Abstract
Background:
Prescribing psychotropic drugs often leads to long-term use in people with intellectual disabilities. In this population, polypharmacy is also common, likely because of the high frequencies of comorbid somatic, behavioural and mental disorders, while the vulnerability for medication side effects is high. Therefore, there is a risk of inappropriate prescribing of psychotropic drugs in people with intellectual disabilities.
Objectives:
This study aimed to develop a tool to assess the appropriateness of psychotropic drug prescriptions in people with intellectual disabilities and to test its psychometric properties.
Design:
In this study, we used a mixed-methods design. In a qualitative phase, we developed the tool, followed by a quantitative phase, to evaluate its psychometric properties.
Methods:
We used a modified Delphi procedure consisting of a preparation phase and three Delphi iterations to develop the tool and to determine which items encompass the concept of appropriate psychotropic drug prescribing. A weighting round was conducted to determine a summated index score. Finally, the test–retest reliability, the interrater reliability and the convergent validity of the tool were investigated.
Results:
The Delphi panel, with 37 field expert participants, agreed on the content of the tool, including seven domains (indication, dosage, duration, duplication, interactions, evaluation of effect and evaluation of side effects) that should be assessed regarding appropriate psychotropic drug prescribing. The test–retest reliability and the interrater reliability turned out to be moderate to perfect for five of the seven domains, except the domains ‘evaluation of effect’ and ‘evaluation of side effects’, although no interrater agreement was found in the domain ‘duration’. The convergent validity was slight.
Conclusion:
In conclusion, this study resulted in the development of a reliable tool to support prescribers in clinical practice in the appropriate prescribing of psychotropic drugs in people with intellectual disabilities.
Plain language summary
Keywords
Introduction
Psychotropic drugs (PDs), in particular antipsychotics and antidepressants, are frequently prescribed to people with intellectual disabilities (ID). 1 Just like in the general population, PDs are prescribed for the treatment of mental disorders. However, in people with ID, they are also often prescribed outside a licenced indication (off-label) for the treatment of challenging behaviours,2,3 like aggressive and injurious behaviours, irritability, hyperactivity, mood swings and temper outbursts. Mainly, antipsychotics are prescribed for challenging behaviour and often for long-term use, although this long-term use of PD may be controversial because the effect in this indication has not been proven.4,5 Also, people with ID are more likely to suffer from psychiatric and somatic comorbidities,6,7 which may lead to the need for pharmacological treatments. Consequently, polypharmacy is often present in people with ID,8–10 whereby PDs, anti-epileptics and analgesia are the most used drug classes.9,11 Polypharmacy is associated with increased risks for medication side effects and medication interactions.12,13 People with ID are vulnerable to side effects of PDs, especially movement side effects 14 and metabolic side effects,15,16 which risks will likely increase by polypharmacy. These side effects, and their potential to harm the bodily and mental functioning, may negatively affect the quality of life of people with ID.17,18
The high frequency of prescribing outside the licenced indications, the long-term use, and polypharmacy may indicate inappropriate PD prescribing in people with ID. Therefore, several countries have introduced programmes to address the issue of inappropriate PD prescribing,19–22 for example, the STOMP and STAMP projects of the Royal College of Psychiatrists in the United Kingdom. 19 These programmes highlight the importance of careful prescribing of PD in people with ID, considering the lack of evidence regarding the effectiveness in case of unlicenced indications, the increased risk of occurrence of side effects and the increased prevalence of comorbidities and polypharmacy. Therefore, medication reviews are a useful tool to assess the appropriateness of medication use and to identify drug-related problems. However, these reviews and tools retrospectively assess the medication use rather than assessing appropriateness from the start of prescribing. Several tools have been developed to assess the appropriateness of prescribing for use in medication reviews, such as the Systematic Tool to Reduce Inappropriate Prescribing (STRIP) 23 and the Medication Appropriateness Index (MAI). 24 The STRIP has been tested in a pilot study (N = 27) on medication use of adults with ID and turned out to be feasible. 25 In patients with dementia, the MAI was used to develop the Appropriate Psychotropic drugs use In Dementia (APID) index, an index for clinical studies to evaluate appropriateness of PD prescription for neuropsychiatric symptoms in dementia. 26 From the APID, the self-assessment tool for prescribers, the Psychotropic Drug Monitor, was derived, with an accompanying smartphone application. 27 To our knowledge, there are two tools especially developed for adults with an ID: the Tool for Optimising Prescription in Intellectual Disability (TOP-ID) 28 and the tool Optimising Pharmaco-Therapy and Improving Medication for Ageing with Intellectual Disability (OPTIMA-ID). 29 These two tools guide prescribers in optimising their drug prescribing in (older) people with ID, starting from the anatomical system or clinical situations.28,29 Although the tools include the nervous system 29 and challenging behaviour, 28 they are not explicitly focused on PD prescribing.
