Abstract
Background:
Fever is a common postoperative complication, typically caused by aseptic inflammation or infection. However, drug-induced fever (DIF) is an underdiagnosed etiology that should be considered in the differential diagnosis, especially in patients receiving complex medication regimens post-surgery.
Objectives:
This systematic review aims to assess the current literature on DIF in post-surgical patients to improve diagnostic accuracy and patient care.
Design:
Systematic review of case reports and case series.
Data sources and methods:
This systematic review was conducted following the PRISMA 2020 guidelines. We included case reports and series involving post-surgical patients with fever linked to drug administration. Studies were retrieved from the PubMed, Scopus, Embase, and Web of Science databases, as well as gray literature sources. Quality and bias were assessed using the Joanna Briggs Institute (JBI) critical appraisal tools.
Results:
A total of 16 studies (14 case reports and 2 case series) involving 23 patients were included. The most frequently implicated drugs were propofol, morphine, and cephalosporins. Fever onset ranged from immediate postoperative to several days later, with varied patterns, including intermittent, remittent, and continuous fever. The majority of patients recovered after drug discontinuation, although two cases resulted in death.
Conclusion:
DIF is one of the causes of postoperative fever. Early identification and cessation of the offending drug are crucial for resolving the fever and preventing severe complications. Clinicians must remain vigilant in diagnosing DIF to improve patient outcomes post-surgery.
Plain language summary
• Why was the study done? The study aimed to assess the current literature on drug-induced fever in postsurgical patients, with the goal of improving diagnostic accuracy and patient care. The study sought to raise awareness about this often-overlooked complication.
• What did the researchers do? The researchers systematically reviewed case reports and case series that involved drug-induced fever in postsurgical patients. They searched several databases, including PubMed, Scopus, and Web of Science, and assessed the quality of the studies included in the review.
• What did the researchers find? The review included 16 studies, with 23 patients. The most frequently implicated drugs in causing fever were propofol, morphine, and cephalosporins. In most cases, the fever resolved after stopping the offending drug. However, a few cases resulted in severe outcomes, including death.
• What do the findings mean? The findings underscore the importance of considering drug-induced fever as part of the differential diagnosis for postoperative fever. Identifying and discontinuing the drug causing the fever can help resolve the issue and prevent severe complications.
Keywords
Introduction
Fever is a common complication following surgery, occurring in nearly half of patients across various operations. 1 While aseptic causes due to inflammatory responses and infectious causes are the most frequent explanation, drug-induced fever (DIF) represents a significant, yet often underdiagnosed etiology, and is primarily considered a diagnosis of exclusion.2,3 Although the exact mechanism of DIF is not well established, previous studies have suggested it may involve a type III hypersensitivity reaction that interferes with peripheral heat dissipation, triggering immune responses.4,5 DIF typically manifests 7–10 days after drug administration; however, there are no definitive criteria regarding the pattern or timeline of the fever, which can vary from a few hours to several months. 6 The diagnosis of DIF is generally confirmed when the patient’s body temperature decreases following discontinuation of the suspected drug. 7 This condition is particularly relevant in post-surgical patients, who often receive a complex regimen of medications, including antibiotics, anesthetics, and analgesics—all of which have been implicated as potential culprits.6,8 Despite its clinical significance, DIF remains under-recognized and underreported in the medical literature. 9 This underdiagnosis is partly due to the nonspecific presentation of DIF and the absence of specific diagnostic tests to confirm it.
Previous case reports and case series have identified various drugs associated with post-surgical fever and described the clinical features and management of such cases. However, no comprehensive review has been conducted to clarify the patterns, common involved drugs, and outcomes of DIF in post-surgical patients.
This systematic review aims to comprehensively assess the current literature on DIF in post-surgical patients. By doing so, we seek to provide clinicians with a clearer understanding of the characteristics of DIF, enhance diagnostic accuracy, and improve patient care in the postoperative setting.
