Abstract
Background:
Congenital anomalies represent a significant public health concern, with maternal drug exposure during pregnancy being a potential modifiable risk factor. While certain medications, such as antiepileptics, are well-documented for teratogenic risks, the safety profiles of many other drug classes during pregnancy remain understudied.
Objective:
Leveraging the FDA Adverse Event Reporting System (FAERS), this study aimed to systematically evaluate associations between specific medications and neonatal (<28 days old) congenital anomalies potentially related to maternal drug exposure through large-scale pharmacovigilance analysis.
Design:
A retrospective pharmacovigilance study was conducted by comprehensively searching the FAERS database to identify drugs potentially associated with congenital anomalies.
Methods:
We reviewed all reports from the FAERS database from January 2004 to December 2022, using the criteria of age less than 28 days and outcomes of congenital anomalies. Signal detection methods, including the Reporting Odds Ratio, Proportional Reporting Ratio, Bayesian Confidence Propagation Neural Network, and Multi-Item Gamma Poisson Shrinker, were employed to determine the associations between specific drugs and congenital anomalies, with a focus on potential teratogenic effects.
Result:
We reviewed 22,263,916 cases in the FAERS database and identified the association between 91 drugs and congenital anomalies, yielding 610 positive signals. Notably, specific drugs, including olanzapine and quetiapine among antipsychotics, valproate and topiramate among antiepileptics, sertraline, paroxetine, citalopram, escitalopram, and bupropion among antidepressants, and amlodipine and sartans among antihypertensives, exhibited higher teratogenic risks compared to their counterparts. Additionally, the use of omeprazole and metronidazole may lead to an increased risk of teratogenesis.
Conclusion:
Our research offers a thorough examination of teratogenic risks across different drug categories using data from the FAERS database, which can enhance the safety of medication use during pregnancy and inform clinical decision-making.
Plain language summary
Why was the study done? Birth defects are a major health concern. While we know some medications taken during pregnancy can increase this risk, the safety of many others isn’t fully clear. This study aimed to find out which medications might be linked to a higher chance of birth defects by looking at real-world safety reports.
What did the researchers do? The research team analyzed over 22 million reports collected in the US Food and Drug Administration’s (FDA) safety database during 2004–2022. They specifically looked for reports involving babies less than one month old where a birth defect was reported. They used special analysis methods to identify which medications appeared more often in these reports than would be expected by chance, suggesting a potential link to birth defects.
What did the researchers find? The study identified 91 different medications showing a potential link to an increased risk of birth defects. This included some medications used for: Mental health conditions (like olanzapine, quetiapine, sertraline, paroxetine, citalopram, bupropion), Seizures (like valproate, topiramate), High blood pressure (like amlodipine and “sartans” – e.g., losartan, valsartan), Other conditions (like the heartburn drug omeprazole and the antibiotic metronidazole). These medications showed stronger signals for potential risk compared to other similar drugs.
What do the findings mean? This large study using real-world safety reports highlights specific medications that may need closer scrutiny regarding their safety during pregnancy. While it doesn’t prove these drugs cause birth defects, the findings provide valuable information.
Introduction
Congenital abnormalities, also known as congenital anomalies, refer to structural or functional defects that arise during intrauterine life and can be identified prenatally, at birth, or sometimes only later in infancy. According to the World Health Organization, approximately 240,000 neonates die globally from congenital anomalies within the first 28 days of life each year. 1 These anomalies may result from a variety of factors, including genetic, environmental, maternal, and pharmacologic influences. 2 In recent years, the impact of pharmacologic factors on congenital anomalies has garnered significant research interest.
A historical example of drug-induced congenital anomalies is the thalidomide tragedy. Between its market release and subsequent withdrawal in 1962, thalidomide caused over 10,000 cases of teratogenic effects worldwide, resulting in more than 4000 fetal deaths in utero. 3 This incident underscored the inadequacy of relying solely on animal testing to ensure human safety and highlighted the potential risks even high-quality drugs may pose during pregnancy. It emphasized the necessity for comprehensive understanding and rigorous safety studies to address drug teratogenesis.
Although drug-related congenital anomalies are estimated to account for less than 1% of all congenital anomalies, only a few drugs have been definitively shown to be teratogenic in humans. 4 Due to the ethical constraints of conducting randomized controlled trials in pregnant populations, many drugs have not been systematically studied for their potential teratogenic risks. Consequently, real-world studies have become a crucial source of evidence in assessing medication safety during pregnancy. Regulatory agencies globally employ registry studies—prospective observational cohort studies on the risks of drug exposure during pregnancy—to augment the available safety data.
The advent of electronic information technology has revolutionized pharmacovigilance, with large database studies becoming a focal point for research. Techniques such as data mining and signal detection within pharmacovigilance databases have been particularly instrumental.5–8 Our study leverages the open and accessible FDA Adverse Event Reporting System (FAERS) database to identify drugs potentially associated with congenital anomalies. We aim to provide a comparative analysis of these drugs across various pharmacological classifications, thereby contributing valuable insights into the safety of medication use during pregnancy.
Methods
Data extraction
We conducted a retrospective pharmacovigilance study based on the FAERS database. FAERS is a publicly accessible database that contains adverse event (AE) reports submitted to the US FDA. These reports, which are voluntarily submitted by healthcare professionals, manufacturers, and the general public, include information on adverse drug reactions, product quality issues, and demographic data. The database is an essential tool for drug safety surveillance and signal detection.
Initially, the raw data obtained from the FDA official website were managed using Navicat Premium (Version 16.0, https://www.navicat.com/). Structured Query Language (SQL) statements were employed to extract and manipulate the data. Subsequently, the extracted data underwent manual review and normalization. Given that the FAERS database does not account for duplicate reports in its earlier iterations, and due to potential outliers or gaps in data collection attributable to human error, the study involved a comprehensive data cleaning process. This included the removal of duplicate entries, correction of identifiable data discrepancies, and elimination of records with missing or unverifiable critical information. These steps were essential to ensure the integrity and reliability of the data for subsequent analytical procedures.
