Abstract
This is a case report of a 75-year-old immunocompromised male who developed encephalopathy while undergoing treatment for disseminated herpes zoster with peripheral nerve involvement. While his initial presentation involved primarily profound lower extremity weakness, he developed progressive confusion to the point of obtundation only after initiation of standard therapy with intravenous acyclovir. The evaluation of his altered mental status was largely unremarkable. It was only after his acyclovir was discontinued that his symptoms resolved and he returned to his baseline mental status. His presentation was most consistent with acyclovir-induced neurotoxicity, which can present in patients with renal impairment and those who are immunocompromised.
Introduction
The Herpesviridae family of viruses are highly prevalent infections in humans. Within this family, varicella zoster virus (VZV) is particularly common in children with initial infection presenting as a rash with blisters, fever, and fatigue. 1 Approximately 50% of persons who live to age 85 years will have experienced herpes zoster. 2 This condition typically resolves without developing any serious sequelae. The virus, rather than being cleared by the immune system, becomes latent in the dorsal root ganglion of spinal and cranial nerves which can later be reactivated, typically consolidated to a specific body dermatome. Risk factors for reactivation include increased age, steroid use, immunosuppression, malignancy, and organ or stem cell transplantation. 3 While the typical presentation of a patient with reactivation of varicella, also known as shingles, includes pain, paresthesia, and a vesiculated rash, certain patients can even develop extra-dermatological manifestations of the disease. For example, varicella zoster has been known to cause encephalitis, aseptic meningitis, myelopathy, central and peripheral nerve manifestations, Ramsay Hunt syndrome, and retinal necrosis in some cases. 3 Polyneuritis and polyradiculitis are infrequent complications. In the case of disseminated VZV and central nervous system involvement, treatment usually involves intravenous acyclovir.
Acyclovir is a nucleoside analog that must be activated by a viral enzyme within infected cells in order to be effective. It is predominantly cleared by glomerular filtration and tubular secretion in the kidneys. 4 The most widely known adverse reaction from acyclovir toxicity is profound kidney injury secondary to crystal nephropathy. 5 However, a lesser studied effect of this drug is the development of neurotoxicity. In someone with known renal disease, especially end-stage renal disease (ESRD) and transplant patients, the propensity for neurotoxicity is increased though data are limited. Neurological symptoms can appear within 24-h of administration of acyclovir.6–9
Case report
A 75-year-old Caucasian man with history of hypertension, atrial fibrillation, and renal transplant 5 years prior was transferred to UNC hospital for urgent management of esophageal perforation and pneumomediastinum following an episode of emesis. He had presented from long-term care facility, where he had recently been placed due to profound lower extremity weakness. The etiology of his debility was unknown at the time of his admission to UNC hospital. Following repair of his esophageal tear, he was transferred to medical team for medical management of mediastinitis and evaluation of his weakness.
The patient reported that several months prior he had developed lower back pain and a rash and was diagnosed by his primary care provider with herpes zoster reactivation involving the L2-L3 dermatome. He was subsequently treated with acyclovir and prednisone. One month later, he began to experience new lower extremity weakness that progressed to paralysis. Before this point, he had been very functional, frequently running 3 miles a day.
At this point in his care, he was alert, interactive with no mental status changes. His exam revealed severe lower extremity weakness with overt lower motor neuron findings. In addition, he was found with signs of atrophy in the lower extremities with fasciculations and hyporeflexia. Following a comprehensive evaluation, both his cerebrospinal fluid (CSF) and serum by polymerase chain reaction (PCR) were found to be positive for VZV. Electromyography (EMG) studies showed signs of active denervation in lower extremities with upper extremity motor nerves concerning for multifocal motor neuropathy or VZV polyneuritis.
This is an extremely rare condition and the patient was deemed to be at an increased risk of dissemination due to his immunocompromised state. Treatment involved intravenous acyclovir on day 3 of admission at an initial dose of 10 mg/kg every 8 h for 3 weeks as recommended by the manufacturer’s data sheet and cefepime for mediastinitis. Baseline creatinine and weight at the time of admission was 0.44 mg/dL and 57.7 kg, respectively.
