Abstract
Background:
Immune checkpoint inhibitors (ICIs) have transformed cancer therapy but are linked with immune-related adverse events (irAEs), including cardiac events.
Objective:
This study aims to assess the reporting frequency of atrial fibrillation with ICIs using data from the Food and Drug Administration Adverse Event Reporting System (FAERS).
Design:
It is an observational, retrospective, pharmacovigilance study.
Methods:
Individual Case Safety Reports (ICSRs) were retrieved from FAERS up to September 24, 2024. Cases reporting one or more ICIs (atezolizumab, avelumab, cemiplimab, dostarlimab, durvalumab, ipilimumab, nivolumab, pembrolizumab, and tremelimumab) and atrial fibrillation were selected. Disproportionality analyses were performed by applying the reporting odds ratio (ROR) and the Informational Component (IC) with a 95% confidence interval (95% CI).
Results:
A total of 1228 ICSRs were retrieved, of which 218 (17.75%) were related to combinations of ICIs. Most ICSRs (N = 812; 66.1%) referred to male patients and the age group most represented was ⩾65 years (N = 772; 62.9%). Atrial fibrillation was serious in 99.3% (N = 1220) of cases and had a fatal outcome (N = 248; 20.3%). Atezolizumab, avelumab, durvalumab, nivolumab, and pembrolizumab were associated with a statistically significant higher reporting frequency of atrial fibrillation compared to all other drugs (ROR: 1.90, IC: 0.91; ROR: 1.94, IC: 0.92; ROR: 1.52, IC: 0.60; ROR: 1.30, IC: 0.38; ROR: 1.66, IC: 0.72, respectively). The anti-CTLA-4 ipilimumab showed a statistically significant lower reporting frequency of atrial fibrillation compared to all other drugs (ROR: 0.69, IC: −0.53) and to all other ICIs (ROR: 0.45, IC: −1.02). Moreover, anti-PD-L1 (ROR: 2.60, IC: 0.47) and anti-PD-1 (ROR: 2.12, IC: 0.16) were associated with a higher reporting of atrial fibrillation compared to anti-CTLA-4.
Conclusion:
ICI-induced atrial fibrillation was serious and had severe outcomes. The anti-CTLA-4 showed a lower likelihood of reporting atrial fibrillation, while higher reporting was found with anti-PD-1 and anti-PD-L1. Further studies are needed to confirm this safety aspect.
Plain language summary
Why was the study done? Immune checkpoint inhibitors (ICIs) have significantly transformed cancer treatment by stimulating the immune system to fight cancer. However, these drugs can cause side effects, including heart problems such as atrial fibrillation, which is a type of irregular heartbeat. This study aimed to understand how commonly atrial fibrillation occurs in patients using ICIs. What did the researchers do? The research team evaluated the characteristics and reporting frequency of ICIs-related atrial fibrillation by analyzing data from the Food and Drug Administration Adverse Event Reporting System (FAERS) up to September 24, 2024. What did the researchers find? The study found 1,228 reports of atrial fibrillation in patients using ICIs. The majority of cases were serious and involved adult males. The anti-PD-1 and anti-PD-L1 were associated with a higher reporting of atrial fibrillation compared to anti-CTLA-4. Among the individual ICIs assessed, ipilimumab was associated with a lower reporting frequency of atrial fibrillation. On the other hand, atezolizumab showed a high reporting frequency of this event compared to all other drugs. What do the findings mean? The study highlights the importance of monitoring and further investigating the safety profile of ICIs, particularly regarding cardiac events such as atrial fibrillation, which is a serious adverse event with severe outcomes. Further studies are needed to confirm these findings and better understand the underlying mechanisms of ICI-induced atrial fibrillation.
Introduction
Immunotherapy with immune checkpoint inhibitors (ICIs) has revolutionized cancer therapy. 1 ICIs are particularly effective in several advanced cancers, such as metastatic melanoma, non-small-lung cancer, head and neck cancer, breast cancer, pancreatic cancer, prostate cancer, renal cell carcinoma, and nasopharyngeal carcinoma.2,3 ICIs take advantage of immune system components to fight tumor cells. They unleash the body’s immune system to recognize better and attack cancer cells. ICIs work by blocking various targets: cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) or programmed cell death protein-1 (PD-1) or its ligand (PD-L1). 4 Currently, 12 ICIs have been approved by the US Food and Drug Administration (FDA), including CTLA-4 inhibitors (ipilimumab and tremelimumab), PD-1 inhibitors (cemiplimab, dostarlimab, nivolumab, pembrolizumab, retifanlimab, tislelizumab, and toripalimab), and PD-L1 inhibitors (atezolizumab, avelumab, and durvalumab). Since their marketing introduction, ICIs have also been associated with the development of various immune-related adverse events (irAEs). 5 These events are the direct consequence of their mechanism of action, and they are directly linked to the T-cell response against common antigens shared by both tumor and healthy tissues and organs. 6 Among irAEs, those cardiac, although rare, are associated with unfavorable outcomes, becoming a matter of safety concern over the years.7,8 Several studies have indeed evidenced an association between ICI therapy and cardiac events, finding higher reporting of myocarditis for anti-PD-1 and anti-PD-L1.9,10 These findings were also observed for the reporting of arrhythmia as cardiac manifestation.11–13 However, studies focusing on the reporting of atrial fibrillation are meager, probably due to its higher rarity and difficulty in data collection than myocarditis.
