Abstract

Introduction
Pharmacovigilance (PV) is a discipline that encompasses all activities relating to the detection, assessment, understanding, and prevention of adverse effects and other risks associated with the pharmaceutical products use because the scope of PV covers other risks, other than adverse effects, such as quality defects, misuse, drug interactions, medication errors, counterfeiting, etc. 1 Pharmacoepidemiology involves the study of the uses and effects (beneficial and/or harmful) of drugs in large well-defined populations.2,3
These disciplines pursue a primary mission of ensuring the safety of individuals worldwide who use pharmaceuticals, including biologics, such as vaccines, plasma-derived therapies, and/or medical devices, which are in clinical development or in the postmarketing setting. Pharmacovigilance is constantly adapting to a rapidly evolving regulatory landscape and dizzying pace of advancement of new therapies and technologies. The pace of change, speed of drug development, and increase in numbers of medicinal products in development and on the market stress the traditional approach to the development of pharmacovigilance professionals, which is through ‘on-the-job’ apprenticeship at large pharmaceutical companies.
Developing structured educational programs and teaching pharmacovigilance to undergraduate- and graduate-level medical and pharmacy students, as well as offering postgraduate education on pharmacovigilance and pharmacoepidemiology to healthcare professionals, has been emphasized by several authors, as key leaders in this fast evolving field, and professional societies such as International Society of Pharmacovigilance (ISoP).4–17 It is evident that novel, flexible, scalable approaches are needed to address regional needs and also to expand efficiently pharmacovigilance education efforts internationally. The Am2P program, which provides remote training opportunities, has capabilities and expertise to offer scalable solutions to address these needs, and at least in part contribute to cost-effective model for further regional extensions in order to support and expand education in the field of pharmacovigilance internationally.
Pharmacovigilance is a highly regulated discipline, which has to adapt to the rapid advancement of new therapies. In the many decades since the establishment of the first formal public health system to demonstrate the safety of drugs before market approval, the clinical development of new therapies has specifically required close monitoring of emerging adverse events, and PV regulations have been constantly evolving. 18 In the United States, medication consumption and rates of adverse events continue to increase along with the prevalence of chronic diseases such as cancer, diabetes, cardiovascular and respiratory disorders, and obesity in an aging population, and the availability of novel pharmaceuticals for previously unmet medical needs.
During the coronavirus (SARS-CoV-2, or COVID-19) pandemic, pharmacovigilance took a central role with the development of new vaccines and new treatments for COVID-19, which require close monitoring of adverse events to promptly identify, assess and manage emerging safety signals.18,19 Pharmacovigilance of these vaccines and treatments came suddenly into the spotlight when several received emergency authorization and were rolled out, resulting in rapid exposure in millions of individuals in late 2020. 20 The increased importance of pharmacovigilance has been identified by a group of economists who studied the US market since 2016 and projected the pharmacovigilance market until 2030. 21 As shown in Figure 1, significant growth of the PV market by life cycle is expected from 2020 to 2030 and is predicted to be driven by an increase in phase IV postmarketing surveillance. Pharmacovigilance is important in all phases of a product life cycle and no more so than after the market launch of a product.
