Abstract
Background:
Valproate (valproic acid, sodium valproate) is authorised in Montenegro for epilepsy and bipolar disorder treatment. Due to known teratogenicity, risk minimisation measures were introduced in 2014 and further reinforced in 2018 by the implementation of the Pregnancy Prevention Programme (PPP). Despite these measures, consumption of valproate in Montenegro increased in the period 2016–2022.
Objectives:
To investigate the effects of risk minimisation measures on valproate prescription in Montenegro.
Design:
A retrospective, observational, 7-year, nationwide study.
Methods:
The Primary Health Care Information System (PHCIS) was used as a data source. The health records of women of childbearing potential (12–55 years) for the period 2016–2022 were analysed. Additionally, unstructured data were reviewed to determine the number and characteristics of valproate-exposed pregnancies. The software PASW, version 25.0 (SPSS Inc., Chicago, IL, USA) was used for the statistical analysis.
Results:
A total of 2247 women of childbearing potential using valproate were identified during the observed period. The number of patients using valproate for epilepsy treatment decreased by 24% while the use of valproate in psychiatry increased by 45% over the observed period. The age of the patient was the only predictive factor for successful PPP implementation (chi-square = 35.811, df = 4 and p < 0.001). The odds ratio (OR) for the age category was 1.22 (95% CI: 1.10–1.35). Contraception prescription was recorded in only 1.5% of patients following the PPP implementation. A total of 11 cases of exposed pregnancies were identified, while epilepsy was the indication in 5 (45%) cases.
Conclusion:
Regulatory risk minimisation measures had a limited impact on reducing the risk of valproate teratogenicity in Montenegro, with the most success in the group of youngest patients. Measures were more effective in the epilepsy indication. The rising use of valproate for psychiatric indications is concerning. Targeted education, better preconception care and improved coordination among healthcare professionals are necessary.
Plain language summary
Why was the study done: Valproate is an effective medicine used for epilepsy and bipolar disorder treatment. However, it is known that the use of this drug during pregnancy is harmful to the unborn baby. Valproate increases the risk of malformations as well as many developmental disorders. Many measures have been taken to prevent the use of valproate during pregnancy and its side effects. The goal of the research was to determine the effects of measures implemented so that they could be improved and more effective in the future.
What did the researchers do: The researchers analysed the prescription of valproate over seven years (2016–2022) to determine the effects of the measures taken. The factors that influenced the success of the measures were determined, as well as the number and characteristics of cases in which the measures were not successful and pregnancy occurred during the use of valproate.
What did the researchers find: The researchers found that during this period valproate was prescribed to 2,247 women of reproductive age. The number of women using valproate to treat epilepsy decreased by 24%, but the number of women using this drug as treatment for psychiatric disorders increased by 45%. The measures were more successful with very young women, aged 12 to 20. Contraception was prescribed to 1.5% of women during this period, and 11 women became pregnant during the use of valproate.
What do the findings mean: The conclusion of the research is that although the implemented measures have produced some results, it is necessary to further educate patients about this topic, especially when it comes to the use of reliable contraception. It is necessary to improve preconception care and cooperation between health workers and patients in order to avoid unnecessary exposure to valproate during pregnancy.