To fill this knowledge gap and to improve the evidence on the cardiac safety profile of ICIs, we used the Food and Drug Administration Adverse Event Reporting System (FAERS), an American database that collects spontaneous reports of adverse reactions or medication errors associated with drugs and therapeutic biologics. FAERS enables the identification of potential safety signals in real-world settings, allowing for early detection of rare adverse events (AEs), such as ICI-associated atrial fibrillation. However, while FAERS offers several advantages, including accessibility, a validated methodology, and the ability to identify safety signals quickly and cost-effectively, it also has inherent limitations, such as the possibility of underreporting, reporting biases, duplicates, and a lack of detailed clinical information. Despite these challenges, FAERS remains an important resource for identifying safety signals, as evidenced by recent studies.14–17 In this context, we decided to conduct a pharmacovigilance study focusing on the reporting frequency of atrial fibrillation with ICIs by using data collected in the American spontaneous reporting system.
Methods
Study design
An observational, retrospective, pharmacovigilance study evaluating the characteristics and reporting frequency of ICIs-related atrial fibrillation. The study followed the REporting of A Disproportionality analysis for drUg Safety signal detection using individual case safety reports in PharmacoVigilance (READUS-PV) guidelines. 18 The READUS-PV checklist is illustrated in Supplemental Table 1.
Data source
FAERS is a database collecting Individual Case Safety Reports (ICSRs) sent by healthcare and non-healthcare professionals or pharmaceutical manufacturers. FAERS data were retrieved using a highly interactive and publicly available web-based tool (the FAERS Public Dashboard) for the period up to September 24, 2024. 19 FAERS can also contain duplicate reports, a situation occurring when the same report is submitted by both the consumer and sponsor. However, the information in FAERS can evolve daily, and the number of ICSRs may increase or decrease over time due to the work of regulatory agencies. Data from the FAERS Public Dashboard are quarterly updated. FAERS data include information about patient sex, age, weight, country, reporter, suspect and concomitant drugs, type of AEs, seriousness, and outcome.
ICSRs selection
Using the FAERS intuitive public dashboard, we retrieved ICSRs reporting an ICI as the suspected drug and the preferred term (PT) of the Medical Dictionary for Regulatory Activities (MedDRA) version 24.1 “Atrial Fibrillation” as the AE. MedDRA is a standardized medical terminology globally used for the classification of AEs and it is structured into five levels, reported below from the most specific to the most generic: lowest-level terms, PT, high-level terms, high-level group terms (HLGT), and system organ class (SOC). ICIs considered for selection were atezolizumab, avelumab, cemiplimab, dostarlimab, durvalumab, ipilimumab, nivolumab, pembrolizumab, and tremelimumab. ICIs authorized recently were excluded from analyses due to their short commercialization period (retifanlimab, toripalimab, and tislelizumab).
Descriptive analysis
ICSRs referred to atrial fibrillation and an ICI as the suspected drug were analyzed for characteristics of patients (sex, age group, and weight), reporter type, reporting year, primary source country, seriousness of the case, number of reported suspected drugs and concomitant drugs. ICSRs reporting more than one ICI were grouped into “combination of ICIs.” The seriousness was classified as “serious” or “not serious.” Serious ICSRs were codified according to the FAERS into hospitalized, died, life-threatening, disabled, required intervention, or other outcomes.
All other AEs, reported in addition to atrial fibrillation, were categorized according to the SOC level of MedDRA. Oncological disorders reported among ICSRs were tabled and classified according to the level of HLGT and PT of MedDRA.
Concomitant drugs were classified according to the second level of the Anatomical Therapeutic Chemical (ATC) classification system of the World Health Organization.
The quantitative variable (weight) was displayed as median and interquartile range (IQR), while all other qualitative variables as numbers and percentages.
