Abstract
In 2019, intranasal esketamine gained approval as a promising therapy for those individuals grappling with treatment-resistant depression. Both clinical trials and real-world studies have underscored its efficacy in alleviating and remitting depressive symptoms, with sustained benefits observed for nearly 4.5 years. As the S-enantiomer of ketamine, esketamine’s dosing guidelines and strict medical supervision stem from prior research on ketamine’s use in depression and history as a recreational drug. Despite initial concerns, long-term clinical studies have not documented instances of abuse, misuse, addiction or withdrawal, and the same was found in case reports or subsamples of high-risk populations with comorbidities such as substance use disorder or alcohol use disorder. Esketamine has proven to be safe and well tolerated without fostering new-onset substance use in vulnerable groups. Real-world studies reinforced these observations, reporting no adverse events (AEs) related to pharmacological interactions of esketamine with any other substance, and no new-onset drug or alcohol misuse, craving, misuse or diversion of use. Reports of esketamine craving remain rare, with only one case report documented in 2022. Most drug-related AEs reported in pharmacovigilance databases are those identified in the product’s technical data sheet and with known reported frequency. More importantly, no register of illicit acquisition of esketamine or its tampering for obtaining ketamine or other altered products was found in our search. Overall, our review confirms esketamine’s safety across diverse patient populations, reassuring its responsible use and the scarcity of reports of abuse or misuse since its introduction to the market.
Plain language summary
Why was the review done?
Some patients with treatment-resistant depression (TRD) often don’t get better even after trying different medications. A new nasal spray called esketamine was created to help these patients. But because esketamine is similar to a drug that some people use for fun, ketamine, doctors wanted to make sure it wouldn’t be misused by patients who are already struggling.
What did the authors do?
The authors reviewed the literature for many studies about esketamine. These studies talked about side effects and whether people used it correctly. They also checked if people with a history of drug or alcohol problems had any issues with esketamine. The review included studies conducted for esketamine’s commercial approval, long-term studies, and reports from regular doctor’s offices.
What did the authors find?
The authors found that esketamine treatment is safe to use and doesn’t cause many problems. There was no evidence that people were using it incorrectly or taking other drugs because of it. This was true even for people who had struggled with alcohol or drugs before. These findings confirm that esketamine is safe for many different types of patients, both in research studies and in everyday medical care.
Keywords
Introduction
Approximately 5% of the world’s population has depression, 1 with 175 million diagnosed specifically with major depressive disorder (MDD). 2 In Spain, MDD is one of the most common psychiatric diagnoses in the general population, and its prevalence is comparable with the trend worldwide and in Europe,3,4 with a yearly prevalence of around 4%, and a lifetime prevalence of 10.6%. 5
The first-line treatment for MDD targets the monoaminergic system, mainly selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs).6,7 These antidepressants require several weeks to achieve measurable therapeutic responses and months to achieve full remission of symptoms, a shortfall especially in patients at high risk of suicide.8,9 Therefore, approximately one-third of patients with MDD fail to respond and/or achieve remission after treatment with monoaminergic antidepressants like SSRIs and SNRIs. 10 These patients are diagnosed with treatment-resistant depression (TRD), defined by The European Medicines Agency (EMA) 11 and the multinational European research consortium ‘Group for the Study of Resistant Depression’ 12 as patients who failed at least two different antidepressant treatments (of the same or different classes), either consecutively or as a combination or augmentation therapy, with an average duration of 6–8 weeks each one. This treatment failure leads to severely ill and resistant patients, as determined by the 17-item Hamilton Depression Rating Scale (HDRS). 10
Even when patients with TRD respond to monoaminergic antidepressants, the relapse rate is high (approximately 70%) within 6 months. 13 Therein lies the reason why depression-related disorders, such as MDD and TRD, require the greatest number of sick leaves with a high associated cost due to their chronic nature, high prevalence and recurrence over time. 14 The Spanish Foundation for Psychiatry and Mental Health (FEPSM) estimated in 2017 that the average annual cost per year for treatment of psychiatric/mood disorders in Spain was €9–€10 billion, which was around 1% of the gross domestic product. 15 When treating patients with MDD, the average annual cost for a patient with TRD is almost twice the cost of a patient without TRD (€6096 vs €3846). 2 An aggravating factor appears when at least one substance use disorder (SUD) is diagnosed together with a mental disorder in the same person (dual disorder). 16 In the case of SUD and MDD (dual depression), 17 these patients have a worse prognosis when compared with patients with only TRD or SUD. 18
As an alternative when monoaminergic antidepressants fail, the general anaesthetic ketamine was found by researchers to be a valid option for the treatment of TRD, due to its potential effects in reducing depressive symptoms in a sustained fashion over time, especially in those TRD patients with suicidal ideation (SI).
19
Ketamine is an N-methyl
Methods
It should be noted that this article is not meant to be an exhaustive review of the literature concerning esketamine, which requires a systematic approach, but to address the present evidence of the risks for its misuse or abuse in both clinical and real-world settings. To this end, we searched the PubMed and Google Scholar databases to identify articles in the English or Spanish languages published as of February 2024 combining in two-by-two the keywords ‘esketamine’ and ‘ketamine’ with the terms: ‘misuse’, ‘treatment-resistant depression’, ‘TRD’, ‘MDD’, ‘abuse’, ‘diversion’, ‘routine clinical practice’, ‘clinical studies’, ‘real word’, ‘substance use’, ‘substance use disorders’, ‘suicidal ideation’ and ‘high-risk populations’. The results of the search were further screened for relevance, focusing on reports of side effects, potential abuse, off-label use, craving, misuse and diverted use, as well as high-risk populations with substance or alcohol abuse. All five clinical trials used for the FDA’s and EMA’s marketing approval of esketamine (TRANSFORM 1, 2 and 3 studies, SUSTAIN 1 and 2 studies)22–26 were thoroughly reviewed for treatment-emergent adverse events (TEAEs), followed by the successive long-term and real-world studies from 2019 on (with patients included in September 2015 the earliest and followed up until February 2023 the latest).
