Abstract
Background:
Lacosamide and perampanel are two novel antiepileptic drugs with distinct mechanisms of action. However, the real-world evidence of adverse events (AEs) related to these drugs in children is limited.
Objectives:
This study aims to explore the AE profiles of lacosamide and perampanel in children by mining the FDA Adverse Event Reporting System (FAERS).
Design:
A retrospective disproportionality analysis of the FAERS database was conducted.
Methods:
Reports were collected and analyzed from the first quarter of 2009 to the third quarter of 2024. Four disproportionality analysis were employed in data-mining process to detect signals in children, including the reporting odds ratio, the proportional reporting ratio, the Bayesian confidence propagation neural network, and the multiitem gamma Poisson shrinker algorithms.
Results:
A total of 1017 AE reports of lacosamide and 349 AE reports of perampanel deemed as the “primary suspect” were retrieved in children. After being categorized and summarized by the number of reports in system organ class, the top 3 for lacosamide were nervous system disorders, general disorders and administration site conditions, injury, poisoning and procedural complications, the top 3 for perampanel were psychiatric disorders, nervous system disorders, infections, and infestations. Both lacosamide and perampanel had significant effects on the nervous system. Lacosamide was more prone to multidrug resistance and cardiac disorders, while perampanel was more likely to affect psychiatric system.
Conclusion:
Our study identified potential new AE signals for lacosamide and perampanel. The findings warned the need for cautious therapeutic decisions during pregnancy and emphasized the importance of rational use of drugs and correct medication education. This postmarketing study provided reference data for the clinical monitoring and treatment of lacosamide and perampanel in children. Our study merely proved the statistical correlations; further research is needed to clarify the causal relationship between the drug and the AEs.
Plain language summary
Why was the study done? Lacosamide and perampanel are two novel antiepileptic drugs with distinct mechanisms of action, offering therapeutic options for adults and pediatric patients. Although both lacosamide and perampanel have demonstrated good therapeutic efficacy and tolerability in children, the safety in the real world is still uncertain, especially for the off-label use in children under 4 years of age.
What did the researchers do? This study collected approximately 5 years of adverse event (AE) reports from the FDA Adverse Event Reporting System (FAERS) database, and conducted an in-depth analysis of the reports to summarize the safety of the two antiepileptic drugs in the pediatric population. Besides, the study investigated the situation of off-label drug use, as well as whether there were any new drug-related AEs. The aim was to provide reference data for the clinical drug safety.
What did the researchers find? A total of 1017 AE reports of lacosamide and 349 of perampanel deemed as the “primary suspect” were retrieved in children. Lacosamide was more commonly associated with nervous system disorders, while perampanel was more likely to result in psychiatric disorders. Both lacosamide and perampanel had significant effects on the nervous system, including ataxia (a syndrome characterized by impaired coordination of voluntary movements), drop attacks, and dizziness. In addition, the signals of AEs were similar in the age under 4 years group and 4–17 years group.
What do the findings mean? Our findings provided safety evidence for the off-label use of lacosamide and perampanel in children under 4 years old, and highlighted the need for cautious therapeutic decisions during pregnancy and emphasized the importance of rational use of drugs. However, our study merely proved the statistical correlations between the drug and the AEs, further research is needed to clarify the causal relationship.
Introduction
Epilepsy is a common chronic disease of the nervous system that characterized by recurrent seizures, which affects more than 70 million people worldwide. 1 The research from the Global Disease Burden 2021 indicated that the global prevalence rate of epilepsy was 308.9 (95% UI: 236.2–390.1) and the mortality rate was 1.7% (95% UI: 1.5–1.9) in age-standardized rates (per 100,000 people). 2 Epilepsy is also one of the most common neurologic disorders in children, especially with the highest incidence at the age of 1 year. 3
Currently, the well-recognized treatment approaches for pediatric epilepsy encompass pharmacological interventions and nonpharmacological therapies. Antiseizure medications (ASMs) stand as the cornerstone of epilepsy drug treatment. These drugs exhibit structural and functional diversity, encompassing membrane stabilizers, auxiliary sodium-channel blockers, neurotransmitter release inhibitors, GABA mimetics, calcium-channel modulators/inhibitors, and so on.4,5 Most pediatric patients with epilepsy are likely to achieve good seizures control with ASM treatment. But unfortunately, it is reported that some patients still experience seizures despite administrating a series of ASMs, and their seizures are more frequently associated with drug-resistant or intractable epilepsy. 6 For patients with seizures poorly controlled by drugs, nonpharmacological therapies are another efficient method, including ketogenic diet, surgical procedures, surgical implants, or gene therapy.7,8
Lacosamide and perampanel are two ASMs with distinct mechanisms of action, offering therapeutic options for adults and pediatric patients with epilepsy. Lacosamide is a functionalized amino acid derivative with unique anticonvulsant activities, which was approved by the US Food and Drug Administration (FDA) in 2008, used for partial seizures with or without secondary generalization. Lacosamide is a third-generation ASM that selectively enhances the slow inactivation of voltage-gated sodium channels rather than the fast inactivation of the channels by traditional sodium-channel blockers, thereby regulating the long-term availability of sodium channels.9,10 In recent years, lacosamide is also widely used off-label in children with drug-resistant epilepsy, which has shown good therapeutic efficacy in children, with acceptable side-effects.11–13 Different from lacosamide, perampanel is a noncompetitive, selective AMPA receptor antagonist, used for partial seizures with or without secondary generalized seizures, which initial approved for patients aged ⩾12 years by FDA in 2012, and subsequently expanded usage to pediatric patients ⩾4 years of age in 2018.14,15 It inhibits an allosteric site which is distinct from the glutamate recognition site, and blocks glutamate-mediated fast postsynaptic excitatory neurotransmission in the central nervous system, thereby exerting antiepileptic activity. Perampanel is currently widely used in clinical practice, and has also revealed good efficacy in children with refractory epilepsy and drug-resistant epilepsy.16,17 Although both lacosamide and perampanel have demonstrated good therapeutic efficacy and patient tolerance in children,18–21 further research of their safety in pediatric patients is still needed based on large-scale data in real world.
