Abstract
Background:
The risk factors related to the adverse drug reactions (ADRs) of anlotinib have been rarely investigated. In addition, a corresponding risk prediction model has not been established in China pertaining to anlotinib-related ADRs.
Objectives:
To manage ADRs more efficiently and improve the prognosis of patients administered anlotinib.
Design:
A retrospective analysis was conducted using the medical records of patients diagnosed with cancer who were administered anlotinib after hospitalization between January 1, 2020, and December 31, 2023.
Methods:
We performed a combination of univariate analysis and multivariate binary logistic regression analysis to identify significant factors that can accurately predict ADRs. Model fitting was performed using forward selection. The accuracy of the prediction model was expressed as the area under the receiver operating characteristic curve (AUC). The final ADR risk model was validated.
Results:
In this study, 300 patients who were administered anlotinib were included. Among them, 238 (79.33%) patients experienced at least one ADR. Diagnosis, combination treatment, distant metastasis, treatment lines, and cumulative dose were independent risk factors for the ADRs of anlotinib. The AUC and the concordance index of the nomogram constructed from the above five factors were 0.790 and 0.789, respectively. The results of the Hosmer–Lemeshow test showed that the model was a good fit (p = 0.811). In addition, the decision curve analysis demonstrated a significantly higher net benefit of the model. The external validation indicated that the prediction nomogram was reliable.
Conclusion:
We developed and validated a simple model to use the ADR risk score in patients who were administered anlotinib. This risk prediction model was well-calibrated and discriminative. It can be used as a reference for clinical decision-making. It has clinical significance for preventing ADRs, improving the prognosis of patients, and providing support for the rational use of drugs.
Plain language summary
Why was the study done? With the widespread use of anlotinib along with other anticancer regimens, the associated ADRs that occur may interrupt treatment and cause patients to refuse further treatment. The risk factors related to the ADRs of anlotinib has been rarely investigated. Additionally, corresponding risk prediction model has not been established in China pertaining to anlotinib-related ADRs. Therefore, the safety of anlotinib in patients with cancer needs to be paid special attention in clinical practice.
What did the researchers do? We conducted a retrospective study using data from hospitalized patients from January 2020 to December 2023 by strict inclusion and exclusion criteria. The patients were categorized into training cohort and validation cohort. The results of the study were analyzed using the R software (version 4.3.2).
What did the researchers find? The study finally included 300 patients. We developed a model to predict the occurrence of anlotinib-related ADRs. Among them, diagnosis, distant metastasis, combination therapy, line of therapy, and cumulative dose were found to be the main predictors of anlotinib-related ADRs. External validation data also confirmed the fitting and predictive capabilities of the model.
What do the findings mean? This study has found that the model is suitable for application by doctors and clinical pharmacists to quickly assess the risk of anlotinib-related ADRs. Such a real-world model can not only be used as a reliable tool for the early identification of ADRs associated with the application of anlotinib but also facilitate early intervention, treatment, and care. Thus, it can improve the quality of life of patients.
Introduction
Anlotinib is a highly potent and multi-targeting tyrosine kinase inhibitor. It exerts strong antitumor effects in different types of tumors by inhibiting angiogenic and proliferative signaling.1–4 In the clinical trial ALTER303, anlotinib-related adverse drug reactions (ADRs) had an incidence of more than 10% and included hypertension, fatigue, hand–foot syndrome, hypertriglyceridemia, proteinuria, diarrhea, loss of appetite, high levels of blood thyrotropin, hypercholesterolemia, hypothyroidism, etc. 5 Most ADRs occurred within 50 days (within two cycles of treatment). However, the exact mechanism of occurrence was uncertain. 6 Due to the high selectivity of patients and a single drug used in clinical studies, certain differences existed between study participants and actual clinical users.7–12 Either the sample size of the real-world studies on the safety of anlotinib was small or the studies conducted involved a single ADR.13,14
The ADRs obviously place extreme pressure on clinicians, with clinicians having to treat the consequences of ADRs in addition to the primary pathology. And ADRs have brought a huge burden to patients both physically and psychologically. Early identification and intervention of ADRs are the key to effective treatment. The risk factors related to the ADRs of anlotinib have been rarely investigated. In addition, a risk prediction model has not been established in China pertaining to anlotinib-related ADRs. The factors that influence anlotinib-related ADRs in multiple tumor species in real-world scenarios require clinical research and attention.
