Abstract
Introduction
The objective of this project was to assess the percentage of interoperability compliance within our pediatric hematology/oncology patient care areas for intravenous chemotherapy medications before and after the implementation of circle priming.
Methods
We conducted a retrospective quality improvement project at an inpatient pediatric hematology/oncology floor and outpatient pediatric infusion center before and after implementation of circle priming.
Results
There was a statistically significant increase in percent interoperability compliance for the inpatient pediatric hematology/oncology floor from 4.1% prior to implementation of circle priming to 35.6% after (odds ratio 13.1 (95% CI, 3.96−43.1),
Conclusion
Implementation of circle priming has significantly increased the percentage of interoperability compliance for intravenous chemotherapy medications in our pediatric hematology/oncology patient care areas.
Keywords
Introduction
Interoperability is a function of BD Alaris™ smart medication infusion pumps, a term used to describe how the smart pump communicates with the electronic health record (EHR). When utilizing interoperability, a provider places an order for an intravenous (IV) medication in the EHR and information imbedded in the order such as dose, rate, and duration of infusion are communicated to the smart pump wirelessly. The nurse administering the medication is required to scan barcodes on the patient, medication, and smart pump, and then any action completed on the smart pump will automatically be transmitted back to the EHR to be documented within the patient's chart. 1 Interoperability eliminates the need for manual programming of the smart pump, which in turn should reduce the risk of medication errors and simplify documentation.2,3 After the implementation of interoperability at our institution, it was identified through compliance reports that medications classified as IV chemotherapy had poor compliance with the use of interoperability, specifically in our pediatric hematology/oncology patient care areas. Meetings with nursing representatives were held to identify their potential barriers to utilizing the interoperability function for these medications. One modifiable barrier identified was the utilization of a process called “hyper-priming” in these patient care areas, described below.
Hyper-priming is a manual process performed by nursing staff to prime IV tubing with medication, at a rate faster than prescribed, from the chemotherapy bag to get the chemotherapy to the end of the tubing prior to starting the prescribed rate. Historically, the chemotherapy prepared in the pharmacy sterile processing and hazardous medication IV laboratory had an attached, closed-system tubing set that was pre-primed with compatible IV fluid. To hyper-prime a medication prior to administration, there are two potential methods. One method involves a nurse connecting the IV tubing to the patient and utilizing the smart pump to manually enter a rapid rate and volume to be infused to flush the pre-primed IV fluids from the tubing into the patient. Alternatively, a nurse could manually “open” the closed system and allow the fluid to flow freely into an unapproved receptable, such as the sink or trash bin. Both hyper-priming methods require estimation of the volume of pre-primed fluid in the IV tubing and are not compatible with interoperability due to the lack of utilization of the prescribed rates that have been entered into the EHR. Additional risks include the potential for administration errors if the chemotherapy reaches the patient during the priming process at an unprescribed rate, as well as improper hazardous drug handling posing a risk of exposure to nursing staff. These methods bypass safety mechanisms imbedded into the medication administration process and place unnecessary risks on both patients and nursing staff.
Circle priming is a novel process for priming medication tubing, utilizing medication from the IV bag, rather than pre-primed compatible fluid tubing. This process also takes advantage of a closed system for the entirety of the priming process. Circle priming sets consist of three components: (a) ChemoClave® Bag Spike with additive port, (b) an empty, BD Alaris™ primary infusion set (with or without a 0.2-micron filter) with a back check valve, and (3) a Spiros™ IV connector (Figure 1). The pharmacy department assembles the three components and attaches it to the IV chemotherapy bag in the pharmacy sterile processing and hazardous medication IV laboratory prior to sending the assembled IV chemotherapy bag and circle prime setup to the floor. Prior to attaching any tubing to the patient, the nurse will attach the circle prime tubing to the smart pump and select the “circle prime” function on the smart pump library. Once this is selected, the smart pump will begin to run and pull chemotherapy from the IV bag to prime the empty tubing with medication. Once the chemotherapy reaches the Spiros™ IV connector attached to the ChemoClave® adapter, the volume of chemotherapy used to prime the tubing will deposit back into the bag, maintaining a closed system during the entire process. Once the priming process is complete, the nurse will have medication-primed tubing and can proceed with utilizing appropriate safety checks to administer the IV chemotherapy as they would normally after attaching the tubing to the patient. Circle priming is compatible with interoperability because it occurs prior to any tubing being hooked up to the patient and does not require estimation of tubing volume. Additional benefits of circle priming include improved safety for both patients and nursing staff, as well as eliminating the need for pre-primed tubing. The objective of this quality improvement project was to assess the percentage of interoperability compliance within our pediatric hematology/oncology patient care areas for IV chemotherapy medications both before and after the implementation of circle priming.