Material and methods
Eligibility criteria
We conducted a systematic review registered on PROSPERO (ID: CRD42024508209). One amendment was made after registration to correct a wording error in the original review question, which had led to misinterpretation. The revised question now accurately focuses on the clinical characteristics, implicated drugs, time to fever onset, fever patterns, management strategies, and outcomes of DIF in post-surgical patients. This amendment was necessary because estimating prevalence and incidence is not feasible based on case reports and case series. All modifications made after protocol registration have been clearly identified and justified within the manuscript to ensure transparency. This study was performed in accordance with PRISMA 2020 guidelines 10 (Figure 1). The inclusion criteria were as follows: (A) case report or case series, (B) a history of surgery within the past 4 weeks, (C) immediate postoperative fever (within 24 h of surgery) or late postoperative fever (4–30 days post-surgery), (D) initiation of new drugs either before or after surgery (within 48 h before or after surgery), (E) potential symptoms accompanying fever such as rash or, in some cases, hypotension, 11 (F) fever resolving after discontinuation of the suspected drug, and (G) no other evident cause for the fever, as determined by the original case report authors at the time of patient care based on clinical evaluations and diagnostic findings.

The PRISMA flow diagram illustrating the study selection process.
The exclusion criteria included: (A) studies that were not case reports or case series, (B) no history of surgery, (C) absence of a suspicious new drug, (D) fever did not resolve after drug discontinuation, or (E) presence of other identifiable causes for the fever. Finally, all included articles were critically appraised using the Joanna Briggs Institute (JBI) critical appraisal tools (Joanna Briggs Institute, Adelaide, Australia).
Information sources
A systematic search was conducted using PubMed, Scopus, Embase, and Web of Science from inception through December 2023. To ensure comprehensive data collection and minimize publication bias, gray literature sources (e.g., allconferences.com and conferencealerts.com) were also searched, in line with PRISMA 2020 and Cochrane Handbook recommendations. In addition, the reference lists of the included articles were reviewed, and related articles identified through miscellaneous searches were included as well.
Search strategy
MeSH and non-MeSH keywords used to find related studies included: (1) “drug fever” OR “febrile drug reaction” OR “case report of drug fever”; AND (2) “surgical patients” OR “surgery” OR “postoperative fever” OR “fever after surgery.” The full search strategies for all databases are available in Supplemental File 1: S1.
Selection process
The titles and abstracts of the retrieved articles were independently reviewed twice by three researchers (F.A., M.A., and S.T.). In cases of disagreements, the articles were reassessed by other authors (F.A. and B.S.), with B.S. taking the lead in resolving conflicts and making final decisions. No automation tools were used to include or exclude records during this process. For the final inclusion and exclusion stage, the researchers independently examined the full texts of the articles that passed the initial screening, conducting the review twice. Disagreement was resolved by consulting other authors (F.A. and B.S.), with B.S. taking the lead in resolving conflicts and making final decisions. The full texts of the final studies were thoroughly examined to evaluate their quality, assess the risk of bias, and extract the necessary data. Three researchers independently extracted data from the eligible studies, with any discrepancies being resolved by other researchers (F.A. and B.S.). No automation tools were used for data extraction.
Data extraction and risk of bias assessment
We developed a data extraction form to collect patient information (e.g., age, gender, type of surgery, and history of allergies), details of suspected drugs (e.g., drug name, pharmacological category, dosage, and method of administration), fever patterns (intermittent, remittent, continuous/sustained, hectic, and relapsing), fever onset (immediate: within hours of surgery; early: 0–3 days post-surgery; late: 4–30 days post-surgery; delayed: >30 days post-surgery), and fever grade (low grade: 38.0°C–39.0°C; moderate: 39.1°C–40.0°C; high grade: 40.1°C–41.0°C; hyperpyrexia: >41.1°C), other signs and symptoms (e.g., onset, severity, pattern, clinical, and laboratory data), and treatment details (e.g., treatment plan, patient outcomes, and re-challenge of suspected drugs).
Data extraction from the articles was carried out by four researchers (F.A., M.A., S.T., and S.J.) using a standardized checklist. In cases of unresolved disagreements, another researcher (B.S.) made the final decision. The extracted data were then independently rechecked by two researchers (B.S. and A.A.).
To evaluate the risk of bias and study quality, we used the 2020 version of the JBI critical appraisal tools for assessing case reports 12 and case series. 13 Case reports had to meet at least 6 out of 8 criteria, and case series needed to fulfill 8 out of 10 items for inclusion in this review. All included articles were reassessed twice for quality by M.A. and S.T.