We extracted individual case safety reports from the FAERS database with an outcome of “Congenital Anomaly” and an age of 28 days or less, spanning reports from the first quarter of 2004 to the fourth quarter of 2022. Following the exclusion of combination formulations and primary suspect drugs with fewer than three reported cases, we conducted a descriptive analysis of the extracted reports, detailing the year of presentation, gender, route of administration, country of reporting, occupation of the reporting individual, and identifying the top 20 suspected drugs and AEs based on the number of reports.
To standardize terminology for AEs, we used the Medical Dictionary for Regulatory Activities (MedDRA 25.1, https://www.meddra.org). In MedDRA, a Preferred Term (PT) represents a single medical concept (e.g., ventricular septal defect), and PTs are grouped into broader System Organ Classes (SOCs; e.g., “cardiac disorders”). Each PT within the extracted reports was then individually subjected to disproportionality signal detection to identify positive signals. Subsequently, these positive signals were categorized by their corresponding SOCs to facilitate stratified analysis. This approach allowed for precise identification of signals related to congenital anomalies at the detailed PT level prior to aggregation at the SOC level for overall statistical interpretation.
Drugs were classified according to the Anatomical Therapeutic Chemical (ATC 2023, https://www.whocc.no/atc_ddd_index/) at the therapeutic class level.
Disproportionality analysis
Disproportionality analysis is a statistical technique used to identify potential safety signals by comparing the observed frequency of AE reports for a specific drug to the expected frequency based on the overall reporting of AEs. The primary objective of this analysis is to detect any disproportionate reporting of specific AEs for a given drug, which could indicate a potential safety concern.
To enhance the accuracy of the analysis, we combined four methods: Reporting Odds Ratio (ROR), Proportional Reporting Ratio (PRR), Bayesian Confidence Propagation Neural Network (BCPNN), and Multi-Item Gamma Poisson Shrinker (MGPS). An AE was considered a positive signal if it demonstrated a positive correlation in all four methods, meeting the following criteria9,10: Number of AE occurrences (n) ⩾3, Lower bound of the 95% confidence interval of ROR (ROR05) >1, PRR ⩾2, Chi-squared (χ²) ⩾4, Lower bound of the 95% confidence interval of the Information Component (IC05) >0, and Lower bound of the 95% confidence interval of the Empirical Bayes Geometric Mean (EB05) ⩾2. Signals were categorized using the first tier of SOCs and the second tier of PTs in MedDRA. We focused on the top 20 signal values and signals indicative of teratogenic effects. It is essential to note that these PTs do not directly indicate congenital anomalies. Instead, the primary aim of this analysis is to investigate potential teratogenic effects associated with specific drugs, rather than to provide a broad evaluation of congenital anomalies as a general category.
We further categorized and analyzed these signals using MedDRA’s SOCs and PTs, with particular attention to those signals that rank high and indicate teratogenic effects. This process allowed us to identify and further investigate drugs potentially associated with congenital anomalies and to refine our understanding of the teratogenic risk linked to specific drug exposures.
We drafted this manuscript in accordance with the STROBE reporting guideline 11 and used the STROBE checklist 12 during editing, which is provided in the Supplemental Material (Supplement A).
Results
Clinical characteristics of congenital abnormality reports
From the first quarter of 2004 to the fourth quarter of 2022, the FAERS database recorded a total of 22,263,916 reports. After data cleaning, 15,881,398 reports were deemed valid for analysis. Among these, 21,104 reports were related to neonates, accounting for 0.13% of all reports. Analysis revealed that 6208 of these neonatal reports had a clinical outcome of “Congenital Anomaly.” Additionally, 6014 reports were associated with a primary suspect drug. Thus, these 6014 reports were categorized as drug-related neonatal congenital anomaly reports (see Figure 1 for the detailed workflow).