The FDA Adverse Event Reporting System (FAERS), a publicly accessible pharmacovigilance database established by the FDA, which collects substantial adverse event (AE) reports spontaneously submitted by healthcare professionals, pharmacists, pharmaceutical manufacturers, and consumers, representing one of the largest global pharmacovigilance databases. 22 Considering the lack of data on AEs related to lacosamide and perampanel in children at the real-world level, we conducted a postmarketing surveillance to assess and compare lacosamide and perampanel-related AEs in FAERS from the first quarter of 2009 to the third quarter of 2024. The results of this study can investigate potential red flags and unreported signals, thereby guide physicians in monitoring AEs of lacosamide and perampanel, and provide references for their appropriate use in clinical practice to ensure patient medication safety.
Methods
Data sources
The data used in this study were obtained from the FDA’s publicly available database FAERS, which includes seven datasets: demographic and administrative information (DEMO), drug specifics (DRUG), adverse drug reactions (REAC), patient outcomes (OUCT), report sources (RPSR), medication start and stop dates (THER), and drug use indications (INDI). 23 We extracted data on lacosamide from the FAERS database during the first quarter of 2009 to the third quarter of 2024, and on perampanel during the fourth quarter of 2012 to the third quarter of 2024. To ensure the uniqueness of the data, we deduplicated reports by a two-step process. First, we deleted AE reports according to the list of delete reports provided by FAERS database, and then removed reports according to the FDA recommendations by selecting the latest FDA_DT when the CASEID were the same, and selecting the higher PRIMARYID when the CASEID and FDA_DT were the same. 24 We further filtered the data to extract reports in which lacosamide or perampanel was deemed as the PS (primary suspect) drug to improve the reliability of this study.
Procedures
The FAERS database and AE terminology used in this study were coded by preferred terms (PTs) outlined in the MedDRA, and mapped to their corresponding highest system organ classes (SOCs) level in MedDRA to standardize coding and classification. Signal detection analysis focused on pediatric population, so our inclusion criteria were (1) reports in which the patient’s age was explicitly stated as less than 18 years; (2) reports in which lacosamide or perampanel was identified as the PS drug. Reports that did not meet the above criteria were excluded (e.g., lacosamide or perampanel was defined as the “Secondary Suspect,” “Concomitant,” or “Interacting” drug, or reports lacking patient’s age information).
Signal mining and statistical analysis
Disproportionality analysis is a key tool in pharmacovigilance research which can analyze the potential association between drugs and AEs to mine the AE signals of target drugs. When the incidence of a specific AE for a target drug was significantly higher than the background frequency in the database and reached a certain threshold or criterion, a significant signal is considered to be generated. 25 Four algorithms commonly used in disproportionality analysis were employed in this study: proportional reporting ratio (PRR), reporting odds ratio (ROR), Bayesian confidence propagation neural network (BCPNN), and multiitem gamma Poisson shrinker (MGPS). PRR and ROR are non-Bayesian methods that have better efficiency in early signal detection, while BCPNN and MGPS are Bayesian methods which can detect signals from rare events. 26 Information components (IC) are used in the BCPNN to determine disproportionality. MGPS analysis is a mature technique for reducing the occurrence of false-positive reports by using a Bayesian shrinkage estimator to adjust observed/expected ratio, which can obtain smaller risk estimates with narrower confidence intervals (CI) although the event counts are less. 27 Generally, an increasing parameter value results in a corresponding increase association. The detailed formulas and the criteria of the four disproportionality analysis methods were provided in Table 1. To ensure the reliability of the findings, AE signals that met the thresholds of all four methods simultaneously were selected for subsequent studies. Furthermore, PTs associated with off-label use, epilepsy, and other epilepsy-related diseases were deleted and not included in the analysis. AE signals with higher ROR, PRR, IC, or EBGM values indicated stronger statistical correlation. Data processing and statistical analysis were conducted by Microsoft Excel 2019 and R software (version 4.4.2).
Four major algorithms used for signal detection.

a, number of reports containing both the target drug and target adverse event; b, number of reports containing other adverse events of the target drug; c, number of reports containing the target adverse event of other drugs; d, number of reports containing other drugs and other adverse events; ROR, reporting odds ratio; PRR, proportional reporting ratio; BCPNN, Bayesian confidence propagation neural network; MGPS, multiitem gamma Poisson shrinker; 95% CI, 95% confidence interval; N, the number of reports; χ2, chi-squared; IC, information component; IC025, the lower limit of 95% CI of the IC; EBGM, empirical Bayesian geometric mean; EBGM05, the lower limit of 95% CI of EBGM.
