Abstract
Non-cystic fibrosis bronchiectasis is a long-term lung disease characterised by abnormal dilatation of the bronchi, with patients experiencing chronic productive cough and recurrent exacerbations. Currently, there are no licensed drugs for use in bronchiectasis while clinical trials have been conducted to either test new drugs or repurpose existing ones. These drugs target the underlying pathophysiology of bronchiectasis which is known to include infection, inflammation, mucus hypersecretion and retention. Most of the drugs used in daily clinical practice for bronchiectasis are off-label with no randomised trials exploring their safety. This review aims at exploring the safety profile of drugs frequently used in clinical practice to manage bronchiectasis, including antibiotics (e.g. macrolides, aminoglycosides, polymyxins, fluoroquinolones, aztreonam), mucoactive therapy (e.g. hypertonic saline, mannitol, DNase and carbocisteine), anti-inflammatory therapy (inhaled corticosteroids) and drugs currently in development for use in bronchiectasis (e.g. brensocatib, benralizumab and itepekimab).
Plain language summary
This review aims to detail the safety aspects of drugs that are currently prescribed to patients with bronchiectasis. These drugs are used in bronchiectasis without some of the high quality trials seen for other lung conditions. The drugs used have shown clinical benefits in patients who are suffering from infective exacerbations or worsening of the disease. The idea behind the use of these drugs is that they target the pathological processes in bronchiectasis such as inflammation, infection and excess mucus production. In this review, we have included the results from clinical trials that assessed the use of antibiotics (both oral and inhaled) during pulmonary infections and long-term antibiotics to prevent infections. Mucus production is a major symptom of bronchiectasis, and hence the drugs that target mucus secretion and consistency are used in an attempt to improve the quality of life and prevent infections. Inflammation is a key component of bronchiectasis, and we report on the safety of inhaled steroids in bronchiectasis. Some new drugs are currently being tried in clinical trials worldwide and are discussed. The occurrence of multiple other medical problems are recognized in people living with bronchiectasis has been seen to increase symptoms and linked with higher infection rates and hospitalizations. This means patients are often on lots of different medications for multiple conditions; we highlight the importance of considering the fact these drugs in combination can lead to potential issues and side effects linked to polypharmacy.
Introduction
Non-cystic fibrosis bronchiectasis is an inflammatory lung disease characterised by chronic cough, sputum production and recurrent infective exacerbations resulting in a decreased quality of life and increased mortality. 1 The prevalence of bronchiectasis has increased worldwide over the past 20 years, partly due to the increased availability of advanced radio imaging and an increase in the disease prevalence in the elderly due to longer life expectancy. 2 However, there is limited data available to quantify the burden of bronchiectasis worldwide. A systematic review by Polverino et al. showed that the prevalence of non-cystic fibrosis bronchiectasis in the general population was calculated as 39.9 cases per 100,000. 3
Ever since Laennec first described this disease in 1819, physicians have tried to understand the pathophysiology of bronchiectasis, yet the disease remains poorly understood. Bronchiectasis is a disease without a robust evidence base to inform care and without any specifically licensed drug therapies. 4 Multiple clinical trials have been conducted in recent years to either test new drugs or explore the efficacy and safety of existing drugs to repurpose for use in bronchiectasis. These drugs target the underlying pathophysiological mechanisms of the disease to prevent/suppress acute and chronic bronchial infection, improving mucociliary clearance and reducing the impact of structural lung disease. 5 A systematic review is required on the safety profile of these drugs as they are used off-label and have issues with cost and tolerability.
Novel drug therapies are being developed due to a better understanding of the disease pathophysiology. Although there are multiple aetiologies for bronchiectasis, most of them lead to airway inflammation, remodelling and dilation with infection to varying degrees. Impaired mucociliary clearance and mucus retention result in an imbalance of host defences making the airways vulnerable to infection and inflammation. Repeated episodes of infection and inflammation result in permanent airway dilatation and remodelling, leading to worsening clinical features and quality of life. Novel drugs such as brensocatib (DPP-1 inhibitor), itepekimab (IL-33 monoclonal antibody) and benralizumab (IL-5 receptor monoclonal antibody) are being studied to target these pathophysiological mechanisms. 6
A major challenge associated with novel drugs, as well as those used off-label, is identifying their safety profile and mitigating any adverse effects to ensure patient safety. Clinicians must focus on possible adverse effects of both novel and existing drug therapies especially in patients with coexisting heart, kidney or liver disease. In addition, as bronchiectasis often requires long-term therapy, the focus needs to be on reducing any possible long-term drug-related complications.