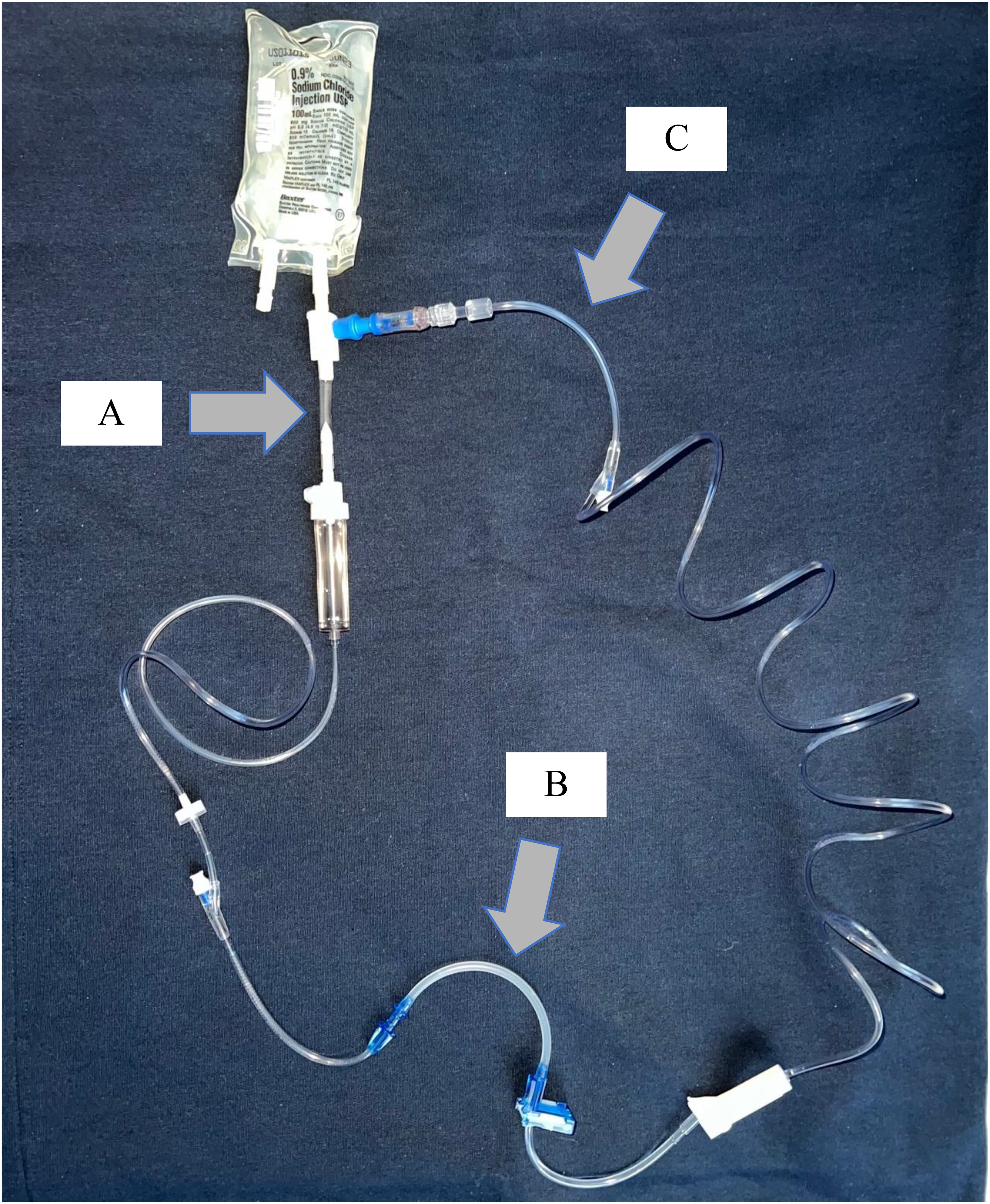
Circle priming setup.A—ChemoClave® bag spike adapter with additive port; B—empty BD Alaris™ primary infusion set (with or without a 0.2-micron filter) with a back check valve; C—Spiros™ intravenous connector (creates a closed system).
