Abstract
Background:
Statins have been demonstrated to decrease cardiovascular events in high-risk patients. Statin-induced myotoxicity is a major contributor to statin intolerance and often the leading cause of statin discontinuation. Studies on the association between statin use and rhabdomyolysis risk remain limited.
Objectives:
This study aimed to compare the risk of rhabdomyolysis in patients who used statins versus those who did not.
Design:
A population-based case-control study was conducted.
Methods:
Data were collected from the Taiwan National Health Insurance Research Database between 2011 and 2020, involving 186,604 individuals with rhabdomyolysis and 746,416 without. Each patient with rhabdomyolysis (case group) was matched with four control patients based on the index year. Statins were assessed in both groups.
Results:
Approximately 50% of study participants were male, with an average age of 53 years. After confounding variables were adjusted for, patients who used statins exhibited a higher risk of rhabdomyolysis than those who did not (adjusted odds ratio (OR): 1.70, 95% confidence interval (CI): 1.68–1.73). Psychiatric disorders, alcoholism, generalized epileptic seizure, heat stroke, and crush injury were independent risk factors of rhabdomyolysis. Patients with psychiatric disorders who used statins exhibited a substantial risk of rhabdomyolysis (adjusted OR: 2.30, 95% CI: 1.95–2.71) compared with the reference group of patients without psychiatric disorders who did not use statins.
Conclusion:
Statin use was associated with a higher risk of rhabdomyolysis, and patients with psychiatric disorders who used statins exhibited an additive risk of rhabdomyolysis. These findings emphasize the need for clinicians to remain attentive to the potential risk of rhabdomyolysis in patients prescribed statins, especially in those with psychiatric disorders. Proactive monitoring, early recognition of symptoms, and individualized risk-benefit assessments are crucial to optimize treatment outcomes while minimizing adverse effects.
Plain Language Summary
Statins are medicines that help lower cholesterol. They can reduce the chance of heart problems. But some people stop taking statins because of muscle side effects. One rare but serious problem is muscle breakdown, called rhabdomyolysis.
We looked at health records from Taiwan between 2011 and 2020. We compared people with rhabdomyolysis to people without it. We checked how many in each group had used statins.
We found that people who used statins were more likely to get rhabdomyolysis than those who did not. The risk was higher for people with mental health conditions who also used statins.
Doctors should watch for signs of muscle problems in people taking statins. This is especially important for people with mental health conditions. Careful monitoring can help catch problems early and support safer treatment.
Introduction
Rhabdomyolysis is a rare but serious condition characterized by the breakdown of muscle tissue, resulting in the release of muscle proteins into the bloodstream. Common clinical presentations of rhabdomyolysis include muscular pain, weakness, reddish-brown discoloration of urine, and an increase in creatine phosphokinase levels. 1 Rhabdomyolysis can lead to critical complications if not promptly and effectively managed, such as myoglobinuria, acute kidney injury, hyperkalemia, and even death.2–4 A range of causes have been identified for rhabdomyolysis, including crush injury, excessive exercise, psychiatric disorders, alcohol abuse, use of certain medications, and toxicity.5,6
Statins, which are 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitors, are frequently prescribed to reduce cholesterol levels and prevent cardiovascular diseases.7,8 However, the side effects of these medications may include myotoxicity.9–13 The clinical spectrum of statin-induced myotoxicity includes an asymptomatic rise in creatine kinase (CK) levels as well as myalgia, myositis, myoglobinuria, and, in severe cases, rhabdomyolysis. Several studies suggest that the association between statins and myotoxicity may stem from statin-induced inhibition of coenzyme Q10 (CoQ10) synthesis, leading to reduced CoQ10 levels.14–16 Emerging evidence suggests statin-induced myopathy is linked to mitochondrial dysfunction and disrupted Akt/mTOR signaling pathways.12,17 Statins impair mitochondrial respiratory chain function, reducing adenosine triphosphate (ATP) production and increasing reactive oxygen species (ROS), which may trigger mitochondrial permeability transition, cytochrome c release, and apoptosis. Additionally, statins inhibit Akt activation, primarily due to reduced mTOR complex 2 (mTORC2) function, which is associated with mitochondrial dysfunction. This also leads to AMPK activation and impaired mTOR complex 1 (mTORC1) signaling, promoting muscle protein degradation, reducing protein synthesis, and further stimulating apoptosis. Collectively, these molecular disruptions drive clinical manifestations such as muscle fatigue, weakness, pain, and, in severe cases, rhabdomyolysis. 12
Statin-induced myotoxicity is a major contributor to statin intolerance and often the leading cause of statin discontinuation. 18 Numerous case reports have documented instances of statin-induced rhabdomyolysis, a critical condition associated with severe muscle damage.19–22 Despite the potentially severe consequences of rhabdomyolysis, studies investigating its risk ratio in patients using statins are limited. To address this gap, we conducted a population-based case-control study to evaluate the association between statin use and rhabdomyolysis risk.