Even though there are similarities in the included drug classes or among the different patient groups, none of the tools mentioned above was developed specifically to assess the appropriateness of prescribing PDs in people with ID. The development of a tool based on scientific and clinical knowledge and on the consensus of experts in the field may contribute to an appropriate prescribing of PDs in people with ID. The tool may, therefore, help to prevent prescribing outside the licenced indication, long-term use, polypharmacy and the unnecessary occurrence of side effects. Hence, this study aimed to develop an assessment tool for measuring appropriate PD prescribing in people with ID and to test its psychometric properties.
Methods
The study was approved by the Medical Ethics Review Board of the University Medical Center Groningen (2017.183). All other Dutch data protection rules and research codes of conduct were adhered to as applicable. To test the tool’s psychometric properties, written informed consent was obtained from all participants or their legal representatives to collect data on PD use in their medical and pharmaceutical records.
Study design and procedures
The study took place between January 2018 and October 2019 and consisted of two parts, that is, the development of the tool (part A) and the testing of the psychometric properties of the tool (part B). The project was led by the research team, composed of an elderly care physician involved in the development of the APID, an ID physician with clinical experience in prescribing PD in people with ID and a psychiatric nurse who was also a PhD student. In the Netherlands, an ID physician is a medical specialisation, a 3-year postgraduate training programme to meet the special health needs of people with ID. A consultant pharmacist was part of the team to advise on detailed information regarding PDs. We used a Delphi method to develop the assessment tool. The Delphi method, originally developed by Dalkey and Helmer of the Rand Corporation (1963), is a widely used and accepted method for consensus building. 30 In an iterative process, Delphi panellists (experts on a specific topic) are asked to respond independently to statements or answer questions. Traditionally, the first Delphi iteration begins with an open-ended questionnaire as a basis for the content area and is followed by structured questionnaires. However, a modified Delphi procedure starting with a structured questionnaire in the first Delphi iteration, as we did in this study, is also commonly used. 30 In each subsequent iteration, the panellists receive a questionnaire summarising the previous iteration and are asked to revise their judgement. Usually, consensus is reached in three or four Delphi iterations. After the development of the final draft of the tool, we investigated its psychometric properties in a validation study.
Part A of the study: Development of the tool using a Delphi procedure
To develop a first draft of the assessment tool (step 1), we used the Psychotropic Drug Monitor 27 as a base. This self-evaluation instrument has resulted from the research index, Appropriate Psychotropic drugs use In Dementia (APID). 26 Data and criteria from the NICE guidelines 31 and the monodisciplinary Dutch guideline of the Dutch association of ID physicians ‘Prescribing psychotropic drugs in people with Intellectual Disabilities’ (which is in 2019 incorporated in the multidisciplinary guideline ‘Challenging behaviour in adults with Intellectual Disabilities’) 32 were used to assess which aspects of appropriate prescribing (the domains of the tool) are considered relevant to include in the draft and which domains should be adapted. In defining the elaborated content of the domains for each PD which would be included in the tool, information from the ‘Farmacotherapeutisch Kompas’, a Dutch drug database with independent pharmaceutical information for medical professionals, 33 and the Dutch Children’s formulary, a drug database for paediatric dosing, 34 was used in addition to these guidelines. The first draft of the Tool for Appropriate Psychotropic Drug Prescribing in people with Intellectual Disabilities (TAPP-ID) contained an introduction and instruction section in which the way of using the TAPP-ID is explained. At first, a user should determine the route that should be followed to assess the quality of the prescribing of the PD in question. In route A, either there is a diagnosed mental disorder or it is unclear whether the behavioural symptoms can be attributed to a mental disorder. Route B is chosen if the behavioural symptoms do not result from a mental disorder but may warrant treatment. After choosing a route, a prescriber (or other assessor) scored per PD prescription each domain with a 2, 1 or 0 (2 for appropriate prescribing, 1 for less appropriate prescribing and 0 for inappropriate prescribing).
To determine the final tool, the following steps have been taken based on this first draft (step 1): step (2) a semi-structured questionnaire for a first expert assessment in the preparation phase, step (3) a Delphi procedure to determine the content of the tool, step (4) a weighting round to determine a summated index score.
Recruitment of field experts for the development of the tool took place among members of the Dutch association of ID physicians, members of a specialised platform on ID psychiatric care and among the research teams’ network of pharmacists with knowledge or experience in the field of ID care. Potential participants could choose to participate in the preparation phase and the Delphi procedure (panel 1) or the weighting round (panel 2). Participants received a small fee for participating in this study.
