by acute myocardial infarction (35.24%) and cardiac failure (35.10%). Myocarditis also demonstrated a substantial mortality rate of 31.34%, while congestive cardiac failure showed a mortality rate of 27.72%. Lower mortality rates were observed for cardiomyopathy (17.56%), acute coronary syndrome (17.08%), pericardial effusion (15.72%), atrial fibrillation (15.44%), and left ventricular dysfunction (14.58%) (Figure 5).

Mortality rates associated with cardiovascular adverse events following antineoplastic therapy.
Time to onset of cardiovascular adverse events
Figure 6 depicts the time to onset of cardiovascular adverse events for various antineoplastic agents. The median time to onset was notably shorter for fluoropyrimidines, with 5-Fluorouracil demonstrating the earliest onset at 11 days (IQR: 2.75–50.25 days), followed by Capecitabine at 17 days (IQR: 6–62 days). In contrast, Mitoxantrone exhibited the longest time to onset at 457 days (IQR: 104.5–836 days), followed by Imatinib at 178.5 days (IQR: 32.75–816.5 days). Among the most commonly used agents, Trastuzumab and Pomalidomide both had median onset times of 98 days (IQR: 36.5–242 days and IQR: 23–299.75 days, respectively), while Thalidomide showed 92 days (IQR: 28–253 days). Anthracyclines demonstrated intermediate onset times, with Doxorubicin at 72 days (IQR: 12.5–255.5 days) and Epirubicin at 64 days (IQR: 20.5–216 days).

Time to onset of cardiovascular adverse events by antineoplastic agent.
Multiple agents showed median onset times between 20 and 45 days, including Docetaxel (24 days), Bortezomib (28 days), Pembrolizumab (28 days), Paclitaxel (28 days), Durvalumab (31.5 days), Panitumumab (32 days), and Sorafenib (34 days). Immune checkpoint inhibitors demonstrated relatively consistent onset patterns, with most ranging from 37 to 43 days. This analysis reveals significant variability in the onset timing of antineoplastic-induced cardiovascular toxicity, providing valuable insights for clinical monitoring strategies and highlighting the importance of agent-specific cardiovascular surveillance protocols.
Comparison between serious and non-serious groups for antineoplastic agent-related cardiovascular AEs
Comparison of serious and non-serious cardiovascular adverse events (Table 6) revealed significant reporting differences across various event types. Several events showed higher prevalence in serious cases (p < 0.05), including cardiac failure (0.23% vs 0.14%), acute myocardial infarction (0.065% vs 0.04%), cardiogenic shock (0.039% vs 0.017%), cardiopulmonary failure (0.042% vs 0.0015%), and myocarditis (0.068% vs 0.048%). Conversely, other events were more frequently reported as non-serious (p < 0.05), such as atrial fibrillation (0.22% vs 0.12%), pericardial effusion (0.073% vs 0.041%), cardiomyopathy (0.056% vs 0.036%), and left ventricular dysfunction (0.03% vs 0.016%). No significant differences were observed in 16 cardiovascular events, including cardiac tamponade, supraventricular arrhythmia, and restrictive cardiomyopathy. These patterns highlight important distinctions in the clinical severity of different cardiovascular toxicities and emphasize the need for tailored monitoring approaches based on the specific cardiovascular adverse event profile.
Comparison between serious and non-serious cardiovascular adverse events.

PT, preferred term.
Stratified and subgroup analyses
To assess the robustness and generalizability of our findings, we conducted four pre-specified subgroup analyses (Tables S4–S12): (1) Temporal period analysis: Cases were stratified into two periods (2004–2016 vs 2017–2024) to evaluate consistency of signals over time and identify emerging safety concerns with newer agents. (2) Geographic analysis: Reports were categorized by country of origin into high-reporting countries (top seven countries by case volume: United States, Japan, United Kingdom, France, Germany, Canada, Italy) and low-reporting countries (remaining countries with ⩾10 reports) to assess regional reporting patterns and potential geographic differences in surveillance practices. (3) Sex-stratified analysis: Cardiovascular adverse events were analyzed separately for male and female patients to identify sex-specific toxicity patterns and differential susceptibility. (4) Age-stratified analysis: Cases were divided into three age groups (0–17.9 years (pediatric), 18–64.9 years (adult), ⩾65 years (elderly)) to characterize age-related risk profiles and inform age-appropriate monitoring strategies. For each subgroup analysis, disproportionality metrics (ROR, PRR) and temporal patterns (median time-to-onset) were recalculated. Statistical comparisons between subgroups were performed using chi-square tests for categorical variables and Mann-Whitney U or Kruskal-Wallis tests for continuous variables, with statistical significance set at p < 0.05.