Our study aims to develop and validate (using a three-step process) a risk prediction model for ADRs. We retrospectively analyzed the data of patients in hospitals to identify the factors that influence ADRs. Then the concise and simple-to-use risk prediction model was established to quantitatively predict the probability of ADRs. Finally, it was externally validated. Such a real-world model can not only be used as a reliable tool for the early identification of ADRs associated with the application of anlotinib but also facilitate early intervention, treatment, and care to improve the quality of life of patients.
Methods
We conducted a retrospective study using data from hospitalized patients. The study was reported as per the TRIPOD checklist. 15 In total, 321 patients with cancer who were administered anlotinib in Minhang Branch, Fudan University Shanghai Cancer Center, Shanghai, China, between January 1, 2020, and December 31, 2023, were included; 21 did not meet the inclusion criteria and have incomplete records. Finally, 300 patients were enrolled in this study (Figure 1). The training cohort consisted of 212 patients, whereas the validation cohort consisted of 88 patients. The flowchart of the study design is shown in Figure 1.

Flowchart of the study.
The inclusion criteria were as follows: (1) Pathological diagnosis of the tumor was performed according to the latest clinical guidelines. (2) The minimum duration of treatment was two cycles, which enabled adequate observation and data collection. (3) Availability of complete clinical data for each patient, ensuring a comprehensive analysis and robust findings of the study.
The exclusion criteria were as follows: (1) Anlotinib was not administered during the treatment cycle. (2) Treatment cycles were less than two cycles and follow-up records were lacking. (3) The records of the patients were incomplete.
Data collection
All data sets of the study were obtained through a combination of a review of the medical notes. Thus, the candidate predictor variables considered for the risk score were included the general conditions of patients (age, gender, BMI, clinical stage, Eastern Cooperative Oncology Group-Performance Status (ECOG-PS), base disease, distal metastasis, diagnosis, and degree of differentiation), clinical treatment information, and laboratory-related indicators (combined treatment, treatment line, dose, prior treatment, prior surgery, prior radiotherapy, previous Epidermal Growth Factor Receptor-Tyrosine Kinase Inhibitors (EGFR-TKI) treatment, cumulated dose, and ADRs).
Identification and documentation of ADRs
First, the Trigger Tool (IHI Global Trigger Tool) methodology was used to detect the occurrence of ADRs. Subsequently, a physician and two senior pharmacists reviewed each event and determined the causal relationship between drug intake and the suspected ADRs using Naranjo’s adverse drug reaction criteria. Finally, the severity level of ADRs was classified by CTCAE 5.0.
Definitions
The diagnosis was reached according to the pathological diagnosis records of the medical record system. Age was categorized as >65 years old, between 18 and 65 years old. BMI was calculated and classified as >18.5 and ⩽18.5. The stages of the disease corresponded to level IV or below. ECOG-PS was classified as >1 and ⩽1. The distal metastasis, prior surgery, prior radiotherapy, and previous EGFR-TKI treatment were categorized as yes or no. The dose of anlotinib was 10 or 12 mg. Treatment line was assessed as first, second, third, and multi-line. The degree of differentiation was assessed as low, middle, and high. Prior treatment included chemotherapy, chemotherapy combined targeting, chemotherapy combined targeting and immunization, and chemotherapy combined immunization. Combined treatment included chemotherapy combined targeting, chemotherapy combined targeting and immunization, targeted combination immunization, targeted combined radiotherapy, and target. The cumulated dose was calculated and classified as ⩽336 and >336 mg (two cycles).