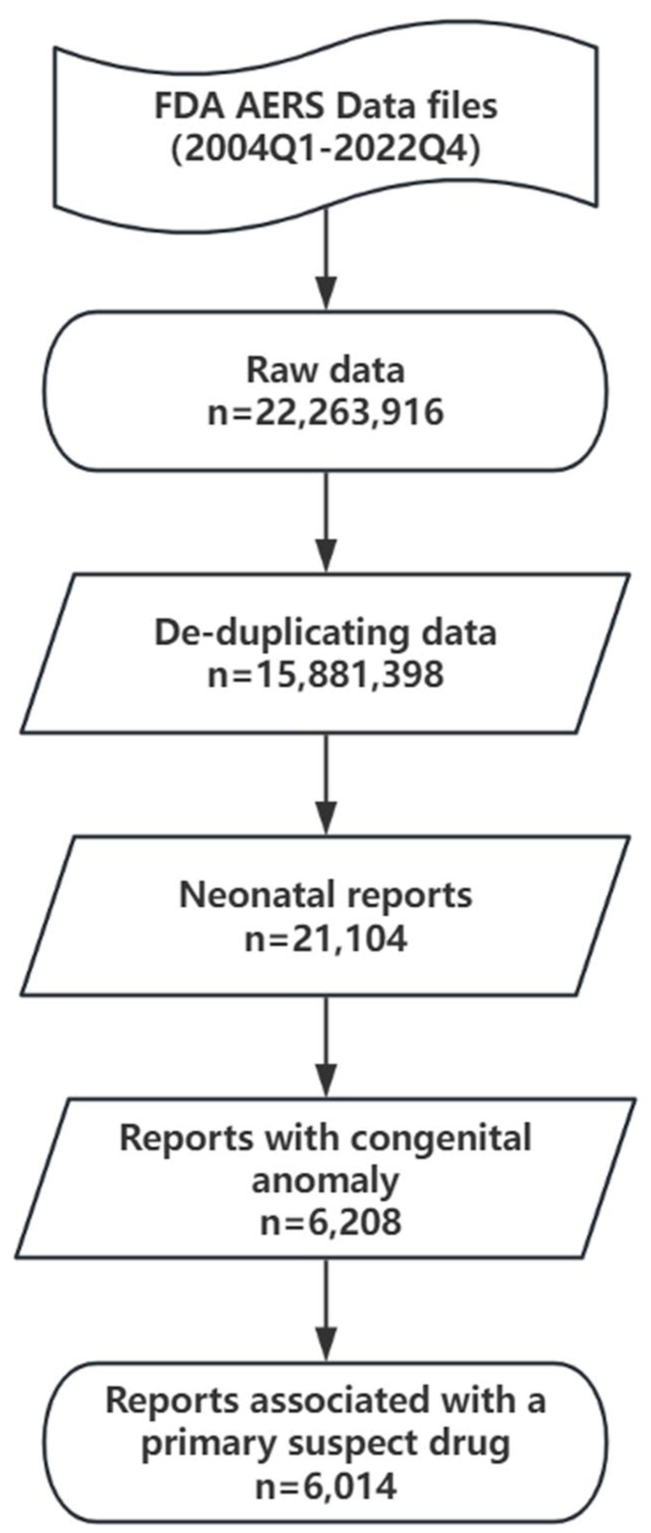
Data processing flowchart for reports of the FAERS database reporting from Q1 2004 to Q4 2022.
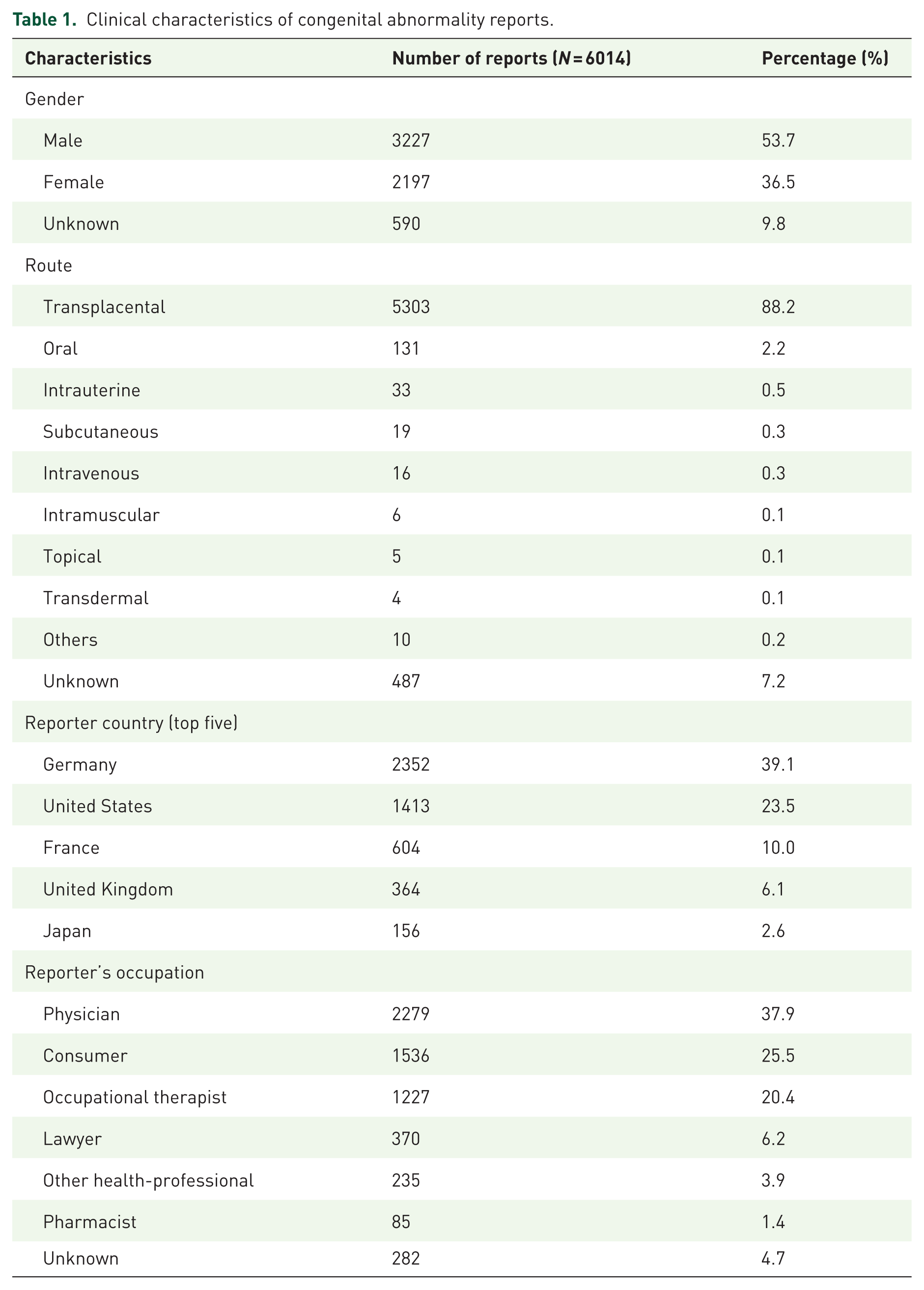
Gender distribution among the analyzed reports showed 3227 reports (53.7%) for male neonates and 2197 reports (36.5%) for female neonates. The remaining reports did not specify the gender. Nearly 90% of the reports indicated that the congenital anomalies were due to transplacental drug exposure. Reports from European and North American countries predominated, with Germany contributing the highest number of reports (see Table 1 for detailed distribution).