Introduction
Valproate (valproic acid, sodium valproate) is a well-established antiseizure medication that is authorised in Montenegro for the treatment of epilepsy and bipolar disorder. 1 In some countries, valproate products are also approved for migraine prophylaxis. 2 Valproate is a prescription-only medicine, and the treatment is reimbursed by the Health Insurance Fund (HIF) of Montenegro. Valproate is known to have a high teratogenic potential – approximately 11% of children exposed to valproate in utero will have a congenital malformation, and the risk of neurodevelopmental or behavioural disorders is 30%–40%.3 –5 In addition, exposure to valproate in utero is associated with the risk of lower intelligence quotient, autism and attention deficit hyperactivity disorder.6 –11 Regulatory authorities around the world have introduced risk minimisation measures to reduce exposure to valproate during pregnancy. In Montenegro, as well as in the European Union (EU), a Direct Healthcare Professional Communication (DHPC) was distributed in 2014 to inform healthcare professionals that girls and women of childbearing potential should not be prescribed valproate unless other treatments are ineffective or not tolerated.12,13 These measures had not achieved satisfactory results in Europe.14,15 In 2018, the European Medicines Agency (EMA) introduced new risk minimisation measures, including the Pregnancy Prevention Programme (PPP). 16 The use of valproate to treat migraine and bipolar disorder during pregnancy is contraindicated, while the use of valproate to treat epilepsy during pregnancy is contraindicated unless there is no other effective treatment. In addition, valproate should not be prescribed to women of childbearing potential unless the conditions of the PPP are met. These measures included an assessment of each patient’s potential to become pregnant, pregnancy tests before starting and during treatment with valproate, advice on the risks of valproate treatment, the need for effective contraception throughout treatment, an annual review of ongoing treatment by a specialist and adding a visual warning on the packaging of the medicine. The PPP was introduced in Montenegro in December 2018. 17 Educational materials, consisting of a guide for healthcare professionals, a patient guide, a patient card, a pharmacy poster and an annual risk acknowledgment form, were distributed together with respective DHPCs for prescribers and pharmacists. Distribution of the medicine packaging with a visual warning on teratogenicity began in May 2019. Since 2018, the educational materials have been redistributed once a year. Nevertheless, drug consumption data collected by the Institute for Medicines and Medical Devices (CInMED), as the regulatory authority, show an increase in the consumption of valproate from 0.64 DDD/1000 inhabitants/day in 2016 to 0.87 DDD/1000 inhabitants/day in 2022. Studies conducted in EU countries have shown that the PPP implementation had positive effects, but careful monitoring of its implementation is needed.18 –21 This study aimed to evaluate the impact of the introduction of the PPP on valproate prescribing in Montenegro, to identify the factors influencing the implementation of the PPP and to determine the number and characteristics of valproate-exposed pregnancies after the introduction of the PPP. Our research highlights the use of valproate in psychiatry and the indication-specific education needs. This study provides valuable insight into the conditions and impact of the PPP implementation in developing countries of Southeast Europe.
Methods
A retrospective, observational, 7-year, nationwide study was conducted.
Data source
The Primary Health Care Information System (PHCIS) of Montenegro was used as a data source, with the consent of the HIF as the owner of the data. The complete primary healthcare setting in Montenegro was covered. PHCIS holds healthcare data for more than 90% of the population in Montenegro. The data were analysed anonymously, with a unique code assigned to each patient and doctor. Data on the patient’s gender and age, prescribed and dispensed medicines (international non-proprietary name and brand-name), diagnoses, the prescribing doctor’s specialty and the location of the prescription were available. Moreover, non-structured textual data entered by general practitioners and gynaecologists during visits were also available.
Time period
The study covered the period from January 1, 2016, to December 31, 2022. For evaluating the success of PPP implementation, the period from January 1, 2016 to December 31, 2018 was considered the period before the introduction of the PPP. The period from January 1, 2020 to December 31, 2022 was considered the period after the implementation of the PPP, while 2019 was considered the period of the introduction of the PPP and the adaptation of clinical practice to the new risk minimisation measures.
Patients
The total number of female patients who were prescribed valproate at least once during the observation period was determined. Women aged 12–55 years were considered patients of childbearing potential. Female patients born between January 1, 1961, and January 1, 2011, who had received at least one valproate prescription in the period from January 1, 2016, to December 31, 2022, were selected. The electronic healthcare data for these patients were extracted in an Excel file. Patients younger than 12 or older than 55 at the time of valproate prescription were excluded from the study. Patients who were prescribed valproate, but did not have any valproate dispensed during the observation period, were also excluded.
Study approach
The data from the PHCIS were migrated to a data warehouse, validated, cleansed and extracted into an Excel file. The ATC code N03AG01 was used to select the valproate prescriptions. The International Classification of Diseases, Tenth Revision (ICD-10) was used to describe the diagnoses. Due to the range of indications for valproate prescriptions, these were categorised into four groups: diagnoses related to epilepsy; psychiatric diagnoses; off-label diagnoses; and diagnoses related to migraine and headaches. A hierarchy of importance was created so that epilepsy was treated as the most important diagnosis, followed by psychiatric diagnoses, migraine and off-label use. Patients prescribed valproate for multiple diagnoses were assigned to a higher-priority group of diagnoses. The same diagnostic categories related to pregnancy, in vitro fertilisation, abortion and childbirth were used to identify potential cases of exposure during pregnancy. Diagnoses related to contraception and prescriptions for contraceptives and intrauterine devices were used to assess contraceptive use. In addition to valproate, the prescription of other medicines approved in Montenegro for the treatment of epilepsy and bipolar disorder was analysed to assess the prescription of alternative treatments before and after the introduction of the PPP. The patients were divided into five age groups (12–20, 21–30, 31–40, 41–50 and 51–55 years). The specialties of the prescribing physicians were divided into six groups (psychiatrists, neurologists, general practitioners, paediatricians, gynaecologists and other specialties). According to the location of prescribing, the capital city was considered as one entity, and other parts of the country were considered as central, northern and southern regions.