Time-to-onset analysis
A time-to-onset (TTO) analysis was conducted by Weibull distribution to study the time-related risk dynamics of AEs associated with lacosamide or perampanel in children. TTO was defined as the interval between the AE onset date (EVENT_DT) and the drug start date (START_DT). Missing dates and cases where EVENT_DT preceded START_DT were excluded to ensure the integrity and accuracy of data. The Weibull model was selected for its flexibility in modeling various hazard patterns, as it can indicate the risk rates of increasing or decreasing over time. 28 Whereas other distributions commonly used in survival analysis assume constant hazard rates or limited shapes, the parameters of Weibull distribution represent more complex risk patterns. 29 This makes it suitable for capturing the potentially variable AE onset times associated with pediatric drug use. We calculated median, quartiles, the Weibull scale parameter (α), and the shape parameter (β). When the shape parameter β < 1, and its 95% CI: <1, the risk of AE is considered to decrease over time (early failure type curve); while β > 1 indicates an increasing risk over time. 30 TTO analysis can provide information for clinical monitoring and comprehensive guidance for effective treatment strategies with lacosamide or perampanel in pediatric patients.
Subgroup analysis
Lacosamide is currently approved for partial-onset seizures in patients ⩾1 month of age or as adjunctive therapy for primary generalized tonic-clonic seizures in patients ⩾4 years of age, and perampanel is approved only for patients ⩾4 years of age with epilepsy. However, they were used as off-label drugs in patients <4 years of age in real world. Thus, we performed subgroup analyses according to the age (aged <4 years group and aged 4–17 years group) of the patients to study the differences in AEs due to lacosamide versus perampanel across different age. We also performed subgroup analyses according to the gender of the patients to study the differences in lacosamide and perampanel-related AEs between male and female. We used the ROR method to identify PTs with disproportionate AE incidence between different subgroups.
This retrospective study is observational and descriptive in nature. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Table 4) and follows the READUS-PV guidelines.31,32
Results
Descriptive analysis
A total of 20,099,557 AE reports were recorded in the FAERS database during 2009(Q1) to 2024(Q3), and then 17,037,498 AE reports were remained by removing duplicates. A total of 560,466 AE reports aged 0–17 years were finally obtained after further screening, among which 1017 reports identified lacosamide as the PS, and 349 reports identified perampanel as the PS. The detailed data screening process and workflow diagram are shown in Figure 1. In this study, the proportions of females, males, and unknown gender of lacosamide-related reports were 45.72%, 48.87%, and 5.41%, respectively, while perampanel-related reports were 46.70%, 50.43%, and 2.87%, respectively. The mean age of pediatric patients treated with lacosamide and perampanel were 8.71 and 11.08 years, respectively. Compared to other pediatric group, 6–12 years age group had the greatest number of AE reports for lacosamide (n = 358, 35.20%), while for perampanel, 13–17 years age group reported most AEs (n = 169, 48.42%). In addition, most reports were submitted by physician, with 423 reports (41.59%) for lacosamide and 255 reports (73.07%) for perampanel. The AE reports related to lacosamide were primary from the United States (36.18%) and Japan (14.06%), and similarly, reports of perampanel were most from Japan (19.77%) and United States (18.91%), see detailed information in Table 2.

The flow diagram of screening lacosamide and perampanel-related AEs in patients under 18 years of age from the FAERS database.
Clinical characteristics of lacosamide and perampanel AE reports from the FAERS database (Q1 2009–Q3 2024).