The first draft (step 1) was submitted to those participants who had consented to participate in steps 2 and 3 (panel 1) with a request to comment on it, using a semi-structured questionnaire. After that, the research team reviewed and discussed the comments on the first draft and developed a second draft of the assessment tool. Based on this second draft, a Delphi procedure took place (step 3). For this purpose, the research team defined statements, which could be scored on a 9-point Likert scale, with a range from strongly disagree (1) to strongly agree (9) and space for comments by the panellists of panel 1. After each iteration, the research team discussed the statements on which no agreement was achieved and the comments of the panellists. After that, they summarised their findings and defined new statements for the next Delphi iteration. After the last iteration, agreement on the assessment tool was achieved.
After this Delphi procedure, the panellists of panel 2 weighted the identified domains of the assessment tool for relevance (step 4) to create a summated index score, enhancing its use in clinical research. This resulted in the final draft of the tool: a TAPP-ID, incorporating the seven domains ‘indication’, ‘dosage’, ‘duration of use’, ‘duplications’, ‘interactions’, ‘evaluation of effect’ and ‘evaluation of side effects’. The domains ‘indication’, ‘dosage’, ‘duration of use’ and ‘interactions’ present information specific to each agent. The domains ‘duplications’, ‘evaluation of effect’ and ‘evaluation of side effects’ are described in general terms and thus do not differ from each agent. Figure 1 shows the complete procedure.

The four steps of the development of the tool.
The Online survey software Unipark of Cleverbridge (Germany) was used to conduct the semi-structured questionnaire, the Delphi iterations and the weighting round. Participants received an email with a link to the online survey and a request to respond within 2 weeks. After 1 week, a reminder was sent by email.
Part B of the study: Psychometric properties of the final draft of the TAPP-ID
In part B, the validation study of the final draft, that is, the testing of the psychometric properties, took place. We used data from medical and pharmaceutical records of patients with ID to investigate the convergent validity and the test–retest and interrater reliability of the TAPP-ID. The recruitment of participants in this part of the study took place in an ID care organisation in the northern part of the Netherlands, providing 24 h a day care for approximately 400 residents. This ID care provider sent an information letter and informed consent form to all their residents who used at least one PD or to the legal representatives of those, to ask permission to collect data on PD use in their residents’ medical and pharmaceutical records. The prescribers of this ID care organisation also received an information letter and were asked to participate in this study. By participating in this study, they received a small fee to compensate their time. The participating prescribers received oral and written instructions on how to use the TAPP-ID as an assessor. In the absence of a gold standard instrument, we defined the judgement of an expert panel consisting of an experienced ID physician and an experienced pharmacist in the field of ID care as the gold standard.
We defined two time points for assessing the psychometric properties of the TAPP-ID. To investigate the interrater reliability, at timepoint one, the TAPP-ID was scored for each PD prescription of each participating client by two independent assessors. Simultaneously, the expert panel scored the appropriateness of the prescribed PD based on patients’ records. To investigate the test–retest reliability, the TAPP-ID was scored once more by the same assessors at timepoint two, 2 weeks after timepoint one.
Statistical analyses
Delphi procedure
In the three Delphi iterations, the level of agreement was calculated for all statements according to the RAND/UCLA appropriateness method. 35 Therefore, we calculated the median to measure central tendency, and the inter-percentile range adjusted for symmetry (IPRAS) 35 as a measure for the level of dispersion. Statements with a median between 7 and 9, and on which there was agreement (if the IPRAS was greater than the inter-percentile range), were included in the TAPP-ID.
To calculate the total index score, we used the mean weights of the domains multiplied by the minimum and maximum scores of a domain. If a domain consisted of two subdomains, a mean composite score of the two subdomains was calculated. Each domain is scored with a 2, 1 or 0, where 2 means appropriate prescribing, 1 means less appropriate prescribing and 0 means inappropriate prescribing. So, the minimum and maximum of each domain are 0 and 2, respectively. We defined the quality of a prescription as appropriate if the weighted total index score was above the cut-off score of 70% of the total score (i.e. summated scores of all seven domains) of the tool, and as inappropriate when the score was below the cut-off score of 70%.