The implementation of PPP was considered successful in patients who were switched from valproate to another therapeutic option. Moreover, the implementation of the PPP was also considered successful in patients who were prescribed valproate, followed by the prescription of contraceptives, which was recorded in the period after the introduction of the PPP.
Patients with at least one prescription for valproate in the period after the introduction of PPP and at least one prescription or diagnosis indicating pregnancy, abortion, childbirth or in vitro fertilisation in the same period were selected. Their electronic medical records were manually reviewed to identify cases of valproate-exposed pregnancies. Analysis of unstructured data entered by gynaecologists and general practitioners was performed to supplement the structured information and reliably identify cases of exposure.
We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline (Supplemental Material). 22
Statistical analysis
The demographic and clinical baseline data of the patients, as well as other relevant data, were divided into groups as described in section “Study approach.” In descriptive statistics, absolute numbers are used to describe the population, and percentages are used to describe categorical variables. Valproate-exposed pregnancy cases are described in text form.
The software PASW, version 25.0 (SPSS Inc., Chicago, IL, USA) was used for the statistical analysis. The relationship between age and disease categories was associated with the successful completion of PPP using the chi-square test or Fisher’s exact test. Binary logistic regression was used to evaluate the predictions for successful PPP implementation. The logistic regression model was constructed using the stepwise model, excluding variables with a threshold of 0.1. The results are presented as odds ratios (OR) and their 95% confidence intervals (CI). A p-value < 0.05 was considered statistically significant.
Results
Trends in prescribing
During the observation period, 3918 female patients received at least one prescription for valproate, while 2433 (62.1%) of them were born between January 01, 1961, and December 31, 2011. Patients younger than 12 or older than 55 years at the time of exposure to valproate and patients without dispensed valproate were excluded. We identified 2247 patients for further analysis. Of these, 403 (17.9%) patients were prescribed valproate in each of the 7 years observed.
The total number of female patients of childbearing potential exposed to valproate increased, with some fluctuation, approximately 12.5%—from 983 in 2016 to 1106 in 2022. However, our study has shown that the prescribing of valproate for epilepsy diagnosis decreased while the use of valproate for psychiatric diagnosis increased over the observed period. Valproate prescription for off-label diagnoses maintained a similar trend, and a slight decrease was observed in the use for migraine (Figure 1).

Number of patients prescribed valproate according to diagnosis.
Our analysis of valproate prescribing by patient age group showed that patients older than 40 years of age predominated throughout the observed period. After the introduction of PPP, a decrease was observed in the youngest age group (12–20 years), while no significant change was observed in the second (21–30 years) and third (31–40 years) groups. The number of patients in the older age groups (41–50 and 51–55) increased during the observation period, although there were some fluctuations (Figure 2).
