Abstract
Background:
Self-treatment of dietary supplements may contribute to interactions and severe side effects. Limited studies have constructed a scale that can measure the disclosure practice of supplements to healthcare providers and the influencing factors.
Objective:
The study aims to investigate the supplement disclosure practice among the public in the UAE using a developed and validated supplement disclosure assessment scale tool.
Design:
A cross-sectional survey study that targeted those residing in the United Arab Emirates (UAE) aged 18 years and above from both genders through an online survey.
Methods:
A novel scale tool was developed and examined for its validity and reliability through three pilot studies.
Results:
The study included three validity and reliability pilot studies before the main study evaluation: pilot 1 (n = 104), pilot 2 (n = 101), pilot 3 (n = 37), and study data (n = 407). A total of 407 respondents provided feedback from which 137 stated that they consumed supplements. A significant indirect effect of healthcare provider initiation of enquiry (HPE) on patient-informing practice (PI) was observed through two mediating variables, patient’s beliefs (PB) and pharmacist counseling regarding supplements interactions (PC) (B = 0.106, t = 2.120, p = 0.03 and B = 0.077, t = 2.011, p = 0.04, respectively). Most respondents were not asked about their supplement consumption by the hospital and community pharmacists (52.94 and 50.74, respectively). Most respondents (54.89%) stated that pharmacists did not counsel them about any possible interaction of supplements with laboratory tests. The mean construct scores were 1.096 for PI, 2.618 for PC, 1.552 for HPE, and 1.412 for PB.
Conclusion:
The instrument demonstrates desirable validity and reliability. The study results revealed a direct effect of PB and PC on the supplement disclosure practice. HPE indirectly affected PI through two mediating variables: PB and PC. The results showed a moderate HPE and PC and an excellent PB and PI construct.
Plain language summary
Why was the study done? Self-treatment and overuse of dietary supplements have been a longstanding concern and have notably escalated during the COVID-19 pandemic. This may contribute to interactions and severe side effects. Limited studies have constructed a scale that can measure the disclosure practice of supplements to healthcare providers and the influencing factors.
What did the researchers do? We studied the supplement disclosure practice among the public in the UAE using a developed and validated supplement disclosure assessment scale tool. We asked several questions to UAE residents through an online survey. The survey explored supplement intake and disclosure practice to physicians and pharmacists. The survey also assessed novel influencing factors of supplement intake disclosure, which contributes to preventing drug-supplement interactions.
What did the researchers find? This study found that the developed tool showed desirable validity and reliability. We found that the novel suggested hypotheses stating that pharmacists’ counseling regarding supplement interactions (PC) with drugs, food, and lab test results directly affected patients’ supplement informing practices (PI) to their healthcare providers. Similarly, the patient’s beliefs regarding the need to inform healthcare providers (PB). Most of the respondents were not asked about their supplement consumption or counseled at all regarding any possible interactions of their supplements, especially with lab test results, which indicates a gap in practice that requires further attention, especially during pandemics in which the self-treatment behavior escalated significantly, and lockdown restricted the accessibility to healthcare institutions. Interestingly, healthcare providers’ initiation of enquiry (HPE) did not directly affect disclosure but indirectly through two mediating factors, PB and PC. The scale results showed moderate HPE and PC and excellent PB and PI practices in the UAE.
What is the impact of the findings? The study has identified important factors influencing the disclosure practice of supplement consumption. The current scale provides a quality healthcare service evaluation tool that can be used in healthcare facilities to improve the counseling and communication services of supplement consumption between patients and healthcare providers to assure drug safety and avoid interactions.