Psychometric properties
For the reliability and validity analysis, we dichotomised the classes 0 and 1 in ‘inappropriate’, and 2 in ‘appropriate’. We analysed the interrater reliability as well as the test–retest reliability using Cohen’s kappa. Since the draft of the TAPP-ID has two routes and the content of the domains ‘indication’, ‘dosage’ and ‘duration’ differs between these routes, we first determined the agreement of the ratings of two independent assessors (interrater) or the two assessments of one assessor (test–retest) on the chosen route for each prescription. For these three domains, we calculated Cohen’s kappa only for the prescriptions with the same chosen route. For the other domains, that is, duplications, interactions, evaluation of effect and evaluation of side effects, we calculated Cohen’s kappa for all prescriptions since the content of these domains does not differ per route. We interpreted a kappa of lower than 0.20 as slight, 0.21–0.40 as fair, 0.41–0.60 as moderate, 0.61–0.80 as substantial and higher than 0.80 as almost perfect agreement. 36 To assess the convergent validity of the TAPP-ID, we also calculated the correlation between the judgements of the expert panel (gold standard) in comparison to the assessor rating using Cohen’s kappa.
All statistical analyses of the psychometric properties were performed using IBM Statistics version 28 for Windows.
Results
Participants
In total, 37 professionals with expertise in ID healthcare participated in part A of this study. Fifteen ID physicians, five psychiatrists and five pharmacists participated in panel 1 and nine ID physicians, one psychiatrist and two pharmacists in panel 2. Table 1 shows the number of participants per phase of the Delphi procedure.
Participants per step for the development of the tool.

In the preparation phase, the participants responded to a semi-structured questionnaire.
In each Delphi iteration, the participants responded to a questionnaire with predefined statements.
In the weighting round, the participants weighted the identified domains.
Preparation phase
The semi-structured questionnaire about the agents and domains, as suggested by the research team to be included in the tool (i.e. the TAPP-ID), was sent to 25 experts in total and had a response rate of 92%. Topics of this questionnaire were the kind of drug class and agents, type of use, indication, predefined domains by the research team and the design of the TAPP-ID. Most comments were made on the psychotropic agents that should be included in the TAPP-ID. Based on these comments, the research team added six and removed two PDs. In addition, the research team simplified the flow chart on the route choice and modified the domain dosage according to the Delphi panellists’ (panel 1) suggestions. Finally, there were comments about including the domain interaction and the terminology regarding (in)appropriate use. These comments conflicted with each other, so the research team decided to include these topics in the first Delphi iteration for judgement by the whole panel 1.
Delphi iterations
In total, three iterations were needed to achieve agreement on the content of the assessment tool. In the three iterations, the respondents reached agreement on 75 statements with a median score between 7 and 9, and when controlled for the IPRAS (Table 2). The statements were arranged per topic, successively addressing the topics’ terminology and flowchart, including agents, domains and response categories in general, and elaborated domains. For example, ‘The flowchart clearly shows the route to be followed within the TAPP-ID when a PD is prescribed’ or ‘The question “Has it recently been evaluated whether the agent has the desired effect?” aligns with the domain evaluation of effect’.
Results of the Delphi iterations.

Statements with a median of 7–9 with agreement were classified as content for the tool, on which agreement was achieved. Statements with a median of 4–6 or any median with disagreement were classified as uncertain and were included in the sequel iteration. Statements with a median of 1–3 with agreement were classified as irrelevant or inappropriate for the tool.
Content of the TAPP-ID
After the Delphi iterations, the content of the TAPP-ID could be determined. Seven domains related to PD prescribing were included: indication, dosage, duration of use, duplication, interactions, evaluation of effect and evaluation of side effects. The TAPP-ID starts with a section introduction and instructions, followed by a short explanation per domain, and ends with an elaboration on each domain for each PD. In total, 60 PDs were included in the TAPP-ID. After the route has been determined using the indication flowchart (Figure 2), the domains can be scored per PD. Five out of seven domains consist of one question per domain; the domain evaluation of effect and evaluation of side effects have two questions each (Table 3).
