Abstract
Background:
Pharmacovigilance (PV) inspections are critical regulatory assessments that evaluate the robustness and compliance of a company’s drug safety system. Despite their significance, there is limited published guidance on how organizations—particularly Medical Safety functions—can prepare operationally for such inspections.
Objectives:
To share practical, experience-based insights and organizational strategies for inspection readiness, execution, and follow-up, based on a recent large-scale European Medicines Agency PV inspection.
Design:
Descriptive case-based manuscript outlining the stepwise activities undertaken before, during, and after a regulatory PV inspection, with a focus on Medical Safety coordination.
Methods:
An internally coordinated approach was implemented, including preparatory meetings, document request management, cross-functional mock inspections, role assignments, and communication planning. A structured system was established for Medical Safety responses involving on-site and remote collaboration among safety leaders.
Results:
The inspection was successfully completed. The coordinated system enabled timely, consistent, and quality-controlled responses. Visual tools, including timelines and workflows, supported operational efficiency and stakeholder alignment. Post-inspection debriefs further informed process improvements across Medical Safety functions.
Conclusion:
This manuscript provides a practical, case-based framework for PV teams to approach regulatory inspections with strategic foresight and cross-functional coordination. The experience shared can serve as a useful reference, particularly for Medical Safety professionals and organizations seeking to strengthen inspection readiness and compliance operations.
Plain language summary
• Regulatory inspections assess whether pharmaceutical companies are effectively monitoring the safety of medicines.
• These inspections are complex and require detailed preparation, especially for drug safety teams.
• The article presents real-world lessons from a recent pharmacovigilance (PV) inspection. It outlines how one company organized and managed the inspection process.
• Practical tips are provided on team coordination, handling document requests, and maintaining clear communication. The content is especially useful for professionals new to the field of drug safety.
• The goal is to support better inspection readiness and improve overall safety management practices.
Introduction
Inspections by regulatory health agencies such as the US Food and Drug Administration,1–3 European Medicines Agency (EMA), 4 Japan Pharmaceutical and Medical Device Agency, 5 and UK Medicines and Healthcare products Regulatory Agency (MHRA), 6 as well as other regulatory agencies from around the world, are conducted to assess compliance with health authority regulations and company procedures to ensure patient safety and maintain public trust. Pharmacovigilance inspections typically focus on key areas of safety data management throughout the drug lifecycle. These areas include adverse events collection, management, and reporting, standard operating procedures (SOPs), signal detection and management, safety labeling changes, risk management, aggregate safety reporting, Post-Authorization Safety Studies (PASS), and the quality of safety data management processes. Understanding these focus areas allows drug safety teams to prepare effectively, ensuring their processes and documentation are inspection-ready and aligned with regulatory requirements.
Health authority inspections are generally classified into two main categories 3 : routine and for-cause. Routine inspections are conducted to ensure ongoing compliance with regulatory standards. In contrast, for-cause inspections are prompted by specific events or concerns, such as but not limited to noncompliance with reporting obligations, delays or failure to communicate safety updates with the health authority, or fulfillment of commitments, or whistleblower complaints, making them more targeted. A risk-based approach is fundamental in selecting Marketing Authorization Holders (MAHs) for pharmacovigilance inspections. This method ensures that regulatory resources are efficiently allocated, focusing on situations with the highest potential impact on public health. The health authority’s prioritization of inspections is guided by key considerations, including potential risk to public health, the complexity and nature of the products involved, and the extent of product use across diverse patient populations. In addition, the number of products overseen by a company might be a consideration in inspection risk profiling. A larger portfolio often signifies increased complexity in pharmacovigilance operations, prompting regulators to focus on system robustness, resource adequacy, and compliance across diverse products and regions. Other risk factors, such as the MAH’s compliance history, the novelty of therapeutic mechanisms, or vulnerabilities identified in previous inspections, further inform this prioritization. 7
The objectives of pharmacovigilance inspections include ensuring companies have the necessary personnel, systems, and facilities to fulfill their pharmacovigilance obligations; identifying and addressing noncompliance that may pose a risk to public health; and to use the inspection results as a basis for enforcement actions when necessary. 4 Inspectors may select specific products as examples to trace and verify critical information through the MAH’s processes. This approach not only ensures alignment with regulatory expectations but also provides a tangible means to evaluate the robustness of the MAH’s systems, identifying potential gaps or risks that could compromise compliance. Different regulatory agencies have their own set of standards and expectations for drug safety practices, although many commonalities exist. Drug safety teams must remain informed of each agency’s specific guidelines to ensure comprehensive compliance.