Statistical analysis
The sample size was estimated using PASS version 15.0 (NCSS LLC., Kaysville, UT, USA). All available samples were utilized in this study. The whole data set was divided into training and validation cohorts at the ratio of 7:3 according to a random number table. All data are represented as categorical variables. All statistical analyses were performed using the R software (version 4.3.2, R Foundation for Statistical Computing, Vienna, Austria). The statistical tests conducted in this study were two-sided and p < 0.05 was considered significant.
The process of developing the risk prediction model consisted of three steps. In the first stage, a univariate analysis was performed to generate a framework of variables. The statistically significant variables (p < 0.2) were used in the next step of the multivariate analysis. In the second step, the multivariate analysis was conducted using binary logistic regression analysis. The statistically significant variables (p < 0.05) were used in the final risk model. Finally, the selected independent risk factors and clinically significant factors were included to build a nomogram of the ADR risk prediction model. Discrimination of the model was further assessed by an analysis of the area under the operator curve. The overall fit of the ADR risk prediction model was assessed by the Hosmer–Lemeshow test. The clinical effectiveness of the model was evaluated by the decision curve analysis (DCA).
Results
General characteristics
According to the inclusion and exclusion criteria, a total of 300 patients who were administered anlotinib were included in this study. The training cohort used to construct the ADRs risk-prediction model consisted of 212 patients, whereas the validation cohort consisted of 88 patients. Their characteristics are given in Table 1. There was no statistical difference between the training cohort and the validation cohort (p > 0.05). The median age was 43 years, and males accounted for 202 cases (67.3%). In total, 229 cases (76.3%) had stage IV cancer at the time of diagnosis. There were 16 tumor types in the patients, the five most common types were bone and soft tissue tumors (43.66%), gynecologic malignancies (29.58%), lung malignancies (8.92%), digestive malignancies (6.57%), and other malignancies (11.27%). Among all participants, 215 (71.6%) were treated with targeted combination chemotherapy, and 159 (53.0%) experienced distant metastasis of the tumor.
Baseline characteristics of all patients in the training cohort and the validation cohort.

+, Yes; −, no; c, chemotherapy; c + i, chemotherapy combined immunization; c + t, chemotherapy combined targeting; c + t + i, chemotherapy combined targeting and immunization; t, target; t + i, targeted combination immunization; t + r, targeted combined radiotherapy.
After a preliminary assessment of the treated patients, we found that 238 patients experienced at least one ADR (79.3%) during treatment with anlotinib. The reported common ADRs were associated with anemia (n = 87, 29.0%), proteinuria (n = 62, 20.7%), decreased white blood cell count (n = 62, 20.7%), decreased platelet count (n = 45, 15%), hematuria (n = 44, 14.7%), and hypertension (n = 32, 10.7%). A detailed analysis of the types and severity of the ADRs is provided in Supplemental Table 3.
Variable selection of influencing factors
Univariate and multivariate binary logistic regression analyses were performed to investigate the risk factors of ADRs among patients who were administered anlotinib. Eight variables (Diagnose, Treatment line, Prior treatment, Distal metastasis, Previous EGFR-TKI treatment, Combined treatment, Base disease, Cumulated dose) showed a significant difference (p < 0.2) between ADRs and non-ADR patients from the univariate analysis; these variables were used in the next stage of multivariate analysis (Table 2). The results of the multivariate logistic regression analysis showed that four factors were independent predictors of ADRs, including diagnosis, distal metastasis, combined treatment, and treatment line. Although the variable “cumulated dose” showed minimal association in this dataset (p > 0.05), it was identified as important and relevant in predicting ADR risk by other studies 6 ; thus, it was also included in this model.
General characteristics of the patients and a summary of the univariate and multivariate logistic regression analyses performed for screening predictors of ADRs.

Indicates a significant difference.
+, Yes; −, no; ADR, adverse drug reaction; c, chemotherapy; c + i, chemotherapy combined immunization; c + t, chemotherapy combined targeting; c + t + i, chemotherapy combined targeting and immunization; t, target; t + i, targeted combination immunization; t + r, targeted combined radiotherapy.