Introduction
Dietary supplement intake is consuming one or more minerals, vitamins, amino acids, and traditional herbal medicines. 1 The phenomenon of self-treatment and overuse of dietary supplements has been a longstanding concern and has notably escalated significantly during the COVID-19 pandemic.2–6 In addition to the issue of self-treatment, a significant and enduring issue exists, namely, the non-disclosure of supplement intake to healthcare providers. This phenomenon has been observed for years and contributes to the ramifications of self-treatment practices. 7 This conduct raises significant safety concerns as it can lead to serious health issues, such as interactions, 8 improper dose intake, and various other complications. 9 Furthermore, the misuse and overconsumption of dietary supplements can result in serious adverse effects such as seizures, allergies, and some hematological issues. 10
At this juncture, providing effective counseling and communication by healthcare providers is pivotal and vital. 11 The predicament lies in the fact that in countries such as the United Arab Emirates (UAE), pharmacist counseling is limited to pre- and post-meal supplement intake, in contrast to other countries such as the United Kingdom, where pharmacists are more adept at counseling regarding supplements, with skills to identify interactions and side effects.12,13 In the existing body of literature, prior investigations have primarily concentrated on appraising medication disclosure 14 or the practice of supplement disclosure in general. Specifically, these studies have examined the names of taken supplements and the reasons for non-disclosure.15,16 Alternatively, specific research endeavors have aimed to construct surveys to assess the pharmacists’ general counseling practices, though lacking a quantifiable audit scale and a direct connection to non-disclosure. 17 Notably, some endeavors have been undertaken to formulate an index designed to assess the relative importance of factors influencing the disclosure and non-disclosure of complementary medicines. However, this index has predominantly focused on assessing the relative importance of reasons for non-disclosure. It has not furnished an auditing instrument for healthcare quality service evaluation in health institutions and general scale evaluation. 18
Given the critical nature of the issue and the foregoing gap in the literature connecting patients’ non-disclosure of supplement consumption to practical actions exerted by physicians and pharmacists, there arose an urgent demand for an auditing tool capable of assessing both healthcare providers’ interventions and patients’ beliefs and disclosure practice, thereby ensuring health service quality and public safety. The current tool assesses the physicians’ and pharmacists’ initiation of supplement intake inquiry (healthcare providers enquiry (HPE)) and pharmacists’ counseling on interactions and maximum doses (PC). It directly correlates these actions to patients’ informing practice (PI) and beliefs regarding disclosure (PB). The present tool represents an inaugural audit scoring instrument capable of collecting data on supplement disclosure from patients and evaluating the healthcare providers through the lens of patient experience. The study aims to investigate the supplement disclosure practice among the public in the UAE using a developed and validated supplement disclosure assessment tool.
Methods
Study design and setting
This study conforms to the STROBE guidelines for reporting information related to cross-sectional studies (see Supplemental Materials). 19 The current study employed a cross-sectional online survey-based design. The study included two phases: instrument development and validation and the main study data collection. This approach aligns with recent trends in research methodologies during the COVID-19 pandemic. 20 The current study followed the validity phases as per the literature recommendations.21–24 Three pilot studies were performed before the main study data collection commencement: pilot 1, pilot 2, and pilot 3 (test–retest) study, as shown in Figure 1. Pilot 1 was held to perform factor analysis and item reduction to obtain participants’ initial feedback and comments and to identify the number of items to be added to the developed tool. Following the necessary modifications based on pilot 1 results (n = 104), pilot 2 (n = 101) was performed. Pilot 2 included another set of collected data to confirm the clarity of the instrument items and perform convergent validity, discriminant validity, and composite reliability analyses. Pilot 3 (n = 37) was a test–retest reliability study to confirm the instrument’s reliability across different time intervals, indicating internal consistency.