This paper aims to provide drug safety teams with practical strategies to effectively navigate pharmacovigilance inspections, ensuring seamless and efficient inspection management, with a particular focus on Medical Safety functions. By highlighting real-life preparation strategies, communication techniques, and collaborative approaches surrounding a recent major regulatory inspection, it offers a structured framework for inspection readiness and execution. Beyond serving as a resource for experienced pharmacovigilance professionals directly involved in regulatory inspections, this paper also aims to support new professionals entering the field, as well as those considering a career in drug safety. In addition, it seeks to facilitate clearer communication between disciplines, including those less familiar with the operational intricacies of safety oversight.
Key insights from a recent major regulatory inspection
Pre-inspection preparation
The company’s EU-QPPV (Qualified Person Responsible for Pharmacovigilance) received notification of a routine EMA inspection scheduled to take place in approximately 2 months’ time. This initial notification, which set the preparatory activities into action (Figure 1), included document requests covering a broad scope of pharmacovigilance processes and deliverables, including case processing and reporting, and medical safety. Documents requested at that time included SOPs, cases, and line listings, risk management plans (RMPs), and records of safety signals and labeling changes from the preceding 3.5 years. The response deadline was set at 30 days, and the inspection focus was indicated to be on Centrally Authorized Products, with the potential to extend to Nationally Authorized Products. No products or topics were identified at this stage.

This figure outlines the major phases of pharmacovigilance inspection readiness and execution. The timeline begins with the notification of inspection, followed by sequential activities: initial document request response, preparatory activities, mock inspection, on-site inspection execution, post-inspection debrief, and follow-up. Each phase is mapped against a calendar timeline to illustrate the flow and overlap of critical activities, emphasizing the need for proactive planning, cross-functional coordination, and timely execution to support a successful inspection outcome.
A month after submitting the initial responses, the company received a second round of document requests with a more targeted scope. This included PASS protocols and interim reports, specific product RMPs, safety data exchange agreements and pharmacovigilance agreements, and job aids. The inspectorate also provided a draft “Pharmacovigilance Inspection Plan” detailing the inspection topics and session schedules, requesting input from the MAH on participant assignments. The proposed topics were broad and aligned with the Good Pharmacovigilance Practices (GVP) Module III on pharmacovigilance inspections. 4
To prepare for the inspection, multiple cross-functional teams covering various aspects of drug safety—including pharmacovigilance operations, case processing, medical review, individual case safety report (ICSR) reporting, signal management, risk management, periodic safety reporting, quality management systems, global medical safety, and the EU-QPPV office—began preparing relevant documents, systems, and personnel. The preparations were closely aligned with EMA expectations and regulatory requirements, incorporating lessons learned and findings from previous inspections and associated corrective and preventive actions (CAPAs). The preparation activities also focused on identifying any potential inspection risks and ensuring that the proper mitigation strategies were in place to minimize the risks. The Audit and Inspection Readiness team, in conjunction with the Pharmacovigilance and Medical Quality Compliance teams, had the central role in coordinating inspection-related activities, liaising with the EMA inspectorate, and conducting a mock inspection to ensure the team’s inspection readiness.
Multiple preparatory sessions were held to ensure that the Medical Safety Subject Matter Experts (SMEs), comprised all products’ Global Safety Leads (GSLs) and Pharmacovigilance Scientists (PVSs) across the organization, had a clear understanding of their roles and responsibilities during the inspection. These sessions covered key topics, including an overview of the inspection background, the EMA Pharmacovigilance Inspection Plan, previously requested documents from EMA, anticipated areas of focus, and logistical details for the inspection period. Specific guidance was provided on expected attendance in the Front Room, where inspection interviews occurred, and the Back Room, where the company’s inspection team coordinated activities and responses. Some sessions were led by the Medical Safety Therapeutic Area Heads (TAHs), who provided specific guidance on how GSLs and PVSs should conduct themselves professionally, reinforcing key dos and don’ts during the interviews. These sessions emphasized the importance of concise and accurate responses, the necessity for a quick turnaround on all agency requests, and a review of prior inspection topics and questions. In addition, the TAHs provided oversight of the development of structured inspection readiness storyboards, summarizing key events and activities related to an identified risk or gap, activities undertaken to address the risk, along with high-level key messages to ensure consistency and alignment across the teams. In addition, specific guidance and instructions were provided to ensure high-quality inspection request responses. One week before the inspection, additional practice sessions were held, focusing on the SMEs outlined in the inspection agenda. These sessions ensured that each SME was well-prepared, aligned with their respective responsibilities, and equipped to contribute effectively to a seamless and efficient inspection process.