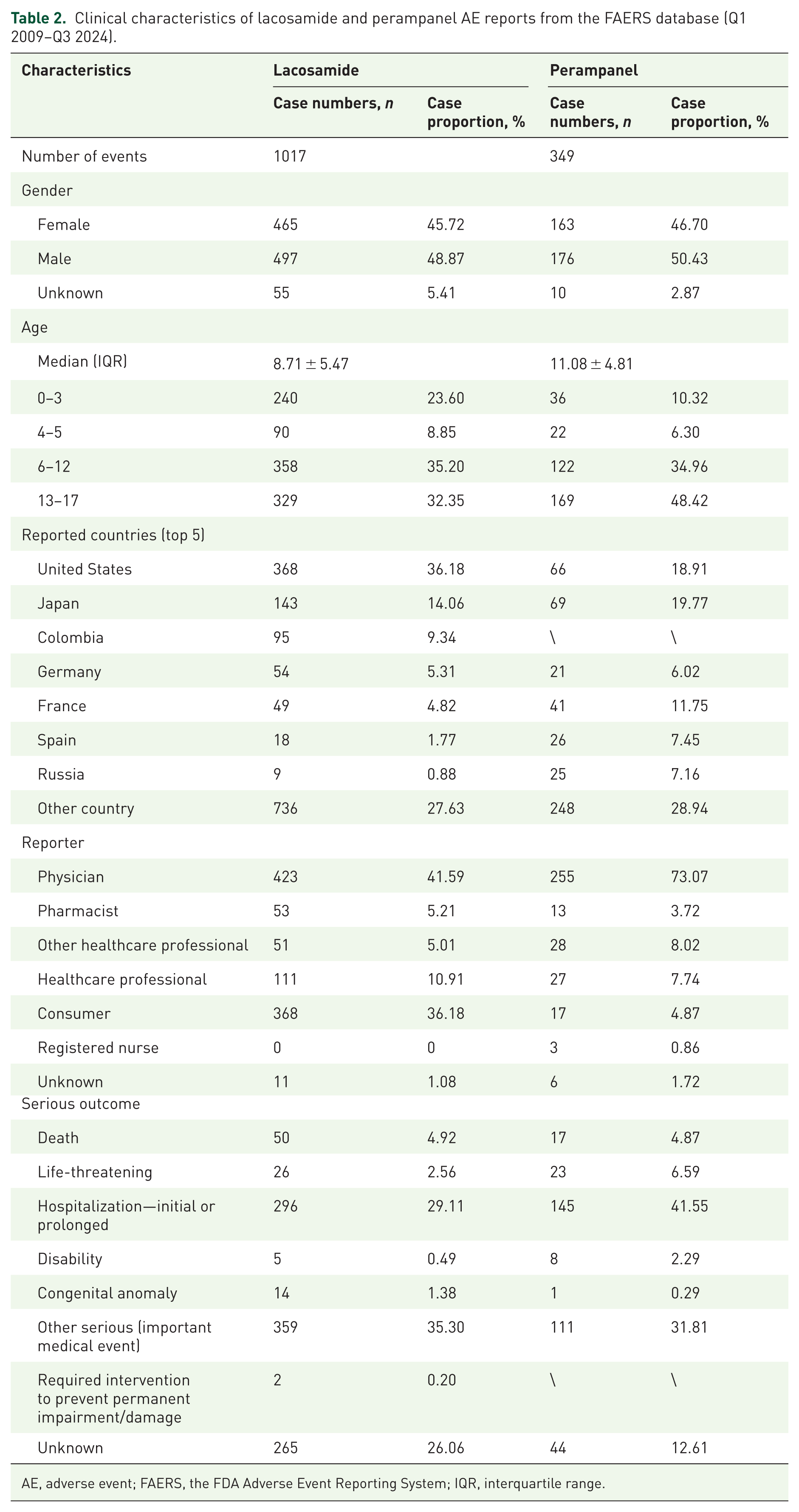
AE, adverse event; FAERS, the FDA Adverse Event Reporting System; IQR, interquartile range.
Signal detection at the SOC level
In this study, we performed SOC-level classification of the mined positive PT signals and obtained 328 AE signals of lacosamide involving 13 SOCs, 337 AE signals of perampanel involving 10 SOCs. The proportion of AEs at each SOC level were shown in Figure 2, the top 3 for lacosamide were nervous system disorders (n = 149, 45.43%), general disorders and administration site conditions (n = 52, 15.85%), injury, poisoning, and procedural complications (n = 48, 14.63%), and the top 3 for perampanel were psychiatric disorders (n = 160, 47.48%), nervous system disorders (n = 103, 30.56%), infections, and infestations (n = 22, 6.53%). These results indicated that both lacosamide and perampanel were prone to induce nervous system disorders, and perampanel was more likely to cause psychiatric disorders.

The proportion of lacosamide and perampanel-related significant AEs at SOC level.
Signal detection at the PT level
A summary analysis was employed to study the obtained AE positive signals at the PT level, and the results showed that there were some differences in the signals mining results between lacosamide and perampanel. We summarized AE signals that met the signaling criteria of all four methods simultaneously at PT level, excluding AEs as possible indications for lacosamide and perampanel or AEs clearly not related to the drug. Three hundred twenty-eight AE signals of lacosamide and 337 AE signals of perampanel that we finally obtained were illustrated in Supplemental Tables 1 and 2 with detailed information. The PT with the greatest number of reports related to lacosamide was multiple-drug resistance (n = 52), followed by somnolence (n = 45) and dizziness (n = 31), as for perampanel, aggression (n = 59), somnolence (n = 36), and suicidal ideation (n = 18) were the most reported PTs. Most of these AEs above are consistent with the drug insert, but the perampanel-related AE of suicidal ideation are different, in which suicidal ideation was indicated as a rare AE. In addition, the disproportionality analysis of the PTs has different results from the number of PTs. The top 5 most significant signals of lacosamide were cardiac arrest neonatal (n = 3, ROR: 95.13 (28.25–320.32)), multiple-drug resistance (n = 52, ROR: 76.64 (57.35–102.42)), drop attacks (n = 3, ROR: 55.96 (17.18–182.3)), atrioventricular block second degree (n = 9, ROR: 49.31 (25–97.26)), and cell death (n = 4, ROR: 39.65 (14.43–108.96)), interestingly, among which cardiac arrest neonatal and cell death were unexpected signals. We also considered the IC025 value because of the increased stability of calculated results offered by the Bayesian methods when the numbers of AE reports are low, 33 so despite the low number of cases, high IC025 values were found in cardiac arrest neonatal (IC025: 4.81), multiple-drug resistance (IC025: 5.65), and cell death (IC025: 3.88), indicating a strong association with the use of lacosamide. Given that lacosamide is currently approved only for patients ⩾1 month of age with partial seizures, we separately mined signals of lacosamide in patients <1 month at PT level. The most significant signals with high-ROR value were atrioventricular block second degree and cardiac arrest neonatal, detailed results are shown in Supplemental Table 3. Considering the approved indications for lacosamide, we speculate that the cardiac arrest neonatal signal may be related to off-label use or accidental exposure during pregnancy in clinical practice. As to perampanel, the top 5 PT were drop attacks (n = 4, ROR: 230.27 (81.41–651.3)), homicidal ideation (n = 4, ROR: 25.33 (9.42–68.06)), ataxia (n = 13, ROR: 23.27 (13.42–40.37)), altered state of consciousness (n = 12, ROR: 19.43 (10.96–34.43)), and psychotic disorder (n = 11, ROR: 19.2 (10.56–34.9)), which were all warned in the drug insert.