On day 11 of admission, the patient began to develop encephalopathy. His symptoms started with mild confusion, disorientation, and intermittent hallucinations. Over time, the patient progressed to near complete obtundation and was responding only minimally to noxious stimuli.
The differential diagnosis for the abrupt deterioration in his mental status included viral encephalitis from uncleared VZV in his CSF, cerebrovascular accident (CVA), cefepime toxicity, nutritional deficiency, and seizure activity. Computed tomography (CT) of the head at the time was concerning for CVA with hypodensities in the corona radiata and insula. Magnetic resonance imaging (MRI) was unable to be performed due to retained metal from a prior injury. Neurology was consulted and felt that his acute CVA was unlikely to be related to his obtundation. Electroencephalogram (EEG) illustrated no seizure activity and was consistent with moderate bilateral cerebral dysfunction and encephalopathy. Additional infectious studies performed, including fungal cultures, human herpesvirus 6 (HHV-6), herpes simplex virus (HSV), enterovirus, and cytomegalovirus (CMV) studies, were all negative. He did have evidence of mild ketoacidosis, which prompted concern for possible contribution from starvation though his mental status did not improve with enteral feeding. Throughout this period, his serum creatinine ranged from 0.44 to 0.70 mg/dL.
Following this exhaustive search for causal encephalopathy, it was felt that his presentation was either due to herpes encephalitis or medication toxicity. It was thought that zoster encephalitis was less likely the cause given that he had intact mental status prior to treatment and his presentation began while on high-dose acyclovir. Cefepime toxicity was felt to be less likely given the degree of his encephalopathy, and his failure to improve following his completion of cefepime.
After discussion with several medical teams, the possibility of acyclovir neurotoxicity was raised, which is a rare side effect. A 24-h urine collection was obtained and reflected a creatinine clearance of 30, which prompted a reduction in the total daily dose of acyclovir to 10 mg/kg/day. Interestingly, following this change, the patient showed minor signs of neurological improvement, though he still remained largely obtunded. He continued empiric treatment with intravenous acyclovir for a 3-week total course, and he cleared his CSF of VZV, although remained positive in his blood.
The day following completion of his acyclovir therapy, he had a dramatic recovery in mental status. He was able to answer questions, follow commands, and move his lower extremities albeit with weakness. Fortunately, after a long and complicated hospital course, he had returned to his baseline mental status and was discharged to a rehab facility.
Discussion: herpes zoster encephalopathy versus acyclovir neurotoxicity
In the aforementioned case, high doses of intravenous acyclovir at 10 mg/kg were required for his polyneuritis and disseminated infection. However, in reviewing the case, it was felt that acyclovir neurotoxicity was the most likely explanation for his marked mental status changes, especially given the temporal relationship with his acyclovir therapy. While not confirmed, his clinical presentation was very suggestive of this uncommon drug reaction. Another challenge was that a definitive duration of treatment for VZV encephalitis with acyclovir has not been clearly defined, which had prompted extensive discussion once the idea of drug toxicity was considered.
This case highlights the difficulty of treating a patient with disseminated VZV infection. For our patient, the risk for dissemination to his CSF was likely increased due to taking immunosuppressant medication following transplantation. The patient had been on a maintenance regimen of tacrolimus, prednisone, and mycophenolate mofetil prior to admission and required close care coordination with the renal transplant team. Mental status changes observed in these patients could be related to treatment failure and subsequent worsening of infection or, as seen in this case, direct neurotoxicity from acyclovir. There are limited data to guide differentiation of these two entities and treatment protocols, for each condition completely oppose each other. As a result, inaccurate diagnosis can lead to devastating results. There is some discussion about visual hallucinations and speech changes being more closely linked to acyclovir neurotoxicity.6,7 Prior studies have shown that assessing for high concentrations of the effective metabolite of acyclovir, 9-carboxymethoxymethylguanine, in the serum and CSF is predictive of neurotoxicity.10–12 Treatment of these patients requires thorough and frequent neurological exams to monitor for subtle changes in mental status. Involvement of neurology early can be beneficial as changes in mental status can be subtle and they can assist with alternate diagnoses. If acyclovir neurotoxicity is suspected, it is important to monitor the serum and CSF and to consider evaluation of metabolites and PCR of VZV. Given the renal clearance of acyclovir, dose adjustment for renal impairment is important (Table 1). We recommend a 24-h urine collection if possible in order to obtain a more accurate representation of creatinine clearance. A reduced dose for patients with known renal disease is recommended; however, cases of neurotoxicity despite appropriate dose reduction have been described. 13 Additionally, the role of hemodialysis, while a complicated operation, has been linked to immediate improvement in mental status.