Pharmacology of esketamine
Esketamine, also known as S-ketamine, is the S-enantiomer of ketamine. Ketamine exists in two optical enantiomers, R-ketamine and S-ketamine, which are the mirror images of the same molecule – (2-[2-chlorophenyl]-2-[methylamino]-cyclohexanone) – one right-facing (R-ketamine) and the other left-facing (esketamine). 27 Ketamine is pharmaceutically manufactured as a 1:1 racemic mixture of S and R enantiomers. 28
Both ketamine and esketamine started being prescribed as anaesthetics in 196529,30 and 1970, 31 respectively. Esketamine has been produced from the racemic mixture of ketamine and its resolution 32 since its introduction as an anaesthetic. However, the method of enantioselective synthesis was reported in 2019 as more efficient for obtaining esketamine 33 and employed in the development of the drug as an antidepressant. While esketamine had previously been obtained from ketamine, the latter, as a racemic mixture, cannot be produced from esketamine.33–35
As a main mechanism of action for their anaesthetic effect, both enantiomers work as non-competitive NMDA receptor antagonists.36,37 NMDA receptors are tetrameric ionotropic glutamate receptors that are present throughout the central nervous system (CNS), including the cerebral cortex, cerebellum, brain stem and spinal cord. 38 NMDA receptors and their agonist or binder, glutamate, are important players in synapsis communication, neuronal plasticity and memory function. Ketamine blocks the NMDA channel in a non-competitive manner, 39 preventing neuronal activation, a prerequisite for the conscious state and consequently resulting in an anaesthetic effect.
The NMDA receptor has been implicated in the pathophysiology of a variety of neurological and neuropsychiatric diseases. 40 Glutamate, the agonist, and binder of NMDA receptor, is recognized as the principal excitatory neurotransmitter in the CNS 41 since it is estimated to participate in 90% of all the synapses in the brain.42,43 Excessive glutamate and subsequent over-stimulation of NMDA receptors have been implicated in the pathophysiology of many neurodegenerative diseases.44,45 By contrast, in the context of depression diseases, reduced glutamate levels have been reported in the prefrontal areas of TRD subjects. 46
A study of the antidepressant properties of ketamine was conducted due to the involvement of glutamate and NMDA receptors in neural circuitry.47,48 Racemic ketamine was administered intravenously in subanaesthetic doses (0.1–0.5 mg/kg) in unipolar and bipolar TRD patients, with a measurable antidepressant effect emerging 2–4 h after treatment (by a reduction in the Montgomery-Asberg Depression Rating Scale (MADRS) score), and a substantial mood improvement (after observing a decrease in the Beck Depression Inventory and HDRS scores) at 24 h.49–51 Administrations took place twice a week for 2 weeks in two studies,19,51 but the reduction in MADRS score lasted up to 7 days after treatment even when only a single administration was provided. 52 In these studies, a change in anhedonia (inability to feel pleasure), helplessness and suicidality was observed after ketamine infusion, rather than inducing a nonspecific mood elevating effect, and the clinical benefits appear in 50% 53 –80%54,55 of patients.
Although ketamine use disorder is rarely reported in the clinical setting, 56 ketamine administration at subanaesthetic doses can lead to vivid dreams (both pleasant and frightening), hallucinations, 57 feelings of mind-body dissociation (out of the body or near-death experiences), difficulty in movement, sedation, sensations of floating, severe disorientation, delirium, 38 urinary tract toxicity, 58 cognitive impairments, including severe impairments of working, episodic and semantic memory. 59 These repeatedly observed side effects and addiction potential justify why ketamine started as an off-label treatment option for TRD,21,60 as a last resource for depression, mainly bipolar disorder (BD) and continues to be so. 61
The results with ketamine in unipolar and bipolar TRD patients had a striking contribution to the development of esketamine as a nasal spray (Spravato®; Janssen Pharmaceuticals Inc., Titusville, NJ, USA), approved in 2019 for the treatment of TRD.20,21,62 Esketamine is a more potent antagonist of the NMDA receptor, presenting a fourfold higher affinity for it when compared with the R-enantiomer.63–65 The S-enantiomer has also been reported to have a higher affinity for opioid receptors, leading to specific effects on their activation and a consequently greater potential for abuse. 66 This characteristic suggested prescribing lower amounts to reduce its dose-dependent dissociative properties. 67 The intranasal route of administration is easier and less invasive than intravenous racemic ketamine.68,69 In addition, the intranasal route allows the release of drugs directly into the CNS, being absorbed through the nasal mucosa and thus avoiding the blood–brain barrier, first-pass metabolism and gastrointestinal absorption, with advantages such as rapid onset of action and minimization of systemic side effects. 70
Previous reports of ketamine side effects have highlighted the possibility of abuse for intranasal esketamine, which is why the drug’s treatment guidelines mandate its use in a healthcare setting with an observation period of at least 40 min after administration (in a first instance for blood pressure assessment) and until the healthcare practitioner considers the patient clinically stable and ready to leave the facility. 20 Special attention to symptoms such as dissociation and sedation was given in the clinical trials with esketamine, whose safety results are analysed in the next section.