Clinical characteristics of congenital abnormality reports.
The annual number of drug-related neonatal congenital anomaly reports from 2004 to 2022 showed an increase from 128 in 2004 to a peak of 808 in 2014, followed by a gradual decline to 247 in 2022 (see Figure 2 for detailed distribution).

Annual number of congenital anomaly reports recorded in the FAERS database from 2004 to 2022.
In terms of the professional background of the reporters, physicians accounted for the highest proportion at 37.9%, followed by consumers at 25.5%, and occupational therapists at 20.4% (see Table 1 for detailed distribution).
Top 20 drugs and AEs of congenital abnormality reports
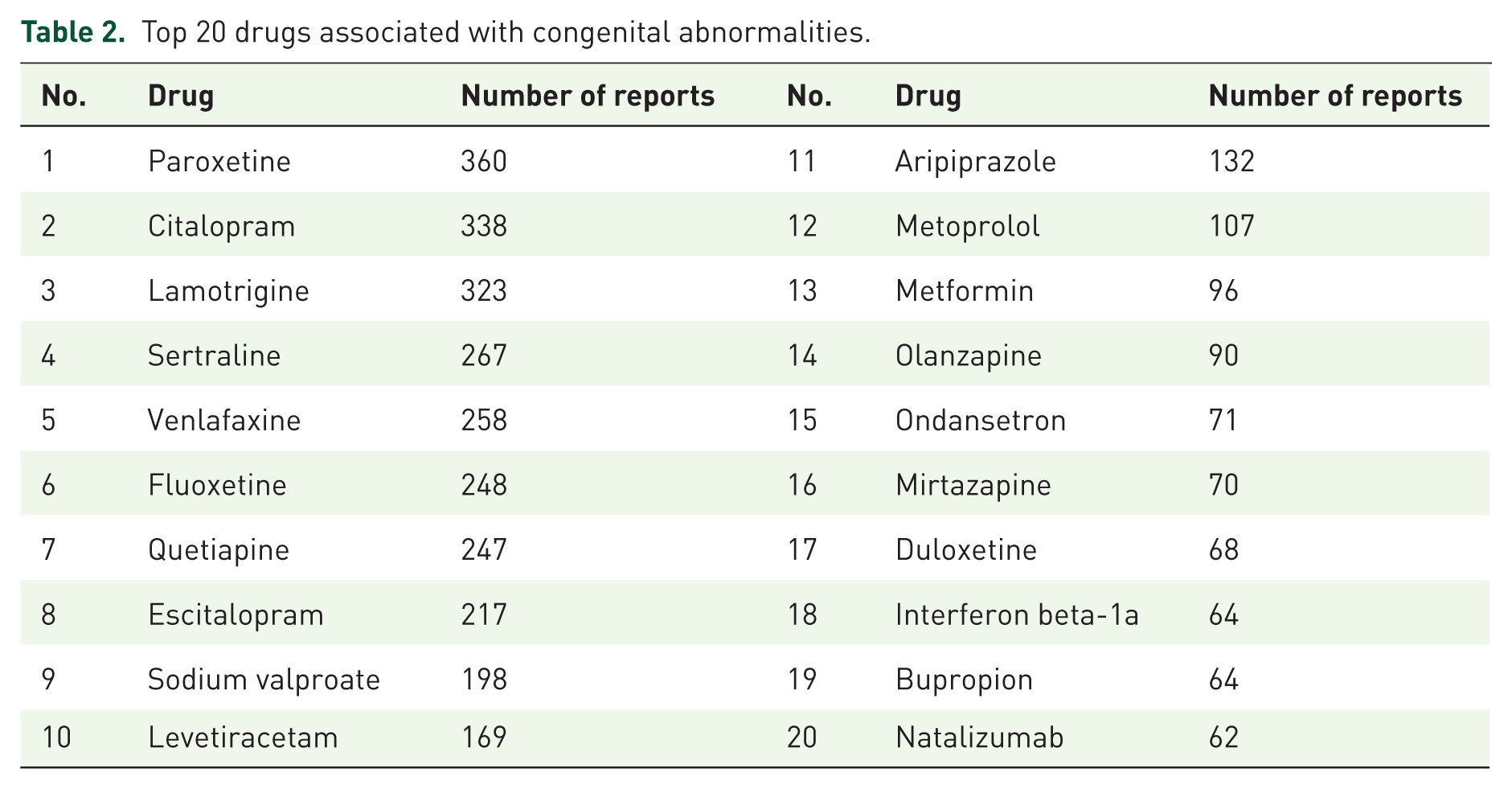
After excluding combination drugs and drugs with fewer than three reports, we identified a total of 195 primary suspect drugs. Table 2 presents the top 20 drugs by the number of reports. Among these, 15 drugs belonged to the nervous system category, further classified into three subcategories: antidepressants (Paroxetine, Citalopram, Sertraline, Venlafaxine, Fluoxetine, Escitalopram, Mirtazapine, Duloxetine, and Bupropion), antipsychotics (Quetiapine, Aripiprazole, Olanzapine), and antiepileptics (Lamotrigine, Sodium valproate, Levetiracetam). The remaining drugs included the antihypertensive Metoprolol, the antidiabetic Metformin, the antiemetic Ondansetron, and the immunomodulatory drugs Interferon beta-1a and Natalizumab.
Top 20 drugs associated with congenital abnormalities.