Disproportionality analyses
To assess disproportionality in the reporting of atrial fibrillation with ICIs, the reporting odds ratio (ROR) and its 95% confidence interval (95% CI) were computed. The ROR was calculated as (a/c)/(b/d): “a” as the number of cases with atrial fibrillation reported with the drug of interest; “c” as the number of cases with atrial fibrillation reported with the comparator; “b” as the number of all other events reported with the drug of interest; and “d” as the number of all other events reported with a comparator. Furthermore, to assess and quantify the strength of the association between ICIs and atrial fibrillation, and decrease false-positive safety signals, the Bayesian confidence propagation neural network (BCPNN) with the information component (IC) and its 95% CI was computed. 20 The IC was used to compare the observed versus expected number of reports for a drug-AE combination and reflects the strength of the association. A positive IC value means that the number of observed reports exceeds the number of expected reports. Conversely, a negative IC value means that the number of observed reports is less than the number of expected reports. In our study, a significant signal was when the value of ROR was >1, the lower limit of the 95% CI for ROR (ROR025) was >1, and the lower limit of the 95% CI for IC (IC025) was >0. IC025 between 0 and 1.5 was defined as a weak signal, an IC025 between 1.5 and 3 was defined as a moderate signal, and an IC025 > 3 was defined as a strong signal. 21 These analyses were performed for the following comparisons: each ICI compared to all other ICIs (excluding the one of interest); each ICI compared to all other ICIs belonging to the same drug class based on their mechanism of action (anti-PD-L1, anti-PD-1, or anti-CTLA-4); each ICIs’ drug class compared to another ICIs’ drug class (anti-PD-L1 vs anti-PD-1, anti-PD-L1 vs anti-CTLA-4, and anti-PD-1 vs anti-CTLA4); each ICI compared to all other drugs reported in the FAERS; each combination of ICIs compared to the single ICI of the combination. At least three events must be reported for each treatment to perform disproportionality analyses. Data management and analyses were performed using Excel 365 (Microsoft Office, Microsoft Corporation, Redmond, WA, USA) and R (version 4.2.2; R Development Core Team, R Foundation for Statistical Computing, Vienna, Austria).
Results
Descriptive results
During the study period, a total of 1228 ICSRs with atrial fibrillation and an ICI as suspected drug (168 for atezolizumab, 20 for avelumab, 11 for cemiplimab, 2 for dostarlimab, 61 for durvalumab, 79 for ipilimumab, 326 for nivolumab, 343 for pembrolizumab, and 218 for combination of ICIs) were retrieved from FAERS. Tremelimumab was reported only in combination with durvalumab (N = 6). Among combination, the most reported was ipilimumab–nivolumab (N = 203; 93.1%). Characteristics of ICSRs for all groups are presented in Table 1 and specifics of ICSRs with a combination of ICIs were reported in Supplemental Table 2. As presented in Figure 1, the main reporting country was America (N = 475; 38.7%), followed by Europe (N = 437; 35.6%), Asia (N = 155; 12.6%), Oceania (N = 32; 2.6%), and Africa (N = 1; 0.1%). In 10.4% of ICSRs (N = 128), the primary source country was unknown (Supplemental Table 3). The highest reporting number of ICSRs was during the years 2021, 2022, and 2023 (N = 162; 13.2%, N = 170; 13.8%, and N = 165; 13.4%, respectively). The reporting annual trend is shown in Supplemental Figure 1. Most ICSRs (N = 812; 66.1%) referred to male patients and the age group most represented was ⩾65 years (N = 772; 62.9%). The weight was available in 611 ICSRs (49.8%), with a median of 77.6 (IQR: 90.8–65.8) and a similar distribution among ICSRs with different ICIs’ drug classes (anti-PD-L1: 81.5, 92.2–66.2; anti-PD-1: 73.9, 89.0–64.0; anti-CTLA-4: 77.9, 85.0–69.4; combination of ipilimumab-nivolumab: 80.0, 91.5–71.6). Healthcare professionals were the main source of reporting (N = 1090; 88.8).
Characteristics of ICSRs reporting atrial fibrillation with ICIs and retrieved from the FAERS up to September 24th, 2024.

Data are expressed as N (%).
Specific atrial fibrillation ICSRs with ICIs combination are reported in the electronic Supplemental Table 1.
FAERS, Food and Drug Administration Adverse Event Reporting System; ICI, immune checkpoint inhibitor; ICSR, Individual Case Safety Report.