GSLs and PVSs play a critical role in evaluating and managing the safety and benefit-risk profiles of the products they are responsible in addition to identifying and investigating safety signals on an ongoing basis. Although several SMEs planned and ultimately traveled to the inspection site, given the large number of products potentially in scope of the inspection, it was logistically impractical for all GSLs to travel to the inspection site. To streamline processes, optimize resource allocation, and enhance inspection readiness, a structured system was devised to centralize document preparation, information flow, and team coordination. A single GSL was designated as the “Lead GSL,” traveling to the inspection site in another country to serve as the primary liaison for medical safety-related requests throughout the inspection. To support this role, two additional GSLs were appointed as “Coordinator GSLs” based in the United States (the primary location of the Medical Safety personnel). These individuals acted as intermediaries between the broader GSLs and PVSs teams in the United States and the Lead GSL and inspection team in the EU. This approach maintained strict control over document and information flow, simplified communication channels, and helped manage time zone differences between the EU (inspection site) and the United States.
During the inspection
A large team of SMEs from various functions traveled to the inspection site in the EU as in-person participants representing their functions to respond to inspectors’ questions/requests. The EU-QPPV and deputy QPPVs were also on-site, providing expertise and support as needed. The inspection followed the Pharmacovigilance Inspection Plan outlined by the inspectors. The active sessions took place in the “Front Room,” where each session included EMA inspectors (in-person or via teleconference), the EU-QPPV or Deputy QPPV, SMEs, and representatives from the Pharmacovigilance and Medical Quality Compliance teams serving as hosts. A designated scribe carefully documented the details of the discussions.
A large team of SMEs and support staff from various functions operated in the adjacent “Back Room,” where they could view real-time scribe notes from the sessions displayed on monitors. This team functioned as the Command Center for the inspection, proactively planning and preparing for any necessary actions or contingencies throughout the day.
Before each inspection session, SMEs convened to review the topics to be covered, align presentation strategies, and coordinate responses to potential questions. A designated SME led the discussion with inspectors, presenting relevant information and answering inquiries while deferring to other SMEs in the room, when necessary. This structured approach facilitated a smooth and efficient flow of discussions, ensuring that responses were provided by the most appropriate expert in the room.
During the inspection, all medical safety-related inspection requests were triaged to the Lead GSL rather than to individual product GSLs. These requests were logged into a tracking system and relayed to the US-based Coordinator GSLs for further triage. The Coordinator GSLs then directed each request to the appropriate product GSL, who provided a response. Before submission, the Coordinator GSLs reviewed responses for accuracy and completeness, consulting with the respective TAH for final approval. The responses were subsequently uploaded into the tracking system for review by the compliance team, which assessed whether they fully addressed the inspector’s request or required further clarification. If additional information or revisions were necessary, the compliance team communicated with the Lead GSL, restarting the cycle as needed (see Figure 2).

Illustration of the structured process used to manage medical safety-related document requests during the EMA inspection. The Lead GSL on-site in Belgium received and logged inspector requests, which were relayed to the US-based Coordinator GSLs. These coordinators triaged requests to the appropriate product GSLs, whose responses were reviewed by the Therapeutic Area Head, Compliance, and QA reviewers before submission. If clarifications were needed, the cycle restarted, ensuring timely, accurate, and consistent handling of all requests.
This structured approach proved highly effective, resulting in significant time savings and consistent handling of all inspection requests within required timelines. By centralizing the communication channels within the Medical Safety function, utilizing coordinator expertise, and involving key stakeholders such as TAHs and compliance teams, the company efficiently managed the high volume of inspection requests without compromising response quality. This experience underscores the importance of strategic planning, clear role delegation, and efficient cross-functional collaboration during inspections, particularly when operations are geographically dispersed.