Results
The initial systematic search identified a total of 3390 records from the PubMed, Scopus, Embase, and Web of Science databases. The searches were conducted between December 30, 2023, and January 7, 2024. In addition, 36 more records were retrieved from gray literature sources. After removing 244 duplicate records, 3182 unique records remained for screening. Titles and abstracts were reviewed, leading to the exclusion of 3075 records for not meeting the inclusion criteria. Full texts of 83 out of the remaining 107 articles were assessed for eligibility. Ultimately, 16 articles were included in this study, while 70 records were excluded for the following reasons: duplication (n = 3), not being a case report or case series (n = 4), not involving postoperative cases (n = 15), not being a DIF (n = 31), and other reasons (n = 14). In addition, the full texts of 24 articles were not accessible (Figure 1).
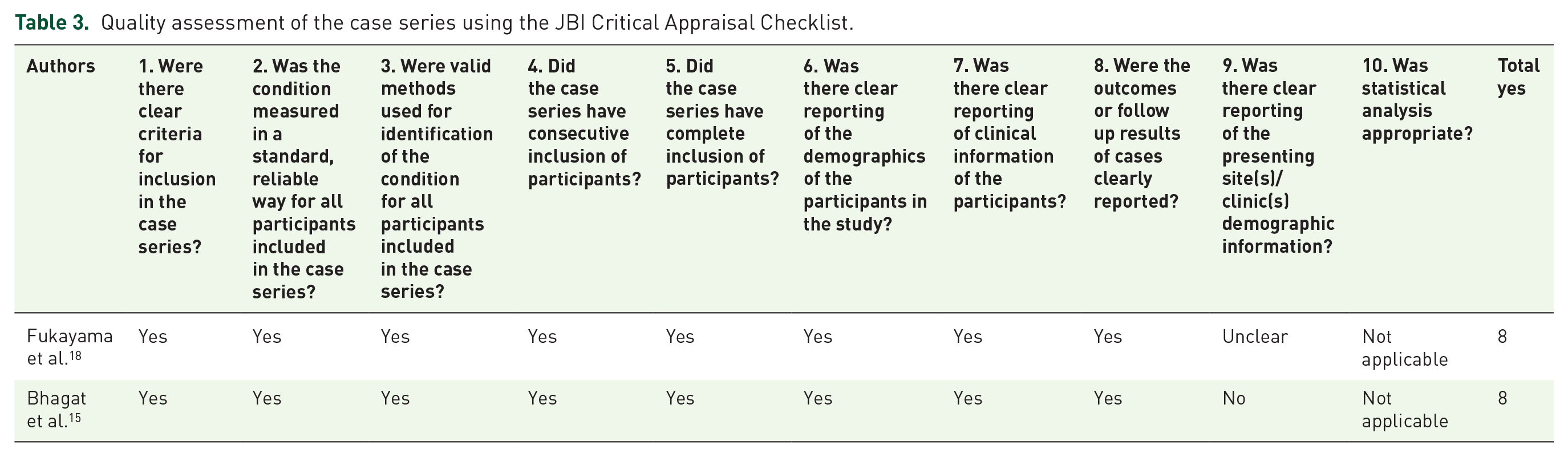
The included studies consisted of 2 case series and 14 case reports, with a total of 23 subjects who developed postoperative DIF (Table 1). The articles were evaluated using JBI criteria. Seven (50%) case reports and both case series met 8 out of the required criteria (Tables 2 and 3).
Demographic, clinical, and laboratory characteristics of patients with DIF.

AC, anticoagulant; CABG, coronary artery bypass grafting; CK, creatine kinase; CPK, creatine phosphokinase; CRP, C-reactive protein; D/C, discontinuation; DIF, drug-induced fever; ECMO, extracorporeal membrane oxygenation; ESR, erythrocyte sedimentation rate; F, female; GA, general anesthetic; HTN, hypertension; ICP, intracranial pressure; inf., infusion; IV, intravenous; LMWH, low molecular weight heparin; M, male; ORIF, open reduction with internal fixation, PCT, procalcitonin; PO, per os (oral administration); SC, subcutaneous.