Results from clinical trials and real-world studies with esketamine for TRD
Table 1 summarizes the safety results of intranasal esketamine reported to date in clinical studies and the real-world clinical setting since its marketing in 2019. Five studies were presented for marketing authorization of intranasal esketamine; three TRANSFORM studies (TRANSFORM 1, 2 and 3) with short-term treatments up to 4 weeks (Table 1) and two SUSTAIN studies (SUSTAIN 1 and 2) where the participants had follow-up lasting 92 and 52 weeks, respectively.22–26 The efficacy-related primary outcome in the TRANSFORM studies was assessing esketamine’s antidepressant effect as the change at day 28 versus baseline in the MADRS total score, and only the TRANSFORM 2 study reported a statistically significant difference when compared to the placebo group. 25 The SUSTAIN 1 study reported a statistically significant reduction in its main endpoint of relapse for those patients in the experimental group when compared to placebo. 22
Safety results from clinical and real-world studies with intranasal esketamine.

Non-responsive to at least two antidepressants in the current episode, without psychotic features, SI, SUD, obsessive-compulsive disorder, bipolar or related disorders in the last 6 months.
The three more reported TEAEs are shown.
After esketamine intranasal administration.
All participants started with a dose of 28 or 56 mg on day 1, and on days 4, 8, 11 and 15, it was permitted an increase to 56 mg/84 mg or a decrease to 28 mg/56 mg in posterior days, at the investigator’s discretion.
Computerized cognitive battery.
Hospitalization ⩾5 days and newly initiated or optimized oral antidepressant(s).
↓, reduction; AE, adverse event; AIFA, Agenzia Italiana del Farmaco; BP, blood pressure; BPIC-SS, Bladder Pain/Interstitial Cystitis Symptom Score; CADSS, Clinician Administered Dissociative States Scale; CGI-SS, Clinical Global Impression–Severity of Suicidality; C-SSRS, Columbia-Suicide Severity Rating Scale; CVA, cerebrovascular accident; DSM-5, Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition; ESK, esketamine; HVLT-R, Hopkins Verbal Learning Test – Revised; IN, intranasal; IV, intravenous; KET, ketamine; MARDS, Montgomery-Asberg Depression Rating Scale; MDD, major depressive disorder; MOAA/S, Modified Observer’s Assessment of Alertness/Sedation; N, number of enrolled patients; ND, not determined; OA, oral antidepressant; SAEs, serious adverse events; SI, suicidal ideation; SNRI, serotonin-norepinephrine reuptake inhibitor; SOC, standard of care; SSRI, serotonin reuptake inhibitor; SUD, substance use disorder; TEAEs, treatment-emergent adverse events; TRD, treatment-resistant depression.
Concerning safety, most TEAEs were mild or moderate in intensity, transient, manifesting generally in up to 4 h after esketamine administration, and resolved in the same day. The most reported TEAEs for the TRANSFORM studies were nausea, dissociation, dizziness and vertigo, occurring 3–8 times more frequently in those treated with esketamine (Table 1). In the TRANSFORM 3 study, with a population older than 65 years, an increase in blood pressure was the third most reported TEAE. 24 For the SUSTAIN 1 study, dysgeusia, vertigo and dissociation were the most commonly reported TEAEs, occurring 4–23 times more often in the experimental group when compared to the placebo (Table 1). All mentioned TEAEs in the three TRANSFORM and SUSTAIN 1 studies are identified to occur with very common frequency in the drug’s summary of product characteristics (SmPC). 20 The Clinician Administered Dissociative States Scale (CADSS) indicated a peak at 40 min after dosing and presented a resolution in approximately 1.5 h in all three TRANSFORM studies (Table 1). The sedative effect after esketamine administration, assessed by the Modified Observer’s Assessment of Alertness/Sedation (MOAA/S), was registered for 10- and 8-fold more patients in the esketamine group in the TRANSFORM 1 and 3 studies, respectively.23,24 To verify whether an association may exist between esketamine’s antidepressant and dissociative effects, Chen et al. performed a comprehensive analysis of three phase III studies: TRANSFORM 1, TRANSFORM 2 and SUSTAIN 1. 71 The post hoc analysis suggests that there is no significant correlation between the antidepressant efficacy of esketamine and the occurrence or intensity of dissociative experiences reported by patients in these 4-week trials. While the dissociative effects tended to diminish over time with repeated esketamine administration, this does not lead to an accompanying impact on the drug’s antidepressant benefits. 71 This was further confirmed by a thorough review of the pharmacological action of R- and S-ketamine enantiomers, as well as classic psychedelics. 72 Once more, compiled studies lead to the conclusion that the antidepressant effects of both ketamine and esketamine are independent of their dissociative side effects, holding true across various patients’ groups, as those with MDD, TRD and BD. 72
Since the long-term use of ketamine at subanaesthetic doses was previously shown to provoke urinary tract toxicity,58,73 the SUSTAIN 2 study employed the Bladder Pain/Interstitial Cystitis Symptom Score (BPIC-SS) to assess it. Only 1.7% of the patients reported a BPIC-SS > 18 (threshold for cystitis, Table 1). Specifically, in the SUSTAIN 2, there was no indication of abuse of esketamine, indicating that its rapid clearance from plasma, short half-life and low dosing frequency did not allow esketamine to reach steady state.