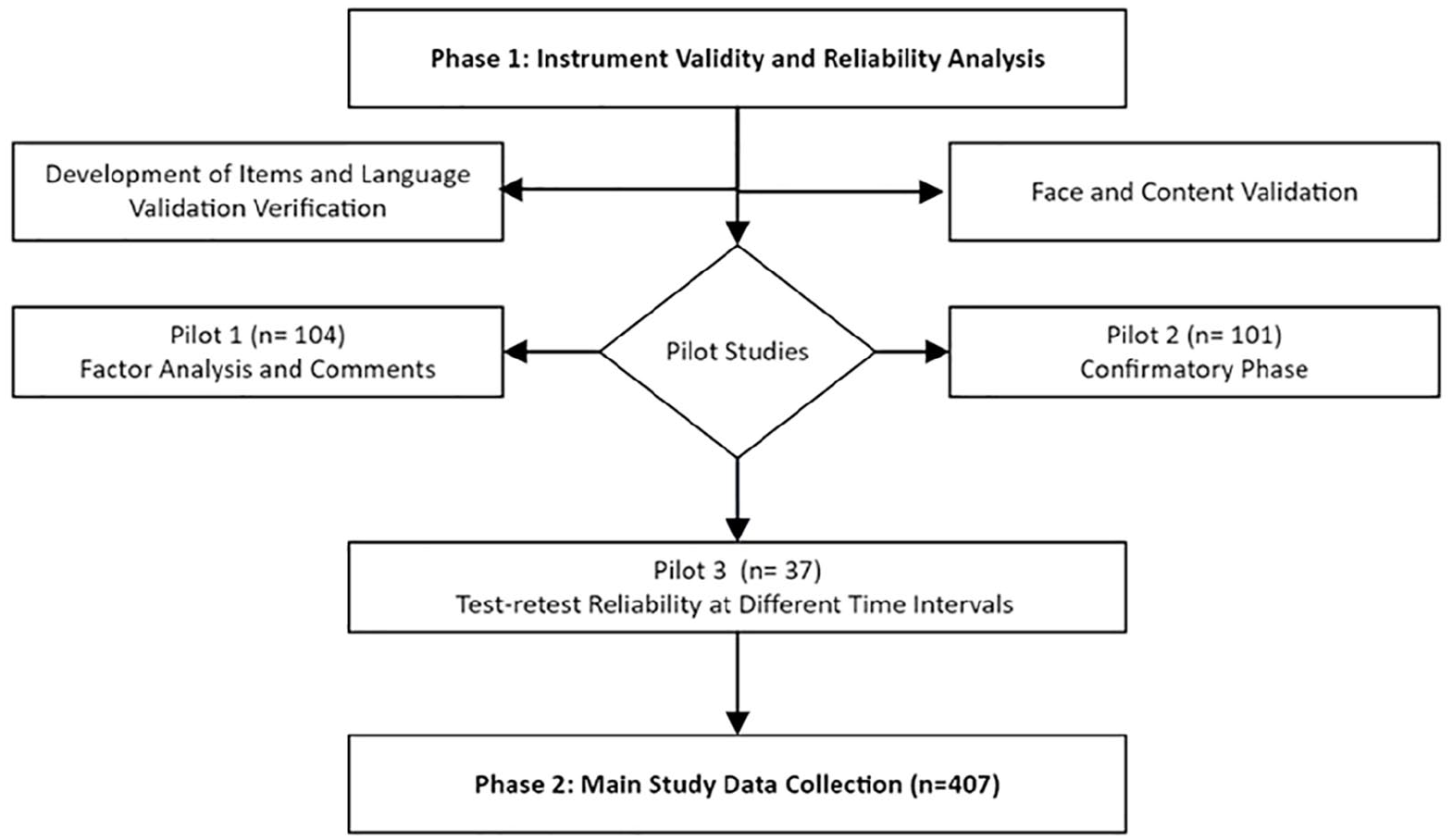
Flow chart of study phases including instrument validity and main study data collection.
The participants were recruited through an online platform (Google Forms) for data collection, targeting the general public in the UAE. The survey link was sent to respondents through social media platforms, and participation was voluntary. Regarding the pilot studies’ collection period, pilot 1 was held in July 2022, pilot 2 in October 2022, and pilot 3 in November 2022. The recruitment of participants and data collection for the main study were from March 2023 to December 2023.
Study participants and instrument variables
The study participants included individuals residing in the UAE aged 18 years and above from both genders. Individuals who expressed unwillingness to participate were excluded. The study used the well-known Cochran adjusted formula for finite populations to calculate the minimum required sample size when the population size is known. 25 The Z-score represents the confidence interval extracted from the Z-table, which was set as 95%. The margin of error “e” value was set as 5%. The ideal sample size would be about 385 respondents. The instrument items were arranged and presented as dichotomous questions. The study instrument consists of four primary constructs besides the general participants’ characteristics and supplement intake. The PC construct includes five items: PC16, PC17, PC18, PC19, and PC20. The HPE construct includes three items: HPE13, HPE14, and HPE15. The PB construct includes two items: PB11 and PB12. PI construct included PI9 and PI10.