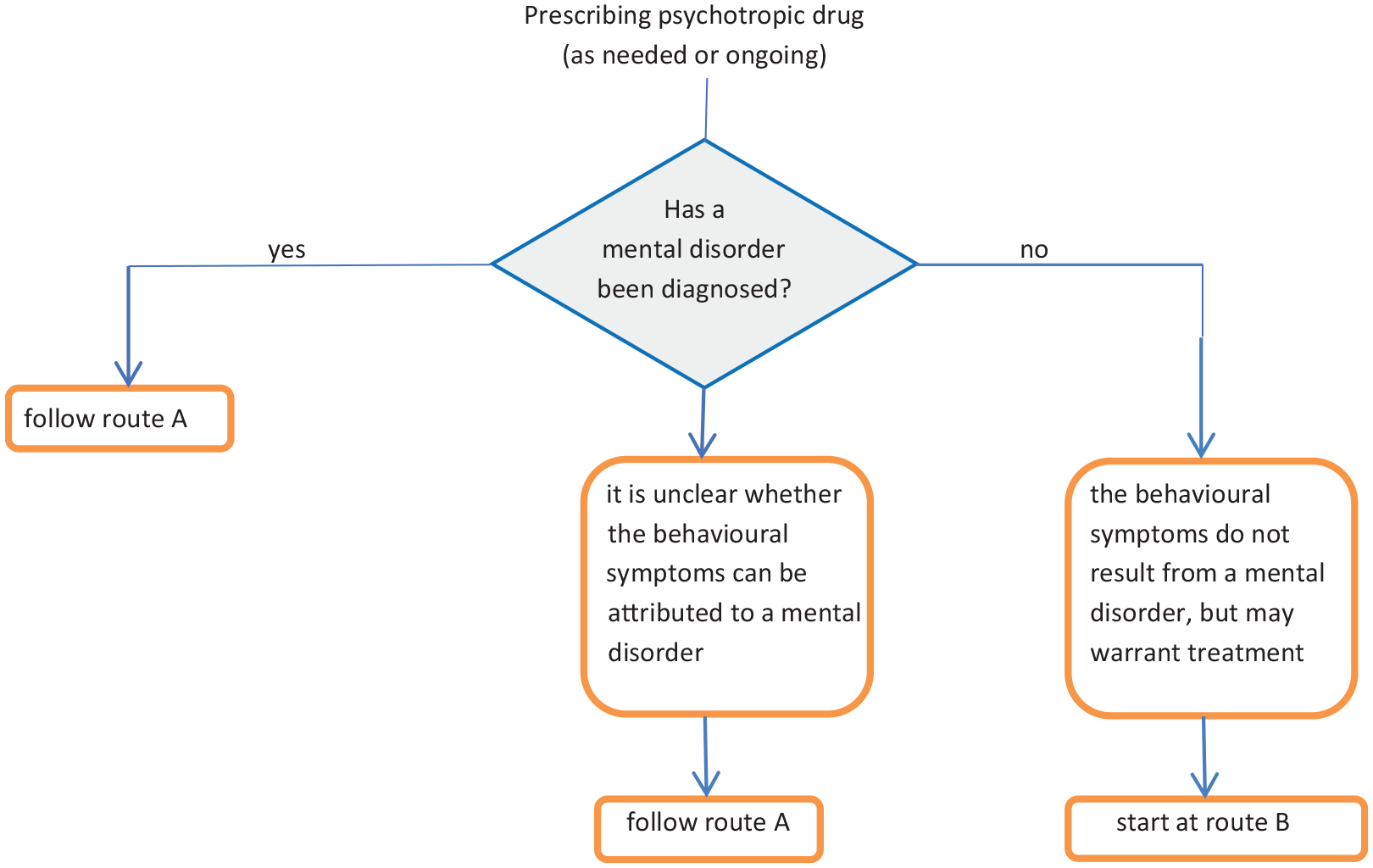
Flowchart to determine the route within the tool based on the indication for prescribing.
Question per domain of the tool: appropriate psychotropic drug prescribing in intellectual disabilities.
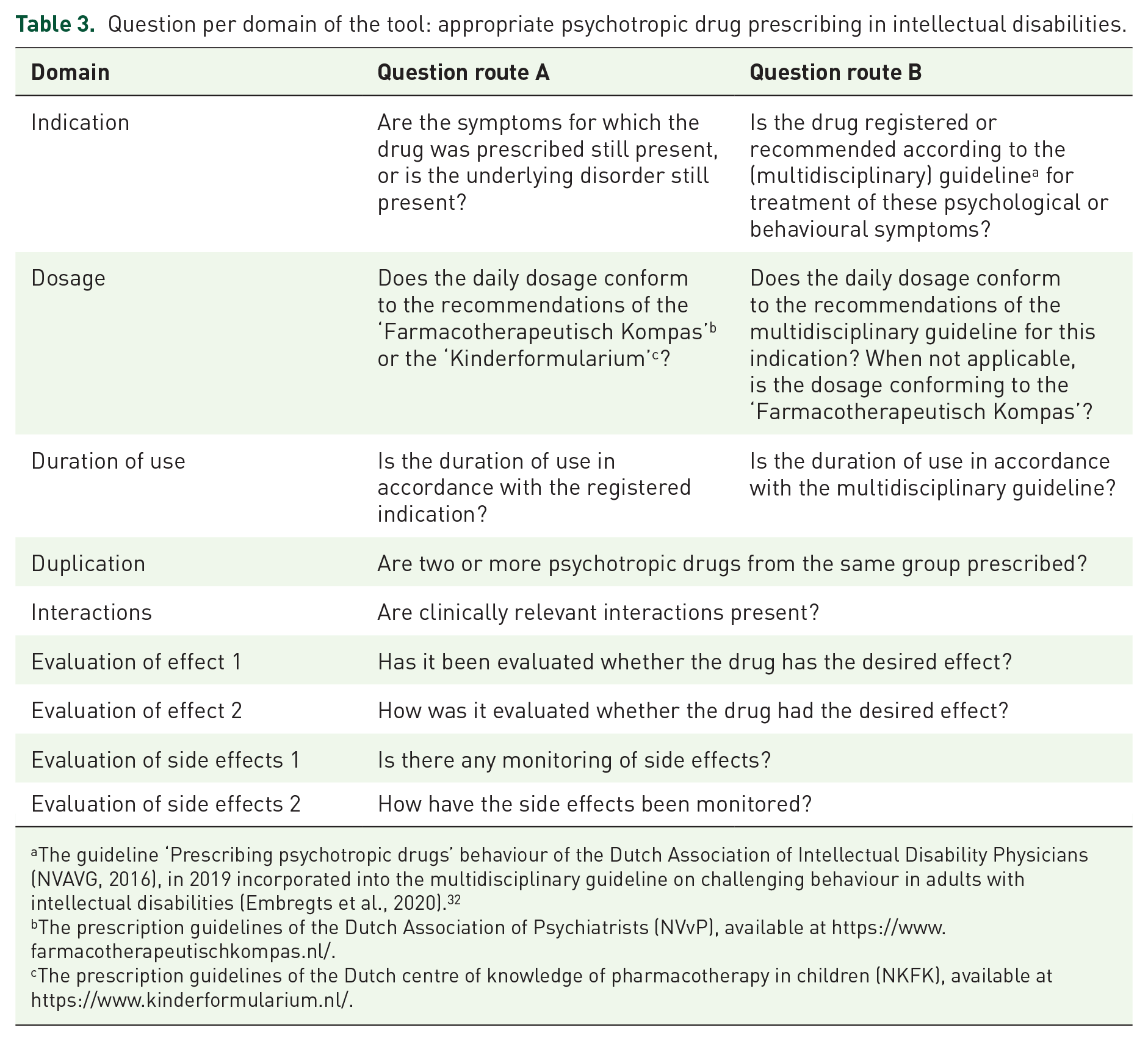
The guideline ‘Prescribing psychotropic drugs’ behaviour of the Dutch Association of Intellectual Disability Physicians (NVAVG, 2016), in 2019 incorporated into the multidisciplinary guideline on challenging behaviour in adults with intellectual disabilities (Embregts et al., 2020). 32
The prescription guidelines of the Dutch Association of Psychiatrists (NVvP), available at https://www.farmacotherapeutischkompas.nl/.
The prescription guidelines of the Dutch centre of knowledge of pharmacotherapy in children (NKFK), available at https://www.kinderformularium.nl/.
Weighting of the domains
Panel 2 weighted the domains of the assessment tool on a scale of 1–10. Table 4 shows the mean weights of the domains. The mean weights of the domains ‘evaluation of effect’ and ‘evaluation of side effects’ are a mean composite score because these two domains consist of two questions. Since the scores per domain have a maximum of 2, the sum of the total weighted domains, and thus the total index score, ranges from 0 to 111.6. Higher scores indicate more appropriate PD use.
Results of domain weighting of the draft of the TAPP-ID by Delphi panel 2.

The mean weight with standard deviation of all panellists’ scores (n = 12) of the domains on a scale from not at all important to extremely important (1–10).
TAPP-ID, Tool for Appropriate Psychotropic drug Prescribing in people with Intellectual Disabilities.
Psychometric properties
The sample of 34 participating clients whose PD use was assessed by the participating prescribers with the TAPP-ID included 15 males and 19 females. Of all participants, written informed consent was given by their legal representative. Their mean age was 56 years (range 27–89 years). They were prescribed a total of 63 PDs (mean number of 1.85 prescriptions and range 1–4 prescriptions per participating client). Most prescribed PDs were antipsychotics (47.6%) and antidepressants (33.3%), followed by anticonvulsants and benzodiazepines (both 7.9%). Mean weighted total index score of the TAPP-ID (range 0–111.6) of the prescriptions at baseline was 80.78 (SD 11.56).