Methods
Data source
Taiwan’s National Health Insurance program was launched on March 1, 1995. We collected data from the National Health Insurance Research Database (NHIRD), which is maintained by the Health and Welfare Data Science Center under the Ministry of Health and Welfare. The NHIRD covers 99% of Taiwan’s residents and includes comprehensive data on reimbursement claims for most healthcare services, encompassing inpatient and outpatient records and prescription drug information. Diseases are coded according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) and Tenth Revision, Clinical Modification (ICD-10-CM). Prescription drugs are classified using the Anatomical Therapeutic Chemical (ATC) system. For privacy protection, the NHIRD encrypts all personal beneficiary information. This controlled study, in which we analyzed NHIRD data from 2009 to 2020, was approved by the Research Ethics Committee of China Medical University Hospital (CMUH111-REC2-109(CR-2)). The reporting of this study conforms to the STROBE statement. 23
Study population
Case patients included those diagnosed with rhabdomyolysis, defined according to the codes ICD-9-CM: 728.89 and ICD-10-CM: M62.82. To minimize diagnostic bias, a confirmed case required at least two outpatient visits or one hospitalization with a diagnosis of rhabdomyolysis. Physicians diagnosed rhabdomyolysis on the basis of clinical symptoms—such as muscle pain, weakness, and reddish-brown urine—and elevated CK levels, typically defined as levels exceeding five times the upper normal limit. 24 The date of initial diagnosis with rhabdomyolysis was designated as the index date. Control patients were selected among individuals who were alive and free of rhabdomyolysis during the observation period using 4:1 matching on the basis of their index year for each case patient. A random index date within the study period was assigned to control patients. Individuals were excluded if their index year was outside the range of 2011–2020, their age or sex was unknown, they had incomplete records of statin usage, or they were aged over 90 or under 20 years.
Statins
Statin use was determined based on the ATC codes C10AA01 to C10AA05, C10AA07, C10AA08, C10BA01 to C10BA03, and C10BX03. The standard defined daily doses (DDD) for statins were as follows: simvastatin 30 mg, lovastatin 45 mg, pravastatin 30 mg, fluvastatin 60 mg, atorvastatin 20 mg, rosuvastatin 10 mg, and pitavastatin 2 mg. Statin use prior to the index date was analyzed for both the case and control groups. We calculated cumulative defined daily doses (cDDD) by summing the total amount of medication prescribed during the study period and dividing it by the DDD for each statin. Furthermore, we categorized statin users into four groups based on cDDD: non-users, ⩽100 DDD, ⩽400 DDD, and >400 DDD. These thresholds were defined using the 33rd and 66th percentiles to facilitate comparisons of therapeutic effects between groups.
Confounding variables
The following confounding variables were included in the study: schizophrenia (ICD-9-CM: 295; ICD-10-CM: F20, F25), psychosis (ICD-9-CM: 298.8, 298.9; ICD-10-CM: F23, F28, F29), paranoia (ICD-9-CM: 297.1 to 297.3; ICD-10-CM: F22, F24), bipolar disorder (ICD-9-CM: 296; ICD-10-CM: F30-F33, F34.8, F34.9, F39), alcoholism (ICD-9-CM: 571.0-571.3; ICD-10-CM: K70), generalized epileptic seizures (ICD-9-CM: 345.10; ICD-10-CM: G40.309, G40.401, G40.409), heat stroke (ICD-9-CM: 992.0; ICD-10-CM: T67.0XXA), and crush injury (ICD-9-CM: 926.8, 928.8, 928.9, 929.0, 929.9; ICD-10-CM: S77.20XA, S77.21XA, S77.22XA, S28.0XXA).