Methods
This was a retrospective quality improvement project that was deemed not human subject research by our Institutional Review Board because no patient-identifying data was collected. It was completed on an inpatient pediatric hematology/oncology floor at a single academic medical center and at an attached pediatric outpatient infusion center. Interoperability compliance was assessed between the smart pump and the EHR prior to the initiation of circle priming (January–March 2021) and the months following initiation of circle priming (August–October 2021) with a washout period in between. All data were collected by a single investigator utilizing a standardized data collection form. Each monthly interoperability compliance report was generated via an enterprise software company, known as SAP® BusinessObjects™. Compliance reports included data such as average compliance, non-compliant administrations, total number of administrations utilizing interoperability, and total number of attempts for each IV chemotherapy agent. To determine the total number of monthly administrations of each IV chemotherapy agent, a second report was generated via a query of the EHR. IV chemotherapy that is not circle primed (i.e., prepared in a syringe) was excluded from the analysis. The primary outcome of this project was to assess the percentage of interoperability compliance within our pediatric hematology/oncology patient care areas for IV chemotherapy medications before and after the implementation of circle priming. All data analysis were performed utilizing SPSS Version 28.0 (IBM Corp., Armonk, NY). Data were presented using descriptive statistics, including number and percentage. Compliant administrations were compared using the Chi-square test or Fisher exact test. Odds ratio and 95% confidence intervals were estimated. All comparisons were two-tailed and a
Results
Results for the primary outcome, showing total percent interoperability compliance both before and after the implementation of circle priming, are shown in Figure 2. There was a statistically significant increase in percent interoperability compliance for the inpatient pediatric hematology/oncology floor from 4.1% prior to implementation of circle priming to 35.6% after [odds ratio 13.1 (95% CI, 3.96−43.1),
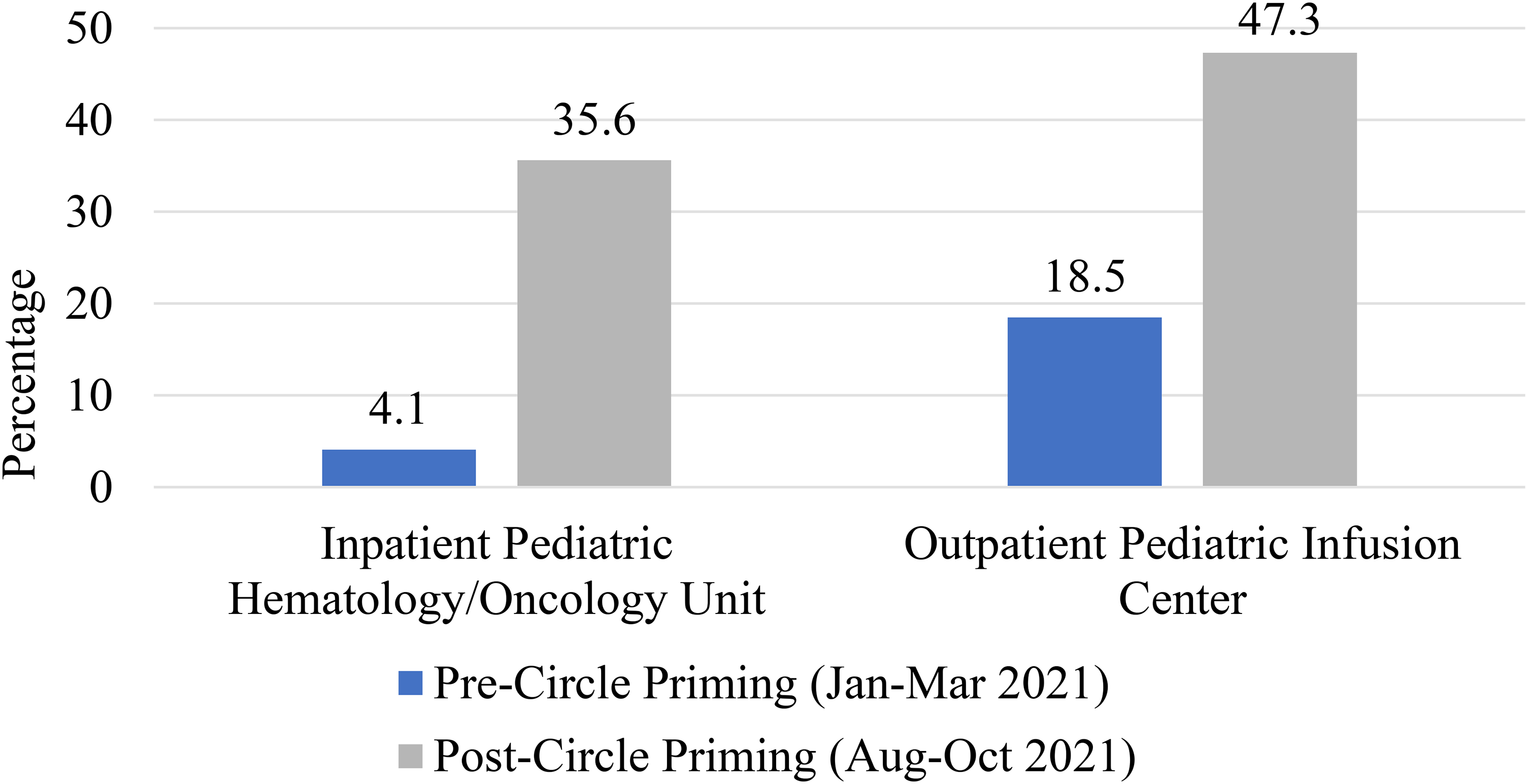
Combined percent interoperability compliance.
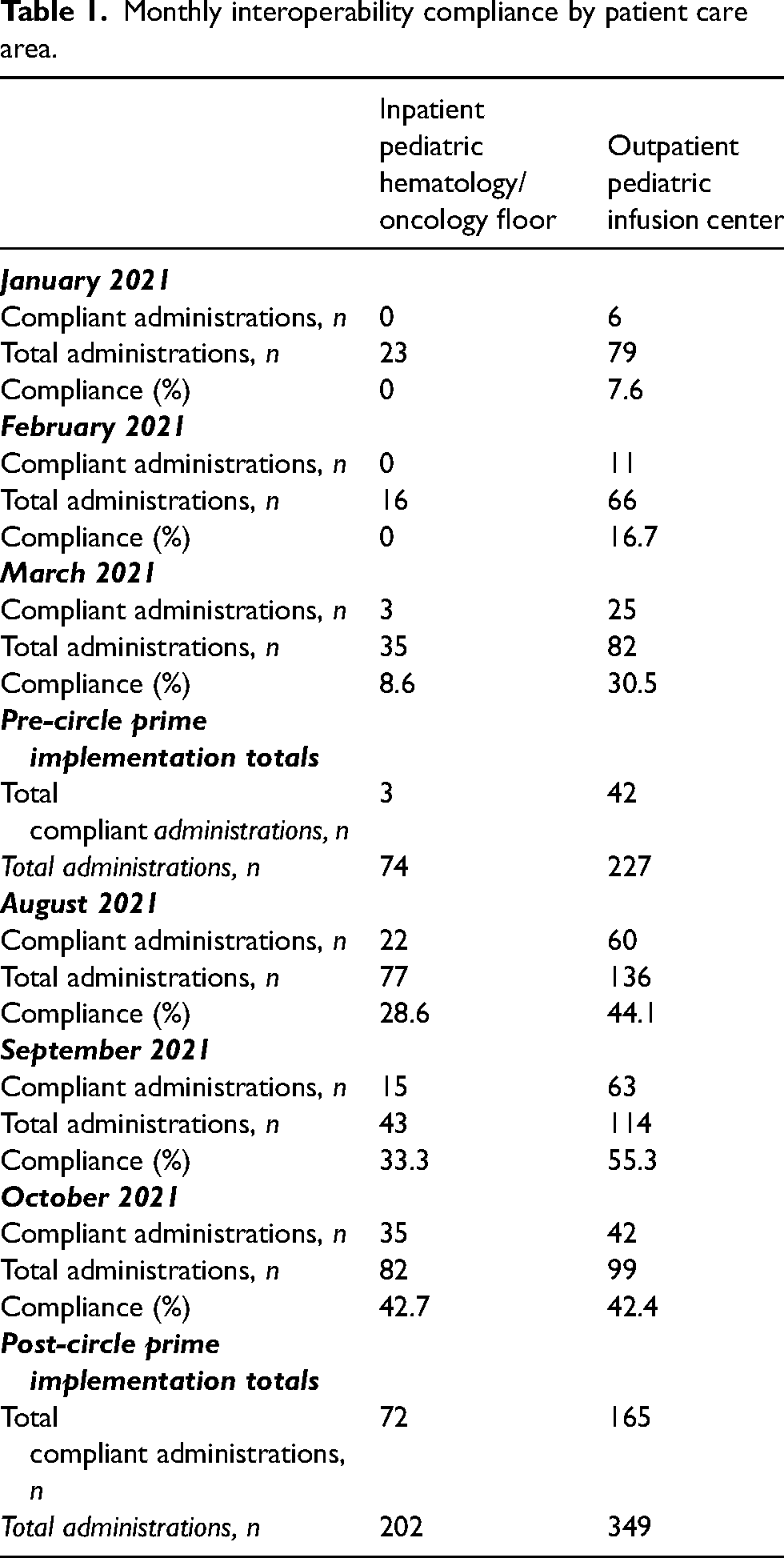