Onset of fever based on: Immediate: within hours of the suspected drug initiation, Early: 0–3 days after the suspected drug initiation, Late: 4–30 days after the suspected drug initiation, Delayed: more than 30 days after the suspected drug initiation.
Grade of fever based on: Low grade: 38°C–39°C, Moderate: 39.1°C–40°C, High grade: 40.1°C–41°C, Hyperpyrexia: more than 41.1°C.
Fever pattern based on: Intermittent, Remittent, Continuous or sustained, Hectic, Relapsing.
Quality assessment of the case reports using the JBI Critical Appraisal Checklist.

Quality assessment of the case series using the JBI Critical Appraisal Checklist.
The drugs associated with the development of DIF in postoperative patients are listed according to their frequency. Propofol was the most commonly reported, linked to seven cases. Enoxaparin and dexmedetomidine were each involved in two cases, followed by cefazolin and piperacillin/tazobactam, which were also implicated in two cases, with one of those involving a combination of cefuroxime. Heparin, morphine, procainamide, and sulprostone were each implicated in one case.
The median age of the patients was 40 years, with age ranging from 3 to 68 years. In one case, the patient’s age was not mentioned. 13 Among the 23 reported cases, the prevalence of male patients (56.5%) was higher than female patients (43.5%). The youngest patient was a 3-year-old female who developed DIF after receiving morphine following an elective exploratory laparotomy with tumor excision. 14 The oldest patient was a 68-year-old male who received dexmedetomidine after a coronary artery bypass graft (CABG) operation. 15
Different fever patterns were observed among the studies, including intermittent fever in four cases (25%),8,13,16,17 remittent fever in five cases (31.2%),15,17–20 hectic fever in three cases (18.7%),2,17,21 and continuous fever in four cases (25%).17,22–24 There was no dominant pattern in the remaining cases. Even among cases where the suspected drug was the same, the fever pattern varied. The pattern was not clear in seven cases.14,25,26
The onset of fever following the initiation of the suspected drug varied across cases. Ten patients (45.4%) experienced immediate fever onset (within hours after drug administration),14,15,18 19,23 eight patients (36.3%) had early onset (0–3 days post-administration),15,17,22,25 and four patients (18.1%) developed fever later than 3 days after drug initiation.2,8,16,20,21,24,27 Fever onset was not reported in one case. 26
The fever grade ranged from low grade (38°C–39°C) in six cases (27.3%),8,16,17,20,24 to moderate grade in seven cases (31.8%),13,14,17,18,21 and to high-grade fever in seven other cases (31.8%).2,14,15,19,23 Hyperpyrexia was also reported in two cases.22,26 Additional clinical data, such as rash, 8 chills,20,22 tachycardia,22,25,26 bradycardia,2,25 and hypertension, 22 were observed in some patients who developed DIF.
The patients underwent various types of surgeries, including 7 emergencies and 16 elective operations. They received different classes of medications during the perioperative period, including anticoagulants,2,16,24 sedatives and anesthetics15,17,18,23,25,26 antibiotics,8,13,19,21 prostaglandins, 22 antiarrhythmics, 20 and opioid analgesics, 14 all of which were suspected to have induced post-surgical fever.
The management of DIF primarily involved discontinuation of the suspected drug in most cases, except for one case where morphine was suspected to have induced the fever. 14 Supportive care, including the administration of antipyretics such as acetaminophen, was provided to alleviate symptoms in various cases.8,13,14 Re-challenge was attempted in two cases (with dexmedetomidine and procainamide), and fever recurred upon re-challenge.20,23 No further complications were observed apart from chills and rigors following procainamide reinitiation. 21 In both cases, the fever resolved again after discontinuation of the suspected drug.21,24 Most patients recovered from the fever after discontinuation of the drug; however, two cases resulted in patient death, both involving propofol infusion,25,26 consistent with Propofol Infusion Syndrome (PRIS).
