Abstract
Background:
Chimeric antigen receptor T-cell (CAR-T) cell therapy represents a significant advancement in cancer treatment, offering remarkable responses in certain hematologic malignancies. However, the risk of secondary primary malignancies (SPMs) associated with CAR-T therapy is a growing concern. Recent studies suggest that antibiotics, which are frequently used in CAR-T patients, may influence this risk, yet their effects remain poorly understood.
Objective:
This study aims to systematically evaluate the association between antibiotics and the incidence and timing of SPMs in patients receiving CAR-T cell therapy, using data from the FDA’s Adverse Event Reporting System (FAERS) database.
Design:
We analyzed reports from FAERS spanning from Q2 2017 to Q1 2024, focusing on SPMs associated with various CAR-T therapies.
Methods:
A comprehensive signal analysis was conducted to explore the associations between antibiotic usage and specific SPMs for different CAR-T products. In addition, we employed cumulative hazard curves to evaluate the time to onset of SPMs in patients receiving antibiotics versus those who did not.
Results:
We have provided a comprehensive summary of all signals for CAR-T-associated SPMs. In addition, our analysis identified significant variations in the association between antibiotics and SPM incidence depending on the CAR-T therapy administered. Antibiotics were associated with a decreased risk of SPMs in patients treated with anti-CD19 CAR-T therapies, particularly brexucabtagene autoleucel. Conversely, a higher risk of SPMs was observed in association with antibiotics for anti-BCMA therapies, with idecabtagene vicleucel showing a notably elevated risk. Notably, antibiotics were associated with an earlier onset of SPMs across CAR-T therapies, suggesting a possible relationship between antibiotics and the timing of these malignancies. Finally, we explored the underlying biological pathways that may be associated with these observations.
Conclusion:
Antibiotics were associated with both the risk and timing of SPMs in patients undergoing CAR-T cell therapy. This study highlights the need for further research to better understand the complex interactions between antibiotics and CAR-T therapies, as well as the potential implications for clinical management and patient care.
Plain language summary
CAR-T therapy has proven to be highly effective in treating certain blood cancers. However, there are concerns about its long-term safety, particularly the risk of secondary primary malignancies (SPMs) – new cancers that develop after treatment for the original cancer. This study examines the association between antibiotics, commonly prescribed to CAR-T patients, and the occurrence and timing of these new cancers, using data from the FAERS database. We analyzed reports from April 2017 to March 2024, focusing on SPMs linked to different CAR-T treatments. Our findings suggest that antibiotics are associated with differences in the risk of SPMs depending on the type of CAR-T treatment. Specifically, a lower risk of SPMs was observed in association with antibiotics in patients receiving anti-CD19 CAR-T therapies, like brexucabtagene autoleucel, while a higher risk was observed in those treated with anti-BCMA therapies, particularly idecabtagene vicleucel. Furthermore, antibiotics were linked to an earlier onset of SPMs, suggesting they might accelerate the development of these new cancers. Overall, this highlights the importance of personalized monitoring strategies for CAR-T patients and indicates a need for further research to understand these complex interactions.
Introduction
Chimeric antigen receptor T-cell (CAR-T) therapy represents a significant advancement in oncology, particularly for patients with relapsed or refractory hematological malignancies. By engineering a patient’s T cells to target and destroy cancer cells, CAR-T therapy has achieved remarkable clinical successes, particularly in treating B-cell leukemias and lymphomas.1–3 However, the long-term safety of CAR-T therapy remains an area of active investigation. 4
As CAR-T therapy has become more widely used, concerns have emerged regarding its safety profile, including the risk of developing secondary primary malignancies (SPMs).5–7 The identification of SPMs is critical for understanding the long-term effects of CAR-T therapy and guiding patient management strategies. To date, pharmacovigilance studies utilizing databases like the FDA’s Adverse Event Reporting System (FAERS) have provided valuable insights into these risks.4,8
Recent studies have shown that changes in the intestinal microbiome were associated with clinical outcomes following anti-CD19 CAR-T cell therapy in patients with B-cell malignancies. 9 One important aspect of CAR-T therapy that has received less attention is the interaction between antibiotics and CAR-T-associated SPMs. Antibiotics are commonly used in patients undergoing CAR-T therapy to manage infections that may arise due to immune suppression. 10 However, the relationships between antibiotics and either the efficacy of CAR-T therapy or the occurrence of SPMs remain poorly understood.
This study aims to address these gaps by utilizing the FAERS database to analyze adverse events (AEs) associated with CAR-T therapy, with a specific focus on the association with antibiotic use. By examining the association between antibiotics and the incidence and timing of SPMs, we aim to provide a comprehensive assessment of how antibiotic use may be related to the safety profile of CAR-T therapy and inform clinical practices to better address these risks.
Patients and methods
The reporting of this study conforms to the READUS-PV 11 statement.
Data source and de-duplications
The data were retrieved from the publicly accessible FAERS database, covering a substantial time period from the second quarter of 2017 to the first quarter of 2024. FAERS comprises seven datasets capturing information on drug utilization and AEs, including demographic and administrative details (DEMO), pharmaceutical substances (DRUG), adverse drug reaction reports (REAC), patient outcomes (OUTC), report sources (RPSR), drug therapy durations (THER), and usage/diagnosis indications (INDI). Preferred terms (PT) within the REAC and INDI datasets are categorized into “high-level terms” (HLT), “high-level group terms” (HLGT), and system organ classes (SOCs) in alignment with the Medical Dictionary for Regulatory Activities (MedDRA, version 26.1, maintained, developed, and distributed by the Maintenance and Support Services Organization (MSSO) under contract to the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), with technical and financial oversight by the MedDRA Management Committee). 12 Our study meticulously followed the FDA directives by implementing a rigorous procedure to identify and eliminate duplicate reports. Following the procedure established by Khaleel et al., 13 we excluded cases that presented consistency in administered drugs and AEs but discrepancies in only one or none of the following fields: sex, age, reporting country, event date, start date, or drug indications. We have encapsulated the entire process within an R package called “faers,” and it has been accepted by Bioconductor.