Statistical analysis
Descriptive variables and reliability analysis were analyzed using IBM SPSS statistics version 25 manufactored by International Business Machines Corporation (IBM). Factor analysis was performed using the software “FACTOR” version 12.01.02.WIN64 manufactured by Rovira i Virgili University. The measurement model structural equation modeling analysis and direct and indirect effects were tested using the software SmartPLS version (4.0.8.4) manufactured by SmartPLS GmbH. 26 A p-value of <0.05 was considered a significant value. The study included two analysis phases: (1) instrument validation and reliability analysis and (2) the main study data analysis. Before the pilot investigation, the face and content validity were analyzed. Experts assessed the instrument’s clarity, relevance, representativeness, and consistency. The content validity index (CVI) assesses the extent to which an item or measure is content valid by a panel of experts. The items were considered satisfactory if the CVI value was between 0.71 and 1.00. 27
A minimum Cronbach’s alpha (CA) value of 0.7 was considered satisfactory. 28 Convergent validity was evaluated through CA and factor analysis, as detailed in Supplemental Material A. The higher the CA, the better the convergent validity of the measures. 29 Regarding the test–retest study, the intraclass correlation coefficient (ICC) was used to determine the reliability, considering that values below 0.4 exert poor agreement followed by good and excellent agreement (0.4 > ICC < 0.75 and >0.75, respectively). 30 All performed tests’ detailed statistical measures and metrics are provided in Table 1 and Supplemental Material B.
Summary of performed validation and reliability analysis tests.

In addressing the issue of missing values within our dataset, we implemented a multifaceted approach to enhance the integrity and comprehensiveness of our data analysis. Initially, we mandated the completion of most of the items in the online survey to minimize the occurrence of missing data. Subsequent to this preliminary step, we explored any missing values and classified the observed missing values into two distinct categories: non-agreements to participate and structurally missing data. Structurally missing data refers to missing values caused by filter questions, which are not missing values but are due to not applicable branched questions. The former category was addressed through the listwise deletion method. However, we acknowledge that this approach led to a slightly reduced sample size, as approximately 1% of the cases were excluded due to missing values. Structurally missing data were not arbitrarily omitted but were inherently linked to the deliberate design of the survey instrument. A filtering mechanism was employed. Moreover, some respondents in the final study did not respond to certain questions, leading to missing data. Still, the provided results were properly calculated and indicated variables with missing data.
Several measures were implemented to mitigate potential biases inherent in online survey-based studies. To counteract selection bias, follow-up reminders were deployed through social media platforms several times, augmenting the response rate. In addition, the study was shared through multiple platforms. In addition, when constructing the survey instrument, including leading questions was meticulously avoided. This was achieved through a rigorous pre-testing phase, which included three pilot studies and involved experts’ and the target population’s feedback. Ethical considerations were stringently adhered to, with participants informed of the study’s intent, their rights to anonymity and confidentiality, and the voluntary nature of their participation. The tool detailed item numbers and domains are provided (see Supplemental Material A).
Results
All validity and reliability tests performed were summarized and clarified for all phases in Table 1 and Figure 1. Detailed validity and reliability results are provided (see Supplemental Material C). A panel of experts performed face and content validity. The results showed that the items met the minimum requirements for CVI and Kappa scores for consistency, representativeness, relevance, and clarity, as recommended in the literature, 27 confirming the instrument’s content and face validity. A CVI of at least 0.8 is acceptable. 44 Kappa value indicates inter-rater agreement that adjusts for chance agreement and supplements CVI.45,46 The values above 0.74, between 0.60 and 0.74, and the ones between 0.40 and 0.59 are considered excellent, good, and fair, respectively.46,47
Pilot 2 included another set of collected data to confirm the clarity of the instrument items by collecting recipients’ feedback and performing convergent validity, discriminant validity, and composite reliability analyses. The results revealed that all latent variables have an average variance extracted (AVE) value above 0.5, and the factor loadings are satisfactory (Table 1). 34 Pilot 3 was performed, the instrument was assessed for intraclass correlation test–retest reliability analysis, and the results ranged between 0.4 and 1. 30 A cutoff point value of 0.4 for the ICC was met (Table 1).