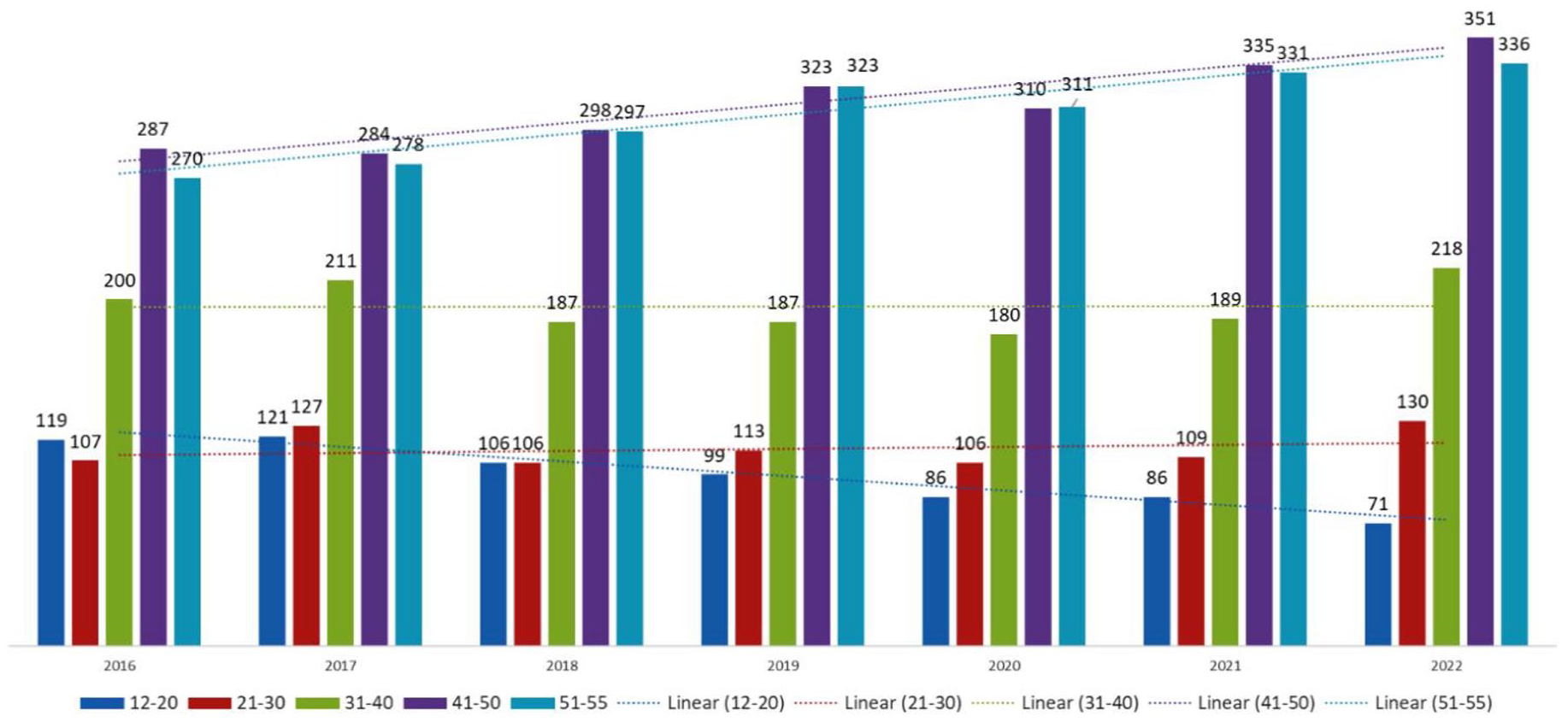
Number of patients prescribed valproate according to age group.
Use of contraception
The number of patients with a prescription for a contraceptive was very low throughout the observation period, but increased from 3 (0.1%) before the introduction of PPP to 35 (1.5%) afterwards. The number of patients with a diagnosis related to contraceptive use, for example, insertion of an intrauterine device or contraceptive counselling, was also very low throughout the period and similar before and after the introduction of PPP: 34 (1.5%) and 30 (1.3%) respectively.
Use of other medicines
In general, the number of patients prescribed other medicines authorised in Montenegro for the treatment of epilepsy and bipolar disorder increased after the introduction of PPP. Before the introduction of the PPP, 702 patients were prescribed alternative treatments for epilepsy; after the introduction of the PPP, 920 patients were prescribed alternative treatments (31% increase). Other medicines authorised for the treatment of bipolar disorder were prescribed to 386 patients before the introduction of the PPP and 679 patients after the introduction of the PPP (76% increase).
Evaluation of the success of the PPP implementation
Out of 2247 patients, 1670 were suitable for evaluating the success of PPP implementation, while 577 patients were without prescription during 2016–2018 or without follow-up during 2020–2022.
Positive effects of PPP implementation, assessed as switching to an alternative treatment or use of valproate together with the evidence of contraceptive use, were recorded in the PHCIS in 301 (18%) out of 1670 patients. The majority of participants (1369, i.e., 82%) whose data were suitable for assessment were receiving valproate without evidence of reliable contraception use, after the introduction of PPP.