AEs data of CAR-T
To maximize the utilization of the FAERS data, we conducted a comprehensive search that included both brand names and generic names of CAR-T products. The included CAR-T products consisted of commercially available anti-CD19 therapies (axicabtagene ciloleucel (axi-cel), brexucabtagene autoleucel (brexu-cel), lisocabtagene maraleucel (liso-cel), tisagenlecleucel (tisa-cel)) and anti-B cell maturation antigen (BCMA) CAR-T therapies (idecabtagene vicleucel (ide-cel), and ciltacabtagene autoleucel (cilta-cel)).14–18 In addition, we retrieved the associated symptoms of these drugs from the DrugBank 19 database to identify cases that received CAR-T therapy.
Disproportionality analysis
Disproportionality analysis was employed to examine the correlation between SPMs and CAR-T therapy. 20 For robustness, we utilized both frequentist and Bayesian techniques, such as the odds ratio (OR) and Bayesian confidence propagation neural network. A statistically significant signal was detected when the lower limit of the 95% confidence interval (CI) for the odds ratio (OR025) exceeded 1, the lower boundary of the 95% CI for the information component (IC025) was greater than 0, and the count (a) of targeted drugs causing the specified AEs was at least 3(4).
Time to onset analysis of SPMs
In the FAERS database, the time to onset (TTO) refers to the duration between the occurrence of AEs (EVENT_DT) and the initiation of drug usage (START_DT). Cumulative hazard curves were used in our study to demonstrate the event-to-onset characteristics of SPMs after CAR-T therapy, comparing patients who received antibiotics to those who did not.
Pathway analysis
To identify the underlying pathways associated with antibiotics and SPMs in CAR-T therapy, we first used the DGIdb database 21 to identify genes targeted by the antibiotics commonly used in CAR-T patients. Since we did not focus on a specific tumor type, we expanded our analysis to include all tissues in the GTEx database, 22 searching for antibiotic-targeted genes that showed a positive correlation with CD19 (for anti-CD19 CAR-T therapies) or TNFRSF17 (for anti-BCMA therapies). We applied strict criteria to select relevant genes: only those with an adjusted p value of <0.05 and a correlation coefficient greater than 0.3 were considered as the intersecting genes between antibiotics and CAR-T therapy. These genes were then subjected to over-representation analysis using the clusterProfiler 23 R package to identify associated biological pathways. For pathway identification, we focused exclusively on the “Cancer: overview” category from the KEGG (Kyoto Encyclopedia of Genes and Genomes) database. 24 This approach ensured that the selected pathways were highly relevant to cancer biology and the potential molecular mechanisms contributing to the development of SPMs in CAR-T therapy patients.
Statistical analysis
We employed the Chi-squared test for comparative analysis to examine the differences between antibiotic usage and non-antibiotic usage. For reliability, we only included AEs with a minimum of three reports that involved the combined use of antibiotics and CAR-T drugs. The Pearson correlation coefficient was calculated to assess the relationship between antibiotic-targeted genes and CD19 or TNFRSF17 expression. To address the issue of multiple comparisons, we controlled the false discovery rate using the Benjamini and Hochberg method.
Results
Characteristics of intravesical CAR-T case reports
The analysis included 11,953,763 reports, spanning from the second quarter of 2017, when the first CAR-T product tisa-cel became available in FAERS, to the first quarter of 2024. After eliminating duplicate reports, 10,019,167 unique reports remained. Out of this population, 11,255 reports received CAR-T therapy, including 9560 reports for anti-CD19 therapy and 1695 reports for anti-BCMA therapy (Figure 1). Separately, we obtained 5574 reports for axi-cel, 1033 reports for brexu-cel, 334 reports for liso-cel, 2646 reports for tisa-cel, 612 reports for ide-cel, and 1083 reports for cilta-cel (Figure 1 and Table 1). The country with the highest number of reports was America (71.1%), followed by France (3.9%), Spain (3.3%), Germany (3.1%), and Italy (2.7%). The majority of AE reports were filed by healthcare professionals (n = 5450, 48.4%; Table 1). Male cases were more prevalent than female cases (48.6% vs 29.7%). The median age was 61 (IQR: 48–69), and the mean age was 55.44 (SD: 19.75; Table 1).