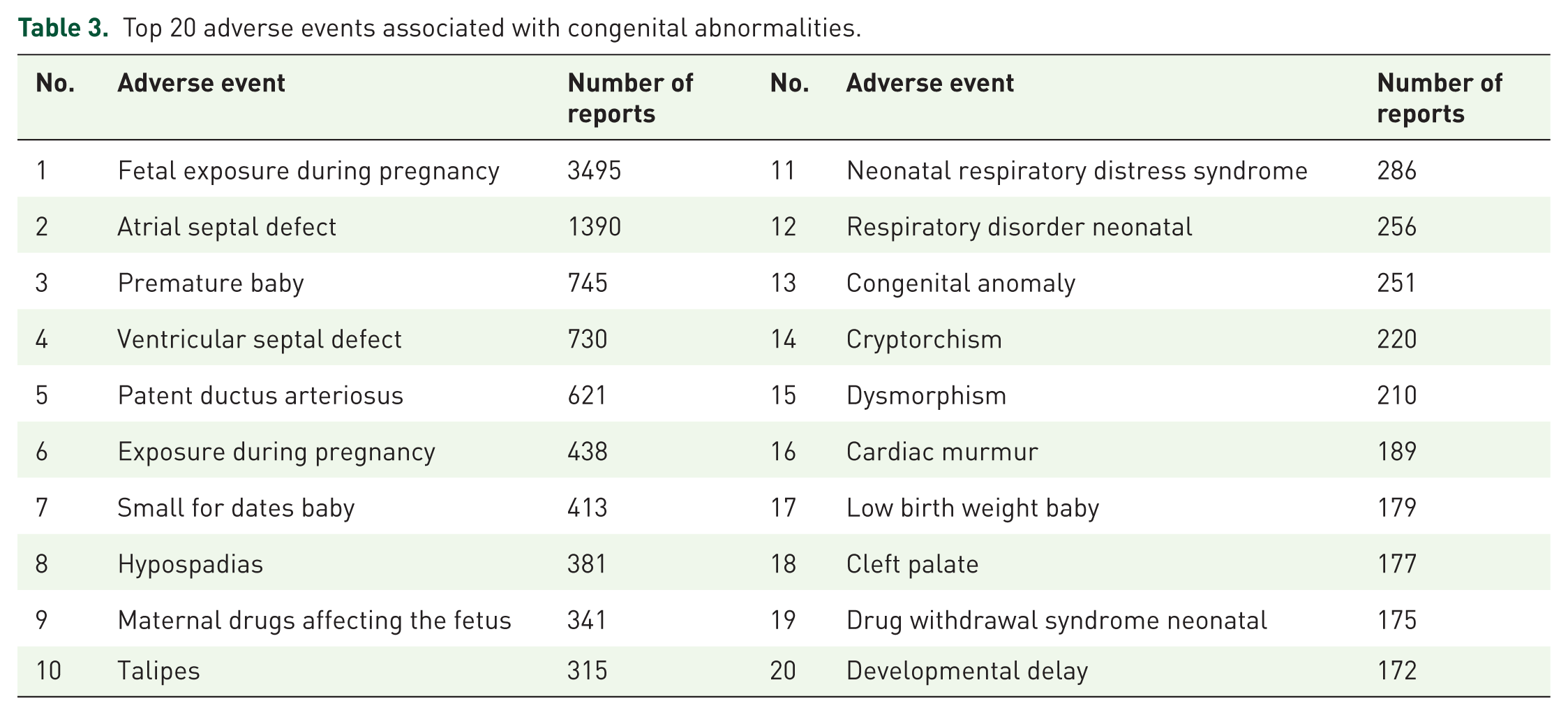
Based on our findings, we observed that terms related to drug exposure during pregnancy appeared with the highest frequency. These terms included “Fetal exposure during pregnancy,” “Exposure during pregnancy,” and “Maternal drugs affecting fetus.” Additionally, congenital heart diseases and adverse pregnancy outcomes were also prominent. Congenital heart diseases reported included “Atrial septal defect,” “Ventricular septal defect,” “Patent ductus arteriosus,” and “Cardiac murmur.” Adverse pregnancy outcomes comprised “Premature baby,” “Small for dates baby,” “Low birth weight baby,” and “Developmental delay.” Other frequently reported AEs included “Hypospadias,” “Talipes,” “Neonatal respiratory distress syndrome,” and others (see Table 3 for details).
Top 20 adverse events associated with congenital abnormalities.
Signal detection of drug-related neonatal congenital anomalies
We performed signal detection on the 195 primary suspect drugs related to neonatal congenital anomalies using four different methods. A total of 91 drugs were found to have signals associated with congenital anomalies, resulting in 610 positive signals. These signals indicate potential risks of specific congenital anomalies associated with these drugs.
Classifying the 91 drugs by the first-level categories of the ATC system revealed that the primary categories generating signals were nervous system (30 drugs, 30.0%), cardiovascular system (16 drugs, 17.6%), antineoplastic and immunomodulating agents (13 drugs, 14.3%), anti-infectives for systemic use (12 drugs, 13.2%), and other categories, albeit contributing to a lesser extent (see Figure 3 for detailed classification).