Establishing a risk prediction model
The results of the stepwise forward logistic regression showed that diagnosis, distant metastasis, combination therapy, treatment line, and cumulative dose were screened and entered into the final model, and the factors were integrated to construct the nomogram (Figure 2, R2 = 0.219, C-index = 0.750). For each patient, higher total points indicated a higher risk of the occurrence of ADRs. For example, if a patient diagnosed with gynecologic malignancies suffered distant metastasis, provided the first cycle of treatment was performed with anlotinib (multi-line targeted combined immunization treatment), then the corresponding score will be approximately 55, 23, 33, 15, and 13, respectively. The total score in such a case would approximately be 139 points, indicating an estimated ADR risk of 0.85 (85%). In addition, the results of the Hosmer–Lemeshow test showed that the model was a good fit and the calibration curve was close to the ideal diagonal line (Figure 3, p = 0.811). The calibration curve of the validation cohort for predicting the probability of ADRs is shown in Supplemental Figure 6.

Nomogram for predicting the risk of ADRs.

Calibration curve of the training cohort for predicting the probability of ADRs.
We used the area under the receiver operating characteristic curve (AUC) to assess the discriminatory performance of the ADR risk-prediction model. The training and validation sets showed good discrimination ability (Figure 4). In the training cohort, the AUC was 0.79 (95% CI: 0.72–0.86; Figure 4(a)). Moreover, 88 patients were used for external validation to test the model. The AUC was 0.65 (95% CI: 0.50–0.81, Figure 4(b)), which indicated that the nomogram had good accuracy. The DCA also showed a significant benefit of the predictive model in the training cohort (Figure 5) and the validation cohort (Supplemental Figure 7). These findings suggested that the model constructed to predict the risk of ADRs can be effectively used in clinical decision-making.
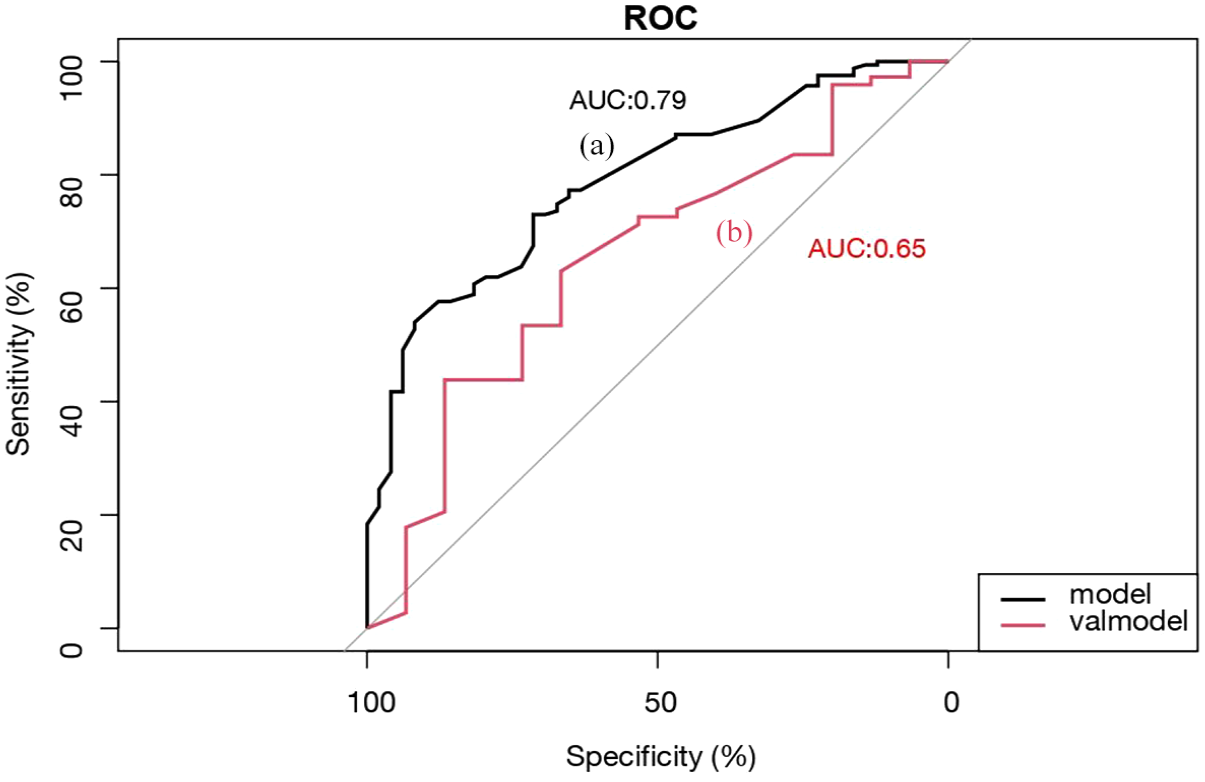
ROC curves. (a) Training cohort and (b) validation cohort.