Our study demonstrated that the presence of epilepsy and migraine, as well as the youngest age group of the patients, were significantly associated with the successful implementation of the PPP. On the other hand, the location of the prescriber and his/her specialty had no significant impact on the PPP implementation. The data are presented in Table 1.
Factors associated with the successful implementation of PPP for valproate.
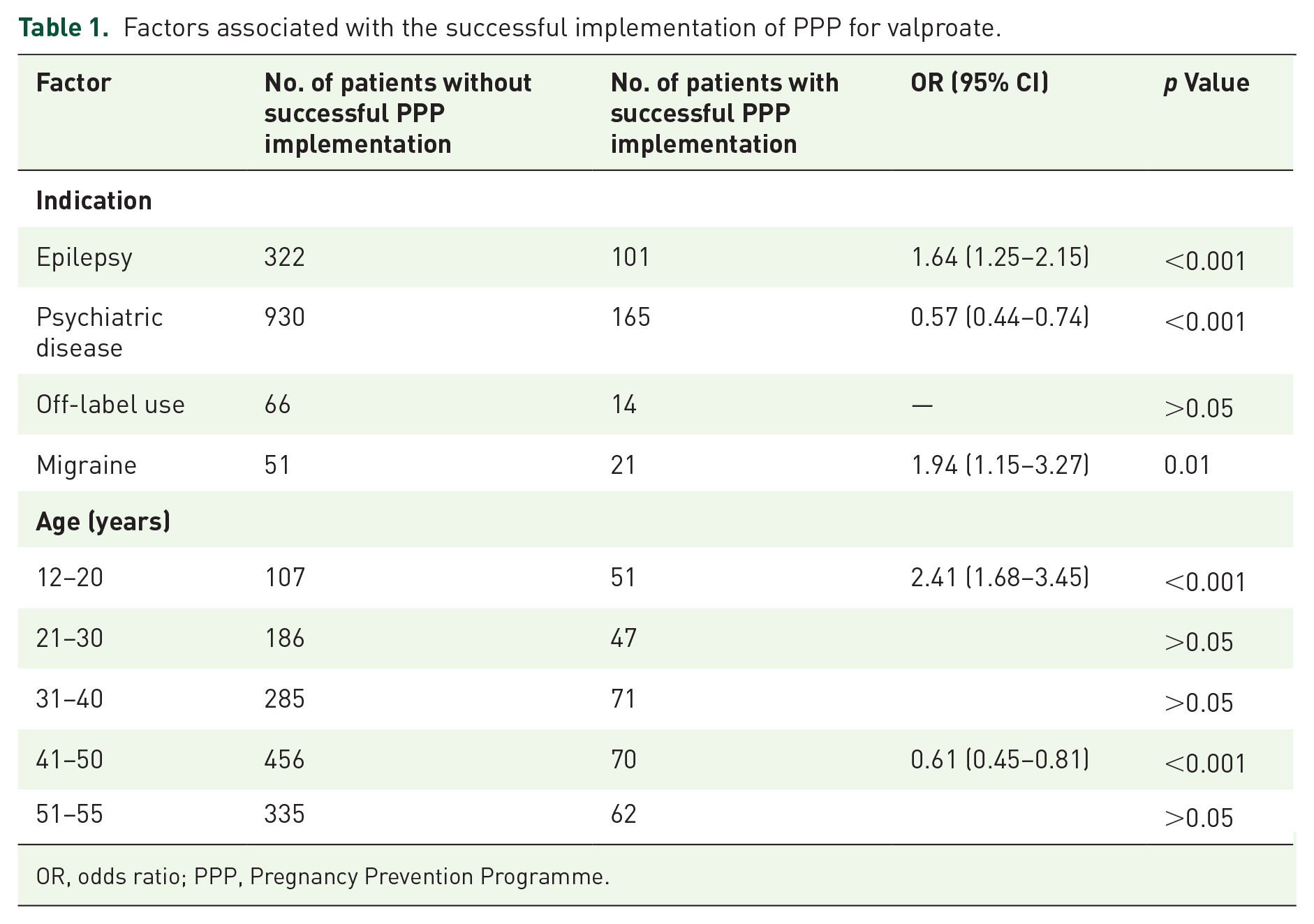
OR, odds ratio; PPP, Pregnancy Prevention Programme.