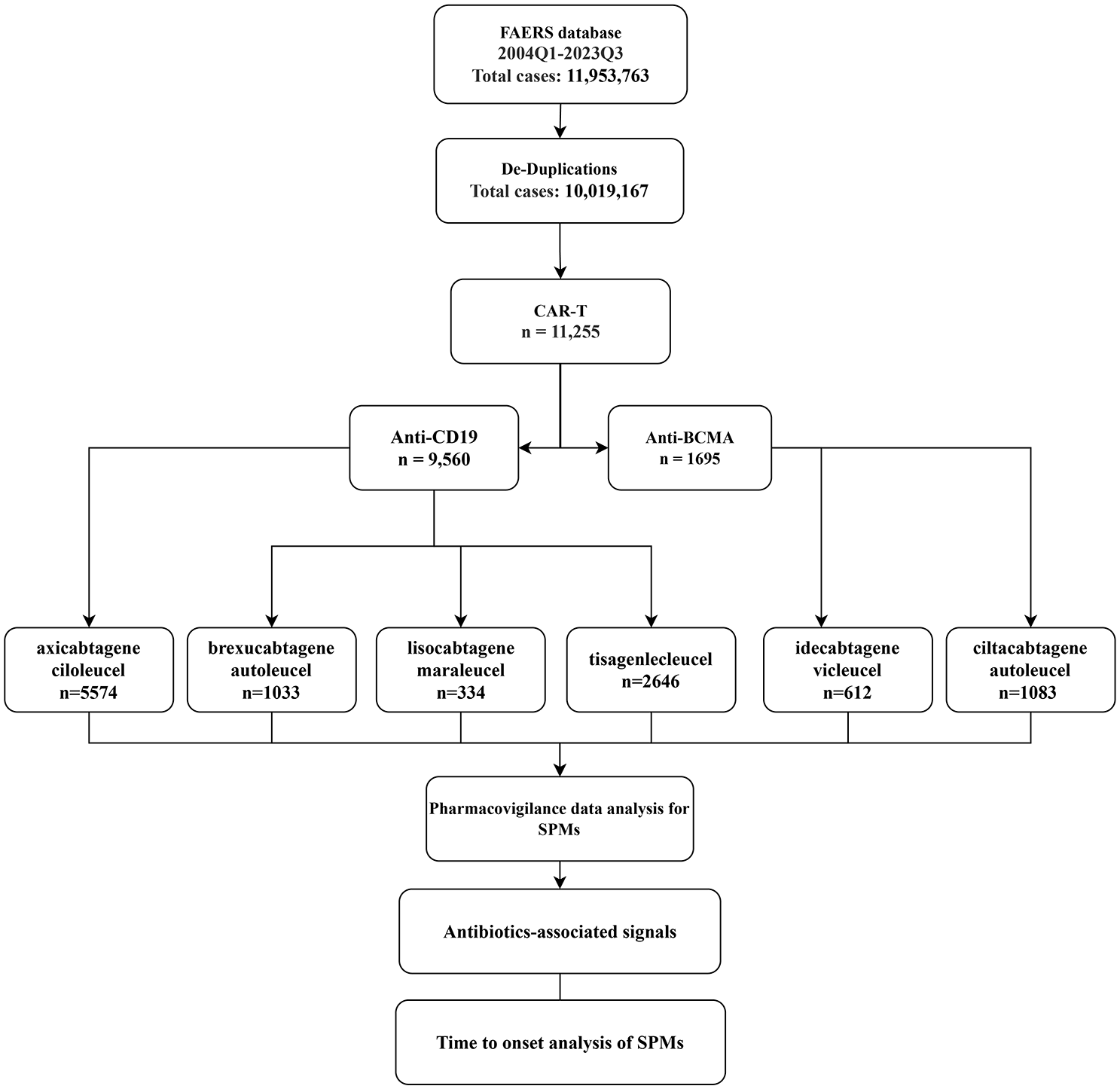
Schematic workflow of the study.
General characteristics of case reports administrated with CAR-T.
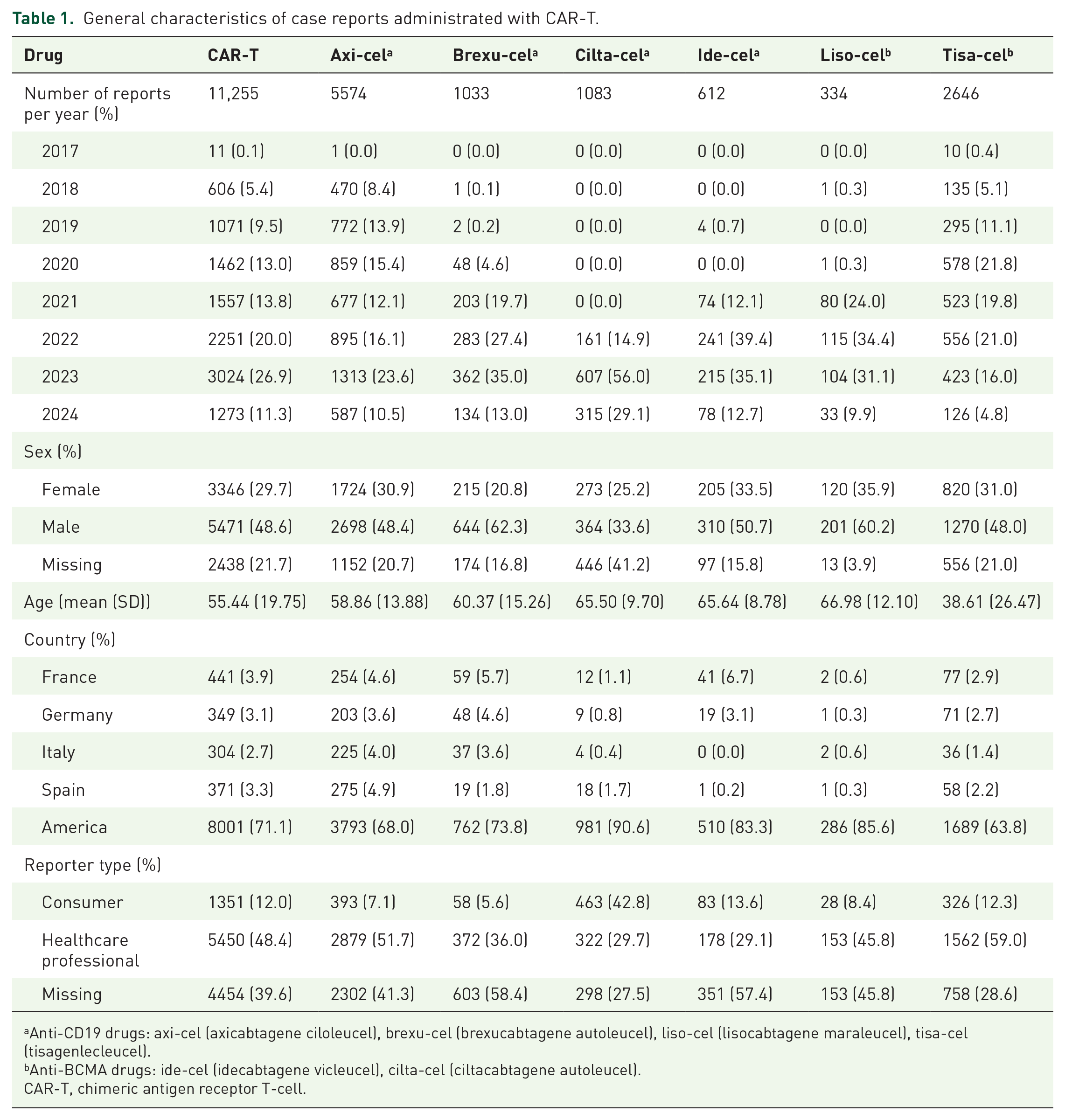
Anti-CD19 drugs: axi-cel (axicabtagene ciloleucel), brexu-cel (brexucabtagene autoleucel), liso-cel (lisocabtagene maraleucel), tisa-cel (tisagenlecleucel).