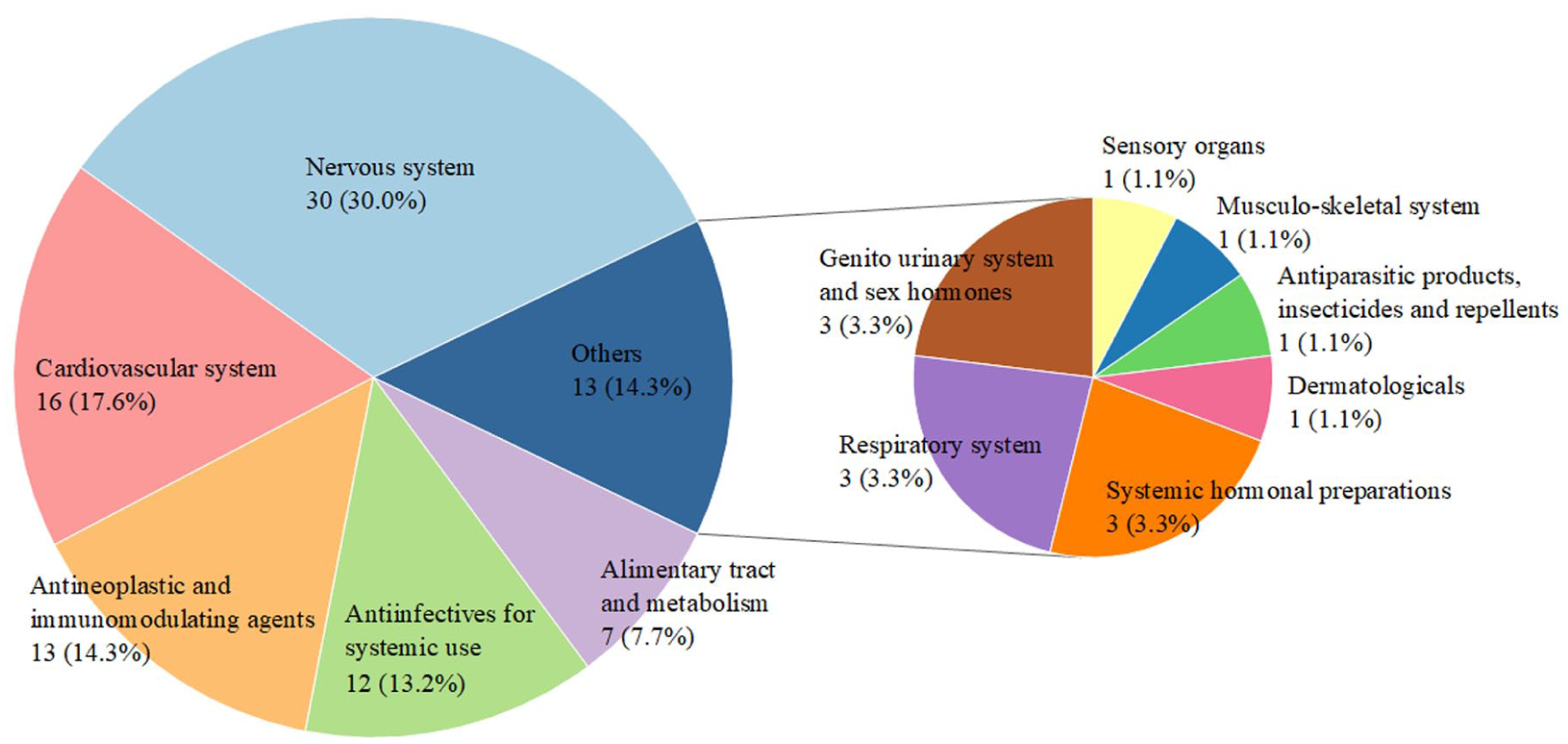
Drugs with congenital anomaly signals based on the first level of the ATC classification.
The 610 positive signals were categorized according to the SOC hierarchy of the MedDRA terminology. The most affected SOCs were cardiac disorders (71 signals), nervous system disorders (68 signals), musculoskeletal and connective tissue disorders (67 signals), and respiratory, thoracic, and mediastinal disorders (64 signals). Further analysis revealed that drugs affecting the nervous system primarily exhibited signals associated with disorders of the nervous system, cardiac system, respiratory system, and musculoskeletal system. Cardiovascular drugs primarily showed signals related to the musculoskeletal, renal, and respiratory systems. Anti-infectives for systemic use predominantly indicate signals involving cardiac, musculoskeletal, and gastrointestinal systems. Antineoplastic agents are primarily linked to fetal development issues and gastrointestinal disorders. Drugs targeting the digestive system and metabolism primarily led to infections, infestations, and gastrointestinal disorders. Other drug categories had fewer signals, affecting various SOCs more dispersedly (see Figure 4 for detailed classification).

SOCs involved in signals classified based on the first level of the MedDRA system.
Signals associated with congenital anomalies with high signal values
We summarized the top 20 signal values from each of the four methods, resulting in a total of 45 strong signals (see Table 4). These signals involved drugs from various categories, including nervous system, cardiovascular system, antineoplastic and immunomodulating agents, and others. The AEs predominantly affected SOCs such as nervous system disorders, musculoskeletal disorders, and respiratory system disorders.
All drugs with congenital anomaly signals.

Values in red indicate that the signal ranks among the top 20 signal values for the corresponding signal detection method.
EB05, Empirical Bayes Geometric Mean; IC05, Information Component; PT, Preferred Term; PRR, Proportional Reporting Ratio; ROR, Reporting Odds Ratio.
Analysis showed a high overlap of signals detected by the ROR, PRR, and MGPS methods, with 15 signals being common among them. However, the signals identified by the BCPNN method did not overlap with those detected by the other three methods. The highest signal values for the ROR, PRR, and MGPS methods were associated with “Pregnancy exposure before pregnancy” for letrozole (ROR05 = 1186.44, PRR = 1855.03, χ2 = 7576.83, EBGM05 = 222.29). In contrast, the highest signal value for the BCPNN method was “Fetal anticonvulsant syndrome” associated with valproate (IC05 = 2.89; see Table 4).