Diagnosis, age category, location and specialty of the prescriber were included in a binary logistic regression model and only age was predictive of successful valproate PPP implementation (chi-square = 35.811, df = 4 and p < 0.001). The odds ratio (OR) for the age category was 1.22 (95% CI: 1.10–1.35). The model had an overall percentage correct prediction rate of 82%.
Cases of pregnancies exposed
We identified 82 patients as potential cases of exposure during pregnancy. In total, we identified 11 cases of pregnancies exposed to valproate after the introduction of the PPP. An average pregnancy rate was 2.6/1000/year (1.91 in 2019, 3.02 in 2020, 2.85 in 2021 and 2.71 in 2022).
Two cases (18%) of exposed pregnancy were registered in 2019, and three (27%) in each of the next 3 years. Valproate was prescribed for the treatment of epilepsy in 5 (45%) cases. Four cases (36%) involved a psychiatric diagnosis, while migraine and off-label use were recorded each in one case (9%) respectively. The majority of patients (54%) were between 30 and 40 years old at the time of pregnancy. Most of the patients (82%) were exposed to valproate during the first trimester of pregnancy. The outcome of the pregnancy after the third trimester was not recorded in the PHCIS in four cases (36%), likely because delivery usually takes place in hospitals. In three cases (27%), information about the delivery was available. In one case (9%), a congenital anomaly was recorded in the newborn. Two cases (18%) ended in abortion, and for two cases (18%), there was no data on the outcome after the pregnancy was confirmed. After the pregnancy was confirmed, the use of valproate was continued only in three (27%) cases, suggesting that these were mainly unintended pregnancies. The detailed results and key observations on healthcare are shown in Table 2.
Cases of pregnancies exposed to valproate after the introduction of the PPP.

PPP, Pregnancy Prevention Programme.
Discussion
Our study provided an overview of valproate prescribing trends in Montenegro as well as a detailed insight into individual cases of exposed pregnancies following the introduction of the PPP. To our knowledge, this is the first study to examine the impact of regulatory measures on valproate prescribing in the Western Balkans. Thanks to the availability of free-text data, often including last menstrual date, expected date of delivery or current pregnancy status, our study provides accurate information on valproate-exposed pregnancies.
Our results show that the total number of women of childbearing potential treated with valproate has increased in Montenegro during the study period. Several studies using different methods and data sources concluded that the implementation of risk minimisation measures in 2014 and 2018 had positive effects on valproate use among women of childbearing potential in European countries.14,15,18 –21 A multinational cohort study on valproate use, conducted in five European countries after the implementation of risk minimisation measures in 2014, showed that the number of valproate prescriptions decreased in all female patients, especially in women of childbearing age. 15 During the same period, a decrease in the incidence rates of valproate-exposed pregnancies in Sweden and the United Kingdom (UK) indicated positive effects of the implemented measures. However, the impact of these measures was limited, and new measures, including PPP, were introduced in 2018.14,15 Following the 2018 EU risk minimisation measures, a cross-database time series study was conducted among women of childbearing age (12–55 years) using electronic medical records from five European countries/regions. 18 This study showed that valproate use had generally declined among women of childbearing potential in Europe between 2010 and 2020. A significant decline in valproate use among women of childbearing age after the PPP introduction was observed in Tuscany (Italy), Spain and the UK, while a non-significant decline was observed in the Netherlands, compared to the previous period. 18
Despite the increase in the total number, we observed a decrease in the prescription of valproate to women of childbearing potential with a diagnosis of epilepsy, although a recent study showed that the incidence of epilepsy in Montenegro is similar to other European countries, with an increasing trend in the period 2011–2022. 23 The overall increase in the number of women of childbearing potential using valproate in Montenegro was associated with valproate use for psychiatric disorder treatment. The analysis of medicines consumption in Montenegro 2018–2022 has shown that consumption of other medicines used for psychiatric disorder treatment (antidepressants, antipsychotics) has also increased. 24 This could be related to the efforts to reduce mental health stigma in Montenegro 25 and improve mental healthcare. 26 In other countries, valproate is also often prescribed for psychiatric indications. 