Anti-BCMA drugs: ide-cel (idecabtagene vicleucel), cilta-cel (ciltacabtagene autoleucel).
CAR-T, chimeric antigen receptor T-cell.
Pharmacovigilance data analysis and signal mining of SPMs
In-depth pharmacovigilance data analysis was carried out to investigate and identify signals of SPMs. We considered AEs from SOC “Neoplasms benign, malignant and unspecified” as the SPMs. 4 This involved analyzing AEs from the complete FAERS database during the period when the specific CAR-T drug was recorded in the database.
The analysis yielded a total of 50 signals at the lowest level term (LLT; Table S1) and 12 signals at the HLGT level when considering all CAR-T drugs collectively (Table 2). When ranked by OR, the top SPM observed was “Reproductive and genitourinary neoplasms, gender unspecified NEC” (Table 2), suggesting a promising area for further investigation into the impact of CAR-T therapy on reproductive and genitourinary malignancies. Focusing specifically on anti-CD19 CAR-T drugs, we identified 12 signals at the HLGT level (Table 3) and 49 signals at the LLT level (Table S2). The top SPM in this group was again “Reproductive and genitourinary neoplasms, gender unspecified NEC” (Table 3) with a notable signal at the LLT level of “Genitourinary tract neoplasm” (N = 9; OR025: 205.661; IC025: 3.085; Table S2). This finding reinforces the clinical relevance of this signal across all CAR-T therapies targeting CD19, suggesting the need for close monitoring of genitourinary health in patients receiving anti-CD19 treatment, especially as these therapies continue to see increased use. Similarly, considering anti-BCMA drugs alone, we identified four signals at the HLGT level (Table 4) and seven signals at the LLT level (Table S3). The leading signal in this case was “Lymphomas, non-Hodgkin’s, T-cell” (Table 4), specifically at the LLT level “T-cell lymphoma” (N = 4; OR025: 19.122; IC025: 1.228) which has also been reported by Shen et al. 25 This points to a potential link between anti-BCMA therapy and the development of specific lymphoma subtypes. The analysis also reveals distinct differences between anti-CD19 and anti-BCMA therapies.
Signals associated with CAR-T.

A statistically significant signal was identified by the lower limit value of the OR 95% confidence interval (OR025) > 1, the lower boundary of the 95% confidence interval for the information component (IC025) > 0, and a ⩾ 3.
CAR-T, chimeric antigen receptor T-cell; HLGT, high-level group terms; OR, odds ratio; NEC: Not elsewhere classified.
Signals associated with anti-CD19 drugs.

A statistically significant signal was identified by the lower limit value of the OR 95% confidence interval (OR025) > 1, the lower boundary of the 95% confidence interval for the information component (IC025) > 0, and a ⩾ 3.
HLGT, high-level group terms; OR, odds ratio.
Signals associated with anti-BCMA drugs.

A statistically significant signal was identified by the lower limit value of the OR 95% confidence interval (OR025) > 1, the lower boundary of the 95% confidence interval for the information component (IC025) > 0, and a ⩾ 3.
HLGT, high-level group terms; OR, odds ratio.
Furthermore, when analyzing the drugs separately, we obtained 30 signals for axi-cel (Table S4), 11 signals for brexu-cel (Table S5), 3 signals for liso-cel (Table S6), 29 signals for tisa-cel (Table S7), 3 signals for ide-cel (Table S8), and 7 signals for cilta-cel (Table S9) at the LLT level. The leading HLGT signals associated with these drugs were “Reproductive and genitourinary neoplasms, gender unspecified NEC,” “Lymphomas, non-Hodgkin’s, B-cell,” “Lymphomas, non-Hodgkin’s, B-cell,” “Lymphomas, non-Hodgkin’s, B-cell,” “Plasma cell neoplasms,” and “Lymphomas, non-Hodgkin’s, T-cell,” respectively. These results point to patterns of specific AEs that may be associated with particular CAR-T products, underlining the importance of tailoring monitoring strategies for different therapies.