Discussion
This systematic review aims to assess reported cases and case series of postoperative DIF in the literature. To date, no comprehensive analysis of these reports on postoperative DIF has been published. This study seeks to analyze these reports to identify the characteristics, patterns, and commonly implicated drugs associated with DIF, and to evaluate patient outcomes to improve diagnostic and care methods in postoperative settings. We examined 23 cases from 16 studies in which patients developed drug fever after surgery. Only methodologically high-quality studies were included in this review.
While antibiotics are often cited as the most common cause of DIF and are frequently linked to nosocomial fever,28–30 this study found that propofol (seven cases), morphine (five cases), cephalosporins (cefazolin and cefuroxime), and penicillins (piperacillin and tazobactam) were the most frequently reported medications causing DIF, accounting for nearly 70% of reported cases. This highlights the need for caution when using these medications in postoperative patients.8,14,15,18–20,22,26,27
A detailed review of all propofol-related cases revealed that two cases demonstrated clinical features of a propofol-related infusion syndrome (PRIS), a condition characterized by rapid onset of fever, metabolic acidosis, and multiorgan failure. In contrast, five other cases—reported in two separate studies—were classified as DIF rather than PRIS, as they did not exhibit metabolic acidosis, rhabdomyolysis, hemodynamic instability, or other hallmark criteria of PRIS. This distinction is crucial for accurate diagnosis and management of drug-related fever. Further research is warranted to better delineate the boundaries between these two conditions.4,17,18,25,26,31,32
Various fever patterns were reported in nearly equal proportions, although remittent fever was more common than other patterns. A narrative review published in 2023 similarly noted that no specific patterns were observed for DIF, with drug fever potentially presenting in various forms. Therefore, fever patterns cannot be considered definitive indicators for diagnosing DIF. 4 Additionally, fever onset occurred immediately (less than 12 h after the drug initiation) or early (between 12 h and 3 days after the drug initiation) in over 78.2% of cases (18 cases), which may be diagnostically useful. However, in some cases, DIF was observed later. Fever severity ranged from low grade to hyperpyrexia, with hyperpyrexia occurring in less than 10% of cases (two cases), specifically due to propofol and sulprostone.23,27
Re-challenge is considered the gold standard for diagnosing DIF. However, due to patient’s condition and the nature of the reaction, this method is not always feasible. In some cases, re-challenge may not be reasonable, and the risks and benefits should be carefully evaluated before proceeding. Moreover, re-challenge should not be performed solely to confirm the suspected diagnosis of drug fever. 6 In this study, re-challenge was attempted in only two cases, both of which resulted in a recurrence of fever.21,24 In another case, where morphine was not discontinued, a similar outcome was reported. 15 Discontinuation of the suspected medication serves as both a diagnostic and therapeutic measure. 4
Although laboratory findings may provide supportive evidence for the diagnosis of DIF, they cannot be used for a definitive diagnosis due to their variability and nonspecificity. Leukocytosis was reported in six cases. Other studies have shown that leukocytosis, with or without a left shift, may be observed in DIF patients. Importantly, if fever is accompanied by leukocytosis, infection must be ruled out.6,33,34 An increase in C-reactive protein (CRP) was reported in 10 cases. Research indicates that elevated CRP levels are commonly observed in patients with drug fever.4,35,36 In a case series, 10 out of 11 patients with antibiotic-induced fever had elevated CRP levels.4,35 A mild increase in erythrocyte sedimentation rate (ESR) may also be observed in DIF patients, with ESR values between 40 and 60 mm/h being more prevalent, although higher levels can occur.5,6,33,37 In this study, an elevated ESR was noted in three cases, with values exceeding 60 mm/h in two cases. Other laboratory findings, such as elevated liver enzyme levels, renal failure, and anemia, have also been observed in DIF patients.4,38,39
The most frequently reported clinical symptoms include tachycardia and chills, with bradycardia, rash, and hypertension also noted. Several case series on antibiotic-induced fever have reported that relative bradycardia is present in 83%–100% of cases,40,41 though in one case series, it was observed in only 11% of cases. 42 It should be noted that the definition of relative bradycardia varied among the studies. 4
According to the reports, the male-to-female ratio of this complication is approximately equal, although the number of male patients is higher. In addition, the age range of patients varied from pediatric to geriatric. These data suggest no significant relationship between the occurrence of DIF and the patient’s age or gender. However, the number of reported cases is insufficient to draw a definitive conclusion.