Statistical analysis
Counts and percentages were calculated for each categorical variable. Continuous variables were summarized as means with standard deviations. Differences in baseline characteristics between case and control patients were evaluated using chi-square tests for categorical variables and t tests for continuous variables. Univariable logistic regression was performed to calculate crude odds ratios (ORs) with corresponding 95% confidence intervals (CIs). Multivariable logistic regression was performed to adjust for age, sex, and comorbidities. Statistical analyses were conducted using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA and R version 4.3.1). A p value of less than 0.05 was considered statistically significant.
Results
Among the 933,020 individuals included in the study, we identified 186,604 with and 746,416 without rhabdomyolysis (Table 1). Compared with the control group, the rhabdomyolysis group had a higher proportion of comorbidities, the most common of which were bipolar disorder (5.29%), schizophrenia (2.34%), and alcoholism (1.69%).
Baseline demographic factors and comorbidity of study population.
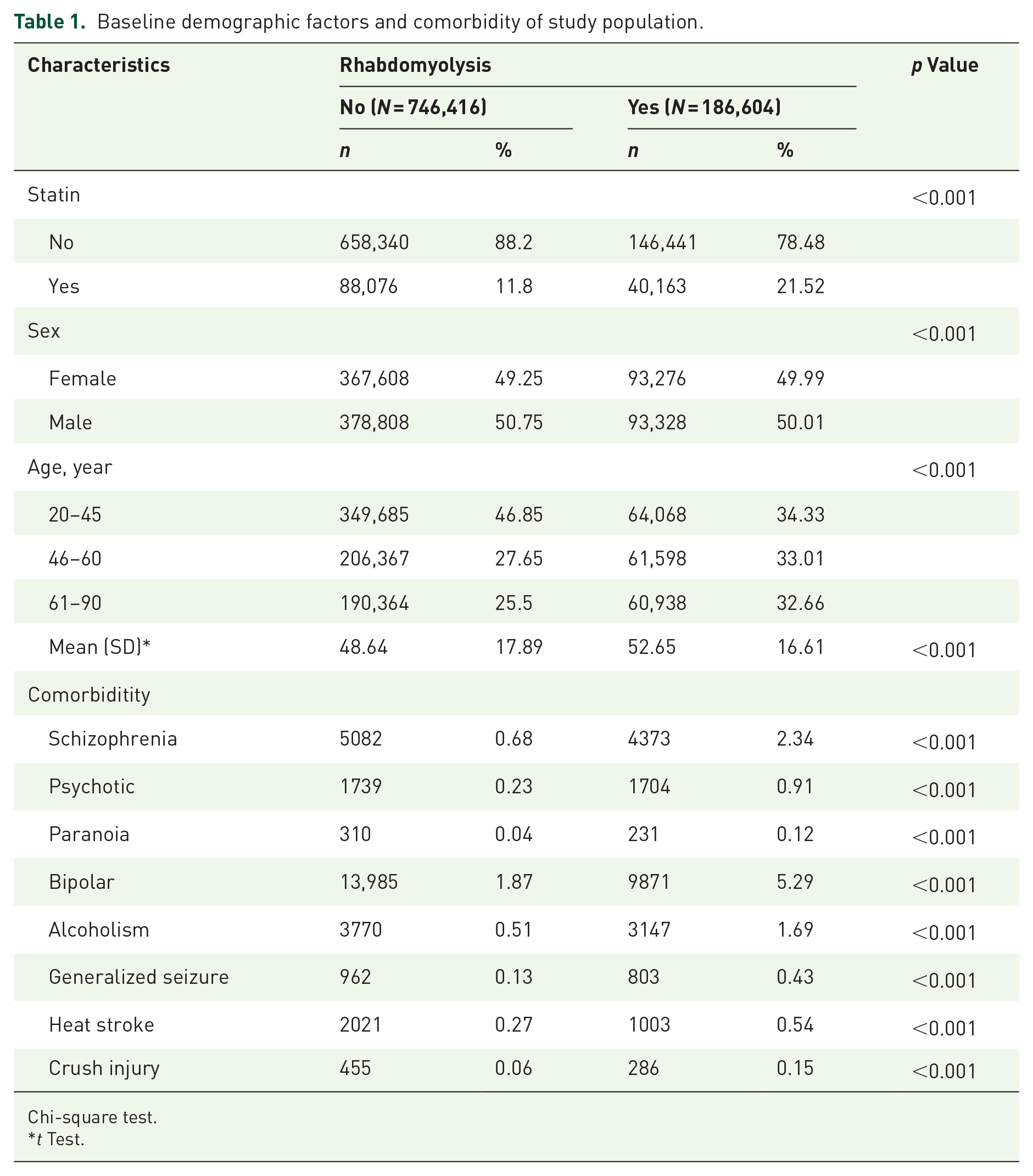
Chi-square test.
t Test.