Antibiotic-associated signals
It is becoming clear that the use of antibiotics might impair CAR-T function.9,26 We further investigated the association between antibiotic usage and the SPM’s signal of CAR-T. Intriguingly, while antibiotics showed no association with an overall difference in SPM incidence when considering all CAR-T drugs collectively (a: 241; OR: 0.94; CI: 0.81–1.10; p value: 0.4.71; Figure 2), they were significantly associated with SPM incidence for both anti-CD19 (a: 220; OR: 0.85; CI: 0.73–0.99; p value: 4.04e–02) and anti-BCMA (a: 21; OR: 2.11; CI: 1.28–3.48; p value: 4.67e−03; Figure 2), albeit in opposite directions. In separate analyses, we found that the association between antibiotics and SPM incidence varies depending on the specific CAR-T therapy administered. Antibiotics were associated with a lower incidence of SPMs in patients treated with post-brexu-cel (a: 13; OR: 0.42; CI: 0.23–0.77; p value: 5.11e−03; Figure 2) but a higher incidence for patients receiving post-ide-cel (a: 7; OR: 3.88; CI: 1.58–9.54; p value: 4.80e−03; Figure 2). However, antibiotics have no associations with the incidence of SPMs in patients treated with axi-cel (a: 97; OR: 0.88; CI: 0.70–1.10; p value: 0.275), liso-cel (a: 11; OR: 1.74; CI: 0.82–3.69; p value: 0.213), tisa-cel (a: 100; OR: 0.88; CI: 0.69–1.14; p value: 0.366), and cilta-cel (a: 14; OR: 1.64; CI: 0.90–3.01; p value: 0.149; Figure 2).

Impact of antibiotic usage on the signal of CAR-T therapy-related SPMs. Anti-CD19 drugs: axi-cel (axicabtagene ciloleucel), brexu-cel (brexucabtagene autoleucel), liso-cel (lisocabtagene maraleucel), tisa-cel (tisagenlecleucel). Anti-BCMA drugs: ide-cel (idecabtagene vicleucel), cilta-cel (ciltacabtagene autoleucel).
We then analyzing the AEs at LLT level, we found antibiotics were associated with a higher frequency of histiocytosis hematophagic (OR: 4.49; CI: 1.26–15.93; p value: 3.79e−02), B-cell lymphoma (OR: 2.33; CI: 1.13–4.79; p value: 3.37e−02), and hemophagocytic lymphohistiocytosis (OR: 2.22; CI: 1.58–3.12; p value: 4.70e−06), but a lower frequency of diffuse large B-cell lymphoma recurrent (OR: 0.39; CI: 0.19–0.79; p value: 1.03e−02), and myelodysplastic syndrome (OR: 0.31; CI: 0.15–0.63; p value: 9.59e−04) for anti-CD19 drugs (Figure 3(a)). Moreover, antibiotics were associated with a higher frequency of myelodysplastic syndrome (OR: 4.13; CI: 1.30–13.14; p value: 3.31e−02) and hemophagocytic lymphohistiocytosis (OR: 2.82; CI: 1.33–5.97; p value: 1.07e−02 for anti-BCMA drugs (Figure 3(b)).
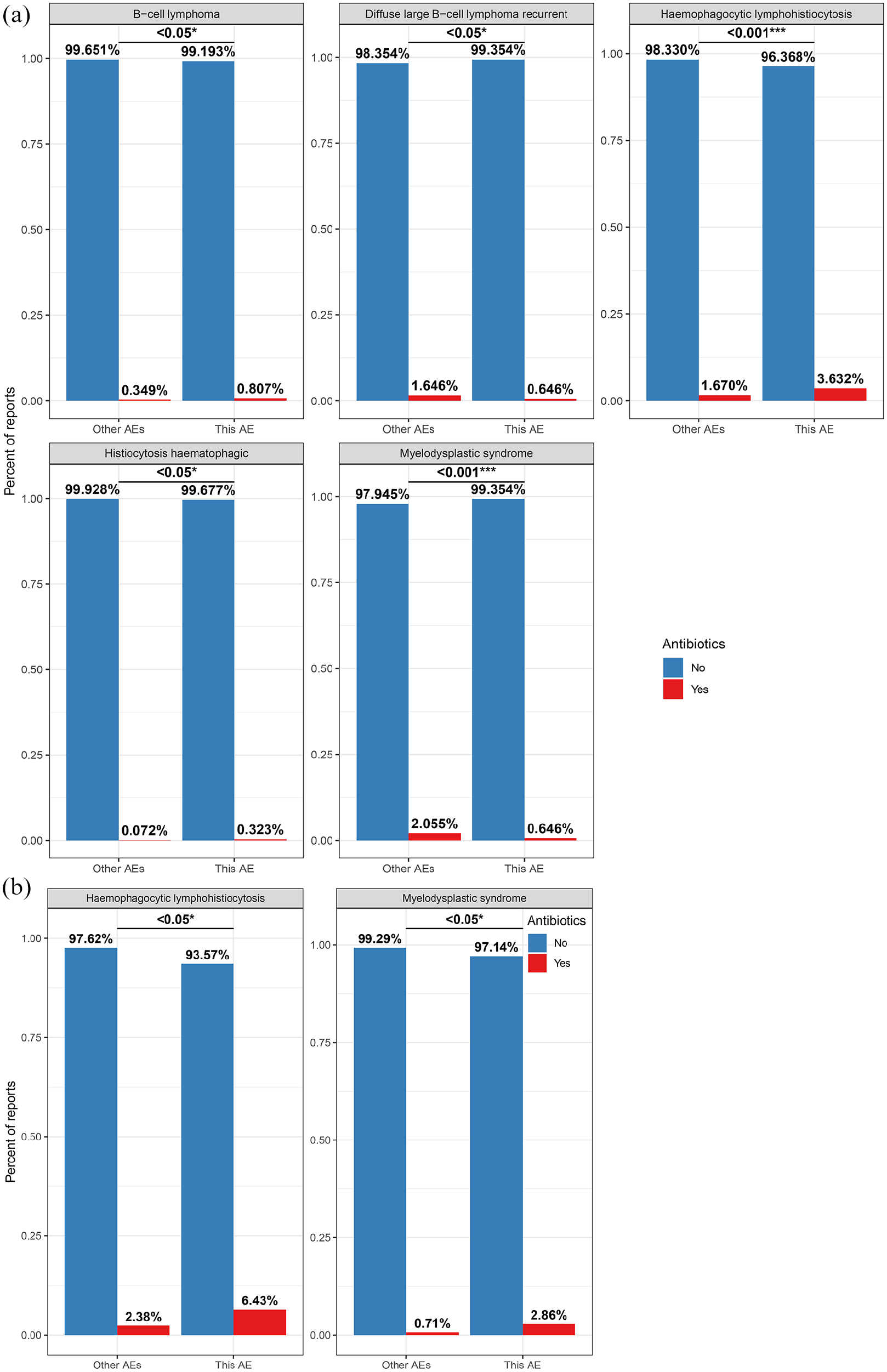
Comparative analysis of antibiotic usage for SPMs at the LLT for (a) anti-CD19 products and (b) anti-BCMA products.