Decision curve analysis of the training cohort in predicting ADRs.
Discussion
It has been proven that the therapy of anlotinib combined with other drugs has high clinical efficacy in a variety of tumors, but several attentions still need to be paid to its safe usage in clinical medication. Safety data based on the use of anlotinib in patients and previous clinical trials showed an incidence of 97.28% for anlotinib ADRs and 47.28% for Grade 3 and higher ADRs.16,17 Most ADRs could be controlled by dose adjustment, dose interruption, and symptomatic treatment. 18 Among them, generalized weakness, increased blood pressure, bleeding, anemia, and gastrointestinal reactions are commonly related ADRs that cause dose reduction or discontinuation of anlotinib. Given the tolerance of different patients and the impact of dose reduction or discontinuation, it is necessary to establish a model to predict anlotinib-related ADRs in cancer patients, which is used to assess the individualized dosing regimen of patients and ensure safety during medication. Therefore, our study selected predictors based on the combination of statistical significance tests and the clinical significance of the variables. Particularly, we have constructed a real-world ADR risk-prediction model associated with the application of anlotinib in terms of five clinical variables, which is capable of identifying cancer patients with an increased likelihood of developing anlotinib-related ADRs. The final ADR risk prediction model is anticipated to provide a simple, quick, and practically applicable scoring system, making its adoption into routine clinical practice easier.
In our study, external validation data also confirmed the fitting and predictive capabilities of the model. The model selected in our study had an AUC of 0.79 which indicated that the model could perform accurate predictions for a clinically complex population. The DCA indicated that a threshold probability of ⩾0.4 has a clinical application value and great clinical benefits. Thus, it can significantly increase the probability of efficacy that a patient will respond to treatment through a planned proactive intervention. We obviously found that diagnosis significantly increased the likelihood of developing ADRs. Anlotinib can be used to efficaciously treat various tumors, such as non-small-cell lung cancer, small-cell lung cancer, soft tissue sarcoma, and medullary thyroid carcinoma. In addition, corresponding indications have been approved in China. In real-world clinical studies of non-small-cell lung cancer, the main adverse effects of anlotinib were considered to be hypertension, fatigue, hand-foot syndrome, proteinuria, hypoalbuminemia, low levels of alanine aminotransferase, and hyponatremia.19,20 The median onset time of hypertension, hypertriglyceridemia, and hand–foot syndrome is 5 days (range: 2–8 days), 20 days (range: 19–38 days), and 30 days (range: 24–41 days), respectively. However, the exact mechanism of anlotinib-related ADRs has not been thoroughly explored. 6 In our study, ADRs of anlotinib mainly occurred within five treatment cycles involved in the blood and hematopoietic system, urinary system, and cardiovascular system. Moreover, there are off-label use cases associated with the use of anlotinib in the real world, and clinical adverse event data from these patients are under-monitored, similar to what is mentioned on the label.21–23 We have found out that the off-label use of anlotinib is the highest (29.38%) for gynecologic malignant tumors in our study, with the main ADR being proteinuria, which is mostly manifested as Grades 1–2 and the incidence of ADRs is 96.83%. However, only a very small percentage of patients may experience ADRs that are Grades 3–4. Thus, it is important to conduct scientific supervision and rational monitoring for the off-label use of drugs.