To visualize the data, we presented the PT signals in the form of a forest plot (Figure 3). It’s worth noting that in SOC: nervous system disorders, lacosamide and perampanel involved five identical PTs. Seven lacosamide-related cases reported ataxia with signal values of ROR 4.13 (1.96–8.69), PRR 4.12 (16.43), EBGM 4.1 (2.2), IC 2.03 (1.01), 13 cases reported balance disorder with signal values of ROR 11.24 (6.49–19.47), PRR 11.19 (118.59), EBGM 11.01 (6.95), IC 3.46 (2.68), 31 cases reported dizziness with signal values of ROR 3.06 (2.15–4.37), PRR 3.04 (42.38), EBGM 3.03 (2.25), IC 1.6 (1.08), 3 cases reported drop attacks with signal values of ROR 55.96 (17.18–182.3), PRR 55.9 (148.64), EBGM 51.45 (19.15), IC 5.69 (4.17), 45 cases reported somnolence with signal values of ROR 3.34 (2.48–4.48), PRR 3.3 (72.01), EBGM 3.29 (2.57), IC 1.72 (1.29). As for perampanel, 13 cases reported ataxia (ROR 23.27 (13.42–40.37), PRR 22.96 (269.93), EBGM 22.7 (14.32), IC 4.5 (3.72)), 3 cases reported balance disorder (ROR 7.63 (2.45–23.75), PRR 7.61 (17.15), EBGM 7.58 (2.93), IC 2.92 (1.47)), 14 cases reported dizziness (ROR 4.14 (2.44–7.01), PRR 4.09 (32.71), EBGM 4.08 (2.62), IC 2.03 (1.28)), 4 cases reported drop attacks (ROR 230.27 (81.41–651.3), PRR 229.27 (810.85), EBGM 204.6 (85.72), IC 7.68 (6.3)), 36 cases reported somnolence (ROR 8.15 (5.83–11.38), PRR 7.87 (215.99), EBGM 7.84 (5.93), IC 2.97 (2.49)).

Differences in AE signals of lacosamide and perampanel at the PT level that involved in same SOC.
TTO analysis
After removing missing and incorrect onset dates, 204 cases of lacosamide and 137 cases of perampanel were included in TTO analysis. Because of insufficient data on the administration route, we did not conduct a separate TTO analysis for the oral and intravenous routes. The median onset time of lacosamide was 69 days (IQR: 15.75–270), while perampanel was 42 days (IQR: 14–130). Figure 4 demonstrates that most AEs occurred within 0–30 days after administration both for lacosamide (n = 81, 39.71%) and perampanel (n = 52, 37.96%); however, a considerable proportion of AEs occurred within 180–360 days (n = 28, 13.73%) or longer (n = 39, 19.12%) for lacosamide, and occurred within 31–60 days (n = 26, 18.98%) for perampanel. In addition, 10 cases reported AEs occurred after 1 year of treatment with perampanel. These findings suggest that the occurrence of AEs during this period should be strictly monitored for timely intervention, and remind patients that AEs could still occur even after 1 year of treatment with lacosamide and perampanel.

Onset time of adverse events related to lacosamide (A) and perampanel (B).
We conducted Weibull distribution to explore whether there is a temporal trend in the risk of lacosamide and perampanel-related AEs, and the results were shown in Table 3. The value of the shape parameter (β) of lacosamide and perampanel were 0.64 and 0.68, respectively, with the upper limit of the 95% CI at 0.71 and 0.77, separately. AEs were defined as early failure types when β < 1, so it indicated a decreasing risk of lacosamide and perampanel-related AEs over time.
Weibull distribution of the lacosamide and perampanel-related AEs.

AE, adverse event; IQR, interquartile range; TTO, time-to-onset; α, the Weibull scale parameter; β, the Weibull shape parameter; 95% CI, 95% confidence interval.
Subgroup analysis of children under 4 years of age
To further investigate the differences in AE signals between the aged <4 years group and 4–17 years group, we stratified the analysis by age for children administrating lacosamide and perampanel, respectively. Figure 5 shows the distribution of AE signals between different groups. Overall, the signals of AEs were approximately the same in the aged <4 years group and 4–17 years group. For lacosamide, maternal exposure during pregnancy (n = 15, ROR: 50.44 (6.65–382.61), adjusted p < 0.05) and maternal exposure before pregnancy (n = 10, ROR: 33.36 (4.26–261.11), adjusted p < 0.05) had higher risk in children aged <4 years group. In addition, oxygen saturation decreased (n = 6, ROR: 9.94 (2–49.37), adjusted p ⩾0.05) and atrioventricular block second degree (n = 6, ROR: 6.62 (1.65–26.56), adjusted p ⩾ 0.05) occurred more frequently in children aged <4 years of age compared to children aged 4–17 years group. As for perampanel, AEs of coma (n = 3, ROR: 7.32 (1.46–36.71), adjusted p ⩾ 0.05) occurred more frequently in children aged <4 years group. The number of AE reports related to perampanel was less than lacosamide in pediatric patients aged <4 years, which possibly reflect the broader use of lacosamide in this age group following its 2021 approval for patients ⩾1 month with partial-onset seizures. Besides, most of the AEs of lacosamide were associated with improper use of drugs or accidental exposure to drugs, reminding medical practitioners that lacosamide should be used cautiously and rationally in clinical treatment.