27 Similar to Montenegro, challenges related to valproate prescription to women of childbearing potential for psychiatric indications have been observed in Estonia. 28 According to the available guidelines, there is no evidence demonstrating the advantage of valproate in terms of effectiveness and acceptability compared to other therapeutic options, while the risk of congenital malformations and neurodevelopmental disorders associated with in utero exposure is higher with valproate. For this reason, other available therapeutic options should be considered in female patients of childbearing potential. Clinical recommendations for switching/discontinuation of valproate in women diagnosed with bipolar disorder suggest that it is mandatory to stop valproate use during pregnancy. 29 General switching techniques and criteria to be considered when making clinical decisions about the use of valproate in women with bipolar disorder at childbearing age are available. 30 New Zealand’s experience shows that targeted interventions in terms of education programmes, warnings and protocols, decreased prescribing of valproate and improved management of its teratogenic risk in women of childbearing age receiving inpatient psychiatric care. 31 A retrospective cohort study of prenatal exposure to valproic acid for various indications covered 69,390 women using valproate in the United States of America (US) between 2005 and 2020. This study found that patients with migraine or headache and mood disorders accounted for the largest proportion of valproate users and had the highest rates of pregnancy during use, while patients with epilepsy had the lowest. 32 A study investigating adherence to PPP in the Grampian region of Scotland found that women treated for psychiatric reasons were less likely to adhere to PPP than other indications. 33 The increasing proportion of patients with psychiatric diagnoses among valproate users in Montenegro requires special attention, taking into account the particular challenges of implementing PPP in this population.33,34
We observed a decrease in valproate prescription among the youngest age group (12–20 years) during the entire study period. According to our findings, the young age of patients was a key predictive factor for the successful implementation of PPP in Montenegro. On the other hand, our study showed that the specialty of the prescriber and the location of prescribing were not decisive factors for the success of PPP introduction. In comparison, a study conducted in Ireland investigating valproate prescriptions between 2008 and 2013 showed an increase in valproate use in indications other than epilepsy, but an address distant from academic specialty centres predicted higher exposure to valproate. 35 In the same country, a drug utilisation study assessing prescribing trends between 2014 and 2019 showed that the decline of valproate use was greatest among those aged 16–24 years following the first intervention in 2014, and among those aged 25–34 years following the second intervention in 2018. 21 A 7-year nationwide study in Latvia also showed a statistically significant decrease in valproate prescribing to young women and girls (<15 years) after the introduction of risk minimisation measures in 2014 and 2018, and it was more pronounced compared to the 15–49 age group. 20 Similar to the EU countries, patients aged 41–55 years accounted for the largest proportion of valproate prescriptions both before and after the introduction of the PPP in Montenegro. 18
Throughout the observation period, contraceptive use was recorded to a small extent. The PPP requires the use of effective contraception during the valproate treatment. This should be applied even to patients who are not currently sexually active, unless the specialist considers that there are compelling reasons to indicate that there is no risk of pregnancy. In Montenegro, recording of contraceptive use was found to be more frequent in the period after the introduction of PPP (from 0.1% to 1.5% of patients), but it is still significantly lower than in other countries. This information should be taken with caution because in Montenegro, contraception is not reimbursed and is mostly purchased privately, outside the primary healthcare system. 36 For that reason, information about contraception is rarely captured in the PHCIS, and details about the type of contraception are not available. However, when introducing a PPP as a risk minimisation measure, we should take into account the generally low rates of contraceptive use in Montenegro. According to the European Contraception Policy Atlas 2022, Montenegro’s score was very low (45.7%), which indicates extremely poor performance. 36 Similar was observed in other Western Balkan countries.36,37 Although effective contraception is not required or suitable for some patients, the fact that contraception is not reimbursed within the national healthcare system, together with insufficient information on effective contraception, could have a negative effect on the PPP implementation.