When analyzing the LLT level for each drug, we observed that antibiotics associated with a higher incidence of B-cell lymphoma (OR: 2.68; CI: 1.29–5.59; p value: 1.27e−02) and hemophagocytic lymphohistiocytosis (OR: 1.98; CI: 1.15–3.42; p value: 1.93e−02), but a lower incidence of myelodysplastic syndrome (OR: 0.27, CI: 0.11–0.67, p value: 3.53e−03; Figure 4) for axi-cel. In addition, antibiotics were associated with a higher occurrence of hemophagocytic lymphohistiocytosis (OR: 2.55; CI: 1.49–4.36; p value: 7.77e−04) and tumor lysis syndrome (OR: 2.46; CI: 1.14–5.30; p value: 3.40e−02; Figure 4), but a lower likelihood of recurrent diffuse large B-cell lymphoma (OR: 0.34; CI: 0.14–0.83; p value: 2.01e−02) for tisa-cel. It should be noted that ide-cel was associated with a higher incidence of hemophagocytic lymphohistiocytosis when combined with antibiotics (OR: 6.23; CI: 1.91–20.34; p value: 3.91e−03; Figure 4).

Forest plot comparing antibiotic usage for SPMs at the LLT level across different CAR-T therapies. Anti-CD19 drugs: axi-cel (axicabtagene ciloleucel), tisa-cel (tisagenlecleucel). Anti-BCMA drugs: ide-cel (idecabtagene vicleucel).
TTO analysis of SPMs
In the FAERS database, the TTO refers to the duration between the occurrence of AEs (EVENT_DT) and the initiation of drug usage (START_DT). In our study, we employed cumulative hazard curves to illustrate the event-to-onset characteristics of post-CAR-T therapy SPMs among patients who received antibiotics and those who did not receive antibiotics. Our analysis showed that antibiotics were significantly associated with the TTO among CAR-T recipients (p value: 0.001; Hazard ratio (HR): 1.314; CI: 1.120−1.541; Figure 5). When combined with antibiotics, both anti-CD19 (p value: 3.73e−04; HR: 1.355; CI: 1.146–1.602) and anti-BCMA (p value: 0.310; HR: 1.329; CI: 0.767–2.303) were associated with earlier manifestation of SPMs, although the associations of anti-BCMA was not statistically significant (Figure 5). In the analysis of specific drugs, axi-cel (p value: 2.95e−03; HR: 1.497; CI: 1.147–1.953), tisa-cel (p value: 0.017; HR: 1.341; CI: 1.053–1.707), and cilta-cel (p value: 0.035; HR: 2.346; CI: 1.064–5.172) were associated with an earlier onset of SPMs when combined with antibiotics (Figure 5).

Cumulative hazard curves illustrating the time-to-onset characteristics of post-CAR-T therapy SPMs among patients who received antibiotics versus those who did not. Anti-CD19 drugs: axi-cel (axicabtagene ciloleucel), brexu-cel (brexucabtagene autoleucel), liso-cel (lisocabtagene maraleucel), and tisa-cel (tisagenlecleucel). Anti-BCMA drugs: ide-cel (idecabtagene vicleucel), cilta-cel (ciltacabtagene autoleucel).
Exploring pathways linking antibiotics to SPMs in CAR-T therapy
Given the significant associations between antibiotics and both the incidence and timing of SPMs in patients receiving CAR-T therapy, we further investigated the underlying pathways linking antibiotic use to SPM development in these patients. KEGG pathway analysis revealed that the combination of antibiotics with anti-CD19 therapy was significantly associated with several pathways, including Viral Carcinogenesis, Transcriptional Misregulation in Cancer, PD-L1 Expression and PD-1 Checkpoint Pathway in Cancer, Proteoglycans in Cancer, and Chemical Carcinogenesis—Receptor Activation (Figure 6(a)). For anti-BCMA therapy, the analysis showed associations with the same pathways observed for anti-CD19 therapy, as well as additional pathways, such as MicroRNAs in Cancer, Central Carbon Metabolism in Cancer, and Chemical Carcinogenesis—Reactive Oxygen Species (Figure 6(b)).
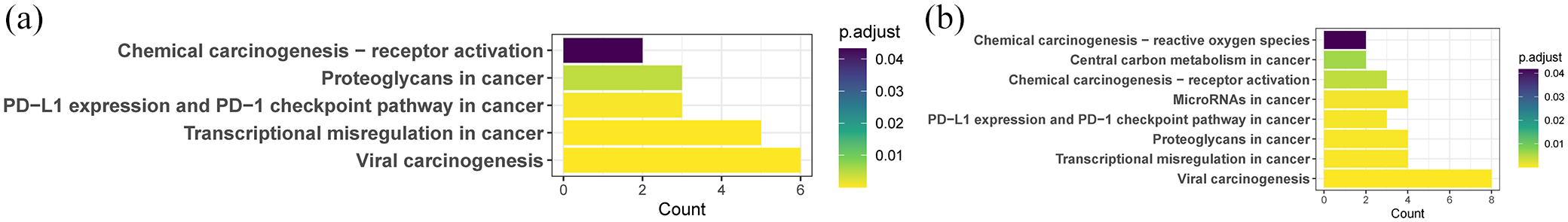
Pathways linking antibiotics to SPMs in CAR-T therapy: (a) anti-CD19 products and (b) anti-BCMA products.