Total numbers of compliant administrations utilizing interoperability, all administrations, and percent interoperability for each of the analyzed months can be found in Table 1. To expand, for the months prior to the implementation of circle priming in our inpatient pediatric hematology/oncology floor, percent compliance rates were 0%, 0%, and 8.6% for January, February, and March 2021, respectively. The outpatient pediatric infusion center had slightly improved baseline percent compliance rates at 7.6%, 16.7%, and 30.5% for the respective months. Subsequently, the monthly percentage compliance rates for the inpatient pediatric hematology/oncology floor increased to 28.6%, 33.3%, and 42.7% for the months of August, September, and October 2021, respectively, after the implementation of circle priming. The outpatient pediatric infusion center also had increased percent compliance rates at 44.1%, 55.3%, and 42.4% for the respective months after the implementation of circle priming.
Monthly interoperability compliance by patient care area.
Discussion
To our knowledge, this quality improvement project is the first of its kind, as there is currently no published literature assessing the implementation of circle priming and its effect on compliance with interoperability. Safety is important, especially in high-risk medication areas such as hematology/oncology floors utilizing chemotherapy/antineoplastic agents. Various technologies, including smart pumps with dose error reduction software (DERS), computerized prescriber order entry, barcode medication administration (BCMA), and most recently interoperability, have been developed and are used by institutions to improve medication safety. Interoperability is one of the safety mechanisms that is yet to be universally implemented. Although smart pumps were created to help improve safety, they are not free of errors. Both the Institute for Safe Medication Practices (ISMP) and The Joint Commission have released statements providing recommendations to allow safe implementation of smart infusion pumps.4,5 The ISMP provides information on the limitations to smart pump technology and guidance on error-reduction strategies. Some of the smart pump limitations discussed in the ISMP guidance include the inability to prevent wrong-patient errors, wrong library selections, overriding alerts, and bypassing the use of the smart pump library altogether. To successfully engage interoperability, the ISMP states that an institution must have high BCMA compliance, a stable wireless connection, and capabilities for the smart pumps to communicate bi-directionally. Lastly, ISMP provides advice on how to implement interoperability and calculate metrics on interoperability compliance, recommending to attempt to achieve a 95% compliance rate for interoperability. 4 Similarly to the ISMP, Vizient Pharmacy Network has released a document on the importance of metrics in implementing interoperability and smart pump safety. They state that sharing interoperability compliance metrics with interdisciplinary colleagues, including nursing, can help with target attainment of compliance, which is our future direction for this project. 