The SUSTAIN 3 study has a 5-year follow-up for esketamine use in TRD, the longest term so far. The results published are of interim analyses, up to an interim database lock on 1st December 2020, approximately 4.5 years. Adults with TRD who participated in more than one of the six phase III ‘parent’ studies (TRANSFORM 1, 2 and 3, SUSTAIN 1 and 2 and Chen et al., performed in Chinese patients [NCT03434041]) could continue esketamine treatment, combined with an oral antidepressant, by enrolling in the SUSTAIN 3 phase III open-label study.22–26,74 There was no evidence of decline in cognition associated with long-term treatment among participants <65 years old from baseline to week 160. The results reported indicate that long-term exposure to esketamine yielded no additional concerns or trends related to SI and/or behaviour, drug abuse or drug dependence (Table 1).
Studies focusing on TRD subpopulations of interest were also performed. A clinical development program consisting of two identically designed, phase III double-blind multicentre global studies (ASPIRE I and ASPIRE II) was launched to confirm the antidepressant benefits of esketamine in patients diagnosed with MDD according to the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) and with SI with intent and need for hospitalization. This initiative has a special relevance since this population is usually excluded from antidepressant clinical trials. In ASPIRE-1, both esketamine and placebo groups improved their Clinical Global Impression of Severity of Suicidality (CGI-SS) score after 24 h, with no statistically significant difference. 75 The most reported TEAEs were dizziness, dissociation and nausea reported 2–7 times more in the experimental group (Table 1). The score in MOAA/S also revealed symptoms of dissociation occurring 6–10 times more often in patients who received esketamine. The ASPIRE II study 76 studied another set of 230 participants and reported the same results as ASPIRE I.
The study ESCAPE-TRD aimed to compare intranasal esketamine with extended-release quetiapine, an antipsychotic augmentation agent commonly used for TRD patients with previous failed treatments. 77 The esketamine group presented a higher reporting of adverse events (AEs) (Table 1). The AEs reported in the esketamine group were consistent with the established safety profile of the drug and were generally transient and mild in severity, being limited to the day of dosing. Both treatment groups have similar serious AEs (SAEs), reporting suicidal attempts and deaths. The quetiapine group presented more AEs which led to the discontinuation of the study treatment. Concerning efficacy, the percentage of patients with remission and the percentage with a treatment response presented an odds ratio favouring esketamine, which was considered superior to the comparator in this study. 77
REAL-ESK 78 was an observational, retrospective and multicentric study comprising a total of 116 TRD patients treated with esketamine nasal spray from several Italian mental health services, in compliance with the indications provided by the Italian regulatory agency for drugs (Agenzia Italiana del Farmaco, AIFA) and the common clinical practice of TRD management. Little information was available at the time about esketamine’s safety and effectiveness in routine clinical practice, which can face challenging TRD cases (patients with substance abuse issues or with physical and mental health comorbidities), which raise important safety issues and influence clinical decision-making. Despite the real-world conditions, esketamine determined a rapid and sustained reduction of depressive symptoms and improved remission. Most responders remitted later when compared to the TRANSFORM or SUSTAIN studies, probably due to a sample of patients with more severe depressive and associated factors when compared to those in the clinical trials, and the lower compliance usually observed in real-world settings. Also, there were fewer patients prescribed 84 mg in the first weeks of treatment since clinicians in routine clinical practice tend to be rather cautious. There was no evidence of abuse, misuse, withdrawal or gateway activity (i.e. when the use of a psychoactive substance is coupled with an increased probability of the use of further substances) in 3 months of follow-up (Table 1).
Limitations of studies with esketamine
The TRANSFORM 2 phase III clinical trial was the only TRANSFORM study with a statistically significant result for efficacy; the MADRS score decrease from baseline to day 28 favoured the treatment group when compared with the antidepressant plus placebo (difference of least square means = −4.0, SE = 1.69, 95% CI = 27.31, 20.64; p = 0.020). 25 However, there is a lot of discussion in the scientific community about whether this difference is clinically significant in a questionnaire with a score from 0 to 60.79,80 The authors claim the observed −4.0 difference exceeded the minimum clinically significant difference thresholds reported in the literature.81,82 Accordingly, the EMA’s assessment report for Spravato compiled data for several drugs used in the treatment of depression – such as quetiapine, aripiprazole, brexpiprazole, vortioxetine and the combination olanzapine plus fluoxetine – where a difference of −1.19 to −3.05 to a comparator was shown to be considered sufficient to demonstrate efficacy and support the data for esketamine as clinically meaningful. 83 The discussed borderline effectiveness of intranasal esketamine is of utmost importance when weighed against reported side effects in clinical and real-world studies to level the risks and benefits of prescribing this new treatment for TRD.