At the end of each day, the inspection host reviewed the day’s document requests with the inspectors, while the Back Room manager debriefed the internal inspection team. Together, they determined response strategies and finalized plans for the following day. At the conclusion of the inspection, the inspectors shared a summary of their observations with the team and indicated that the full Inspection Report would be issued within 30 days.
Post-inspection
Upon returning, SMEs from multiple functions within Medical Safety convened to discuss the details of the inspection. They delivered a comprehensive presentation to the broader Medical Safety team, sharing their experiences, key takeaways, and lessons learned. In addition, they proposed several process improvements for future inspections, ensuring that the organization remains well-prepared for subsequent regulatory evaluations.
Medical safety readiness for pharmacovigilance inspections
As articulated by William Edwards Deming, we should “Cease dependence on mass inspection to achieve quality. (Instead, we should) improve the process and build quality into the product in the first place.” This principle underscores the importance of proactive quality assurance for inspection readiness. This philosophy remains pivotal in modern frameworks for ensuring excellence in product development and regulatory compliance. One way to achieve this is through a proactive approach to inspection readiness, which involves establishing a dedicated program within pharmaceutical companies. By embedding inspection readiness into the daily workflow, teams are better equipped to respond quickly and accurately during an inspection (see Figure 3).

This figure summarizes the major actions across the three critical phases of a pharmacovigilance inspection: preparation, execution, and follow-up. “Before the Inspection” focuses on readiness strategies such as SOP maintenance, mock inspections, cross-functional coordination, and risk identification. “During the Inspection” emphasizes structured communication, centralized request handling, real-time document retrieval, and professional conduct. “After the Inspection” includes responding to findings, implementing CAPAs, documenting lessons learned, and sustaining a culture of continuous improvement. Together, these actions reflect a comprehensive, process-driven approach to inspection readiness and compliance excellence.
Principles of pharmacovigilance inspection readiness
Inspection readiness is not an isolated activity, but an ongoing, integrated effort embedded into daily workflows. Robust procedures, monitoring of Key Performance Indicators, proactive internal mock inspections, cross-functional collaboration, robust data management, and structured documentation enable teams to navigate inspections efficiently. These strategies collectively enhance inspection readiness, positioning organizations to successfully demonstrate their commitment to pharmacovigilance excellence.
Establishing and maintaining SOPs that support operational excellence is essential for achieving regulatory compliance in all drug safety activities, forming the foundation for inspection readiness. Regular reviews and updates ensure that SOPs reflect current practices and evolving regulatory requirements. Clearly documented procedures help teams maintain consistency in safety operations and provide inspectors with a structured overview of how pharmacovigilance activities are managed.
A key component of this readiness involves internal audits designed to pressure-test processes, identify gaps, and streamline cross-functional collaboration. Audits assess compliance and help identify and address gaps, which ensures operational transparency, reinforcing compliance readiness. Similarly, mock inspections serve as valuable tools for preparing drug safety teams, ensuring readiness, and improving performance. By simulating realistic inspection scenarios, teams can anticipate common challenges, refine their responses, and build confidence in their ability to address inspectors’ questions. Mock inspections, much like internal audits, help identify potential documentation and process gaps that need correction before an actual inspection. Role-playing exercises assign specific responsibilities—such as primary points of contact, documentation retrieval, and SMEs—clarifying team members’ roles. Post-mock feedback sessions provide helpful insights into areas for improvement, fostering a cycle of continuous enhancement and reinforcing inspection preparedness (see Figure 4).