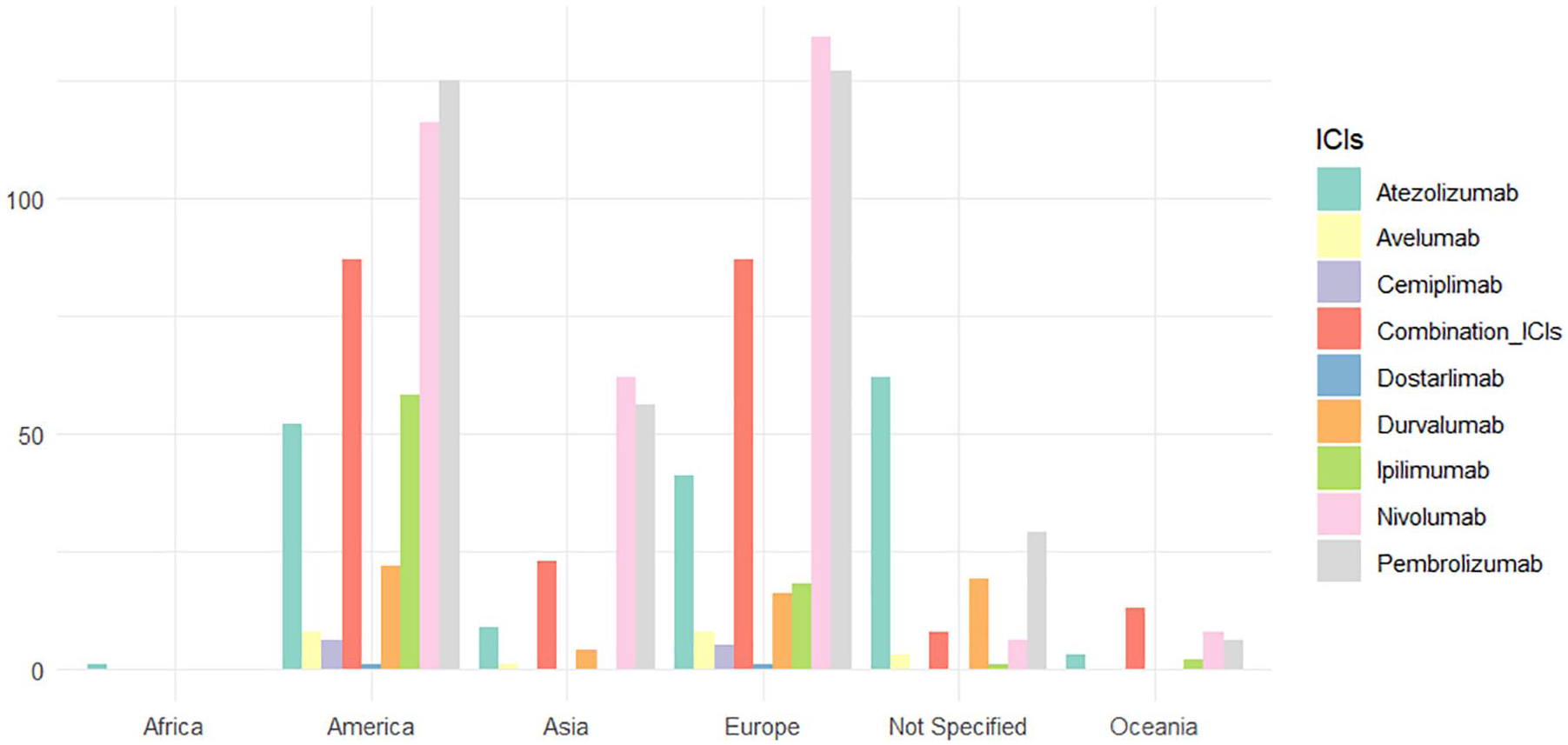
Distribution of primary country source for ICIs.
As shown in Figure 2, therapeutic indications for ICIs included various types of cancer, such as solid tumors (N = 1063; 86.6%), liquid tumors (N = 44; 3.6%), and secondary tumors (N = 5; 0.4%). The therapeutic indication was not specified in 116 cases (9.4%). Most reported oncological disorders belonged to the HLGT “Respiratory and mediastinal neoplasms malignant and unspecified” (N = 406; 33.1%), “Skin neoplasms malignant and unspecified” (N = 218; 17.8%), and “Renal and urinary tract neoplasms malignant and unspecified” (N = 186; 15.1%). All oncological disorders and their specifications are reported in Supplemental Tables 4 and 5, respectively. Moreover, 39.3% of ICSRs (N = 482) reported only the ICI as a suspected drug, and 45.7% (N = 561) presented no concomitant medication. More than half of cases reported at least one concomitant drug. In particular, analgesics (ATC N02) were the most frequently reported (N = 505), followed by drugs for acid-related disorders (A02, N = 320), beta-blocking agents (C07, N = 252), and antithrombotic agents (B01, N = 248). The distribution of concomitant drugs is presented in Supplemental Figure 2.

Distribution of indications for ICIs.
The 99.3% (N = 1220) of ICSRs were classified as serious. The outcomes of seriousness (Table 2) were hospitalized (N = 617; 50.6%), died (N = 248; 20.3%), life-threatening (N = 102; 8.4%), disabled (N = 17; 1.4%), and required intervention (N = 1; 0.1%).
Seriousness outcomes of atrial fibrillation for each ICI and combination of ICIs recognized in the FAERS up to September 24th, 2024.