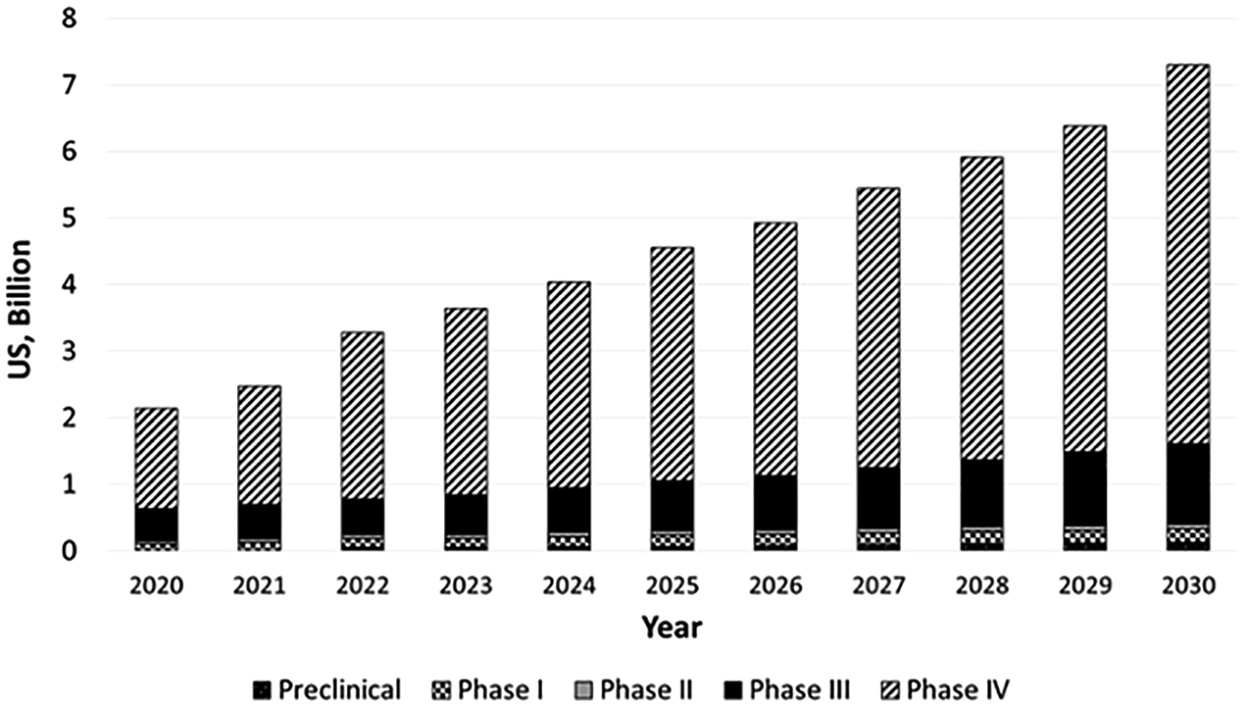
Pharmacovigilance market in the United States by life cycle for 2020–2030 period of time.
Healthcare professionals with training and experience in clinical medicine are key contributors to PV functions in regulatory agencies, academia, and the pharmaceutical industry. 1 Their medical and scientific expertise is essential for adverse events to be accurately characterized and assessed within the context of a product’s benefits and risks, and their knowledge of patient care and healthcare delivery is invaluable for developing effective risk mitigation strategies. 1 Therefore, it is essential that the number of trained PV professionals and the knowledge of each professional practicing PV keep pace with the growth of pharmacovigilance in a rapidly evolving regulatory landscape. The Am2P program will contribute to fulfilling that need.
The purpose of this publication is to review a specific online training resource available to pharmacovigilance trainees/professionals and to encourage colleagues to continue to advance the discipline of pharmacovigilance through distant/online learning and improve their knowledge in this field.
Virtual educational and globalization in pharmacovigilance
The COVID-19 pandemic has triggered a dramatic shift in how knowledge can be accessed in a distance learning environment. It became necessary to develop new ways of interacting with each other due to social distancing, and other restrictions imposed during several waves of COVID-19 associated with new evolving variants. 22 Distance or online learning has become the new norm during this global outbreak of COVID-19.22,23 Meanwhile, several national and international academic and professional societies have modified and/or combined resources to assure that continuing medical education occurs during this difficult time.24,25
Although distance or online learning is not a new concept, for many clinical researchers, physician investigators, and biomedical and pharmacovigilance professionals, it was a shift away from the traditional classroom teaching model for trainees.1,24–26 Among others, Cook and Dupras examined the most effective way to create an online learning platform to be used in medical sciences. 27 The authors highlighted the importance of a user-friendly and intuitive website/online learning platform design that is well maintained and pointed out the need for the integration of self-assessment features into learning modules to ensure that learners are more effectively engaging in the material provided. 27 Recently, Gewin has shared useful tips in the transition to online learning. 28 This author emphasized the importance of creating more focused lessons for online learning that highlight only a few main points. In addition, Gewin noted the utilization of ‘live-video’ conferencing for the follow-up to the previously provided self-learning modules. 28 However, Gewin stated that live conferencing should not be used for all educational activities due to poor connections, limited internet access, and overuse. Authors generally agreed that the most effective online learning platforms should provide interactive capabilities, which can enable learners to interact with the material.20–28 Using this method, learners can pursue the information at their own pace and engage in the course/module through feedback, commentaries in the ‘live chat’, and/or subsequent ‘live video’ or audio conference for follow-up.27–29 These aspects are very important to keep in mind as we transition to online instructions and/or develop/offer new training programs.