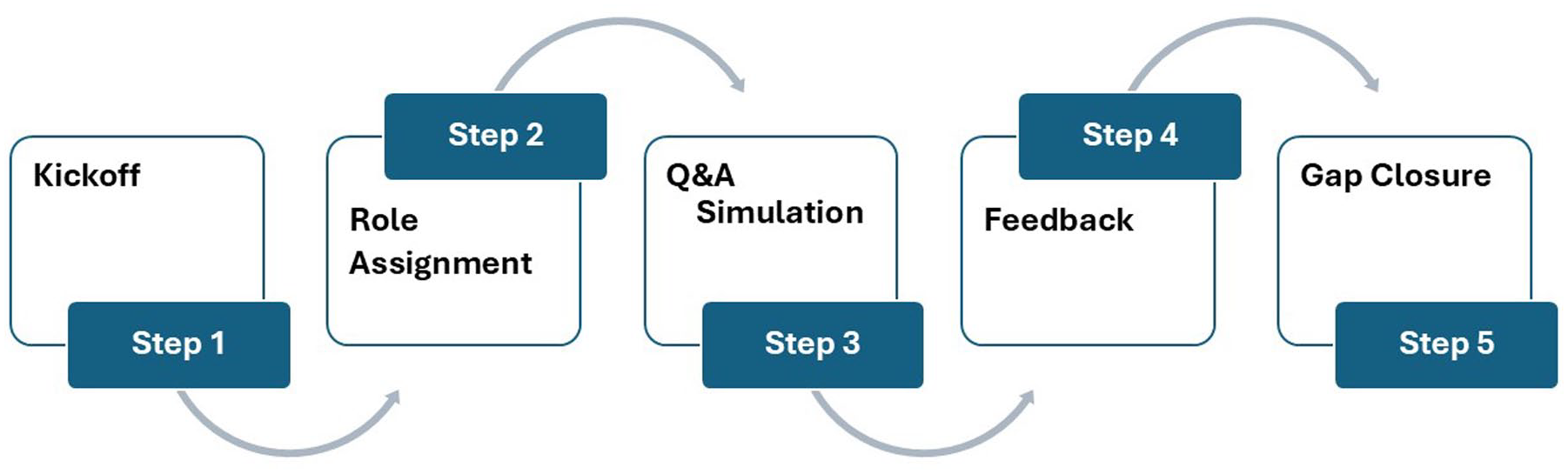
This flowchart illustrates the key steps in conducting a mock pharmacovigilance inspection to support organizational readiness. The process begins with a kickoff meeting and continues through role assignment, Q&A simulation sessions, feedback collection, and closure of identified gaps. This structured approach helps teams clarify roles, build confidence, and proactively address potential weaknesses before the actual regulatory inspection.
Successful inspection readiness also requires cross-functional collaboration within and outside the pharmacovigilance team. Clearly defining roles and expectations across departments creates a unified approach, ensuring alignment among all contributing functions such as Compliance, Regulatory Affairs, and Quality Assurance. Cross-functional training sessions and regular meetings enhance communication and shared accountability, helping teams streamline inspection-related tasks and address compliance requirements efficiently.
In addition, effective data management practices are essential to support all aspects of drug safety compliance. Teams must ensure that data entry, validation, and reporting processes are well-documented and aligned with regulatory requirements. Adverse event reporting, a cornerstone of regulatory compliance, requires particular attention. Teams must accurately report adverse events within mandated timelines, maintaining robust tracking systems to verify data integrity and conduct necessary follow-ups. Audit trails demonstrating adherence to reporting standards reinforce transparency and regulatory compliance.
Another key aspect of inspection readiness is ensuring that periodic safety reports, such as Periodic Safety Update Reports/Periodic Benefit-Risk Evaluation Reports, are submitted on schedule. These reports must include comprehensive benefit-risk assessments and undergo quality checks to ensure accuracy. Contributions from key stakeholders should be verified before submission to maintain regulatory credibility. Signal management and risk management are equally vital components of drug safety inspections. Teams must document and be able to demonstrate how they monitor and evaluate potential safety signals, ensuring timely actions are taken in response to safety concerns.
Finally, overall processes must be structured and summarized into a clear and accessible documentation system. Efficient documentation is vital for successful inspection outcomes. Drug safety teams should implement a well-organized electronic document management system that tracks updates, versions, and approvals. A centralized system prevents delays during inspections, allowing teams to retrieve requested documents promptly. Consolidating information into a coherent structure ensures that organizations present their processes as transparent, systematic, and well-controlled, reinforcing their compliance posture.
Pre-inspection preparation strategies
Centralized point of contact
Appointing a central Medical Safety point of contact as a “gatekeeper” during inspections helps streamline communications between the drug safety team and the inspection management team. This individual can manage requests, facilitate access to documents, and coordinate responses from various team members, ensuring a cohesive and organized inspection experience. A central point of contact minimizes miscommunications and enhances the overall flow of the inspection process. If the SMEs are in another location, especially if there is a large time difference, another central point of contact should be established in that location as a coordinator tasked to identify SMEs relevant to questions and facilitate gathering and reviewing the responses in a timely fashion.