Another challenge whilst targeting the pathophysiological mechanisms of bronchiectasis includes avoiding polypharmacy and its associated complications. Bronchiectasis has a higher prevalence in the older population and with increased global life expectancies, a major increase in the burden of bronchiectasis is expected to be seen.7,8 The impact of multimorbidity (i.e. multiple long-term conditions (MLTC)) on bronchiectasis disease severity and prognosis is poorly understood. McDonnell et al. constructed the Bronchiectasis Aetiology Comorbidity Index demonstrating the significant impact of MLTC. 9 Marsland et al. conducted a systematic scoping review to summarise the existing literature and identify any deficits in MLTC. Data from 40 eligible studies showed that chronic obstructive pulmonary disease (COPD) and hypertension (pooled mean 35% and 34%, respectively) were the most prevalent multimorbidities followed by asthma (pooled mean prevalence 30%) in bronchiectasis patients. 10 There is scant literature on polypharmacy in bronchiectasis. Due to a multi-faceted treatment approach and high rates of MLTCs, this patient population is likely to be affected by polypharmacy. 9
Aim
This review aims to explore the safety profile of novel and existing drugs used in bronchiectasis.
Methods
We present a narrative review of the safety profile of drugs currently used in non-cystic fibrosis bronchiectasis. An extensive literature search was conducted on MEDLINE (PubMed), Google Scholar and Cochrane to identify clinical trials conducted in non-cystic fibrosis bronchiectasis to date. The following Medical Subject Headings (MeSH) terms and their synonyms were used: ‘anti-inflammatory therapy’, ‘clinical trial’, ‘mucoactive therapy’ OR ‘mucolytics’, ‘multimorbidity’, ‘nebulised antibiotics’, ‘non-cystic fibrosis bronchiectasis’, ‘oral macrolide’, ‘phase II clinical trial’ OR ‘phase III clinical trial’, ‘polypharmacy’ and ‘safety profile’ OR ‘adverse effects’ OR ‘adverse events of special interest’. Clinical trials were categorised according to the drug class and initial screening was done by Henil Upadhyay (HU) and Anthony De Soyza (ADS) to assess the suitability of the articles for inclusion in the review. All phase II and phase III clinical trials involving human subjects were included in the review, while animal studies were excluded. Each selected study was read in-depth to understand the study setup, the patient population, primary and secondary endpoints and the key findings from the trial by HU and ADS. Data were extracted from these trials and presented in a concise format. Authors Stefano Aliberti, Katy Hester, James D Chalmers and Andy Husband contributed to reviewing the initial draft, suggesting further references, making necessary revisions and approving the final draft of the manuscript.
Systemic oral antibiotics
Permanently dilated and damaged airways with impaired mucociliary clearance encourage the growth of pathogenic microorganisms in the airway, particularly Haemophilus influenzae and Pseudomonas aeruginosa. 2 Other organisms include Moraxella catarrhalis, Staphylococcus aureus and Enterobacteriaceae sp. Persistent infection of the respiratory tract by these pathogens results in increased frequency and severity of pulmonary exacerbations, decreased quality of life, increased hospitalisations and increased healthcare costs. 11 Long-term macrolide therapy (more than 3 months) aims to reduce these pulmonary exacerbations and is recommended in guidelines for patients who have three or more exacerbations in a year.12,13
Bacterial colonisation and neutrophil-mediated inflammation in the airways result in structural damage and further impairment of mucociliary clearance which leads to increased bacterial load. According to this hypothesis, if bacteria are the primary drivers of inflammation in bronchiectasis, then the use of long-term antibiotics as a suppressive therapy can decrease airway inflammation, enabling airway healing and improving long-term morbidity. A study by Chalmers et al. established a direct relationship between airway bacterial load and airway and systemic inflammation, exacerbations and health-related quality of life in stable patients with non-cystic fibrosis bronchiectasis. Hence, suppressive antibiotic therapy is an important aspect of bronchiectasis management to reduce airway inflammation and improve the quality of life. 14
Azithromycin and erythromycin ethyl succinate are the macrolide therapies studied within robust randomised control trials (RCT; Table 1):
RCTs studying long-term macrolide therapy in bronchiectasis.

RCT, randomised control trial.
Gastrointestinal symptoms included nausea, vomiting, diarrhoea, epigastric discomfort and constipation.