Reliability and validity
Regarding the test–retest reliability, we used 61 prescriptions, because two prescriptions were stopped at timepoint one. For 34 of the 61 prescriptions, the assessor (prescriber) chose the same route at baseline as at timepoint one. For the domains ‘indication’ and ‘duplications’, we found a perfect agreement; agreement on the domains ‘dosage’ and ‘duration of use’ proved substantial, the domain ‘interactions’ moderate, and the domain ‘evaluation of side effects’ fair. We could not calculate Cohen’s kappa of both questions on the domain ‘evaluation of effect’ and of the question ‘How have the side effects been monitored?’ of the domain ‘evaluation of side effects’ because one of the variables was a constant. In all cases, there was one assessor who scored a 0 for all prescriptions.
For the interrater reliability analysis, we used 60 prescriptions, because three prescriptions were stopped at the time of scoring by the second assessor (prescriber). Regarding the interrater reliability, for 27 of the 60 prescriptions, both independent assessors chose the same route at baseline. The agreement was found to be perfect for the domain ‘duplication’, followed by the domain ‘interactions’, in which the agreement was found to be substantial. Agreement on the domains ‘indication’ and ‘dosage’ was moderate, the first question of the domain ‘evaluation of effect’ fair and the first question on the domain ‘evaluation of side effects’ slight. For the domain ‘duration of use’, a negative Cohen’s kappa was found, meaning there was no agreement on this domain. We could not calculate Cohen’s kappa of the ‘how’ questions on both domains ‘evaluation of effect’ and ‘evaluation of side effects’ because one of the variables was a constant. In both cases, there was one assessor who scored a 0 for all prescriptions. Table 5 shows the results of both the test–retest and the interrater reliability.
Reliability of the tool for appropriate psychotropic drug prescribing in people with intellectual disabilities.

Test value using Cohen’s kappa.
No variance in ratings, so no Cohen’s kappa can be estimated.
The agreement between the experts’ panel judgement (gold standard) and the physician score was slight (kappa 0.078).
Discussion
In this study, we developed, with the use of a Delphi procedure, the TAPP-ID, a tool to optimise appropriate prescribing of PDs in people with ID. Of 60 PDs, prescribers can score the appropriateness of their prescription on the seven domains: ‘indication’, ‘dosage’, ‘duration’, ‘interactions’, ‘duplications’, ‘evaluation of effect’ and ‘evaluation of side effects’. Next, the total index score can be calculated by multiplying each domain score by the mean weight score per domain and summing up all these weighted domain scores. The higher the score, the more appropriate the prescribing.
In three Delphi iterations, 37 field experts had reached consensus about the domains and agents included in the TAPP-ID. By investigating the psychometric properties of the TAPP-ID, we found that these were overall sufficient. Five out of seven domains of the TAPP-ID have a moderate to perfect test–retest reliability, one domain showed a fair test–retest reliability, and for one domain, the test–retest reliability could not be calculated. This is equivalent to the interrater reliability, although there was no agreement on the domain ‘duration of use’. The convergent validity, the agreement between the expert’s judgement (gold standard) and the physician’s score, was slight.
With regard to the choice of the route that should be followed when using the TAPP-ID, difficulties may arise. This may be illustrated by the low percentage of agreement between the different assessors of the PD prescriptions. However, diagnosing mental disorders may be difficult in people with ID, especially when there is a lower level of functioning. 37 Diagnostic overshadowing can be a factor here, where psychopathology is attributed to the mental or cognitive condition rather than an underlying mental disorder, although a recent review shows inconsistent findings. 38
The domains of the TAPP-ID established in this study align with domains or items of instruments used in elderly care (Psychotropic Drug Monitor 27 ) and with recently developed instruments in the field for people with ID (TOP-ID and OPTIMA-ID28,29). These three instruments were also developed with the use of a Delphi procedure. The TOP-ID is a prescription and deprescription tool for adults with ID. 28 The TAPP-ID and TOP-ID tools share the topics indication, evaluation of treatment effect and duplication. However, the starting points of the instruments differ. The TOP-ID starts from four clinical situations in medical domains, pain, sleep disorders, gastrointestinal disorders and challenging behaviour, respectively, and also includes a deprescription guide, 28 whereas the TAPP-ID starts from the pharmacological treatment and includes questions on the quality of prescribing PDs. The OPTIMA-ID is developed for older adults with ID and has as a starting point the anatomical system domains and the therapeutic classes, which are elaborated in criteria. 29 The similarities of the OPTIMA-ID to the TAPP-ID can be found in the general principles and criteria for the nervous system, in which domains such as indication, dosage, duration, duplication, evaluation of effect and evaluation of side effects are elaborated. Although the TOP-ID and OPTIMA-ID also aim to support prescribers in optimising their drug prescribing in (older) people with ID, these tools are not explicitly focused on PD prescription and do not include specific elaboration per PD.