As indicated in Table 2, after adjustment for age, sex, and potential confounding variables, statin users were found to have a higher likelihood of developing rhabdomyolysis compared with non-statin users (adjusted OR: 1.70, 95% CI: 1.68–1.73). The patients of older age exhibited a higher risk of rhabdomyolysis compared to the younger patients (adjusted OR: 1.49 for ages 46–60 years; adjusted OR: 1.50 for ages ⩾61 years). In the multivariable logistic regression analysis, additional risk factors for rhabdomyolysis were identified, including schizophrenia (adjusted OR: 2.52, 95% CI: 2.41–2.63), psychosis (adjusted OR: 1.96, 95% CI: 1.82–2.11), paranoia (adjusted OR: 1.27, 95% CI: 1.06–1.52), bipolar disorder (adjusted OR: 2.27, 95% CI: 2.20–2.33), alcoholism (adjusted OR: 2.82, 95% CI: 2.69–2.96), generalized epileptic seizure (adjusted OR: 2.52, 95% CI: 2.29–2.78), heat stroke (adjusted OR: 2.00, 95% CI: 1.85–2.16), and crush injury (adjusted OR: 2.25, 95% CI: 1.93–2.62). Logistic model analysis of the risk of rhabdomyolysis associated with the cDDD of statin use showed that patients using statins had a higher risk of rhabdomyolysis, regardless of the cDDD level (Table 3).
Logistic model analysis on the risk of rhabdomyolysis associated with statin.

Adjusted OR: adjusted for age, sex, and comorbidities in the logistic regression model.
CI, confidence interval; OR, odds ratio.
Logistic model analysis on the risk of rhabdomyolysis associated with cDDD statin use.

Adjusted OR: adjusted for age, sex, and comorbidities in the logistic regression model.
cDDD, cumulative defined daily dose; CI, confidence interval; OR, odds ratio.
Table 4 illustrates the association between psychiatric disorders and statin use in relation to the risk of rhabdomyolysis. Patients with psychiatric disorders who did not use statins (adjusted OR: 2.14, 95% CI: 1.97–2.32) and those using statins without psychiatric disorders (adjusted OR: 1.71, 95% CI: 1.68–1.73) exhibited an elevated risk of rhabdomyolysis compared with patients without psychiatric disorders who did not use statins. Notably, patients with both psychiatric disorders and statin use exhibited the highest risk of rhabdomyolysis (adjusted OR: 2.30, 95% CI: 1.95–2.71) compared with the reference group of patients without psychiatric disorders who did not use statins.
Interaction between psychiatric disorders and statin use on the risk of rhabdomyolysis.

Adjusted OR: adjusted for age, sex, and comorbidities in the logistic regression model.
CI, confidence interval; OR, odds ratio.
Discussion
This study is the first investigation of the association between rhabdomyolysis risk and statin use. Our analysis revealed that a higher proportion of patients with rhabdomyolysis used statins compared with those without the condition. Moreover, we observed a higher prevalence of psychiatric disorders (such as schizophrenia, psychosis, paranoia, and bipolar disorder), alcoholism, generalized seizures, heat stroke, and crush injuries among patients with rhabdomyolysis.
After adjustment for potential confounding variables, statin use emerged as an independent risk factor for rhabdomyolysis, with an adjusted OR of 1.70 (95% CI: 1.68–1.73). Although we did not identify a dose-response relationship between varying cDDDs of statins and the risk of statin-related rhabdomyolysis, an increased risk was consistently observed across statin users with different cDDD levels.