Several drugs were associated with multiple signals: Amlodipine had five signals, including rectourethral fistula, congenital uterine anomaly, bone deformity, vitello-intestinal duct remnant, and arrhythmia neonatal. Topiramate’s signals were pyelocaliectasis, congenital megaureter, and hypospadias, while valsartan’s signals were renal failure, pulmonary hypoplasia, and microcephaly. Valproate, sertraline, and rabeprazole each had two strong signals. Valproate’s signals were fetal anticonvulsant syndrome and congenital nose malformation. Sertraline’s signals were agitation neonatal and hypertonia neonatal. Rabeprazole’s signals were crying and muscle tone disorder (see Table 4).
Discussion
This study provides an in-depth analysis of neonatal congenital anomaly reports and drug-related issues during pregnancy using the FAERS database. From the first quarter of 2004 to the fourth quarter of 2022, neonatal reports accounted for only 0.13% of the total reports in the FAERS database. However, over 6000 neonatal reports were identified as drug-related congenital anomalies, highlighting that the proportion of neonates in drug safety monitoring is relatively small and easily overlooked. The restriction on conducting clinical trials involving pregnant women further limits the available research on drug use during pregnancy and neonatal congenital anomalies.
In examining the characteristics of neonatal congenital anomaly reports, we observed that nearly 90% of these reports involved at least one suspected drug. However, because FAERS data report suspected drugs without confirming actual transplacental exposure or causality, this high proportion should be interpreted cautiously as it reflects reporting characteristics rather than definitive evidence of in utero exposure or a causal relationship. Our study also revealed that male infants had a higher number of congenital anomaly reports than female neonates, which may be attributed to the physiological and biochemical characteristics of male neonates. Previous research has shown that male neonates may be more sensitive to external influences during embryonic development, 13 leading to a higher incidence of developmental abnormalities or malformations. Additionally, some congenital anomalies or genetic diseases are more common in male neonates, 14 which could explain the higher proportion of male infants in congenital anomaly reports.
Our study also highlighted the distribution of reports by country and reporter profession in the FAERS database. Reports from developed countries such as Germany and the United States were relatively high, which may reflect multiple contributing factors, including differences in regulatory frameworks, pharmacovigilance awareness, healthcare infrastructure, access to medicines, and cultural attitudes toward AE reporting, in addition to the FAERS database being established and supported by the FDA. The profession distribution of reporters showed that physicians were the primary reporters, emphasizing the crucial role of medical professionals in detecting and reporting neonatal congenital anomalies. Patients, their families, and occupational therapists also contributed to the reports, indicating the active role of patients’ families in identifying and reporting anomalies and the involvement of occupational therapists in the diagnostic and treatment processes.
“Fetal exposure during pregnancy” and related maternal exposure terms were among the most frequently coded AEs, highlighting the importance of considering in utero exposure in pharmacovigilance assessments.
We identified 195 primary suspect drugs, with antidepressants being the most frequently reported class. The safety of using psychotropic drugs during pregnancy, and which drugs are appropriate, remains a hotly debated topic. Existing data suggest that using antidepressants during pregnancy, particularly in the first trimester, is associated with a potential increased likelihood of congenital anomalies, including heart defects and neurodevelopmental abnormalities.15,16 For example, paroxetine has been linked to ventricular defects, citalopram to musculoskeletal defects, and venlafaxine to respiratory defects. 17 Although escitalopram showed comparatively weaker signal values than some other Selective Serotonin Reuptake Inhibitors (SSRIs), it still ranked eighth in the top 20 suspect drugs (Table 2). Notably, escitalopram was available only as a brand in Germany and the United States from 2002 to 2014, limiting its accessibility and potentially lowering reporting frequency during much of our study period. Thus, not being listed among the highlighted SSRIs does not necessarily indicate a more favorable safety profile.
Using four methods, we performed signal detection on the 195 primary suspect drugs, identifying 91 drugs associated with congenital anomalies, resulting in 610 positive signals. The most common drug categories generating signals were nervous system drugs, cardiovascular system drugs, and systemic anti-infectives.
Discrepancies between BCPNN and other methods (ROR, PRR, MGPS) were observed, with BCPNN detecting fewer overlapping signals. This difference is primarily due to variations in statistical assumptions and detection thresholds among these methods. BCPNN, as a Bayesian approach, incorporates prior probability distributions and tends to be more conservative in detecting signals compared to the frequentist-based methods ROR and PRR, which rely on observed-to-expected ratios. In contrast, MGPS applies empirical Bayesian adjustments to account for variability in reporting rates, leading to further differences in signal detection outcomes. These methodological differences highlight the importance of using multiple disproportionality analysis methods to enhance signal detection robustness, rather than relying on a single approach.
One of the key challenges in FAERS-based signal detection is the presence of concomitant medications, as many reports include multiple drugs. While we focused on primary suspect drugs as designated in FAERS reports, we acknowledge that potential confounding effects from co-administered medications cannot be ruled out. Some congenital anomalies may result from drug interactions rather than a single drug effect. Additionally, reporters may assign a particular drug as the primary suspect based on clinical judgment, but this may not always reflect the true causative agent.
To mitigate this limitation, we excluded combination drug formulations to reduce complexity. However, this approach does not account for potential interactions among separately reported concomitant medications. Given the observational nature of FAERS data, further pharmacoepidemiological studies using structured healthcare databases (e.g., pregnancy registries, electronic health records) are needed to adjust for confounding by indication and polypharmacy.
Our extensive signal detection and analysis of different drug categories potentially associated with congenital anomalies during pregnancy revealed several key points:
Nervous system drugs generated many signals related to anomalies in the nervous system, cardiac organs, respiratory system, and musculoskeletal tissues. Antipsychotics, antiepileptics, and antidepressants were significant contributors. Our findings align with several studies suggesting that using antipsychotics during pregnancy may be associated with musculoskeletal malformations, congenital heart disease, cleft palate, anorectal atresia/stenosis, and gastroschisis.18–20 Antiepileptics have been reported to be linked to congenital heart disease, orofacial clefts, genitourinary defects, and attention-deficit/hyperactivity disorder.21–24 Antidepressants have been suggested to be associated with heart defects, persistent pulmonary hypertension of the newborn, and neurodevelopmental disorders.25–29 For these three drug classes, we found that olanzapine and quetiapine among antipsychotics, sodium valproate (nasal malformation risk), and topiramate (renal and urinary tract malformation risk) among antiepileptics, and sertraline, paroxetine, citalopram, and bupropion among antidepressants showed stronger signal associations with major congenital malformations risks compared to other drugs in their classes.