“Front Room” preparation
Pre-inspection preparation requires a comprehensive review and understanding of relevant regulations established by the inspecting agency. For instance, in an EMA inspection, familiarity with GVP modules, associated addendums, and emerging trends is crucial for addressing regulatory expectations and standards effectively. This ensures that the team is well-versed in the specific expectations and standards governing pharmacovigilance activities, enabling them to confidently address regulatory inquiries and demonstrate compliance. By anticipating potential questions and document requests from inspectors, the inspection process can be streamlined. Teams should identify likely inquiries and ensure relevant documentation is readily accessible, with clear designation of responsibilities to team members. This preparation minimizes delays and enhances the inspection’s efficiency and fosters confidence in the organization’s readiness.
Developing a storyboard for high-profile drugs expected to be involved in the inspection can be beneficial. This approach allows teams to effectively address potential areas of concern by providing a narrative framework to articulate risk identification and mitigation strategies. Storyboards for each product should include both validated and non-validated signals, emphasizing those that led to updates in the Company Core Safety Information or Company Core Data Sheet. Significant regulatory requests, particularly those from the Pharmacovigilance Risk Assessment Committee or those resulting in Warning and Precaution (W&P) actions, should be documented in detail to demonstrate regulatory compliance. Similarly, storyboards for Direct Healthcare Professional Communications should demonstrate their development, distribution, and alignment with regulatory standards and organizational procedures. For the RMP, the storyboard should outline the triggers for its creation or updates, such as emerging safety data, and document the endorsement process by Safety Management Teams, ensuring that all decisions are supported by robust evidence and clear governance. Preparing storyboards for these critical areas allows Medical Safety to offer a comprehensive and transparent account of its processes. This enhances confidence in its pharmacovigilance practices and facilitates smooth interactions with inspectors or other stakeholders.
Identifying risks is a critical element of preparation involving the analysis of historical findings, compliance challenges, and process vulnerabilities. Analyzing past inspection reports to understand previous findings and commitments provides valuable insights into potential topics that may be raised during the inspection. Reviewing deviations/quality events and CAPAs is also relevant to potential inquiries from inspectors. It is also crucial to closely review the information provided in the pre-inspection document requests to anticipate questions during the inspection.
In addition, reviewing company policies and processes closely is fundamental. This includes SOPs, Job Aids, Forms, and other key documents that outline how pharmacovigilance activities are conducted. Mapping regulatory expectations to the company processes can facilitate discussions and responses to questions about it. Any discrepancies or outdated information should be identified and resolved prior to the inspection. It is important to understand how each process is executed and how it interrelates or intersects with other processes. Further, since inspectors often request a demo of various systems, prepare a demo checklist to make sure all systems work and that the system SMEs are on standby. It is also important to ensure that all the relevant mandatory trainings have been completed by appropriate staff in a timely fashion. Approaches to vendor management and oversight are also important for many inspectors, as they show how the company is keeping track of vendors’ performance.
Interview readiness training and guidelines
Preparing key staff for inspection interviews is essential, as inspectors often seek to understand individual roles and responsibilities. Training sessions that address common types of questions help staff anticipate what to expect, providing guidance on how to present clear and concise responses. It is crucial to reinforce that the interview is a professional conversation guided by mutual respect. The focus is not on the individual but on the company’s processes and procedures, and staff should understand that their role is to represent these processes effectively. Ensuring that team members are comfortable discussing their areas of expertise reinforces the impression of an organized and knowledgeable drug safety team. The better prepared team members are, the more confidence they will exude during interviews, ensuring a positive impression and successful navigation of the inspection process.
The staff should recognize that inspectors come well-prepared for their assessments, and understanding their approach is key to effective preparation. Inspectors will have read procedural documents in advance, equipping themselves with detailed knowledge of the organization’s systems and operations. They meticulously review all documents provided to ensure compliance, accuracy, and alignment with established procedures. They may ask questions they already know the answers to, using this tactic to evaluate the consistency, accuracy, and depth of the responses provided. In addition, they may pose the same question to multiple individuals to cross-check information and identify any inconsistencies within the team. Beyond verbal exchanges, inspectors also pay close attention to non-verbal cues, such as body language, which can indicate confidence, hesitation, or discrepancies in understanding. Moreover, they record everything said during the inspection, creating an official record for subsequent analysis and findings. Awareness of these practices allows teams to approach inspections with clarity, consistency, and professionalism, ensuring readiness to meet the inspectors’ expectations.