Concerning the long-term clinical trials with esketamine, the SUSTAIN 1 study enrolled only patients who had previously been randomly assigned to esketamine (not placebo) in a previous short-term trial and achieved stable remission, 80 possibly resulting in an enriched population more likely to respond to the drug. In the post hoc analysis of SUSTAIN 1 and SUSTAIN 2, 84 the study exclusion criteria selected patients with fewer comorbidities than observed in real-world. It is known that pivotal studies must investigate drugs to be marketed initially in a more controlled and homogeneous sample than in the clinical setting, but many experts say that this ‘categorical’ approach does not consider the heterogeneity of patient profiles in TRD.85,86 The diagnosis of TRD may encompass various clinical characteristics and profiles (e.g. depression with comorbid personality disorder, bipolar depression, depression associated with the dysthymic disorder).87,88
Along a similar line, the UK NICE guidelines did not recommend esketamine as a treatment for TRD until its last update in December 2022.79,89 The UK expert committee claims that the clinical evidence is uncertain and that the trials’ evidence excluded people with characteristics of depression like psychosis or recent SI with intent. This limits how well the evidence applies to the English National Health System (NHS) because people having treatment for depression in the NHS may present these excluded comorbidities. 89 However, an advisory panel of psychiatrists and clinical researchers with experience in managing TRD in the United Kingdom convened to develop best practice statements on the use of esketamine nasal spray and recommend its availability as an additional treatment option for TRD to improve long-term outcomes in these patients. 90
As esketamine has been licensed by the Medicines and Healthcare Products Regulatory Agency (MHRA), it remains available for prescription off-label in the United Kingdom. 79
Esketamine use in high-risk populations
Studies and case reports of intranasal esketamine treatment in high-risk TRD patients are summarized in Table 2. In 2023, Chiappini et al. published data from a subsample of 26 patients with SUD, as a post-analysis of the REAL-ESK study. 91 Typical side effects were in line with those previously recorded in both clinical and pharmacovigilance studies,26,92 and all were time-dependent and did not cause significant sequelae. The monitoring of side effects was limited to 2 h as requested by the drug’s SmPC, and they did not require any major medical intervention. The safety of esketamine for the treatment of patients with SUD revealed neither moderate nor severe AEs related to pharmacological interactions of esketamine with any other substance. There were no reports of new-onset drug or alcohol misuse, craving, misuse or diversion of use. Finally, no cases of abuse or misuse of esketamine were reported. An expected limitation of this REAL-ESK post-analysis is the limited number of patients and a short follow-up period, but nevertheless suggesting that esketamine proved to be effective and safe in patients diagnosed with TRD comorbid with a SUD.
Studies with esketamine in high-risk populations.
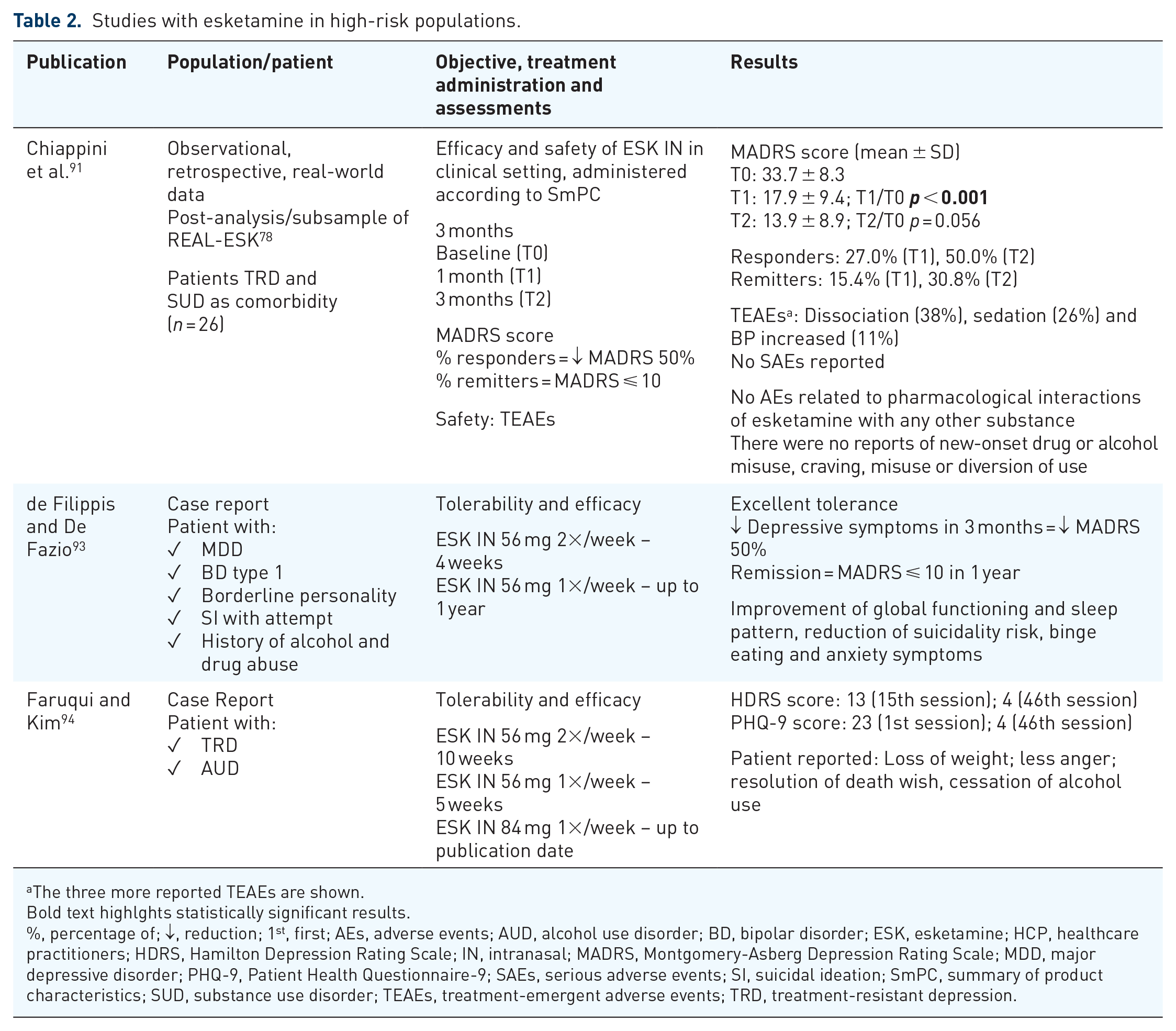
The three more reported TEAEs are shown.
Bold text highlghts statistically significant results.