The evaluation of the proportion of variance of endogenous variables explained by Pearson’s coefficient (R2) indicated the quality of the model (Table 1). Model fit analysis was performed. Fit indices revealed that the model meets the cutoff value requirements.40,42,43 Absolute fit measures were evaluated, and the model chi-square/df showed a value of 1.68, which is less than 3 and considered acceptable. Fit is adequate when the ratio of the chi-square test statistic to the degrees of freedom (χ2/df) is not larger than three.40,41
The goodness of fit index showed a value of 0.91, which is more than 0.9 and reflects an appropriate model fit.40,42 In addition, the Steiger–Lind root means the square of approximation was observed to be 0.08, which is 0.08 or less and is considered appropriate.40,42 The standardized root means a square residual was also measured. This test indicates to what extent the model does not fit the data. The maximum value equals 1, and it is preferred to have values less than 0.08.40,42,43 The results revealed a value of 0.078, which is considered acceptable.
During the pilot 2 phase, a scalability analysis and structural equation modeling analysis were performed for the constructs to confirm the direct and indirect effect of constructs representing the influencing factors of supplement disclosure (PB, PC, and HPE) on the PI (Table 2). A direct effect was observed between the construct PB and PI and between the construct PC and PI (Table 2). Interestingly, the results of the present study did not reveal a direct effect (p = 0.11) between HPE and PI, as assumed in general statements in previous studies. 7 According to available data, we could not observe an effect between HPE and PI (p = 0.11). It is interesting to report that after analyzing the total effect between HPE and PI, the results revealed a significant effect of HPE on PI (p = 0.00). It was encouraging to perform a mediation analysis, which showed an indirect effect of HPE on PI through two mediating variables, namely, PC and PB. All constructs’ scalability coefficient (SC) was excellent and met the cutoff point of above 0.6 39 for construct HPE, PC, PI, and PB (0.92,0.81,0.82, and 0.84, respectively).
Direct and indirect effects of influencing factors on construct PI and mediation analysis in the measurement model.

HPE, healthcare providers enquiry; PB, patients’ beliefs; PC, pharmacists counseling; PI, patient-informing practice.
The main study data collection included 407 respondents. The mean age of participants was about 26 years. This could be attributed to the general population distribution in UAE, as 68.62% of UAE’s population falls within the age group between 25 and 54 years old. 48 A slightly similar response was received from both genders, as females represented 56.3%, and 43.7% of responses were received from males. Most participants were single, followed by married and divorced/widows/widowers (70.8%, 27.3%, and 1.9%, respectively). Most participants were educated, holding bachelor’s, high school, or postgraduate degrees, and less than high school (65.8%, 22.6%, 11.1%, and 0.25%, respectively). This distribution could be attributed to the population’s educational level, as it was observed in the latest 2023 national statistics that 41.18% of the population has a university degree or a postgraduate degree. 49 Most participants were unemployed, 63.1%, as shown in Table 3.
Characteristics of the pilot study and main study respondents.

Furthermore, the participants’ feedback regarding the healthcare providers’ initiation of enquiry about supplement consumption (HPE) was assessed. Most respondents (58.82%) revealed that the physicians initiated the enquiry about their supplement intake. Interestingly, most respondents revealed that they were not asked about their supplement consumption by the hospital pharmacist and the community pharmacist (52.94 and 50.74, respectively). The mean HPE construct value is 1.552, which reflects a moderate healthcare workers’ initiation of enquiry about supplement consumption. The study tool and a detailed scoring scheme are provided (see Supplemental Materials C and D). The literature revealed that the duration of consultation was insufficient, which limited the discussion about supplement use with physicians.16,18 Obstacles related to healthcare workers may include health professionals’ not being interested in patients’ responses,16,50 patients’ fear that a physician would discourage the use of supplements,16,50 and an experience with a physician who discouraged the use of supplements in the past,16,50 as shown in Table 4.
Disclosure practice to healthcare providers, initiation of enquiry, and the beliefs regarding the need to inform.

n = 136 due to missing no response values.