Analysis of age-differentiated risk signals in lacosamide (A) and perampanel (B).
Subgroup analysis by gender
We also performed the subgroup analysis by gender for children administrating lacosamide and perampanel, respectively, to further study the differences in AE signals between male and female. Overall, there was no significant difference in the AE signals between male and female pediatric patients caused by lacosamide or perampanel, and the results were shown in Figure 6. However, it’s worth noting that insomnia (n = 9, ROR: 9.69 (1.23–76.58), adjusted p ⩾ 0.05) more commonly occurred in female children treated with lacosamide, and as for perampanel, dizziness (n = 13, ROR: 14.04 (1.83–107.79), adjusted p ⩾ 0.05) occurred more frequently in female children.

Analysis of gender-differentiated risk signals in lacosamide (A) and perampanel (B).
Discussion
This research is the first postmarketing pharmacovigilance study of lacosamide and perampanel in pediatric patients by using the FAERS database. We compared and comprehensively analyzed the differences in real-world safety between the two ASMs, as well as obtained some unexpected risk signals.
Analysis of clinical characteristics of lacosamide and perampanel indicated a slight preponderation of male in AE reports, although there was no significant gender disparity, it still reminded the importance of gender issue in epilepsy care and treatment strategies. It is reported that hormonal is a significant influent factor in epilepsy management, among which sex hormones are most important for that estrogen and progesterone can affect neuronal excitability. 34 Most of the reports were submitted by medical staff, suggesting professionals were highly focused on the safety of pediatric patients. It is worth noting that the number of lacosamide-related reports submitted by consumer also accounts for a certain proportion, which may be related to the awareness of treatment management and the detailed pharmaceutical care of doctors or pharmacist. In addition, children aged 6–12 years group was the largest proportion of lacosamide-related AE reports compared to other groups, while the adolescent group (aged 13–17 years) was the largest of perampanel-related AE reports. This difference may be related to the treatment strategies of drugs among different ages in clinical practice. It suggested special attention should be paid to the individualized drug selection, and the clinical benefits and potential risks should be comprehensively considered when making the treatment decision for pediatric patients.
Differences in safety signals between lacosamide and perampanel in children
Multiple-drug resistance
Multiple-drug resistance was found to be the most frequently reported lacosamide-related AE with significant signals in this study, while it was not observed in perampanel. This finding was similar to the real-world safety analysis in adults, Ge et al. reported that lacosamide showed the highest ROR for multidrug resistance in their study. 35 Although an increasing number of ASMs have been developed, drug resistance is still a major challenge for epilepsy therapeutics, as it is clearly associated with the increased morbidity and mortality. 36 However, no signal of multiple-drug resistance was found for perampanel in our study, indicating that perampanel may be a better therapeutic choice for children with pharmacoresistant epilepsy compared to lacosamide, for perampanel had less risk of multiple-drug resistance. Nevertheless, active clinical management and long-term monitoring are necessary for pediatric patients administrated with lacosamide or perampanel to ensure the safety and efficacy.
Psychiatric disorders
The signals associated with psychiatric disorders (SOC level) were found both for lacosamide and perampanel, but the involved PTs were different. For lacosamide, staring was the only reported AE of the psychiatric system with significant signal, while it was not found in perampanel. It was also an unexpected signal for lacosamide because it was not mentioned in the drug insert and has not been reported yet. Aggression, suicidal ideation, suicide attempt, agitation, and irritability were most reported AEs for perampanel, which was not found in lacosamide. These results suggested that perampanel was more likely to affect psychiatric system of pediatric patients compared to lacosamide, and these AEs were also all warned in the drug insert, demonstrating the reliability of our study. Besides, our results are consistent with clinical data. A cohort study of 66 children and adolescents with refractory epilepsy showed that 10.6% of patients experienced AEs of irritability and aggression after using perampanel (dose range 2–8 mg) as adjuvant treatment, 37 and an analysis of three phase III clinical studies reported that perampanel was associated with high risk of hostility and aggression, so we recommend healthcare professionals for vigilant monitoring during treatment, especially for patients treated with titrating perampanel and at higher doses.38,39 In addition, a retrospective study suggested that the usage of perampanel combining with multiple ASMs and age <13 years were significant factors for the occurrence of psychiatric disorders. 40 Perampanel is a highly selective, noncompetitive antagonist of AMPA receptors, which reduce glutamatergic transmission by affecting glutamate receptors in the amygdala, hypothalamus, and periaqueductal gray of the midbrain. It is reported that increased levels of glutamate are associated with psychiatric disorders, including aggression and irritability, since glutamate plays a crucial role in the regulation of mood and behavior.41,42 Although a meta-analysis pointed out that there is no current evidence that perampanel increases suicidality in epilepsy, 43 our results still suggested a potential association with suicidal tendency. Therefore, it is necessary to pay special attention to the related suspicious behaviors of pediatric patients when treating with perampanel.