IV acyclovir dosing for immunocompromised patients with renal adjustment.
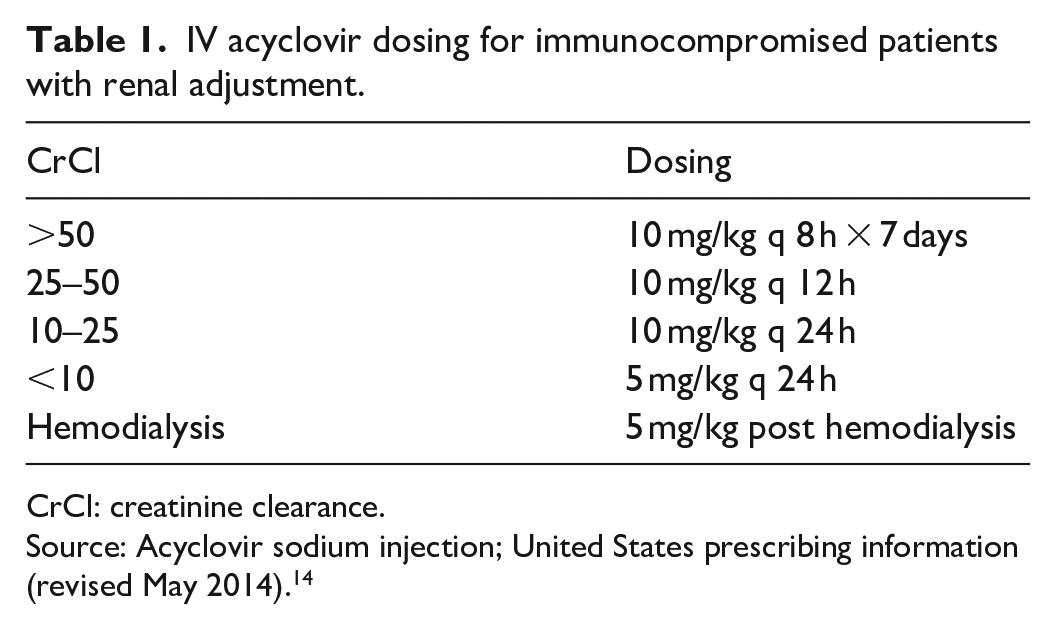
CrCl: creatinine clearance.
Source: Acyclovir sodium injection; United States prescribing information (revised May 2014). 14
Conclusion
Acyclovir-induced neurotoxicity is a rare complication of acyclovir therapy, though it can cause significant disturbances in consciousness and hallucinations. Distinguishing this condition from other more common causes of altered mental status can be challenging. Eliminating herpes encephalitis as the etiology of a patient’s mental status changes is particularly prudent as this impacts treatment strategy. CSF, PCR, and MRI can be useful in determining an accurate diagnosis. Confirmatory testing with acyclovir levels or metabolites may be helpful if available. Unfortunately, this confirmation testing was not routinely performed at our hospital and was thus not obtained. Patients with known renal impairment may be at a higher risk for this toxicity; thus, close monitoring of renal function and proper dose adjustment of acyclovir is important. This case underlines the complexity of care for immunocompromised and transplant patients; it demonstrates that VZV can cause a myriad of neurological complications, and finally, that acyclovir infrequently causes neurological symptoms but should be considered in those with renal disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed verbal and written consent for patient information to be published in this article was obtained. The patient had decisional capacity and the authors of this article took strict measures to ensure anonymity with regard to the patient’s identifying information.