%, percentage of; ↓, reduction; 1st, first; AEs, adverse events; AUD, alcohol use disorder; BD, bipolar disorder; ESK, esketamine; HCP, healthcare practitioners; HDRS, Hamilton Depression Rating Scale; IN, intranasal; MADRS, Montgomery-Asberg Depression Rating Scale; MDD, major depressive disorder; PHQ-9, Patient Health Questionnaire-9; SAEs, serious adverse events; SI, suicidal ideation; SmPC, summary of product characteristics; SUD, substance use disorder; TEAEs, treatment-emergent adverse events; TRD, treatment-resistant depression.
A case report was published by de Filippis and De Fazio where intranasal esketamine was prescribed to a patient with MDD and several high-risk comorbidities. 93 The patient was additionally diagnosed with BD type I, borderline personality, SI with five previous attempts and a long personal history of alcohol and drug abuse. The esketamine SmPC recommends a careful evaluation of risks and benefits when considering prescribing the drug for patients with BD. The patient showed excellent tolerance and a decrease in depressive symptoms measured by a 50% reduction in MADRS score after 3 months of esketamine use (Table 2). At the 1-year follow-up, clinical remission (MADRS < 10) was registered with the improvement of global functioning and sleep pattern, reduction of suicidality risk, binge eating and anxiety symptoms. These results indicate that intranasal esketamine may be safe even alongside multiple co-treatments in BD with severe psychiatric comorbidities.
A patient with TRD and severe alcohol use disorder (AUD) was treated with 20 sessions of 56 mg intranasal esketamine twice a week before increasing to 5 weekly sessions and then 21 weekly sessions of 84 mg. 94 There was an important improvement in depressive symptoms when assessed by HDRS and Patient Health Questionnaire-9 (PHQ-9) as soon as after 7 weeks from the start of the treatment (Table 2). The authors explain that the increasing dosage was to extend the therapeutic effect throughout the entire treatment period (49 weeks until the publication time). What was more surprising was the report of complete cessation of alcohol use by the second week of treatment due to a decreased desire and impulsivity, which was referred to by the authors as incidental. The patient reported feeling less angry and losing a lot of weight since stopping alcohol consumption. This is the first documented example of the safe and effective use of intranasal esketamine with a resolution of a comorbid severe AUD.
Preclinical research with male rats investigated whether esketamine administration (2.5–10 mg/kg intravenously or intraperitoneally) would inhibit cocaine-seeking behaviour after cocaine use and a subsequent abstinence period. 95 These results deserve to be remarked on when considering high-risk populations diagnosed with TRD since the data revealed a reduced cocaine-seeking behaviour after esketamine administration. Cocaine withdrawal triggers depression and anxiety,96,97 and esketamine’s mood-enhancing properties may act on these factors in the results reported in this study. Further clinical investigation is warranted to confirm these results in high-risk patients with cocaine use disorder since previous work showed a significant anticocaine craving after ketamine administration in short-term (14–28 days) 98 but not in the long-term (6 weeks)99,100 follow-up study visits.
Concern regarding esketamine misuse or diversion
A summary of reports highlighting a potential abuse or misuse of esketamine is provided in Table 3. Some of these concerns come from ketamine’s published data and history as an illicit drug 56 since both molecules are optic isomers and act through the same mechanisms in their antidepressant action. A positive aspect of all previously published data concerning ketamine in clinical (subanaesthetic doses of 0.5–1.0 mg/kg for TRD treatment) and recreational settings (doses reported ranging from 100 to 250 mg multiple times per day to up to 4 g/day) is the identification of several side effects associated with the drug.101–103 Most of these identified effects had a thorough follow-up during the phase III clinical studies with esketamine.
Reports of potential abuse or misuse of esketamine.

Assessed by the Emax (peak effect) in the Drug Liking at the Moment scale, a bipolar VAS from 0 to 100, where 0 = dislike a lot, 50 = neutral, and 100 = like a lot.
Assessed by the Emax (peak effect) in the TDA scale, a bipolar VAS from 0 to 100, scored after the question: ‘Would you want to take the drug you just received again, if given the opportunity?’ 0 = ‘Definitely would not’; 50 = ‘Do not care’; 100 = ‘Definitely would’.
AEs, adverse events; ESK, esketamine; FDA, US Food and Drug Administration; FAERS, FDA Adverse Event Reporting System; IN, intranasal; IV, intravenous; KET, ketamine; LSD, Lysergic acid diethylamide; MADRS, Montgomery-Asberg Depression Rating Scale; PT, preferred term; ROR, reporting odds ratio; SAEs, serious adverse events; SD, standard deviation; SI, suicidal ideation; SUD, substance use disorder; TDA, Take Drug Again; VAS, visual analogue scale.