PC regarding supplemental maximum doses and interactions (PC) was assessed. Most respondents (66.91%) stated that the pharmacist had informed them about the maximum dose of supplements. Still, the majority (53.33%) were not informed about the action to be taken if an excessive supplement dose was consumed. Most respondents were counseled about supplements’ food and drug interactions (54.07% and 50.74%, respectively). By contrast, most respondents (54.89%) stated that pharmacists did not counsel them about any possible interaction of supplements with laboratory tests. The mean PC construct value is 2.618, which reflects moderate PC regarding supplement maximum dose and interactions, as shown in Table 5. One solution to ensuring patient safety in supplements could be to mandate education about the supplements and their interactions with other medicines and diseases for all providers. 41
Pharmacist counseling regarding supplement interactions and maximum doses.
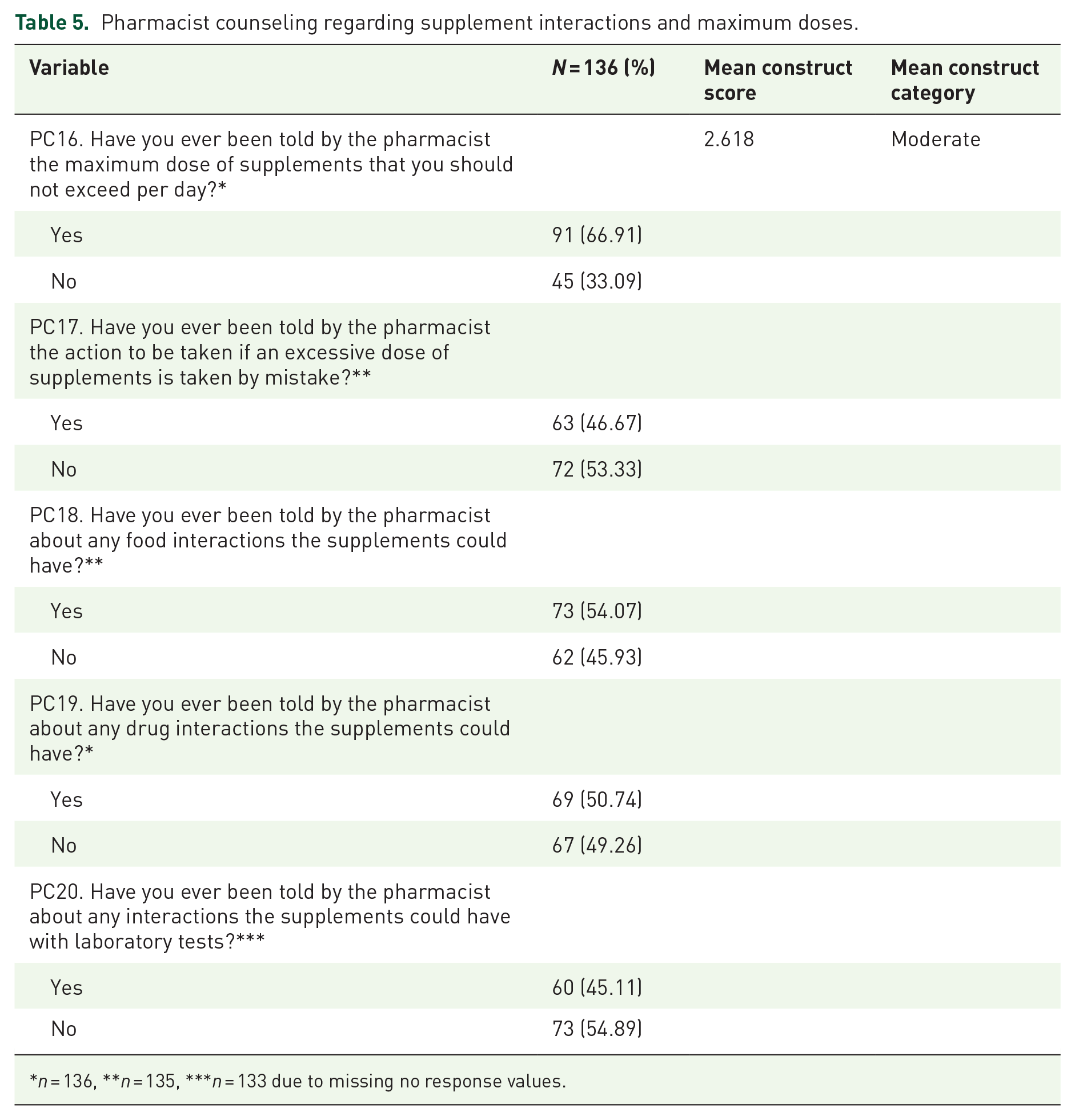
n = 136, **n = 135, ***n = 133 due to missing no response values.