Syncope
Signal of syncope was detected in lacosamide with a relatively large number of reports, while it was not detected in perampanel. The drug inserts also indicated that cases of syncope in patients with lacosamide therapy have been reported in clinical trials, although it is rare, which is consistent with our results. Interestingly, although we detected a high-risk signal of syncope, this AE is rarely reported in real-world and clinical studies. Syncope is often accompanied by dizziness, palpitations, headache, etc., 44 among which dizziness and ventricular tachycardia are common AEs of lacosamide in our mining results. Thus, early identification and management of these related AEs can prevent further deterioration leading to syncope; this may be the reason for the rare reports of syncope in pediatric patients with lacosamide therapy. A systematic review reported that the occurrence of syncope in the pediatric population was most associated with vasovagal dysfunction, other etiologies including cardiac, neurological, and psychiatric disorders. Therefore, when making treatment strategies, it is necessary to consider the benefits and risks for children with these risk factors, and monitor this special population in clinical practice.
Cardiac disorders
Signals associated with cardiac disorders were detected in lacosamide, including atrioventricular block second degree, atrioventricular block first degree, ventricular tachycardia, and cardiac arrest neonatal, while were not observed in perampanel. Consistent with our results, a multicenter cohort study including 714 pediatric patients reported that cardiac disorders were the AEs with the highest incidence in dose-based cohorts for children, including bradycardia, tachycardia, cardiac arrest, and tachyarrhythmia. 45 A case reported that second-degree atrioventricular heart block and cardiac arrest occurred in a 3-week-old female neonate after initiating lacosamide therapy, 46 so the safety of lacosamide for off-label usage in the neonatal population needs further evaluation. Although cardiac disorders have previously reported association with PR prolongation (delayed conduction through the atrioventricular node) in patients administrated lacosamide, 47 the signal of PR prolongation was not detected in pediatric patients across our study, but still worth attention. Based on our findings and available data, the pediatric patients’ cardiac function should be monitored (e.g., ECG) when treating with lacosamide, especially for patients with low baseline heart rate, and if necessary, reduce the dose or discontinue the drug to ensure pediatric safety.
Similarities in safety signals between lacosamide and perampanel in children
Nervous system disorders were reported to be common AEs of ASMs, which are consistent with our study in pediatric patients. We detected five same PTs in this SOC level between lacosamide and perampanel in pediatric patients, that is, ataxia, balance disorder, dizziness, drop attacks, and somnolence, among which drop attacks had the most significant signal both for lacosamide and perampanel. Although drop attacks in pediatric patients was less reported, it is still a meaningful AE because our study indicated its strong correlation with lacosamide and perampanel, which is also warned in the drug insert. Drop attacks may be related to epileptic seizures 48 or indirectly caused by the AE of ataxia, balance disorder, dizziness, muscular weakness, and other risk factors, and these factors were also detected in this study. It is worth noting that dyskinesia may also indirectly lead to drop attacks. Nadine et al. suggested that lacosamide dosing was correlated with the onset and resolution of dyskinesia in their study in children, and monitoring serum levels of lacosamide can help reduce AEs and ensure the safety of patients. 49 Besides, a retrospective study of lacosamide including 79 children (aged <18 years) reported that somnolence (18.2%) was the most common AE and dizziness (9.1%) was also common, and a study including 191 pediatric patients treated with lacosamide also indicated that dizziness and ataxia were most common AEs.13,50 Meanwhile, systematic reviews and studies of perampanel in pediatric patients reported that drowsiness, dizziness, somnolence, and other nervous system-related AEs were most common AEs.18,51,52 Therefore, healthcare professionals and caregivers of pediatric patients should pay attention to the AEs related to nervous system disorders in children and be alert to the occurrence of drop attacks and other serious AEs.
Unexpected safety signals
We obtained several unexpected signals beyond the drug insert, demonstrating the significance of real-world data in detecting rare and potential AEs. For lacosamide, head injury, low birth weight baby, sedation, febrile convulsion, salivary hypersecretion, cell death, underweight, blood sodium decreased, and hypoventilation were unexpected AEs. Among these unexpected signals, head injury may be related to the AE of drop attacks, and the exposure during or before pregnancy may be cause for the AEs of low birth weight baby, so timely management of AEs and ensuring the rationality of drugs were crucial to help prevent more severe or unexpected AEs in pediatric patients. As for perampanel, pneumonia, dysphagia, urinary retention, muscular weakness, hypothermia, hematemesis, and hallucination were new signals, which also need concentration in clinical practice. Interestingly, these unexpected signals we detected in this study were not found in adult patients, it may be speculated that they were associated with the particularity of pediatric population, but need further evaluation and research. 35
Safety signals for children under 4 years of age
Off-label drug use is common in children. In this study, the signals of AE were approximately the same in the age <4 years group and 4–17 years group, which provided safety evidence for the off-label drug use of lacosamide and perampanel for children under 4 years old with epilepsy.
AEs of exposure during and before pregnancy were most common in children aged <4 years treated with lacosamide, it may be related to therapeutic decision-making during pregnancy. High therapeutic concentrations of lacosamide have effects on the human placenta, it could affect folate supply of fetus, although lacosamide is demonstrated not carcinogenic or teratogenic by animal studies. 53 However, a prospective study of lacosamide during pregnancy and breastfeeding showed that newborns were born healthy with no congenital malformations, and breastfed infants had no complications. 54 A case reported that a patient treated with lacosamide during pregnancy and used perampanel at 5–7 weeks had a normal newborn with no malformations. Current case reports and study data provide evidence for the relative safety of lacosamide monotherapy during pregnancy and breastfeeding, but monitoring of supportive therapeutic and plasma concentrations is still significant, and further multicenter study with large sample is necessary to certify its safety and efficacy. Besides, a trial in patients aged ⩾1 month to <4 years and a prospective study (mean age was 2.7 years) reported the occurrence rate of drug-related AE in lacosamide-treated patients was 44.5% and 33%, respectively, including somnolence, irritability, drowsiness, and vomiting, but most of them were mild,55,56 indicating the good tolerability and relative safety of lacosamide in children aged <4 years.