Discussion
CAR-T cell therapy is an innovative and groundbreaking approach in the treatment of cancer. 27 Although it has shown remarkable clinical responses in certain subsets of B-cell leukemia or lymphoma, it faces numerous challenges that impede its therapeutic efficacy in solid tumors and various hematological malignancies. 28 Recently, the FDA has received reports of CAR-positive lymphomas occurring in patients treated with CAR-T cell therapy products.5,6 These concerns emphasize the necessity for more comprehensive characterization of the risk of SPMs following CAR-T cell therapy. In this study, we utilized the FAERS database to assess and quantify the reports of CAR-T cell therapy associated with SPMs.
In this study, we have provided a comprehensive summary of all signals for CAR-T-associated SPMs (Tables 2–4 and Tables S1–S9), which constitutes an invaluable reference for clinicians as it offers crucial insights to support informed decision-making and elevate the standard of patient care. Our analysis includes CAR-T products with varying numbers of reports in the FAERS database. Notably, some products, such as liso-cel (N = 334) and ide-cel (N = 612), have relatively few reports, which may result in less reliable statistical signals. Nonetheless, we aimed to provide a broad overview of signals associated with SPMs across different CAR-T therapies, including those with smaller sample sizes. We acknowledge that smaller datasets may introduce variability, which could affect the reliability of the observed associations. Therefore, caution is warranted when interpreting findings associated with these products. Further studies with larger sample sizes are needed to validate these results and enhance the robustness of the conclusions.
The gut microbiome has emerged as a crucial host factor that could be modulated to enhance responses to immunotherapy. Growing preclinical evidence delineates the differential roles of the gut microbiota, highlighting their potential stimulatory and inhibitory effects on engineered CAR-T cells.9,26,29 Recent studies suggest that antibiotics may influence CAR-T cell function and patient outcomes in complex ways. 9 Therefore, a comprehensive and in-depth characterization of the association between CAR-T therapy-related SPMs and antibiotic treatment is crucial. 30
The role of antibiotics in influencing the incidence of SPMs in CAR-T recipients emerged as a critical factor in our analysis. The relationship between antibiotic use and the incidence of SPMs in patients undergoing CAR-T cell therapy is complex and multifaceted. Our analysis of pharmacovigilance data reveals significant variations in the association between antibiotic use and the risk of SPMs depending on the specific CAR-T therapy administered. While antibiotics were not significantly associated with SPM incidence when all CAR-T drugs were considered collectively, the association between antibiotic use and SPM incidence varied significantly when specific CAR-T products were analyzed. For anti-CD19 CAR-T therapies, antibiotics were associated with a decreased risk of SPMs (OR: 0.85; CI: 0.73–0.99; p value: 0.040). Specifically, antibiotics were linked to a lower incidence of SPMs in patients treated with brexu-cel (OR: 0.42; CI: 0.23–0.77; p value: 0.005), but no significant associations were observed with other anti-CD19 therapies such as axi-cel, liso-cel, or tisa-cel. By contrast, for anti-BCMA CAR-T therapies, antibiotics were linked to an increased risk of SPMs (OR: 2.11; CI: 1.28–3.48; p value: 0.005). Notably, ide-cel was associated with a particularly high likelihood of increased SPM incidence when antibiotics were used (OR: 6.23; CI: 1.91–20.34; p value: 0.004). This suggests that the association between antibiotic use and SPM incidence may vary depending on the type of CAR-T therapy administered. At the LLT level, antibiotics were associated with an increased incidence of several AEs for anti-CD19 CAR-T therapies, including histiocytosis hemophagocytic, B-cell lymphoma, and hemophagocytic lymphohistiocytosis. This observation may align with findings that exposure to antibiotics is associated with worse survival and increased toxicity in patients with B-cell malignancies treated with anti-CD19 drugs. 9 Conversely, antibiotics were associated with a lower occurrence of recurrent diffuse large B-cell lymphoma in this cohort. For anti-BCMA therapies, antibiotics were linked to an increased incidence of myelodysplastic syndrome and hemophagocytic lymphohistiocytosis, reinforcing the variability in associations with specific CAR-T therapies. Clinicians should be aware of these potential associations and consider them when prescribing antibiotics to patients undergoing CAR-T therapy. Further research is needed to clarify the mechanisms underlying these associations and to develop strategies to mitigate any adverse outcomes.
TTO analysis provided significant insights into the association between antibiotics and the manifestation of SPMs in CAR-T cell therapy. Our findings demonstrate that antibiotics were significantly associated with the TTO of SPMs in CAR-T recipients. Specifically, antibiotics were associated with an earlier onset of SPMs across the board, with an HR of 1.314 (CI: 1.120–1.541; p value: 0.001). This suggests that patients receiving antibiotics may experience SPMs sooner compared to those not receiving antibiotics, pointing to a potential association with an accelerated manifestation of these malignancies. The earlier onset of SPMs associated with antibiotics underscores the need for healthcare providers to carefully consider the use of antibiotics in patients undergoing CAR-T cell therapy. This association may warrant closer monitoring and potentially revised treatment strategies to address the accelerated risk of SPMs.