Discussion
This comprehensive pharmacovigilance analysis of the FAERS database (2004–2024) provides critical insights into cardiovascular toxicity patterns associated with antineoplastic therapies. Our examination of over 18 million adverse event reports, yielding 51,402 cases of antineoplastic-related cardiovascular events, reveals distinct cardiotoxicity signatures essential for clinical practice.
The 4.6% incidence of cardiovascular adverse events among all antineoplastic-related reactions represents a substantial clinical burden. The dramatic rise in reporting—from 748 cases in 2004 to 3239 in 2024—reflects both expanded pharmacovigilance efforts and the growing complexity of cancer treatment regimens. Our classification into six distinct therapeutic categories provides a framework for class-specific toxicity monitoring.
Our disproportionality analysis reveals compelling cardiotoxicity patterns with important implications for clinical management. Mitoxantrone (15.9%), carfilzomib (10.0%), and doxorubicin (9.2%) demonstrated the highest proportional cardiovascular event incidence. While lenalidomide, nivolumab, trastuzumab, and bevacizumab generated the highest absolute numbers of reports, their proportional incidence was substantially lower—a critical distinction for risk-benefit assessment.
The distinctive cardiotoxicity signatures we identified across drug classes demand consideration amid ongoing healthcare reforms aimed at optimizing patient outcomes while controlling expenditures. Anthracyclines’ strong association with structural cardiac damage17,18—exemplified by doxorubicin’s link to cardiomyopathy (ROR = 20.6) and left ventricular dysfunction (ROR = 20.3)19,20—assumes new relevance as health systems navigate the tension between therapeutic innovation and fiscal sustainability. Similarly, the remarkably high signals for immune-mediated cardiac events with checkpoint inhibitors—such as pembrolizumab’s association with myocarditis (ROR = 245.3) and pericarditis (ROR = 469.5)—underscore the vital role of robust pharmacovigilance in an era of rapidly expanding immunotherapy indications.
The distinctive cardiotoxicity profiles we uncovered for other drug classes gain particular significance against the backdrop of evolving healthcare accessibility initiatives. Fluoropyrimidines’ strong association with vasospastic effects—including 5-fluorouracil’s link to Prinzmetal angina (ROR = 18.3) and coronary artery dissection (ROR = 11.7)—warrants careful consideration as oral formulations increasingly feature in home-based therapy programs, 21 a trend accelerated by the pandemic and now embedded in numerous national cancer strategies.
Our identification of the most frequently reported cardiac adverse events—atrial fibrillation (N = 7453), cardiac failure (N = 6570), and congestive cardiac failure (N = 4560)—carries substantial implications for healthcare resource allocation. With cancer survivorship in the United States alone projected to exceed 26 million by 2040,22,23 our data contributes essential evidence to the national discourse on sustainable healthcare models. Our mortality analysis, revealing strikingly high rates for cardiogenic shock (43.1%), acute myocardial infarction (35.2%), and cardiac failure (35.1%), provides critical insights rarely captured in clinical trials.
The time-to-onset analysis revealed distinct temporal signatures crucial for optimizing monitoring protocols. Fluoropyrimidines showed early cardiotoxicity (5-fluorouracil: 11 days; capecitabine: 17 days), anthracyclines demonstrated intermediate onset (doxorubicin: 72 days; epirubicin: 64 days),24,25 while mitoxantrone (457 days) and imatinib (178.5 days) exhibited delayed manifestation. Immune checkpoint inhibitors showed relatively consistent onset patterns (primarily 28–43 days). These temporal profiles have practical implications for monitoring frequency and duration. Early-onset toxicities require intensive initial monitoring, while delayed effects necessitate extended surveillance beyond treatment completion. The consistent ICI onset pattern suggests a predictable window for heightened vigilance.