In cases where serious deviations are encountered, their potential impact and remediation actions need to be disclosed and should be done transparently and collaboratively. However, discussions with managers, the QPPV, Quality, and other pertinent functions should take place first to ensure alignment. Addressing these matters transparently not only demonstrates good faith and a commitment to improvement but also builds trust with the inspectors, reinforcing the organization’s dedication to regulatory compliance and patient safety.
There are clear guidelines on behaviors and actions that SMEs should avoid ensuring the integrity of the process. During the inspection, challenging questions may arise, and staff should be trained in techniques to address these with professionalism. During interview sessions, taking some time to pause and reflect prior to answering, asking for clarifications, and focusing on factual answers will contribute to a successful session. SMEs should not guess, fabricate answers, or provide misleading statements, as these can compromise the organization’s credibility and invite further scrutiny. Speculating or anticipating questions before they are asked is equally discouraged, as this can lead to inaccurate or irrelevant responses that detract from the focus of the interview.
During-the-inspection preparation strategies
Organizational and logistical planning
A well-organized and equipped inspection environment can impact an inspection’s success. The inspection management team should designate inspection “Back Rooms” with clear communication with support staff in various locations. Managing logistics such as secure file access, meeting spaces, and technical support creates a professional environment that greatly supports the team’s efforts. A dedicated support team available to retrieve documents in real-time is essential as it allows for quick access and production of necessary documents, avoiding delays. This real-time support boosts efficiency and keeps the inspection on track.
Professional communication during inspector interactions
At the beginning of an inspection interview, staff should set a professional tone by introducing themselves clearly. Their introduction should include their name, job title, functional area, and a concise explanation of their responsibilities relevant to the interview session. This establishes their role and expertise, providing the inspectors with context for their answers and demonstrating alignment with company processes. For example, an appropriate introduction might be, “My name is Jane Duo, Senior Director in the Medical Safety group. My team is responsible for the assessment of the safety and benefit-risk profiles of XYZ product(s) throughout their lifecycle.” Tailoring the introduction to the focus of the inspection ensures clarity and fosters productive communication.
Effective presentations by various functions are critical for promoting constructive and transparent interactions with inspectors. A natural and respectful demeanor establishes a professional yet approachable atmosphere, emphasizing the human factor in communication. Respecting the inspectors’ role and questions ensures mutual understanding and sets a positive tone. Self-confidence is vital, showcasing the presenter’s command of the subject matter and readiness to address inquiries. Maintaining a calm and confident approach, even with challenging questions, demonstrates a commitment to regulatory standards and public safety. This confidence should be balanced with thoughtful discretion, focusing on answering the question asked, providing concise and accurate responses, and avoiding extraneous details.
During an inspection interview, staff should adhere to the LTAS format—Listen, Think, Answer, and Stop—to maintain effective and professional communication. Start by listening carefully to the entire question before responding. Active listening helps staff fully understand the inspector’s inquiry and demonstrates attentiveness and respect. Avoid interrupting or answering prematurely, which can lead to incomplete or inaccurate responses. Once the question has been fully understood, take a moment to think and organize your thoughts. This pause helps in structuring a logical and concise response. When answering, address the specific question without offering unnecessary information. Flexibility is also vital, as inspections can shift focus, requiring staff to adapt quickly. If unsure about an answer, openly acknowledge and propose consulting the appropriate SME or providing the information later. By following the LTAS format, staff can navigate the interview effectively, ensuring clear and professional communication throughout the inspection process.
In addition, debating issues with inspectors or discussing them with others in the room during the inspection process is unacceptable. Staff must respect the inspector’s authority and avoid questioning or challenging it, as this can escalate tensions and harm the collaborative nature of the inspection. Arguments or defensive attitudes should be avoided, and staff should not agree to statements of noncompliance without proper review and consultation with relevant personnel.
Finally, staff should be comfortable with silence after answering and resist the urge to fill the silence. Inspectors may take a moment to reflect or record the response, and allowing this silence demonstrates confidence and composure. Purposeful silences by inspectors may prompt further elaboration, but staff should not respond with unsolicited information that could introduce confusion or unnecessary complexity. Similarly, casual conversation during the interview is inappropriate, as it detracts from the professional tone of the discussion. Adhering to these principles contributes to a successful inspection outcome and upholds the organization’s reputation.