6 The Joint Commission Sentinel Event Alert Statement also identifies barriers to utilizing smart pumps, which include limitations in smart pump capabilities, alarm fatigue, availability of pumps, programming workflow, associated risks with secondary infusions, pump data analysis, and persistent deficiencies related to library use and updates. Similarly to the ISMP, The Joint Commission provides guidance on ways to overcome these barriers and optimize the use of smart pumps. Citing the ISMP, The Joint Commission reports that several errors occur because institutions are not optimizing their use of DERS technology. A study from 2013 found that the use of DERS technology could have prevented 28% of infusion errors and that the use of interoperability could have avoided 75% of reported problems. 7 The Joint Commission recommends connecting the EHR system with bi-directional communication smart pumps to implement interoperability and reduce manual programming errors. 5 Both of these organizations recommend utilization of DERS technology when using smart pumps to reduce the risk of errors. One major consequence of not utilizing DERS technology is bypassing pump safety mechanisms, which is what our institution was doing with the hyper-priming process. Schnock and colleagues found that bypassing smart pump safety mechanisms accounted for close to 10% of the total number of errors or policy violations related to infusion administrations. 8 Through implementation of interoperability, there should be less circumventing of safety mechanisms, leading to less errors and minimizing potential harm to the patient.
Misko and colleagues performed a pre-post study on the number of alerts from smart pumps in a hematology/oncology setting to assess the effects of smart pumps on safety with chemotherapy agents. Discussions with nursing staff were conducted to identify potential issues with utilizing the smart pump library and updates were made to the chemotherapy libraries based on their feedback. A statistically significant reduction in alerts related to concentration, fluid rates, and doses of chemotherapy was found after implementation of these changes to the drug libraries. 9 Interoperability is a more novel program that some institutions have been adopting in recent years. Skog and colleagues found the implementation of interoperability led to a significant reduction in total errors, expired medication errors, errors involving high-risk medications, and continuous infusion errors. Overall, these authors found that the use of interoperability resulted in a 16% reduction in medication administration errors. 2 Furthermore, Wei et al. 3 reviewed the impact of smart pump interoperability in a community hospital on patient safety and found that drug library compliance significantly increased from 73.8% to 82.9%. Additionally, they found that the number of alerts overridden within 2 s and the number of reprogramed alerts both decreased significantly.