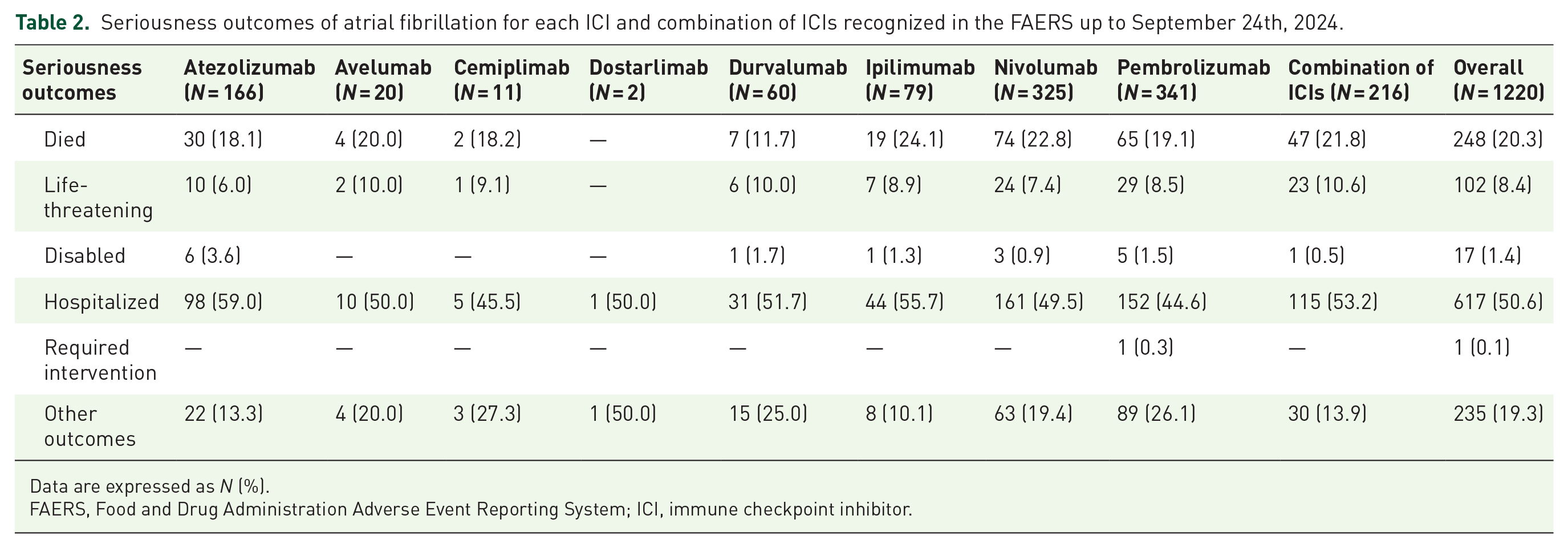
Data are expressed as N (%).
FAERS, Food and Drug Administration Adverse Event Reporting System; ICI, immune checkpoint inhibitor.
The 82.4% of ICSRs (N = 1012) reported an AE other than atrial fibrillation, for a total of 5896 AEs. Looking at the type of other events reported (Figure 3), the most frequent belonged to the SOC “Cardiac disorders” (17.2%, with “Cardiac Failure” as the first represented PT (N = 83; 1.4%)), followed by “General disorders and administration site conditions” (10.2%, with “Pyrexia” as the first represented PT (N = 80; 1.4%)) and “Gastrointestinal disorders” (10.0%, with “Diarrhoea” as the first represented PT (N = 110; 1.9%)). All other AEs are listed in Supplemental Table 6.

Distribution of other adverse events categorized by SOC and reported in the ICSRs related to ICIs and atrial fibrillation.
Disproportionality analyses
RORs of atrial fibrillation compared to all other drugs
Atezolizumab, avelumab, durvalumab, nivolumab, and pembrolizumab were associated with a higher likelihood of reporting atrial fibrillation compared to all other drugs (ROR: 1.90, 95% CI: 1.63–2.21; ROR: 1.94, 95% CI: 1.25–3.01; ROR: 1.52, 95% CI: 1.18–1.96; ROR: 1.30, 95% CI: 1.17–1.45; ROR: 1.66, 95% CI: 1.49–1.84, respectively). The anti-CTLA-4 ipilimumab showed instead a lower reporting frequency of atrial fibrillation (ROR: 0.69, 95% CI: 0.55–0.86; Table 3a).
ROR and IC of ICSRs with atrial fibrillation for each ICI compared to (a) all drugs, (b) all other ICIs, (c) all other ICIs belonging to the same drug class, (d) for each ICI drug class compared to another, and (e) for each combination of ICIs compared to the single ICI.

IC, Information Component; ICI, immune checkpoint inhibitor; ICRS, Individual Case Safety Report; IC025, the lower limit of the 95% confidence interval for IC; IC975, the upper limit of the 95% confidence interval for IC; ROR, reporting odds ratio; ROR025, the lower limit of the 95% confidence interval for ROR; ROR 975, the upper limit of the 95% confidence interval for ROR.
ICs of atrial fibrillation compared to all other drugs
The higher reporting frequency of atrial fibrillation associated with atezolizumab, avelumab, durvalumab, nivolumab, and pembrolizumab compared to all other drugs was confirmed by the IC analysis (IC: 0.91, 95% CI: 0.66–1.10; IC: 0.92, 95% CI: 0.19–1.45; IC: 0.60, 95% CI: 0.18–0.90; IC: 0.38, 95% CI: 0.19–0.51; IC: 0.72, 95% CI: 0.54–0.85). A negative IC value for the ipilimumab compared to all other drugs (IC: −0.53, 95% CI: −0.90 to −0.26; Table 3a) confirmed a lower reporting frequency of atrial fibrillation.