In the EMBRACE multi-centre clinical trial, 141 subjects were randomised to either the azithromycin (500 mg three times per week; n = 71) or the placebo group (n = 70). The most frequent adverse events included gastrointestinal (GI) symptoms (diarrhoea, nausea/vomiting, epigastric discomfort and constipation). These events were reported more frequently in the azithromycin group (n = 19, 27%) than in the placebo group (n = 9, 13%). Only two subjects in each study group discontinued the study drug because of GI adverse events. No symptomatic hearing impairment was reported in this study, although the investigators did not perform any audiometry. The development of GI adverse effects in the placebo group should be taken into consideration by clinicians while discontinuing azithromycin therapy due to fear of GI adverse effects. GI symptoms appeared in this patient population de novo without previous macrolide treatment. Also, there is no mention of whether the participants were counselled to take the drug with food as this simple change is known to substantially reduce GI irritation. 15
The BAT multi-centre trial studied the efficacy and safety of daily azithromycin therapy (250 mg/day) for 52 weeks in bronchiectasis patients. In all, 83 subjects were randomised into treatment (n = 43) and placebo (n = 40) groups. The most frequent adverse effect noted in this trial was nausea in both azithromycin (n = 6, 14%) and placebo group (n = 6, 15%), followed by rash (n = 8, 19% treatment group and n = 4, 10% placebo group). Diarrhoea was noted more frequently in the azithromycin group (n = 9, 21%) compared to placebo group (n = 1, 3%). Self-reported transient hearing loss/tinnitus was noted in 5 (12%) and 4 (10%) subjects in the azithromycin and placebo groups, respectively. These adverse effects were reported to be mild and did not lead to discontinuation from the study. 16
The effect of low-dose, long-term erythromycin (400 mg of erythromycin ethyl succinate twice daily) was studied in the BLESS trial. In all, 117 subjects were randomised (59 erythromycin and 58 placebo group) in this study. Only 107 patients in total completed the study. Overall, the rate of adverse effects was lower than reported in both BAT and EMBRACE. It is unclear if this reflects a better tolerability profile (due to a different formulation) or differences in how adverse events were captured between the studies. This is noteworthy given the GI adverse events in the placebo arms of both BAT and EMBRACE trials. 17
In BLESS, 17 (28.8%) erythromycin and 15 (25.9%) placebo group subjects reported adverse events. One subject in the erythromycin and placebo groups each discontinued the study drug due to QTc prolongation (enrolled despite pre-existing prolonged QTc at screening) and nausea, respectively. Three subjects reported nausea in the placebo group compared to none in the erythromycin group. No subject in the BLESS trial developed a new cardiac arrhythmia. A QTc interval of >450 ms in men and >470 ms in women is a contraindication to macrolide therapy. An increased rate of macrolide resistance was seen in patients after 12 months of therapy with erythromycin ethyl succinate. One strategy to reduce antimicrobial resistance could be to discontinue the drug, preferably in the summer months when infection rates are low, and restart it just before winter. 17
This reversal of resistance on cessation was seen in the ‘azithromycin in severe asthma’ (AZISAST) RCT which showed a decrease in the percentage of macrolide-resistant Streptococci (from 73.8% to 45.9%) in the azithromycin group during the 4-week washout period. 18
Hearing loss is challenging in clinical practice. A similar trial in COPD compared 250 mg daily of azithromycin to a placebo. Although an excess of hearing loss (audiogram confirmed) with long-term azithromycin versus placebo (n = 142, 25% vs n = 110, 20%, p = 0.04) was seen, many cases of hearing loss were temporary in both arms. Not all participants who developed hearing loss discontinued the drug. During the trial, hearing returned to baseline in 21 (34%) patients who discontinued azithromycin versus 6 (32%) who did not. In the placebo group, hearing returned to baseline in 37 (38%) who discontinued versus 2 (25%) who did not discontinue. Hence, improvement in hearing was noted on repeat testing irrespective of discontinuing the drug suggesting that permanent hearing loss was overestimated. Similar data is less readily available in bronchiectasis. 19
Results from these RCTs collectively show that although GI adverse effects such as nausea, vomiting, diarrhoea and epigastric discomfort were more frequent in the active treatment group, these symptoms were commonly reported in the placebo group as well. In the EMBRACE trial, most GI adverse effects were mild, and none were serious. The BAT RCT reported more GI adverse events as compared to other macrolide maintenance therapy RCTs, potentially suggesting that daily azithromycin therapy is linked with a higher incidence of GI events as compared to three times weekly therapy.
Macrolides need to be used with caution in patients with electrolyte disturbances (predisposes to QT interval prolongation) and those with eGFR less than 10 mL/min/1.73 m2. Azithromycin interacts with many drugs including chloroquine/hydroxychloroquine and selective serotonin reuptake inhibitors (SSRIs) increasing the risk of serious cardiovascular adverse effects due to dose-related QT interval prolongation. These drug–drug interactions are particularly relevant due to the association between bronchiectasis and rheumatoid arthritis (where hydroxychloroquine may be used) and the association with bronchiectasis and depression (SSRI).