The exploration of the underlying molecular pathways linking antibiotics to SPMs in CAR-T therapy reveals valuable insights into the complex interplay between antimicrobial agents and CAR-T therapy. A notable finding from our analysis is the strong association between antibiotics and the Viral Carcinogenesis pathway in both anti-CD19 and anti-BCMA therapies. Viral infections have been implicated in the development of various cancers. 31 The use of antibiotics could potentially disrupt the immune system’s ability to control viral infections, thereby facilitating the progression of viral-induced malignancies. In addition, the Transcriptional Misregulation in Cancer pathway, 32 associated with antibiotic use, may lead to the activation of oncogenes or the suppression of tumor suppressor genes, thereby enhancing the risk of SPMs. Our findings also highlight significant associations with the PD-L1 Expression and the PD-1 Checkpoint Pathway in Cancer. The PD-1/PD-L1 axis plays a critical role in immune evasion by tumors, and its modulation can affect the success of immune therapies. The combination of antibiotics with CAR-T therapy could potentially alter the expression of these immune checkpoint molecules, either enhancing or diminishing the immune response. The potential for antibiotics to influence the immune system in this manner may explain the observed alterations in SPM incidence and timing.33,34 As PD-1/PD-L1 inhibition has become a cornerstone of cancer immunotherapy, further investigation into how antibiotics affect this pathway is essential to understanding their impact on CAR-T therapy outcomes.
Limitations
This study acknowledges several limitations. First, the reliance on the FAERS database introduces potential biases inherent in a voluntary reporting system, including fragmented and unsystematic data on patients, diseases, treatments, and outcomes, which limits the ability to fully interpret the data. In addition, underreporting or overreporting of AEs further contributes to bias. Second, while disproportionality analysis was used to identify potential associations, it cannot establish causality. The observed signals may be influenced by unaccounted confounders, including concurrent therapies and underlying conditions. Further studies with more comprehensive clinical data and controlled designs are needed to confirm these findings. Third, TTO analysis assumes the accurate recording of event dates, and reporting delays or inaccuracies in the FAERS database may skew the TTO findings. Further studies with more comprehensive clinical data and controlled designs are needed to validate the TTO results. Lastly, we were unable to differentiate between the prophylactic and therapeutic use of antibiotics, preventing us from evaluating their impacts on the incidence of SPMs.
Conclusion
In summary, our analysis underscores the significant association between antibiotics and both the incidence and timing of SPMs in patients undergoing CAR-T therapy. Specifically, antibiotics were associated with a lower risk of SPMs in patients receiving anti-CD19 therapies, while they were linked to a higher risk in those treated with anti-BCMA therapies. In addition, antibiotics were linked to an earlier onset of SPMs across all CAR-T therapies. These findings highlight the importance of personalized monitoring strategies and emphasize the need for further research to better understand the underlying mechanisms of these interactions.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251340866 – Supplemental material for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies
Supplemental material, sj-docx-1-taw-10.1177_20420986251340866 for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies by Yun Peng, Yuxuan Song, Jiaxing Lin, Caipeng Qin, Yiqing Du and Tao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-10-taw-10.1177_20420986251340866 – Supplemental material for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies
Supplemental material, sj-xlsx-10-taw-10.1177_20420986251340866 for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies by Yun Peng, Yuxuan Song, Jiaxing Lin, Caipeng Qin, Yiqing Du and Tao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-2-taw-10.1177_20420986251340866 – Supplemental material for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies
Supplemental material, sj-xlsx-2-taw-10.1177_20420986251340866 for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies by Yun Peng, Yuxuan Song, Jiaxing Lin, Caipeng Qin, Yiqing Du and Tao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-3-taw-10.1177_20420986251340866 – Supplemental material for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies
Supplemental material, sj-xlsx-3-taw-10.1177_20420986251340866 for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies by Yun Peng, Yuxuan Song, Jiaxing Lin, Caipeng Qin, Yiqing Du and Tao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-4-taw-10.1177_20420986251340866 – Supplemental material for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies
Supplemental material, sj-xlsx-4-taw-10.1177_20420986251340866 for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies by Yun Peng, Yuxuan Song, Jiaxing Lin, Caipeng Qin, Yiqing Du and Tao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-5-taw-10.1177_20420986251340866 – Supplemental material for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies
Supplemental material, sj-xlsx-5-taw-10.1177_20420986251340866 for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies by Yun Peng, Yuxuan Song, Jiaxing Lin, Caipeng Qin, Yiqing Du and Tao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-6-taw-10.1177_20420986251340866 – Supplemental material for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies
Supplemental material, sj-xlsx-6-taw-10.1177_20420986251340866 for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies by Yun Peng, Yuxuan Song, Jiaxing Lin, Caipeng Qin, Yiqing Du and Tao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-7-taw-10.1177_20420986251340866 – Supplemental material for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies
Supplemental material, sj-xlsx-7-taw-10.1177_20420986251340866 for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies by Yun Peng, Yuxuan Song, Jiaxing Lin, Caipeng Qin, Yiqing Du and Tao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-8-taw-10.1177_20420986251340866 – Supplemental material for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies
Supplemental material, sj-xlsx-8-taw-10.1177_20420986251340866 for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies by Yun Peng, Yuxuan Song, Jiaxing Lin, Caipeng Qin, Yiqing Du and Tao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-9-taw-10.1177_20420986251340866 – Supplemental material for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies
Supplemental material, sj-xlsx-9-taw-10.1177_20420986251340866 for Pharmacovigilance analysis of secondary primary malignancies and antibiotic interactions in CAR-T cell therapies by Yun Peng, Yuxuan Song, Jiaxing Lin, Caipeng Qin, Yiqing Du and Tao Xu in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