Discussion
The current study emphasizes that patients’ hesitancy to inform their healthcare providers about their consumption of dietary supplements is a critical factor in controlling and preventing supplement self-treatment and its consequences. Previous studies focused mainly on developing instruments to assess medication adherence and measured patients’ counseling in general, and the evidence supporting their validity varies. 52 Other studies have worked on creating an instrument to assess pharmacists’ general counseling related to dietary supplements without significant details about disclosure factors. 17 Many studies either include observational study questions, fail to mention the underlying principles that guided the instrument’s creation and its components, or do not provide a scale measurement tool besides a limited focus on the influencing factors of patient disclosure. 52
The literature indicated several reasons for patients’ non-disclosure, such as healthcare providers’ interest, knowledge, perspectives regarding the need for supplement intake, and time constraints. 53 The current study focused on identifying other reasons that could directly reflect the communication-related reasons for non-disclosure related to healthcare providers and patients to fit an audit scoring tool intended for a quality service assessment. Four constructs were tested in our model for their validity and reliability. Three variables were identified from the literature as being reasons for non-disclosure, namely, patients’ beliefs regarding the need to disclose (PB), healthcare providers’ initiation of inquiry about supplement consumption (HPE), and patients’ current informing practice (PI). 53 These were only identified and listed as reasons for non-disclosure and have not been used before as an audit tool for quality service assessment. The questions addressing the three variables were developed and validated. The significance of the three construct variables was tested statistically and was proven for its significance in the disclosure of supplements, as summarized in Table 2. The novelty lies in that the questions about the three variables were developed and designed to evaluate the disclosure of supplements to physicians, community pharmacists, and hospital pharmacists. In addition, connecting these constructs and examining these variables’ direct and indirect effects represent a novel validated scale.
The study respondents were asked about their informing practice of supplement intake to healthcare providers (PI), their beliefs about the necessity to disclose their supplement intake to healthcare providers (PB), the healthcare providers’ initiation of enquiry about supplement consumption (HPE), and pharmacist counseling regarding supplements maximum dose and interactions (PC).
Supplement consumption was observed by 33.7% of study participants (Table 3). Most participants who consumed supplements (59.56%) stated that they informed their physician about their supplement consumption, while (50%) said that they told the pharmacist. The mean PI construct score is 1.096, which reflects an excellent disclosure practice. The participants’ beliefs about the necessity of disclosing their supplement intake to healthcare providers (PB) were examined. The results showed that most respondents believed it necessary to disclose supplement consumption to physicians and pharmacists (72.79% and 68.38%, respectively). The mean PB construct is 1.412, which reflects excellent beliefs regarding the necessity of informing healthcare providers about supplement consumption. Previous studies stated certain obstacles related to PB, such as PB that the supplements were safe and would not interfere with the treatment,18,50 patients did not consider supplements as medicines, 50 PB that is not essential to report and that the physician does not need to know, 50 patient’s concerns about a negative reaction from their physician, and PB that the physician had less knowledge about supplement type. 16 Therefore, measuring the PB regarding disclosure is essential to follow up on improvement if any practice change is introduced to mitigate these issues.
The current construct, PC, was suggested by the current study as another novel reason that can be scored in an audit tool and claimed to contribute to the disclosure practice of supplements. This includes pharmacists’ counseling regarding maximum doses of supplements, actions taken if exceeded, and interactions with drugs, food, and laboratory results. The current novel variable claims that if patients were properly counseled regarding drug, food, and laboratory results interactions of supplements as well the maximum dose intake, they would have developed conscious awareness about the seriousness of supplement self-treatment and the potential risk of non-disclosure to their healthcare providers and would have disclosed the intake of supplements. The current novel variable theory was based on psychological theories related to human behavior.54–56 A psychological study reported a relationship between fear and risk-taking. 57 In addition, another study stated that behavior depends on the value placed by an individual on a particular outcome and also on the individual’s estimate of the likelihood that a given action will result in that outcome. 58 PC variable significance was tested statistically and was proven for its significance in the disclosure of supplements (Table 1).