RORs of atrial fibrillation compared to all other ICIs
A higher reporting frequency of atrial fibrillation was found when atezolizumab (ROR: 1.42, 95% CI: 1.20–1.68) or pembrolizumab (ROR: 1.27, 95% CI: 1.12–1.45) were compared to all other ICIs. On the contrary, ipilimumab showed a lower reporting frequency of atrial fibrillation (ROR: 0.45, 95% CI: 0.36–0.56; Table 3b).
ICs of atrial fibrillation compared to all other ICIs
The IC analysis confirmed the higher reporting frequency of atrial fibrillation associated with atezolizumab (IC: 0.43, 95% CI: 0.17–0.61) and pembrolizumab (IC: 0.24, 95% CI: 0.06–0.37) when compared to all other ICIs. By contrast, a negative IC value for ipilimumab (IC: −1.02, 95% CI −1.39 to −0.75; Table 3b) indicated a lower reporting frequency of atrial fibrillation.
RORs of atrial fibrillation within the same drug class
When the ROR was computed within the same drug class, a higher reporting frequency of atrial fibrillation was found for pembrolizumab than anti-PD-1 (ROR: 1.26, 95% CI: 1.09–1.47). A lower reporting frequency of atrial fibrillation was observed for nivolumab compared to anti-PD-1 (ROR: 0.78, 95% CI: 0.68–0.91; Table 3c).
ICs of atrial fibrillation within the same drug class
The lower reporting frequency of atrial fibrillation associated with nivolumab compared to anti-PD-1 was confirmed by the IC analysis (IC: –0.17, 95% CI: –0.35 to –0.04). On the contrary, the IC value for pembrolizumab compared to anti-PD-1 (IC: 0.18, 95% CI: –0.002 to 0.31; Table 3c) was not significant.
RORs of atrial fibrillation among different drug classes
In the comparison among different drug classes, anti-PD-L1 (ROR 2.60; 95% CI 2.02–3.35) and anti-PD-1 (ROR: 2.12, 95% CI: 1.68–2.68) were associated with a higher reporting of atrial fibrillation compared to anti-CTLA-4. Moreover, anti-PD-L1 was also associated with a higher reporting frequency of atrial fibrillation compared to anti-PD-1 (ROR: 1.22, 95% CI: 1.06–1.41; Table 3d).
ICs of atrial fibrillation among different drug classes
The IC analysis confirmed a higher reporting frequency of atrial fibrillation associated with PD-L1 (IC: 0.47, 95% CI: 0.26–0.62) and PD-1 (IC: 0.16, 95% CI: 0.03–0.25) when compared to CTLA-4. Similarly, a positive IC value for PD-L1 (IC: 0.22, 95% CI: 0.008–0.37; Table 3d) indicated a higher reporting frequency of atrial fibrillation compared to PD-1.
RORs of atrial fibrillation compare the combination of ICIs with a single ICI
The combination of ipilimumab–nivolumab and durvalumab–tremelimumab was reported at least three times. Ipilimumab–nivolumab was associated with a higher reporting frequency of atrial fibrillation than ipilimumab or nivolumab alone (ROR: 3.91, 95% CI: 3.01–5.07; ROR: 2.07, 95% CI: 1.74–2.47, respectively). While no difference was observed for durvalumab–tremelimumab compared to durvalumab (ROR: 0.54, 95% CI: 0.23–1.25; Table 3e).
ICs of atrial fibrillation compare the combination of ICIs with a single ICI
The higher reporting frequency of atrial fibrillation associated with the combination of ipilimumab–nivolumab compared to ipilimumab or nivolumab alone was confirmed by the IC analysis (IC: 0.85, 95% CI: 0.62–1.02 and IC: 0.72, 95% CI: 0.49–0.89; Table 3e).