Antihypertensive drugs generated many signals related to renal and urinary system, musculoskeletal tissue, and respiratory system anomalies, especially amlodipine and sartans. We observed stronger signals for certain sartans like valsartan, candesartan, losartan, and telmisartan. For instance, valsartan showed a signal for pulmonary hypoplasia, microcephaly, and renal failure; losartan with right aortic arch; telmisartan with skull hypoplasia; and candesartan with pulmonary hypoplasia and joint contractures. Although there are limited reports on specific teratogenic events caused by individual sartans, studies have reported fetal pulmonary and cranial hypoplasia and subsequent fetal death with losartan exposure, 30 neonatal acute perinatal renal failure with candesartan exposure, 31 neonatal renal insufficiency with valsartan exposure, 31 and limb abnormalities and spontaneous abortion with irbesartan exposure. 32 Amlodipine also had signals indicating associations with rectourethral fistula, congenital uterine anomaly, bone deformity, vitello-intestinal duct remnant, and neonatal arrhythmia. While amlodipine is widely used to treat chronic hypertension during pregnancy, research has mainly focused on its effects on the mother or pregnancy outcomes,33,34 with relatively little attention to its impact on neonates. Given the established contraindication of sartans in the second and third trimesters due to their well-documented fetotoxic effects, their use during pregnancy should be guided by established clinical recommendations, such as those from the American College of Obstetricians and Gynecologists. Furthermore, while amlodipine is generally considered to have a more favorable safety profile compared to sartans, its disproportionality signal for congenital anomalies warrants further investigation. Rather than making definitive clinical recommendations, our findings highlight the need for enhanced pharmacovigilance and further epidemiological studies to better characterize the potential safety profiles of these antihypertensive agents in pregnancy.
Our study found that omeprazole is potentially associated with gastrointestinal malformations, and metronidazole has a high risk of congenital cardiovascular anomalies. However, current research has not established a clear causal relationship between maternal exposure to these drugs and neonatal malformations.35,36 Given these signals, further research is needed to clarify their potential teratogenic effects.
In a word, our research highlights the importance of careful drug selection and monitoring during pregnancy to identify and evaluate potential safety signals related to congenital anomalies. Healthcare professionals should be particularly cautious with the use of antidepressants, antipsychotics, antiepileptics, and antihypertensive medications during pregnancy. Additionally, while our findings suggest a signal for omeprazole and metronidazole in relation to congenital anomalies, these associations require further validation in well-designed epidemiological studies before definitive conclusions can be drawn.
Limitations
Several limitations of this study must be acknowledged. First, as a spontaneous reporting system, FAERS is subject to inherent biases, including underreporting, selective reporting, and reporting exaggeration, which may affect the accuracy of signal detection. The voluntary nature of FAERS reporting means that certain drugs or AEs may be disproportionately reported, while others may be underrepresented, potentially introducing bias in the detected signals. The lack of denominator data (e.g., total pregnancies exposed to specific drugs) prevents the estimation of absolute risk or incidence rates, making it impossible to establish causality between drug exposure and congenital anomalies. Instead, our findings should be interpreted as hypothesis-generating signals that require further investigation through epidemiological studies. In addition, our exclusion of maternal (adult) reports means that some congenital anomaly cases reported via the mother may have been missed, potentially underestimating certain drug–anomaly associations.
Second, restricting cases to neonates ⩽28 days may exclude congenital anomalies diagnosed later in life, particularly neurodevelopmental disorders. Some congenital anomalies, such as cardiac malformations, may not be diagnosed immediately after birth, leading to potential underestimation of their association with drug exposure. This limitation suggests that our study primarily captures anomalies that are more likely to be detected at birth. Future studies should consider integrating data sources with long-term follow-up to assess the full spectrum of congenital anomalies.
Third, the FAERS database does not contain comprehensive clinical details, such as maternal comorbidities, genetic predispositions, or other environmental risk factors that could contribute to congenital anomalies. This limitation restricts our ability to account for potential confounding factors.
Finally, the use of MedDRA terms introduces challenges in accurately defining congenital anomalies, and variations in coding practices cannot be ruled out. While disproportionality analysis is a widely accepted pharmacovigilance approach, future studies should complement our findings with registry-based cohort analyses or active surveillance databases to further validate the observed signals.
Conclusion
Despite these limitations, our study offers insights and data on the relationship between neonatal congenital anomalies and drug safety during pregnancy. It emphasizes the need for healthcare professionals to closely monitor associated risks in clinical practice and highlights the limitations and challenges of researching neonatal congenital anomalies. Strengthening post-marketing drug safety studies and sharing experiences will improve public awareness of neonatal congenital anomalies and drug safety during pregnancy. These efforts are essential for the healthy growth of future generations and sustainable societal development.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251383765 – Supplemental material for Real-world pharmacovigilance study on neonatal congenital anomalies associated with maternal drug exposure using the FDA Adverse Event Reporting System
Supplemental material, sj-docx-1-taw-10.1177_20420986251383765 for Real-world pharmacovigilance study on neonatal congenital anomalies associated with maternal drug exposure using the FDA Adverse Event Reporting System by Jingping Zheng, Zhenpo Zhang, Lin Ma and Ling Su in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