At the conclusion of the presentation and interview, staff should thank the inspector for their time and efforts, demonstrating respect for the inspection process. After the interview, they should promptly leave to allow the inspection to proceed without unnecessary delays. Staff must remain ready to be called up for follow-up questions or clarifications by the inspector, ensuring they are accessible and responsive throughout.
All inspection-related requests should be prioritized with documents ideally within 24 h to avoid delays and demonstrate organizational efficiency and preparedness. Intentional omission or destruction of requested documents is a serious violation of regulatory requirements and ethical standards. Cooperation should also extend to interactions with colleagues, fostering a positive and supportive environment during high-pressure situations such as inspections. If challenges arise, staff should seek help from managers or other relevant SMEs to ensure accurate and complete responses.
Overall, maintaining professionalism, transparency, and open communication is key throughout the inspection. Team members should approach inspectors’ questions and requests with a respectful and collaborative attitude, presenting an image of competence and preparedness.
Post-inspection
Post-inspection activities are vital for reinforcing organizational strengths and addressing areas for improvement identified during the inspection. Start by reflecting on the inspection results and acknowledging the efforts and collaboration that contributed to the outcome. Regardless of findings, this is an opportunity to recognize the team’s dedication and reaffirm their commitment to regulatory excellence.
Addressing findings, observations, or further requests from inspectors should be a top priority. Staff should carefully review each finding, evaluate root causes, and develop a robust CAPA plan. A CAPA plan should outline clear timelines and responsibilities to ensure effective resolution while preventing recurrence. Prompt, transparent responses to the inspectors show a commitment to continuous improvement and regulatory compliance.
Documenting key lessons learned from the inspection provides valuable insights for future readiness and preparations. By recording strengths and identifying areas for improvement, the team can refine processes and better anticipate future inspection requirements. These learnings contribute to a culture of continuous improvement and readiness within the drug safety team.
It is essential to remember that the absence of findings does not equate to perfection. A “no findings” result should not lead to complacency but is instead a reminder that continuous improvement remains essential. Even without explicit concerns, processes and systems should be routinely evaluated and refined to ensure ongoing compliance and adapt to the evolving regulatory landscape. The risks and areas of interest identified during pre-inspection preparation remain relevant, requiring continued vigilance to maintain and enhance the robustness of existing processes.
Conclusion and future considerations
In conclusion, successful regulatory inspection readiness for drug safety teams relies on a proactive, organized approach that incorporates continuous preparation, cross-functional collaboration, and thorough documentation. Key approaches include establishing inspection readiness programs, utilizing mock inspections to build confidence and address weaknesses, and conducting staff training to prepare for interactions with inspectors.
A proactive approach to inspection readiness is critical for navigating regulatory scrutiny effectively. By preparing ahead of time, fostering a culture of compliance, and maintaining transparency, drug safety teams are better positioned to demonstrate their commitment to public health and regulatory excellence.
This manuscript is based on a detailed account of a single, large-scale EMA pharmacovigilance inspection experience. While it reflects the structured preparation, execution, and follow-up processes developed within a specific organizational context, the applicability of these practices may vary depending on company size, internal structure, and regulatory jurisdiction. The intention of this paper is not to generalize across all inspection scenarios but to provide a practical framework and real-world insights that can inform and inspire other pharmacovigilance teams. As the article does not present empirical data or follow a formal research methodology, conventional assessments of bias and generalizability are not applicable. Nonetheless, care has been taken to ensure accuracy, relevance, and alignment with current regulatory expectations.
As regulatory requirements evolve, drug safety teams must adapt to incorporate new technologies and emerging data sources into their inspection readiness strategies. Future trends, such as the integration of artificial intelligence in data management and increased focus on real-world evidence, will continue to shape the regulatory landscape. Therefore, staying informed and agile is essential for inspection preparedness.
Footnotes
Acknowledgements
The authors acknowledge Priyanka Mankar, Head, Audits and Inspections Readiness and Jennifer McDonough, Head, Pharmacovigilance and Medical Quality Compliance, Takeda Pharmaceuticals, for their valuable assistance in reviewing the manuscript and providing thoughtful and constructive feedback.