Previous studies emphasized the need for an instrument to use participants’ responses to assess whether healthcare providers enquire about supplement intake. 59 Limited studies focused on getting the patients’ feedback on counseling provided by pharmacists related to supplement interactions and maximum doses. Therefore, the present study adds to the literature by providing a tool to assess this particular issue and a scaled evaluation of other influencing factors. Limited studies exerted mediation analysis on the patient informing the practice of supplements. The current results emphasize the importance of considering PB and involving them in pharmacy practice change and improvement plans.
Limitations
The limitations of the present study include, first, the fact that indirect reasons affecting the informing behavior were not covered. The current study focused on identifying other reasons that could directly reflect the communication-related reasons for non-disclosure related to healthcare providers and patients to fit an audit scoring tool intended for a quality service assessment. Second, nurses were excluded and not included. As per the medical practice regulations in the UAE, nurses are not allowed to prescribe and have limited direct medication counseling roles, especially for outpatients. Third, the psychometric properties of the measurement scale need to be evaluated further among different age groups and users and in various environments. Fourth, the supplement intake was self-reported by participants and could not be verified due to the nature of the study design.
Conclusion
To conclude, the instrument demonstrates desirable validity and reliability. The constructs’ items passed the face validity, content validity, construct validity, convergent validity, and discriminant validity. The current study has proven that the patients’ informed practice regarding their intake of dietary supplements is directly affected by their beliefs and the pharmacists’ counseling regarding interactions and doses. Interestingly, healthcare providers’ initiation of inquiry about supplement consumption did not directly affect the patient-informing behavior but affected it indirectly through two mediating variables: PB regarding the need to inform and pharmacists’ counseling regarding interactions. The results showed a moderate HPE and PC and an excellent PB and PI construct. The end users of the instrument are the general public and the patients visiting healthcare facilities. It can be used by researchers, healthcare institutions, and policymakers for current and future follow-up and evaluation of improvement in pharmacy practice.
Supplemental Material
sj-docx-1-taw-10.1177_20420986241294150 – Supplemental material for Patient-centric decision-making in supplements intake and disclosure in clinical practice: a novel SIDP-12 tool to prevent drug–supplement interaction
Supplemental material, sj-docx-1-taw-10.1177_20420986241294150 for Patient-centric decision-making in supplements intake and disclosure in clinical practice: a novel SIDP-12 tool to prevent drug–supplement interaction by Sabrina Ait Gacem, Hasniza Zaman Huri, Izyan A. Wahab and Abduelmula R. Abduelkarem in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986241294150 – Supplemental material for Patient-centric decision-making in supplements intake and disclosure in clinical practice: a novel SIDP-12 tool to prevent drug–supplement interaction
Supplemental material, sj-docx-2-taw-10.1177_20420986241294150 for Patient-centric decision-making in supplements intake and disclosure in clinical practice: a novel SIDP-12 tool to prevent drug–supplement interaction by Sabrina Ait Gacem, Hasniza Zaman Huri, Izyan A. Wahab and Abduelmula R. Abduelkarem in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-3-taw-10.1177_20420986241294150 – Supplemental material for Patient-centric decision-making in supplements intake and disclosure in clinical practice: a novel SIDP-12 tool to prevent drug–supplement interaction
Supplemental material, sj-docx-3-taw-10.1177_20420986241294150 for Patient-centric decision-making in supplements intake and disclosure in clinical practice: a novel SIDP-12 tool to prevent drug–supplement interaction by Sabrina Ait Gacem, Hasniza Zaman Huri, Izyan A. Wahab and Abduelmula R. Abduelkarem in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-4-taw-10.1177_20420986241294150 – Supplemental material for Patient-centric decision-making in supplements intake and disclosure in clinical practice: a novel SIDP-12 tool to prevent drug–supplement interaction
Supplemental material, sj-docx-4-taw-10.1177_20420986241294150 for Patient-centric decision-making in supplements intake and disclosure in clinical practice: a novel SIDP-12 tool to prevent drug–supplement interaction by Sabrina Ait Gacem, Hasniza Zaman Huri, Izyan A. Wahab and Abduelmula R. Abduelkarem in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