Our analysis of frequently reported events—atrial fibrillation (n = 7453), cardiac failure (n = 6570), and congestive cardiac failure (n = 4560)—provides insight into the clinical spectrum of cardiotoxicity. The mortality analysis revealed particularly high rates for cardiogenic shock (43.1%), acute myocardial infarction (35.2%), and cardiac failure (35.1%), outcomes rarely captured in clinical trials due to selection biases and limited follow-up. The distinction between serious and non-serious events offers valuable intelligence for care planning. Cardiac failure, acute myocardial infarction, cardiogenic shock, and myocarditis were predominantly reported as serious events, requiring emergency preparedness and critical care resources. Conversely, atrial fibrillation, pericardial effusion, and cardiomyopathy appeared more frequently among non-serious reports, informing outpatient monitoring strategies. The demographic patterns we observed in cardiovascular toxicity reporting have profound implications for health equity initiatives. The female predominance in doxorubicin-associated cardiotoxicity (49.7% females vs 31.1% males) and the even more pronounced gender disparity with epirubicin (79.9% females vs 10.6% males) intersect directly with policy efforts addressing gender-based health disparities. 26 Age distribution showed concentration in adults aged 18–64.9 and 65–85 years, reflecting typical cancer demographics but also highlighting the clinical challenge of managing cardiotoxicity in patients with pre-existing cardiovascular risk factors or established disease.
Limitations
Several methodological limitations warrant acknowledgment. The FAERS database relies on spontaneous reporting, introducing potential underreporting, stimulated reporting (particularly following published safety alerts), and notoriety bias. The database does not capture exposure denominators, precluding true incidence rate calculations. 27 We cannot fully assess confounding by comedications, preexisting cardiovascular conditions, or cancer-related factors. Reporting varies by country and evolves over time, potentially affecting comparative analyses. The wide confidence intervals observed for some signals (e.g., epirubicin-associated immune-mediated pericarditis) reflect small event counts and should be interpreted as hypothesis-generating rather than definitive. Finally, the retrospective observational design cannot establish causality, only associations.28,29 Despite these limitations, the large-scale and real-world nature of FAERS data provide complementary evidence to controlled clinical trials, particularly for rare but serious adverse events.
Future research should integrate prospective registry studies with standardized cardiovascular assessment protocols to address current limitations. Advanced analytical approaches, including machine learning methods for signal detection and identification of complex cardiotoxicity patterns, warrant investigation. Integration of clinical trial data with real-world evidence could enable more accurate risk quantification. Mechanistic studies exploring the biological basis for observed class-specific and temporal patterns would enhance understanding and potentially guide preventive strategies.
Conclusion
This comprehensive pharmacovigilance analysis provides valuable insights into the real-world cardiovascular safety profiles of antineoplastic agents at a pivotal time in oncology care. The distinctive patterns of cardiotoxicity observed across drug classes—from the structural cardiac damage associated with anthracyclines to the immune-mediated cardiac events with checkpoint inhibitors and the vascular complications with fluoropyrimidines—highlight the complex and heterogeneous nature of cancer therapy-related cardiac dysfunction. Our findings regarding the timing, severity, and demographic distribution of cardiovascular events can inform targeted monitoring strategies and contribute to the ongoing development of cardio-oncology practice.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251401122 – Supplemental material for Cardiotoxicity associated with antineoplastic agents: a pharmacovigilance study based on FDA adverse event reporting system
Supplemental material, sj-docx-1-taw-10.1177_20420986251401122 for Cardiotoxicity associated with antineoplastic agents: a pharmacovigilance study based on FDA adverse event reporting system by Xiaohan Qiu, Qinxiao Li, Yiyin Rong, Longyu Wang, Jiahan Ke, Min Wang, Huasu Zeng and Jun Gu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986251401122 – Supplemental material for Cardiotoxicity associated with antineoplastic agents: a pharmacovigilance study based on FDA adverse event reporting system
Supplemental material, sj-docx-2-taw-10.1177_20420986251401122 for Cardiotoxicity associated with antineoplastic agents: a pharmacovigilance study based on FDA adverse event reporting system by Xiaohan Qiu, Qinxiao Li, Yiyin Rong, Longyu Wang, Jiahan Ke, Min Wang, Huasu Zeng and Jun Gu in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The authors acknowledge the FDA for maintaining the publicly accessible FAERS database and thank all healthcare professionals who contributed adverse event reports that made this analysis possible.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