An analysis of all drug-related AEs in the FDA Adverse Event Reporting System (FAERS) database was performed from March 2019 to March 2020, after 1 year of marketing the drug. 104 The FAERS database contained 962 cases of esketamine-related AEs, reported by healthcare professionals in 760 (79.0%) cases and consumers in 202 (20.1%). The most reported AEs were dissociation, sedation, drug inefficacy, nausea, vomiting, depression and SI (Table 3). No case of misuse was reported in the FAERS database; however, the authors consider that AEs like dissociation, as well as others less frequently reported, such as euphoric mood and hallucination, should alert for potential abuse when using esketamine. These effects have been reported for ketamine in previous studies, which turned out as a popular recreational drug.9,105 However, they are known and reported in the tabulated list of AEs in esketamine’s SmPC with the following frequencies: dissociation, nausea and vomiting as very common; sedation, hallucination and euphoric mood as common; depression as rare, and SI is not identified with any frequency. 20 SAEs corresponded to 389, including 22 deaths. Females and patients receiving antidepressants, polypharmacy, co-medication with mood stabilizers, antipsychotics, benzodiazepines or somatic medications were more likely to suffer from SAEs versus non-SAEs. In addition, this analysis detected rare AEs, which were not reported by regulatory trials (or reported in <5% of patients). 104 Some, such as self-injurious ideation, logorrhoea (known in psychology as a communication problem that causes excessive wordiness and repetitiveness, causing incoherence), depressive symptoms, panic attacks, paranoia, ataxia and mania, are extremely relevant in the depression-related disorders context. According to the authors, the total number of reports was disproportional, and the reporting odds ratios (RORs) had higher values when compared with other drugs such as SSRIs (antidepressants) and lurasidone (antipsychotics).106,107 The ROR for SI was 5–9 times higher than venlafaxine, a second-line antidepressant used as a comparator, posing a serious concern, especially in males, as the disproportionality was stronger. 104
In 2022, Guo et al. updated the analysis performed by Gastaldon et al. 2020 in the FAERS database in one additional year, up to March 2021. 108 The three most reported neurological events were sedation (n = 361, 36.35%), dizziness (n = 130, 13.09%) and headache (n = 70, 7.05%). Of the 361 sedation cases, 248 (68.70%) were reported as serious, including six deaths. These results are consistent with those reported in the clinical trials, where sedation was substantially more frequent in the esketamine groups than in the placebo-treated groups (Table 1).23–25,109 Dizziness and headache are identified with a very common frequency and sedation as common in esketamine’s SmPC. 20 Sedation and loss of consciousness were more likely to be reported as SAEs, whereas dizziness, dysgeusia, taste and cognitive disorders were more likely to be reported as non-SAEs. The previous study 104 reported, in 1 year after esketamine approval, a total of 18 esketamine-related neurological AEs, whereas in this following study, the total number increased to 34, representing 16 newly recorded cases. 108 Among those newly reported were amnesia, loss of consciousness, paralysis, disturbance in attention and unresponsiveness to stimuli. The present study revealed that patients taking a higher dose of esketamine (84 mg) were more prone to develop serious neurological toxicity. Other medications patients may be taking should be considered since benzodiazepines were administered more often concurrently with esketamine than other hypnotics. This increased the risk of developing severe neurological AEs, indicating that non-benzodiazepines hypnotics should be the ones recommended to be administered together with esketamine. Healthcare professionals should continuously monitor esketamine safety and ensure timely reporting of any AEs to spontaneous reporting systems.
In 2022, an additional study reported AEs in international pharmacovigilance databases and websites. The pharmacovigilance databases chosen were the FAERS (2141 reports), EudraVigilance (311 reports) and the French National Pharmacovigilance (27 reports). 110 Among the most reported cases that suggest abuse potential in FAERS and EudraVigilance were dissociation, sedation, euphoric mood and hallucination (Table 3). In addition, reported preferred terms (PTs) related to SUD included withdrawal syndrome, substance abuse, dependence and intentional overdose. Terms like ‘off-label’ use, inappropriate schedule of product administration and incorrect dose administered figured as PTs related to misuse. Specifically for the French database, the most reported AEs also reflected an abuse potential (i.e. hallucination, dissociation, euphoria, derealization, feeling drunk and/or somnolence). None of the cases were serious. It was suggested that the AEs could be related to genetic factors, according to this, some polymorphisms in CYP2B6 and CYP2C9 enzymes could lead to reduced esketamine metabolism resulting in the accumulation and AEs. 111 In this way, the necessity was established for further investigation to determine the role of pharmacogenomics in the prediction of AEs and abuse potential.111,112 The authors claim that no report reflected dependence, withdrawal syndrome or diverted use. Two cases were highlighted; one from a woman who asked to switch her treatment to ketamine due to worsening her depression and SI symptoms. The other case referred to a man who reported developing a protocol to dilute ketamine and transfer it to an empty nasal spray, which he called ‘esketamine’. The man reported that this preparation diminished his cravings for cocaine, but at the same time stopped its use after 3 months due to manic symptoms. The study also searched for content in four web-based forums for indications of esketamine abuse: psychonaut.fr, psychoactif.org, reddit.com and drugs.com. Discussions for psychoactive effects of esketamine use (sensation of floating in the air, visual disturbances) were rated by users as positive or negative in a comparable manner. The discussions of the modality of use revealed mainly questions about mixing esketamine with other drugs such as marijuana, cannabidiol, LSD and psilocybin. The clinical consequences of esketamine related to SUD identified users for whom the esketamine ‘high’ was not good enough and searched for other options, while other users were happy and wished to repeat the administration (Table 3). These results advise careful consideration for intranasal esketamine prescription and strict compliance with the guidelines of closely monitoring patients during the entire treatment.
A 2023 update on esketamine-related AEs reported in the FAERS database analysed 5.061 reports, with significantly more cases involving female (52.20%) than male (29.60%) patients. 113 These reports were most frequently communicated by doctors (35.11%), followed by consumers (32.5%) and pharmacists (29.24%). The researchers also highlighted newly reported psychiatric disorder-related events, such as flashbacks and autoscopy (where an individual, while believing they are awake, sees their own body from an out-of-body perspective). 113 Altogether, the numerous esketamine-related reports registered in the FAERS database underscore the need for regular monitoring of patients treated with the intranasal drug by healthcare professionals.104,108,110,113
A clinical case was reported for a patient who, after 1 month of intranasal esketamine treatment, experienced drug-seeking behaviours and craving for esketamine (Table 4).114,115 The patient also reported increased anxiety, irritability, nervousness, tension, insomnia and a subjective need to shorten the interval between the treatment sessions. The craving worsened again when the intranasal esketamine dosing frequency was decreased from twice weekly to every other week. The authors believe this addictive potential needs to be promptly detected and can be managed with slow esketamine de-titration and combination with bupropion, a protocol that was proven to be effective and safe. 115 The patient maintained stable clinical remission with bupropion alone over the 6-month follow-up period.