Chang et al. reported 22% of children treated with perampanel experienced AEs in their study of children aged 0–6 years, including emotional change and lethargy. 17 Renfroe et al. reported that the pharmacokinetics of perampanel is approximately the same in pediatric patients aged 2 to <12 years. 57 However, we recommend that age and weight of children still need cautious consideration when making therapeutic strategies. Meanwhile, drug interactions should also not be ignored because the metabolism of perampanel is primarily mediated by CYP3A4 and/or CYP3A5 58 and drugs affecting these metabolic enzymes may influence its pharmacokinetics. In conclusion, when determining the treatment strategies for children with epilepsy, healthcare professionals should comprehensively consider multiple factors and make personalized treatment plans.
Analysis of safety signals by gender
Although gender may influence the pharmacokinetics and pharmacodynamics of drugs, in our study, the AE signals were approximately the same in male and female pediatric patients treated with lacosamide and perampanel. A study reported that age and sex had no significant effects on the rates of absorption and elimination of lacosamide because the slight numerical differences could be explained by body weight or volume of distribution. 59 Kohn et al. found no significant correlation between lacosamide serum concentration and gender in children, and a study involving 124 children (aged <18 years) also showed that there was no significant difference in the concentration/(dose/kg) ratios between male and female patients.60,61 These results are consistent with our study. As for perampanel, although the results showed similar safety profiles in both male and female patients, it should not be ignored that the number of AE reports involving dizziness and psychotic disorders was higher in female pediatric patients than in male. A phase III randomized clinical study also reported that dizziness and headache occurred more frequently in female group, and the results showed the exposure level of perampanel in females was relatively higher than that in males, which may lead to meaningful differences in efficacy and safety between genders. 62 However, another study involving 124 children (aged 4–15 years) receiving perampanel as primary monotherapy found that the proportion of males experiencing AEs was significantly higher than that of females (50.0% vs 28.1%, p = 0.012). 63 Therefore, considering the differences and limitations of existing researches, further large-scale multicenter clinical trials and systematic analysis are needed to determine whether there are any meaningful gender differences in the safety of perampanel.
Limitations
Our study employed the FAERS database to evaluate the safety of lacosamide and perampanel in the real world, solving the limitations of insufficient sample size and short follow-up time in clinical trials. However, this study also had some limitations. Considering the FAERS database is a spontaneous and open reporting system, its inherent limitations such as underreporting, misreporting, reporting bias, geographical limitations, and a lack of information on drug exposure may affect the analysis results. The FAERS database contains many low-quality reports submitted by nonprofessionals, which may lead to exaggeration of certain adverse reactions, or submission of AEs that are unrelated to the drug. Besides, the reports in the FAERS database are often lacking important information, or the relevant information is incomplete and not standardized, such as dosage, comorbidities, past medical history, concomitant drugs, and other influencing factors. This limits our ability to control complex factors in data analysis. 64 Although signals were detected through disproportionality analysis, they only proved the statistical correlations between the drugs and the AEs, and these signals require further validation through other study types (e.g., controlled clinical trials, epidemiological research) to establish an exact causal relationship. Therefore, our results could merely be considered as warning information. Furthermore, due to the lack of information such as dosage and body weight, it is difficult to evaluate whether the weight and dosage of drugs affect the AEs or lead to other AEs, while these factors are often crucial for assessing the safety profiles of drugs in pediatric populations.
Conclusion
This pharmacovigilance study analyzed the real-world safety of lacosamide and perampanel in children. The results indicated that AEs occurred most frequently within the first month of administration for both drugs, but they could still occur even after 1 year. Both lacosamide and perampanel had significant effects on the nervous system, including ataxia, drop attacks, and dizziness. Lacosamide was more prone to induce multidrug resistance and cardiac disorders compared with perampanel, while perampanel was more likely to affect the psychiatric system. Moreover, the results of subgroup analysis suggested that there were no significant differences in safety with respect to age or gender, but it’s worth noting that accidental exposure during pregnancy and medication errors were more common in children under 4 years of age treated with lacosamide. Our findings highlighted the need for cautious therapeutic decisions during pregnancy and emphasized the importance of rational use of drugs and correct medication education. This post-marketing study offers guidance for decision-making on treatment and provides reference data for the clinical monitoring of lacosamide and perampanel in children.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251389805 – Supplemental material for The real-world safety of lacosamide and perampanel in children: a disproportionality analysis of the FDA Adverse Event Reporting System
Supplemental material, sj-docx-1-taw-10.1177_20420986251389805 for The real-world safety of lacosamide and perampanel in children: a disproportionality analysis of the FDA Adverse Event Reporting System by Yani Tan, Cuilin Li, Qin Yi, Xin Liu, Yuping Yang, Lian Zheng, Long Wei and Li Huang in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
We thank the FAERS database for generously sharing its data, all subjects who submitted adverse event reports to the FAERS database, and all researchers who contributed to this research.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