Discussion
ICIs are widely associated with the occurrence of irAEs due to their mechanisms of action, which enhance immune responses against both tumors and, inadvertently, healthy tissues.22–24 Among these irAEs, particular attention requires cardiotoxicity, which has been widely investigated.10,13,25–27 Cardiac AEs are rare but often associated with serious, life-threatening, and fatal outcomes. 28 These events include myocarditis, pericarditis, acute coronary syndrome, heart failure, and arrhythmias. 29 This study aimed to evaluate the reporting frequency of atrial fibrillation with ICIs using data from the FAERS database. The analysis of large pharmacovigilance databases allows the identification of safety warnings from real-world data.30,31 Atrial fibrillation is the most common form of arrhythmia caused by an abnormal electrical activity within the atria that causes fibrillation. It is characterized by an irregular heartbeat that can lead to serious health issues. 32
The reporting annual trend of atrial fibrillation was found higher during the years of the COVID-19 pandemic, which was generally characterized by a reduction in event reporting, except for those serious that were the ones mostly reported in our study.33,34
From our analysis emerged that most ICI-related atrial fibrillation cases were serious and associated with an unfavorable outcome. This was in accordance with a previous pharmacovigilance study using Eudravigilance data that found most cardiac events, including myocarditis, cardiac failure, atrial fibrillation, pericardial effusion, and myocardial infarction, classified as serious (99.4%) and associated with a fatal outcome (30.1%). 10
Based on the current literature, the exact mechanisms of atrial fibrillation induced by ICIs are not fully understood, but several hypotheses have been proposed, all linked to their mechanism of action and shared by all irAEs. The first mechanism is the development of an autoimmune reaction based on the ability of ICIs to stimulate the immune system by promoting T-cell activity against healthy tissues and organs. In some cases, this immune activation could affect the heart’s electrical system, potentially leading to atrial fibrillation. 35 The second hypothesis is related to the inflammatory changes in the atria of the heart that could disrupt the normal electrical conduction system, triggering atrial fibrillation. 36 In fact, for their mechanism of action, ICIs can lead to increased levels of pro-inflammatory cytokines causing inflammation in various organs, including the heart. 37 Moreover, ICIs may lead to the infiltration of immune cells into the cardiac tissue, disrupting its normal function. 38
In our database, a higher number of ICSRs was found with the anti-PD-1 nivolumab and pembrolizumab. They were the first ICI therapies to receive the FDA authorization, after ipilimumab. In particular, Keytruda® (pembrolizumab) and Opdivo® (nivolumab) were authorized by the FDA in 2014.39,40 The fact that they were among the pioneering ICIs may contribute to the higher reporting of AEs. Indeed, as drugs become more widely used after authorization, the number of reported AEs tends to increase. It could be due to a combination of factors, including enhanced surveillance, heightened awareness among healthcare professionals, and the cumulative effects of prolonged drug exposure over time. We found an increased number of ICSRs referred to elderly male patients, consistent with literature data. In fact, a cohort study explaining 50-year trends in atrial fibrillation has demonstrated a higher incidence in males than females. 41 Moreover, some studies suggested that hormonal changes in women, such as the increase in estrogen, may have a protective effect against atrial fibrillation. 42 A recent study provided an updated safety analysis of ICIs using the FAERS database and revealed a higher proportion of AEs reported in males (57.32%) compared to females. 43 The onset of atrial fibrillation in elderly patients (older than 65 years) can be due to age-related changes and the increase in comorbidities in this subpopulation.44,45 The finding that over half (54.3%) of cases had at least one concomitant drug suggests the presence of underlying comorbidities that may have possibly increased the likelihood of reporting atrial fibrillation. In particular, the use of beta-blockers, antithrombotic agents, lipid-modifying agents, calcium channel blockers, renin–angiotensin system blockers, and diuretics can raise concerns since these medications are prescribed to treat cardiovascular conditions that are themselves risk factors for atrial fibrillation. It means that patients may already have significant comorbidities, such as hypertension or other heart-related issues, which may contribute to the observed reporting probability of atrial fibrillation in this population. Previous studies have reported a higher risk of ICI-related myocarditis in patients with pre-existing cardiovascular risk factors and conditions.10,46,47 Therefore, the presence of concomitant medications, particularly cardiovascular and metabolic agents, may indicate a higher baseline risk for these patients, potentially influencing the reporting frequency of cardiac AEs, such as atrial fibrillation. Unfortunately, due to limitations related to our data source, we cannot be sure of the real quota of concomitants reported among cases since some cases may not report this information just for a lack of quality in filling in the report. Therefore, the quota of concomitants could have been higher than observed. Based on the literature, the evidence on the risk of atrial fibrillation with ICIs is meager. One observational study found that patients with a pre-existing diagnosis of atrial fibrillation are at high risk of recurrence. 48 From our disproportionality analysis, we found signals of reported atrial fibrillation associated with the anti-PD-1 agents nivolumab and pembrolizumab, as well as the anti-PD-L1 agents atezolizumab, avelumab, and durvalumab. These results were also confirmed by the IC analysis. Accordingly, research showed the predominant role of the PD-1/PD-L1 signaling pathway in regulating T-cell activation and secretion of pro-inflammatory factors in atrial fibrillation. 49 Moreover, a recent disproportionality signal analysis using the ROR and the IC for pembrolizumab versus the database for the period 2017–2023 showed a statistically significant association with atrial fibrillation (ROR: 1.43, 95% CI: 1.18–1.72; IC: 0.51, 95% CI: 0.19–1.26). 50 More broadly, a meta-analysis highlighted an increased risk of all-grade cardiovascular events with anti-PD-1 and anti-PD-L1 in cancer patients. 