Factors contributing to an unlikely abuse of intranasal esketamine after its marketing approval for TRD treatment.

EMA, European Medicines Agency; REMS, Risk Evaluation and Mitigation Strategy; RMP, Risk Management Plan; TRD, treatment-resistant depression.
The esketamine abuse potential exists and justifies a REMS in the United States and a Risk Management Plan in the EMA’s European Public Assessment Report (EPAR), with its use confined to healthcare settings followed by monitoring of key side effects. The EMA’s EPAR also recognizes that the TEAEs reported in the five clinical trials for esketamine marketing approval reveal an abuse potential. 83 In this same EMA assessment report, the results of the study NCT02682225 116 are presented and confirm the abuse potential of esketamine as comparable with intravenous racemic ketamine. The study evaluated 34 healthy subjects who were non-dependent recreational polydrug users of perception-altering drugs. The primary endpoint was a measure of Drug Liking at the Moment assessed by a 100-point visual analogue scale (VAS) and indicated that the values for intranasal esketamine 84 and 112 mg were comparable to those for intravenous ketamine (0.5 mg/kg). As a secondary endpoint, the score in another 100-point VAS scale, Take Drug Again (TDA), reported the same comparable abuse potential.
However, after its marketing approval, several factors make nasal spray esketamine abuse unlikely (Table 4). First, each package contains only one dose (of 28 mg) to be administered under the supervision of a physician. The treatment can only be provided by clinics and hospitals, thereby unable to be self-administered at home in the United States, Canada and Europe.60,117 The monitoring of blood pressure, heart rate and side effects such as dissociation and sedation make it a time-consuming intervention with proper phases of induction, preparation and discharge.90,118 Second, the dosing of intranasal esketamine prescribed for TRD patients is far different than those used recreationally for ketamine, where the most serious side effects were reported. 119 Third, contemporary esketamine manufacturing 33 involves intricate chemical synthesis protocols safeguarded by intellectual property rights. Although some may express concern about extracting ketamine from esketamine, given ketamine’s history as an illicit substance,59,102,120 our search yielded no references to manufacturing or synthesizing ketamine from esketamine. Furthermore, considering that ketamine is a racemic mixture of esketamine plus the R-enantiomer, the possibility of obtaining ketamine from esketamine for illicit use is scarce to non-existent. Thereby, caution is necessary when comparing quantities, as a direct 1:1 dose equivalence cannot exist between esketamine (an isolated enantiomer) and ketamine (a racemic mixture). However, for context, recreational users typically consume 1–4 g per ketamine per ‘session’,59,73 and 1 g corresponds to 37 vials of 28-mg esketamine nasal spray. This process would necessitate numerous vials of esketamine (not to mention the absence of the other enantiomer in the racemic mixture), making the diversion of esketamine for ketamine production practically unfeasible. More importantly, no register of illicit acquisition of esketamine or its tampering for obtaining ketamine or other altered products was found in our search, a trend observed with several new psychoactive substances (NPS), including ketamine.121–124
Some authors claim that the requirements for controlled distribution of intranasal esketamine and administration in a healthcare setting are based only on the theoretical risk of addiction/diversion potential and that there is no clear evidence that the addiction potential of esketamine is more serious than other drugs prescribed with due caution in psychiatry, such as stimulants or sedatives.119,125 Some researchers have suggested that certain groups of patients, such as those with TRD but no history of SUDs, might be more vulnerable to developing such abuse disorders, particularly involving opioids and sedatives. This situation also applies to other medications that have the potential for misuse. However, rather than completely avoiding prescribing these medications, a more balanced approach should involve careful and judicious prescribing practices tailored to each patient’s needs and risk factors. 119 However, recent clinical real-world reports have shown esketamine use among patients with drug dependence without risk of misuse.91,93,94 And despite all the highlighted concerns for a questionable clinical significance and TEAEs related to esketamine intranasal treatment for TRD discussed so far, esketamine eliciting abuse or cravings, as well as misuse, has been rarely reported when prescribed for patients with depression.115,119
Conclusion
Novel and additional treatment options for TRD are needed in psychiatry. Not all MDD patients respond adequately to the current therapies, and, when considering those diagnosed with TRD, patient profile heterogeneity with a plethora of comorbidities, plays a fundamental role in clinical response and the possibility of remission. Those patients with dual depression pose a unique challenge for clinicians, and more so when introducing a novel medication raises concerns. To our knowledge, the risk of esketamine abuse or misuse appears to be minimal. This is foreseeably due to both the inherent characteristics of the drug and the stringent regulation for its prescribing and dispensing. As long as these regulatory measures remain in place, the likelihood of substance abuse associated with esketamine remains very low. While the SUSTAIN 3 study provided insights for up to 4.5 years of continuous esketamine use, there remains a need for real-world multicentre studies with long-term follow-up to consolidate the safety profile for this treatment. These long-term effects of esketamine on the potential for addiction need to be carefully monitored in future prospective studies to further evaluate the risk–benefit balance and adjust clinical practice accordingly.