For the different domains, the study of test–retest and interrater reliability shows varying results, but was overall sufficient. To our knowledge, psychometric data are not known from several similar tools, such as the TOP-ID and the STRIP. From some tools, data on psychometric characteristics are available. However, these tools are validated in a different target population. Studies showed a good to excellent test–retest and interrater reliability of the MAI. 39 Of the APID, interrater reliability scores are similar to those from our study regarding the domain duplication, and slightly higher for the interaction and dosage domains, and clearly higher for the duration and evaluation domains. 26
Strengths and limitations of the use of the TAPP-ID
PDs in people with ID may be prescribed by multiple physicians or authorised nurses. In the Netherlands, a study has shown that specialist ID physicians or psychiatrists do not sufficiently adhere to guidelines when prescribing antipsychotic drugs to people with ID, due to insufficient translation of guidelines to meet prescribers’ organisations.40,41 General practitioners are also prescribers of PDs in people with ID, although their knowledge on this topic can be improved. 42 The TAPP-ID could provide opportunities for all prescribers, specialist and nonspecialist, to improve their knowledge on prescribing and monitoring of PDs and to adhere to guidelines.
If there is a diagnosed mental disorder, the available Dutch guidelines that were used as a base for the elaborated domains per PD of the TAPP-ID are very useful with regard to the domains’ indication, dosage and duration. However, based on the scarce evidence for the effect of PDs in the treatment for challenging behaviour of people with ID, guidelines recommend being cautious in prescribing PDs and to prefer non-pharmacological treatment for challenging behaviour as a first choice.31,32 Furthermore, there are also no data on the dosing of antipsychotics for challenging behaviour.
Strengths of the study
The development of the tool took place using a Delphi procedure with 37 experts from the field as Delphi panellists. Expert consensus, the principle of a Delphi procedure, is valuable when no or limited evidence is available on a topic, as is the case when prescribing PDs to people with ID. Besides, in a Delphi procedure, anonymity is guaranteed, and it can overcome issues with group-based processes, such as dominant individuals. 30 The high response rate for all three iterations in our study contributes to the reliability of the Delphi procedure.
Limitations of the study
This study also has some limitations. First, although the Delphi method is a robust, validated method, the determination of the domains’ content was based on the best available evidence regarding the quality of the prescription of PDs in people with ID. However, the evidence for this quality is scarce with regard to PDs for challenging behaviour or when the presence of a mental disorder is difficult to diagnose. Second, a limitation may be that we did not ask patients, their caregivers and relatives to give their views and opinions on the quality of their medication prescriptions, contributing to the development of the tool. The Delphi panel consisted of prescribers (ID physicians and psychiatrists) and pharmacists, as we primarily wanted to know these professionals’ views and opinions regarding the content of the tool to guarantee its medical and pharmaceutical quality. The perspectives of the patients, their caregivers and relatives should be considered in a subsequent implementation study of the tool. With regard to the investigation of the reliability and validity of the TAPP-ID, the study also has limitations. First, testing the psychometric properties of the study was conducted with a limited number of participants, which may impact the generalisability and robustness of the findings. Second, a joint judgement of an experienced ID physician and an experienced pharmacist was chosen as the gold standard, since there was no instrument available measuring a similar construct. This could have influenced the quality of the gold standard. Both the gold standard and the assessors used the medical records as a base for their TAPP-ID domain scoring. Differences in assessment between the assessors may have arisen due to problems with the data extraction in these medical files, which was also the case in the APID study. 26 Because data on PD use of the various participating clients were recorded differently, data could be less accessible and may also have caused problems in data searches, for example, the date when a prescription was started. This may have affected the agreement on the domain ‘duration of use’. Finally, assessors were prescribers for some of the patients or knew the patients, so their assessment was most likely influenced by prior knowledge about the PD prescriptions of these patients, especially for the evaluation domains, which are more difficult to assess objectively.
Conclusion
In this study, a tool for the appropriate prescribing of PDs in people with ID was developed. Using a Delphi procedure, experts agreed on seven domains: indication, dosage, duration of use, duplications, interactions, evaluation of effect and evaluation of side effects. Although further development and implementation of the tool is desirable, scoring the domains of this reliable tool may support prescribers in clinical practice in evaluating their PD prescriptions in people with ID.
Footnotes
Acknowledgements
The authors would like to thank all the professionals and the organisation participating in this study. More specifically, we would like to acknowledge the consultant pharmacist, the field experts who participated in the Delphi procedure, the prescribers of the ID care organisation, and all the clients and their representatives who consented to participate in the study.