51 The higher reporting frequency of atrial fibrillation for atezolizumab and pembrolizumab was also confirmed when compared to all other ICIs. The risk of atrial fibrillation with atezolizumab was not explored in the literature, but as mentioned before a strong association with anti-PD-L1-related arrhythmic events was consistently highlighted in previous studies.12,13,52 In the analyses within the same drug class, pembrolizumab showed a higher reporting frequency of atrial fibrillation compared to all other anti-PD-1, although not confirmed by the IC value. On the contrary, nivolumab was associated with a lower reporting frequency of atrial fibrillation compared to all other anti-PD-1. Pembrolizumab and nivolumab share some chemical and structural differences. Pembrolizumab is a humanized monoclonal antibody (mAb) with the human Fc (fragment crystallizable) and murine antigen-binding regions. Nivolumab is instead a fully human mAb, meaning that its entire structure is derived from human components.53,54 mAbs development has evolved to include more human sequences in the antibody structure based on the hypothesis that the more the human is the antibody less its immunogenicity. 55 However, the up-to-date evidence suggests that immunogenicity is nearly similar for humanized and fully human mAbs.56,57 Other studies compared this risk between ICIs. Specifically, a previous pharmacovigilance study, conducted on data from Eudravigilance, showed that ICI-related cardiac arrhythmias, including atrial fibrillation, were over-reported with anti-PD-1 or anti-PD-L1 versus anti-CTLA-4. 11 Moreover, previous studies conducted on FAERS data, but focusing on the overall arrhythmic events, showed a higher reporting with anti-PD-1 and anti-PD-L1 than anti-CTLA-4.12,13 Accordingly, our analysis showed a lower reporting of atrial fibrillation with the anti-CTLA-4 ipilimumab than all other ICIs and a higher reporting for the drug classes anti-PD-1 and anti-PD-L1 than anti-CTLA-4. A hypothesis explaining this difference may be related to the CTLA-4 pathway, which participates in the early activation of T cells during an immune response, while PD-1 and PD-L1 participate in the later stages of the immune response, which may help in sustaining the immune response in peripheral tissues, leading to autoimmune reactions, inflammation, and atrial fibrillation.58,59
Evidence on the evaluation of ICI combination compared to monotherapy is conflicting and related to the overall arrhythmia. One pharmacovigilance study did not find an increased reporting with a combination of ICIs, 13 while another study found an overall increased reporting compared to monotherapy. 12 In addition, a case report described a 66-year-old woman with advanced renal cell carcinoma who developed non-ischemic dilated cardiomyopathy and ventricular tachycardia while receiving combination therapy with nivolumab and ipilimumab. While this combination therapy improved tumor control, it also carried a higher risk of cardiac toxicity. 60 Accordingly, we found a positive association between the combination of ipilimumab–nivolumab and the reporting of atrial fibrillation and a not statistically significant association for durvalumab–tremelimumab, probably due to the low number of ICSRs.
Strengths and limitations
Our study has several strengths. Overall, pharmacovigilance analyses can contribute to better characterizing the safety profile of medicines when used in daily clinical practice. A pharmacovigilance study is a validated, fast, and cheap method useful to identify a possible link between a medicine and an AE, to identify a new AE, including rare events not identifiable during the pre-marketing phase, such as the irAEs associated with ICIs. In addition, the use of FAERS, the United States AE self-reporting system, allows the analysis of many ICSRs.
Our study also had several limitations related to the spontaneous reporting system, including underreporting, low quality or lack of data, repeated information, and difficulties in controlling confounding factors. The low quality of data can impede the full evaluation of cases. For example, we found that over half of cases reported at least one concomitant drug. However, we cannot be sure of the real quota of concomitant medications as well as the real presence of pre-existing cardiovascular diseases influencing the occurrence of atrial fibrillation. Therefore, our results should be carefully interpreted. Moreover, our data source contains only safety data. There is a lack of information on the treated quota of patients using ICIs. For this reason, we cannot compute incidences or other risk measures, but we reported an overview of cases related to adverse drug reactions useful for the identification of new safety signals. Moreover, another limitation is related to the data source (FAERS dashboard), which can contain duplicate reports of difficult recognition because they do not always report all the necessary information to be identified. Therefore, for this reason too, FAERS data cannot be used to calculate the incidence of AEs. Therefore, further confirmatory studies are required to prove a causal link between ICIs and atrial fibrillation.
Conclusion
This study showed an increased reporting of atrial fibrillation with ICIs that can lead to severe outcomes. The anti-PD-1 and anti-PD-L1 were associated with a higher reporting of atrial fibrillation than anti-CTLA-4. In the evaluation of each ICI, ipilimumab was associated with the lower reporting frequency of atrial fibrillation. On the other hand, atezolizumab showed a high reporting frequency of this event. It is important to underline that medical professionals should consider pre-existing heart conditions in patients who will receive ICIs.
Considering all the limitations of our study and the importance of translational and clinical research, 61 further studies on ICI-induced atrial fibrillation conducted on different data sources are necessary to confirm this safety issue and its trend among ICIs.
Supplemental Material
sj-docx-1-taw-10.1177_20420986241312497 – Supplemental material for Pharmacovigilance study on the reporting frequency of atrial fibrillation with immune checkpoint inhibitors: insights from FDA Adverse Event Reporting System
Supplemental material, sj-docx-1-taw-10.1177_20420986241312497 for Pharmacovigilance study on the reporting frequency of atrial fibrillation with immune checkpoint inhibitors: insights from FDA Adverse Event Reporting System by Nunzia Balzano, Annamaria Mascolo, Donatella Ruggiero, Concetta Rafaniello, Giuseppe Paolisso, Francesco Rossi and Annalisa Capuano in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