There is a unique opportunity to incorporate these novel training approaches into the education of pharmacovigilance professionals so they can develop new skills and maintain professional certifications by pursuing continuing education and/or improving their knowledge in drug safety and pharmacovigilance in an increasingly globalized environment with complex and not always consistent regulations in different parts of the world. Pharmacovigilance remains a dynamic clinical and scientific discipline. In the next decade, pharmacovigilance will continue to face many challenges in developing more efficient interactions with healthcare systems and more effective global outreach. 29 Among the challenges, the World Health Organization (WHO) notes the following: globalization; web-based sales and information; broader safety concerns; public health versus pharmaceutical industry economic growth; monitoring of established products; developing and emerging countries; attitudes and perceptions to benefit and harm, outcomes and impact. 30
Am2P program design, structure, and options
North American PV system is regulated by the FDA and subject to laws, regulations, and other relevant guidelines (e.g. ICH Good Clinical Practice (GCP), Good Pharmacovigilance Practices (GVP) as applicable to biomedical products (e.g. dug, device, biologic, or combinations of these products and vaccines) in development phases and postmarketing.31–36
In November 2021, the North American Society of Pharmacovigilance (NASoP), a chapter of the ISoP, under the leadership of Veronique F. Kugener, Abimbola Cole, Dona Ely, Sepideh Habibi, Pinak Joshi, Marina A. Malikova, John Price, Maribel Salas, and Peggy Webster discussed the gaps in pharmacovigilance and ways to fulfill those gaps. As a result of those discussions, a series of pharmacovigilance courses were developed targeting North American healthcare professionals and stakeholders in collaboration with the Eu2P academically accredited program.
Although internationally recognized, the Eu2P program mainly targets European stakeholders, involved in biomedical product development, who predominantly work in Europe. According to Eu2P internal sources, 54% of learners are based in Europe, while 13% of learners are based in North America. Indeed, stakeholders from overseas are still hesitant to register to the Eu2P program even though its academic accreditation is recognized; they doubt that their training would be accepted by their current or future employers outside of Europe. Therefore, there is a need to build pharmacovigilance programs targeting the geographical and cultural environment that would be more focused on FDA regulatory requirements and processes, which are available within the Am2P training program in order to answer to the expectations of the North American continent demand. Besides, European stakeholders who wish to be employed or enter in collaboration with North American public and private sectors should obtain a specialized training focused on FDA and North American pharmacovigilance expectations, and delivered by North American experts to be reassured they have a solid and strong knowledge recognized by North American stakeholders.
The Am2P program is targeted to all people involved in the multiple processes of pre- and postauthorization evaluation of medicines (e.g. professionals and students). Among the professionals as potential learners are all those directly involved in the medicines evaluation and R&D processes such as industry, regulatory and academic pharmacovigilance experts, pharmacoepidemiology, biostatisticians, and healthcare providers, which can be recruited through the private and regulatory Am2P partners. Among recent or current students, who can be recruited through the Am2P academic partners, are graduate and postgraduate students who specialize in human medicine and pharmaceutical sciences and in other biological science fields.
Thus, the Am2P program was designed by the NASoP chapter to fit the North American pharmacovigilance educational needs and is an extension of the existing Eu2P accredited program, a European education and training program in pharmacovigilance and pharmacoepidemiology. The program follows the WHO-ISoP core elements of a comprehensive modular curriculum and subscribes to the Innovative Medicines Initiative (IMI) education and training quality standards, jointly developed by Eu2P and other IMI education and training projects to foster quality in lifelong learning and continuing professional development.
The Am2P program approval is ensured by the Eu2P Executive Board, including academic representatives of the six Eu2P degree-awarding universities, and provides an access to knowledge and understanding of the concepts and principles in pharmacovigilance with a focus on North America. It enables outreach to online education in pharmacovigilance according to the mission of the ISoP, which aims to foster pharmacovigilance both scientifically and educationally and enhance all aspects of the safe and proper use of medicines, in all countries.
The President of ISoP, Angela Caro Rojas, reinforced the significance of the Am2P program by providing the following statement: ‘To ISoP, it is not only a priority but also a main goal to share information and provide training to the world’s pharmacovigilance community, so that they can also impact patients in real life, contributing to improving the safety in the use of medicines and favoring patient safety, all efforts made to this end will be supported by the governing bodies of ISoP to obtain successful results.’
As mentioned above, the Am2P program was developed as an extension of the Eu2P accredited program to respond to the interest of educational and accredited educational programs in pharmacovigilance with a focus on North America. Below is a display of the total number of individual visits to the Eu2P website reported from the North American sub-continent between 5 December 2021 and 6 December 2022 (Figure 2). It is noted that 2969 individuals have visited the European website Eu2P searching for accredited training in pharmacovigilance. This figure conveys a significant interest from North American professionals to be trained in pharmacovigilance.

Numbering of individual visits to the European Eu2P website coming from North American sub-continent between 5th December of 2021 and 6th December of 2022.
In addition, among the 20,819 individual users who have visited the Eu2P website between 5th December 2021 and 6th December 2022, there were 14,702 users who declared they speak English. Among them, there were 10,262 individuals, who identified as US-based English speakers (Figure 3). These data indicate that there is an additional potential target audience for training in pharmacovigilance in the North American geographic region. Furthermore, an unknown proportion of the 20,819 visitors to the Eu2P website were interested in training in North American pharmacovigilance.