The prognosis for DIF is generally favorable, with most patients recovering after the offending medication is identified and discontinued, along with appropriate supportive care. However, two cases associated with PRIS resulted in death.26,27 A recent case series reported that 97% of patients (out of 167) with DIF recovered without adverse consequences,4,30 suggesting that the likelihood of death due to drug fever in postoperative patients is very low. Despite the lack of reported allergies or drug sensitivities in some patients, a thorough review of drug history and screening for drug sensitivity is crucial to minimizing the risk of DIF and enhancing patient safety.
The mechanisms of drug fever are not well understood, and multiple pathways may be involved depending on the structural and functional properties of the causative medication.6,29,30 While hypersensitivity reactions resembling an immune response are the most prevalent mechanism,4,6,33,34,43–45 drugs can also cause fever through other pathways, such as altering central temperature regulation, interfering with peripheral heat loss, causing direct tissue injury, or introducing exogenous substances.6,29,46 The schematic summary of DIF in postoperative patients is presented in Figure 2.

The schematic summary of DIF in postoperative patients.
This study emphasizes the importance of distinguishing DIF from other postoperative syndromes. A deeper understanding of the pathophysiological mechanisms underlying DIF is necessary to prevent misdiagnosis and ensure appropriate treatment strategies. Future research should focus on exploring the relationship between specific medications, including propofol, and the development of fever. In addition, the development of predictive models for early detection of DIF could improve clinical decision-making. Large-scale studies, including cohort studies and randomized controlled trials (RCTs), are needed to establish robust evidence on the incidence, risk factors, and long-term implications of DIF in clinical practice. Further investigations should also aim to differentiate DIF from other fever syndromes, particularly in postoperative settings where overlapping clinical presentations may complicate diagnosis.
Study limitations: This study had several limitations. First, only studies with available full texts were included, potentially excluding cases of postoperative drug fever where full texts were not accessible. Second, while there is an inherent risk of bias in data extraction, we employed the JBI tools to assess the methodological quality of the included studies, ensuring a rigorous evaluation process. It is important to note that these tools are designed to appraise study quality rather than directly govern the data extraction process. Although subjectivity in data extraction cannot be entirely eliminated, we applied a systematic and standardized approach to minimize potential biases in data collection and analysis.12,13 Third, heterogeneity in the included studies could be an issue, as the studies vary significantly in terms of fever onset and surgical contexts. This variability complicates the comparison of outcomes and makes it harder to draw consistent conclusions across the different cases. Fourth, the limited scope of evidence is a concern, as the study only includes case reports and case series, which are observational in nature and do not provide the high-level evidence that larger studies, such as RCTs, would offer.
Conclusion
DIF is a significant but often-overlooked cause of postoperative fever. This review highlights the diverse presentation of DIF and its association with multiple drugs commonly used in the post-surgical setting, including opioids, antibiotics, and sedatives. Early recognition and discontinuation of the offending drug are crucial for resolution, though some cases can lead to severe complications or even death. Given the risks, clinicians must remain vigilant in considering DIF as a differential diagnosis, particularly in patients with unexplained fever following surgery. Enhanced awareness and a structured approach to diagnosing and managing DIF could improve patient outcomes in the postoperative period.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251335825 – Supplemental material for Drug-induced fever in post-surgical patients: a systematic review of case reports
Supplemental material, sj-docx-1-taw-10.1177_20420986251335825 for Drug-induced fever in post-surgical patients: a systematic review of case reports by Fatemeh Afra, Mona Aboutalebzadeh, Soheila Tayefeh, Sepide Javankiani, Bita Shahrami and Amir Ahmad Arabzadeh in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986251335825 – Supplemental material for Drug-induced fever in post-surgical patients: a systematic review of case reports
Supplemental material, sj-docx-2-taw-10.1177_20420986251335825 for Drug-induced fever in post-surgical patients: a systematic review of case reports by Fatemeh Afra, Mona Aboutalebzadeh, Soheila Tayefeh, Sepide Javankiani, Bita Shahrami and Amir Ahmad Arabzadeh in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