The pathophysiological mechanisms underlying rhabdomyolysis involve decreased ATP production and sarcolemma rupture, elevating levels of free ionized cytoplasmic and mitochondrial calcium. Increased calcium triggers a cascade of intracellular events, ultimately causing muscle cell damage and rhabdomyolysis. 1 The precise mechanism of statin-related rhabdomyolysis remains poorly understood. Statins inhibit HMG-CoA reductase, reducing the biosynthesis of mevalonate, a precursor of cholesterol and CoQ10. CoQ10 is essential for mitochondrial function, particularly regarding bioenergetics and oxidative stress regulation. 25 Statin use has been associated with reduced CoQ10 levels, which may impair mitochondrial energy production and increase oxidative stress. This dysfunction manifests as decreased ATP generation, increased production of ROS, and subsequent muscle cell damage or death.26,27 Emerging evidence suggests that CoQ10 supplementation can mitigate muscle symptoms associated with statin use, highlighting the potential of CoQ10 as an adjunctive therapy for statin-induced myopathy. 14
After adjusting for potential confounders, our analysis revealed that older adults had a higher risk of rhabdomyolysis compared with younger adults. Older individuals remain more susceptible to falls, and fall-related injuries may contribute to their increased risk relative to younger individuals. Patients with psychiatric disorders exhibited a significantly higher risk of rhabdomyolysis than those without such disorders. This heightened risk may be attributable to episodes of agitation and the use of antipsychotic medications, which are known contributors to the multifactorial etiology of rhabdomyolysis.28,29 In addition, we identified independent risk factors, including alcohol abuse, generalized seizures, heat stroke, and crush injuries, in alignment with the findings of previous studies. Our results underscore the necessity of vigilant monitoring and comprehensive risk assessment in vulnerable populations.3,24,30
Notably, psychiatric patients using statins exhibited a higher risk of rhabdomyolysis compared to non-psychiatric individuals not taking statins. The elevated incidence of rhabdomyolysis in psychiatric populations may stem from severe drug-drug interactions between statins, psychiatric medications, and alcohol consumption, which can elevate statin serum concentrations. 31 Collaborative efforts between pharmacists and physicians are critical to mitigate such interactions. These findings highlight a potential synergistic relationship between psychiatric disorders and statin use in rhabdomyolysis risk. Although statin therapy is associated with an increased risk of rhabdomyolysis, the absolute risk remains relatively low. Healthcare providers should carefully balance the cardiovascular benefits of statin therapy against its potential risks, including rhabdomyolysis. 32 Monitoring for symptoms such as muscle pain, weakness, or dark urine is critical, and patients should be encouraged to report any unusual symptoms promptly. Given the variability in individual responses to statins, treatment plans should be tailored to a patient’s overall health, medical history, and risk factors. A thorough risk-benefit analysis is essential when prescribing statins, and close monitoring during treatment is strongly recommended.
Statin-induced myotoxicity is also influenced by genetic factors that modulate drug metabolism, muscle function, and immune responses. Key pharmacogenetic profiles include SLCO1B1 polymorphisms (e.g., rs4149056), which elevate systemic statin exposure by impairing hepatic uptake, increasing myopathy risk. 33 Clinical applications of genetic testing (e.g., SLCO1B1 genotyping) may enable personalized statin selection and dosing, while screening for metabolic or immune-related variants aids risk stratification. 34 These findings underscore the role of pharmacogenomics in balancing cardiovascular benefits against the risk of statin-induced myotoxicity, enhancing patient adherence and outcomes.
The large, population-based cohort in our study enabled a robust analysis of rhabdomyolysis risk among patients in Asia using statins. Consequently, these findings have the potential to be generalizable to similar populations. However, this study has several limitations. First, recall bias may have been introduced due to the case-control study design. To mitigate this bias, we collected data from the NHIRD, which covers 99% of Taiwan’s residents and includes comprehensive information on reimbursement claims. Second, mild cases of rhabdomyolysis without noticeable clinical symptoms may have gone undetected, leading to potential misclassification and underestimation of the observed effect.
Conclusion
Our study demonstrated that patients who used statins had a higher risk of developing rhabdomyolysis compared with those who did not. Furthermore, patients with psychiatric disorders who used statins exhibited an additive risk of rhabdomyolysis. These findings emphasize the need for clinicians to remain attentive to the potential risk of rhabdomyolysis in patients prescribed statins, especially in those with psychiatric disorders. Proactive monitoring, early recognition of symptoms, and individualized risk-benefit assessments are crucial to optimize treatment outcomes while minimizing adverse effects.
Supplemental Material
sj-doc-1-taw-10.1177_20420986251365746 – Supplemental material for Increased risk of rhabdomyolysis in patients using statins: a population-based case-control study
Supplemental material, sj-doc-1-taw-10.1177_20420986251365746 for Increased risk of rhabdomyolysis in patients using statins: a population-based case-control study by Ya-Wen Lu, Jong-Yi Wang, Heng-Jun Lin and Wei-Sheng Chung in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