Prior to implementation of circle priming at our institution, a cost utilization analysis was performed and presented to pharmacy leadership. Although circle priming was estimated to increase annual pharmacy equipment costs by about $9500, this increase was deemed appropriate given the potential patient and nursing safety benefits that circle priming provides. We showed that circle priming significantly increased interoperability compliance in both our pediatric hematology/oncology patient care areas. In addition, implementation of the circle priming process has been estimated to save about 15 h on average per week for our pharmacy staff by eliminating the need to pre-prime the IV tubing with compatible fluid prior to sending chemotherapy to the floor.
Limitations to this project include the retrospective study design and small sample size. Additionally, the data extraction process was very complex, leaving room for the potential for selection bias. Furthermore, the current percent interoperability compliance for IV chemotherapy in our pediatric hematology/oncology areas is still below 50%, demonstrating that there is still room for improvement based on the ISMP percent compliance goal of 95%.
Future directions include investigating additional barriers to utilizing interoperability to further improve compliance. Compliance percentages for IV chemotherapy at our institution significantly increased with the implementation of circle priming, however, there is plenty of room for further improvement. Our hope is to identify specific IV chemotherapy medications with the lowest percent compliance rates, survey our pediatric hematology/oncology nursing staff to determine potential reasons for non-compliance, perform education to address these barriers, and finally, to repeat a quality improvement evaluation.
Conclusion
Implementation of circle priming has significantly increased the percentage of interoperability compliance for IV chemotherapy medications in our pediatric hematology/oncology patient care areas. Although a significant improvement was seen, there is room for further improvement.
Footnotes
Contributor Statement
SW conceived the study idea. KA, SW, KS, and JB were involved with protocol development and running reports. KA was responsible for data collection. RS completed all data analysis. KA and RS wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