Generally, organ metastases show a strong relationship with the worse performance status and the lower survival rate. Our results demonstrated that cancer patients with distant metastasis and multiple lines of chemotherapy are more likely to develop ADRs treated with anlotinib. The potential reason may be the distant metastasis leading to the worse performance status of patients, and previous studies have also shown that three or more prior treatments strongly correlated with worse survival. Another promising finding was that the connection between chemotherapeutic regimen and ADR risk was identified to be another novel risk factor in this study. The risks of chemotherapeutic regimens are mainly related to toxicity caused by the low selectivity of the pharmacological action of the selected chemotherapeutic agents and dose-dependent toxicity caused by the chemotherapy dose. The number of drugs in the combination treatment and previous lines of chemotherapies were identified as an important influencing variable for the risk of ADRs. With the widespread use of anlotinib in combination with other anticancer regimens, the associated ADRs may interrupt treatment and drive patients to refuse the next-step treatment. In fact, the cumulative dose was also included as a risk factor in the model. However, the cumulative dose of anlotinib, as a risk factor investigated previously, has not exerted a remarkable effect in this study. Therefore, a larger sample of real-world clinical comprehensive evaluation studies is still needed to further evaluate and analyze this factor.
Limitations
Although this study had identified some variables that predict the ADRs of anlotinib, the risk model needs further development before it can be adopted for use in clinical practice to test the usability of the model. Our study had certain limitations, including the small sample size and the retrospective nature of the data. In addition, the use of all ADR associations (definite, possible, and probable) in the study data may inevitably have data bias affecting the results. The subjectivity of ADR evaluation criteria is also an important factor affecting the study results. The accuracy and sensitivity of the developed risk model also need to be improved. Moreover, cumulative dose, as one of the important contributing factors, showed a low correlation in this dataset, and further studies are needed to confirm the relationship between the combination regimen and drug dose. Although some important biomarkers have been proposed as predictors and diagnostic indicators of ADRs, they are still currently in the early assessment stage, and their clinical application is greatly limited.24–28
Conclusion
In this study, we developed a model for predicting the risk of ADR based on the real-world clinical use of anlotinib. Our findings showed that diagnosis, distant metastasis, combination therapy, treatment line, and cumulative dose were the most important risk factors for the ADR of anlotinib. The external validation has proved the effectiveness of this model. For each patient, higher total points reflected a greater risk of ADRs. Owing to the model that data are easily accessible and no additional training is required, the use of this predictive model has many advantages and is suitable for clinical application to rapidly assess the risk of anlotinib-related ADRs. However, more real-world factors need to be considered in the model to increase its accuracy and sensitivity. Future clinical studies are also needed to validate the interactions between different variables.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251328687 – Supplemental material for Development and validation of a risk-prediction model for adverse drug reactions in real-world cancer patients treated with anlotinib
Supplemental material, sj-docx-1-taw-10.1177_20420986251328687 for Development and validation of a risk-prediction model for adverse drug reactions in real-world cancer patients treated with anlotinib by Jiajia Qian, Cong Ruan, Yunyun Cai, Weiyi Yi, Jiyong Liu and Rui Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986251328687 – Supplemental material for Development and validation of a risk-prediction model for adverse drug reactions in real-world cancer patients treated with anlotinib
Supplemental material, sj-docx-2-taw-10.1177_20420986251328687 for Development and validation of a risk-prediction model for adverse drug reactions in real-world cancer patients treated with anlotinib by Jiajia Qian, Cong Ruan, Yunyun Cai, Weiyi Yi, Jiyong Liu and Rui Xu in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