Contraceptive use while taking valproate was low in EU countries (<25%), with only the Netherlands showing a significant increase after the introduction of PPP. 18 Contraceptive use among women aged 12–44 years using valproate in the US between 2005 and 2020 was also unsatisfactory: only 22.3% of treatment episodes had a 1-day overlap of valproic acid and contraceptive use. 32 On the other hand, a cross-sectional study which included data from all 141 general practices in three clinical commissioning groups (CCGs) in East London (UK) found that contraception and contraceptive advice increased from 24% to 43% following the 2018 risk minimisation measures, which was in association with a 71% decrease in pregnancy rate in women of childbearing age prescribed valproate. 38
The use of alternative medicines authorised in Montenegro for both diagnoses, epilepsy and bipolar disorder, increased following the introduction of PPP. Nevertheless, our results indicate that the successful implementation of PPP, defined as a switch to another medication or as evidence of contraceptive use, was recorded in only 18% of eligible patients. This is in line with the results from the EU countries, where an increasing trend in the rates of alternative treatments use for epilepsy and bipolar disorder was observed. The monthly rate of switch from a valproate to an alternative treatment was similar across all five observed countries/regions (<5%), with no significant increase in switching rates after the 2018 intervention versus the time before. 18
Similar to other countries, cases of valproate-exposed pregnancies were recorded in Montenegro after the introduction of PPP. The average pregnancy rate for the period after the introduction of PPP was 2.6/1000/year, and in six (54%) cases, valproate was used in indications other than epilepsy. In comparison, the results of a cross-sectional study conducted in East London (UK) showed that the pregnancy rate decreased from 9.9/1000 valproate recipients before the 2018 warning to an average of 2.8/1000 afterwards, while in 89% of detected cases, valproate was used in the indication of epilepsy. 38 In Scotland, the number of conceptions on valproate fell from 15.3 per 1000 women treated with valproate in 2011 to 8.2 per 1000 women treated with valproate in 2019, showing a decrease of 46.4%. 39 In the cohort study of 165,772 valproic acid treatment episodes in 69,390 women of childbearing potential in the US, pregnancy rates during treatment remained unchanged over the 15-year study period (2005–2020) and were more than twice as high in users with mood disorders or migraine compared to those treated for epilepsy. 32
Similar to other countries, the majority of pregnancies were exposed to valproate in the first trimester, and patients were mostly between 30 and 40 years old. A cohort study on valproate use conducted in Catalonia (Spain) also showed a decrease in the prevalence and cumulative incidence of valproate prescriptions as pregnancy progressed, with the highest proportion of exposure occurring in the 3-month period before pregnancy and in the first trimester. 40
There are some strengths and limitations to the study. This nationwide study includes the vast majority of female patients in Montenegro who were prescribed valproate in the outpatient setting during the observation period. The availability of unstructured data allows for a more accurate selection of potentially vulnerable pregnancies and provides insight into the details of drug use and the PPP implementation.
Limitations
The limitations of this study are the following. Accurate data on contraceptive use were not available from PHCIS because contraception in Montenegro is usually privately accessed, outside the public primary healthcare system. As this is routinely collected data, the quality of data in the PHCIS is limited, meaning that specialists’ records were not available, as well as data on valproate dose and dose adjustment during pregnancy. No data were available on the long-term outcome for most cases of pregnancies exposed. Although data on prescribed and dispensed drugs were available, it was not possible to determine whether patients adhered to the prescribed treatment.
Conclusion
Regulatory risk minimisation measures, including the implementation of the PPP, had a limited impact on reducing the risk of teratogenic effects of valproate in Montenegro. The success of the PPP implementation was influenced by patient age, with greater success in younger women. The risk minimisation measures were more effective for epilepsy diagnosis, while the location of valproate prescription had no significant influence. While prescribing for the indication of epilepsy decreased, we are concerned about the increasing prescribing in psychiatry, especially given the challenges in implementing PPP in this population.
Our analysis of pregnancies exposed to valproate indicates that healthcare professionals are aware of its teratogenic potential, but there is still a reactive approach when it comes to risk minimisation. Targeted education of healthcare professionals and patients is required, especially concerning the use of effective contraception in patients using valproate. Greater coordination and collaboration among valproate prescribers, gynaecologists, medical genetics specialists, general practitioners and pharmacists is essential to enhance the effectiveness of risk minimisation measures and improve PPP implementation.
Supplemental Material
sj-doc-1-taw-10.1177_20420986251360888 – Supplemental material for How effective is the implementation of the valproate pregnancy prevention programme in Montenegro? – A 7-year national retrospective study
Supplemental material, sj-doc-1-taw-10.1177_20420986251360888 for How effective is the implementation of the valproate pregnancy prevention programme in Montenegro? – A 7-year national retrospective study by Veselinka Vukićević, Snežana Mugoša, Gordana Stanojević and Sandra Vezmar Kovačević in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