Numbering of individual visitors in the European Eu2P website speaking North American English language between 5th December 2021 and 6th December of 2022.
The Am2P program offers two academic options, the certificate degree and the short course certificate of achievement in pharmacovigilance, which are reinforced (recognized) by the ISoP and organized into four modular course programs, which have been developed in collaboration with Eu2P instructors and benefit from existing course materials, education technology, and academic accreditation background.
These four modular course programs are available for registration on a website platform at the Am2P website (https://www.am2p-courses.com) and provide PV educational training on:
Basic pharmacovigilance (PV) & PV regulations
PV for biologics
External databases/real word data (RWD)/real world evidence (RWE)
Benefit-risk assessment
The program monitoring and periodic reviews are conducted by the partnership between a dedicated sub-group of the NASoP chapter and the University of Bordeaux under the supervision of the Eu2P Executive Board. The program provides the following accredited options:
Currently, information about the program is made available with its promotion through multiple channels (e.g. booth presence and posters at the conferences worldwide including ISoP and the International Society of Pharmacoepidemiology (ISPE) and with leaflets specifically designed on course topics for the Am2P certificates and Am2P short courses, respectively.
This program has unique features and distinct multiple benefits as compared to other offerings/programs in the field of pharmacovigilance such as:
convenience with online studies;
certification covers fundamental concepts/topics but also offers specialized courses; focused on particular area of interest (i.e. pharmacovigilance for Biologics, RWE, etc.);
access to updates in the pharmacovigilance field from experts in relevant areas;
global access and connections;
experts as instructors, networking with PV professionals;
opportunity for career development in the United States;
tax-free options may apply on purchased courses/certifications (only flat fee) for European and international learners. Since University of Bordeaux is collecting the fees, in accordance with article 261-4-4° of the general tax code (CGI), the provision of services and the delivery of goods which are closely related to them, carried out within the framework of university education provided in public establishments are exempt from value added tax.
Currently, 3 certificate degrees and 36 short course certificates have been delivered by Am2P program [e.g. Basic Pharmacovigilance and Pharmacovigilance Regulations; Pharmacovigilance for Biologics; US-EU Regulations: principles and comparison; Principles of Labeling and Description of United States Prescribing Information (USPI); Aggregate reporting in the US; Gene therapy; FDA Adverse Event Reporting System (FAERS); FDA Sentinel System; Concepts in Risk Management; Health care records from large databases as a tool to study the use of medicines; Risk Evaluation and Mitigation Strategy (REMS); Organization for risk management in the industry.]
A formal memorandum of understanding has been signed between parties involved in development and execution of the Am2P program in July of 2022. The course provider partners aim to further develop and implement new research and training activities in medicines utilization, safety and effectiveness as well as to maintain on-going promotion and circulate essential information in a timely manner in ISoP and NASoP annual meetings and at the international congresses [e.g. ISoP, International Society of Pharmacoepidemiology (ISPE), and World Drug Safety Congress].
Limitations and future perspectives
First of all, lack of motivation among learners is one of the main reasons why they fail to complete their online courses. However, in the case of the Am2P program, the learners who register are professionals who need to acquire a skill and a diploma quickly to achieve objectives in their work requested by their management.
Second, in ‘face-to-face’ courses, learners can receive immediate feedback on their questions and address any misunderstandings. This personalized feedback is positive reinforcement for learners because it improves their learning process in terms of quality and speed, while increasing the level of student motivation. The limitation of online training, when sessions are not conducted ‘live’ but prerecorded can be addressed by asking questions in the course forums or conversing with the professors by e-mail or videoconference, even outside of the virtual classroom.
Finally, online training courses focus more on theory rather than practice because it is easier to implement. The recorded courses of the Am2P program are carried out by professionals from the healthcare field, who offer practical solutions to cases and problematic situations, so learners would be to apply theoretical lessons learned and solve them. Also, recorded lectures are followed by quizzes, allowing learners to find a practical solution to problems encountered in real life in their professional environment.
Conclusion
The Am2P program offers two academic options, the certificate degree and the short course certificate of achievement in pharmacovigilance, which are reinforced (recognized) by ISoP. Am2P is a partnership between the NASoP Chapter and Eu2P to build a North America oriented online training course offerings in pharmacovigilance. Am2P certificate courses hold the same academic value as Eu2P Certificates. A regular certificate course involves a total of 75 h of online instructional materials, which are spread over a 3-month period.
These are recognized as three ECTS credits, which are equivalent to 1.5 American credits. Am2P short courses are bite-sized courses designed by Am2P instructors for professionals who want to have up-to-date, efficient, and solid knowledge and strengthen their PV expertise with a focus on the North American continent.