The British Thoracic Society (BTS) recommends azithromycin/erythromycin as an alternative to inhaled antibiotics (if not tolerated) or as an additive to inhaled antibiotics (if high exacerbation frequency >3/year as per guideline definitions) in patients with bronchiectasis and Pseudomonas aeruginosa colonisation. 13 Patients need to be counselled regarding the potential adverse effects of long-term macrolides, that is, GI side effects and strategies to mitigate them such as taking macrolides with food which can significantly reduce GI irritation. In terms of follow-up, patients need to be followed up 6 months with an assessment of the efficacy and development of side effects if any. Although hearing loss is not commonly seen in practice, patients need to be cautioned regarding new development/worsening of hearing loss. An ECG needs to be performed prior to and 1 month post-therapy to rule out QTc prolongation. Since macrolides can affect liver function, liver function tests are recommended 1 month after starting therapy and then in 6 months time. 12
Other oral antibiotics
There are fewer data available on the use of long-term co-trimoxazole (sulfamethoxazole/trimethoprim), doxycycline and amoxicillin in bronchiectasis. Regimens the authors have used include long-term co-trimoxazole 960 mg three times weekly and short-term doxycycline 100 mg daily. There are prior reports of amoxicillin 3 g sachets × 3 days every 3 months, but this is not common practice in the author’s experience. A double-blind randomised study by Currie et al. showed reduced frequency of exacerbations, reduction in purulent sputum volume and reduced morbidity with the use of oral amoxicillin 3 g twice daily for 32 weeks. 20 Stockley et al. reported a statistically significant reduction in sputum purulence and volume with the use of nebulised amoxicillin 500 mg (twice a day) for 4 months in bronchiectasis patients who had not responded to oral amoxicillin (3 g twice a day) and had growth of resistant bacteria in the sputum. 21 These regimens are not however recommended in prevailing guidelines.
The effective use of co-trimoxazole to treat severe bronchiectasis is reported in two articles from Japan.22,23 However, no randomised control trials show the efficacy and safety of this drug in the European population. One reason for this could be the strong evidence of blood dyscrasias with co-trimoxazole and the development of other drugs that have a better safety profile.
A trial by Lam et al. in 1989 showed ofloxacin had higher efficacy and was better tolerated than amoxicillin in patients with infective exacerbation of bronchiectasis. 24 Although there are no recent clinical trials available for the use of amoxicillin in the adult population, the BEST-1 and 2 trials in children with bronchiectasis show that GI adverse effects (nausea, vomiting and diarrhoea) were most frequently seen in the treatment group. However, it is unclear if this translates to the adult population.25,26
Long-term systemic antibiotics used as suppressive therapy therefore have the potential for negative impacts on the patient both directly, such as GI upset, ototoxicity, and indirectly, with increased rates of bacterial resistance. The latter is detectable in studies that have specifically assessed this, but the long-term consequences such as the risk of Clostridium difficile colitis or treatment refractory community-acquired pneumonia are unclear. 27 Clinically relevant and cost-effective strategies for monitoring the harms of long-term antibiotics remain unclear with current guidelines being empirical rather than strongly evidenced based.
An important drug interaction worth highlighting given the associations between rheumatoid arthritis and bronchiectasis is patients with bronchiectasis–rheumatoid arthritis overlap syndrome are at significant patient safety risk if taking methotrexate. Methotrexate and co-trimoxazole are classed as major drug interaction risks due to the occurrence of aplastic anaemia and/or agranulocytosis. 28 Additionally, the treatment for bronchiectasis exacerbations is complicated by the presence of co-morbid conditions such as epilepsy and the interaction between meropenem and valproate is notable leading to a subtherapeutic concentration of valproate and seizures in some patients. However, this can be managed effectively in clinical practice. 29
Airway targeted antibiotics
Nebulised antibiotics such as aminoglycosides (tobramycin, amikacin, gentamicin) and polymyxins (colistimethate sodium/colistin) are potentially a more targeted therapeutic option as compared to oral antibiotics as they offer the advantage of local delivery of the drug into the airway, with minimal systemic adverse effects. 30 Nebulised antibiotics are often delivered to the airways at much higher levels than doses used as breakpoints or minimal inhibitory concentrations when delivering IV antibiotics. Nebulised antibiotics are guideline recommended as individual studies and meta-analyses demonstrate that they show reductions in sputum bacterial density, airway inflammation, frequency and severity of pulmonary exacerbations with improved health-related quality of life. 31
Nebulised gentamicin
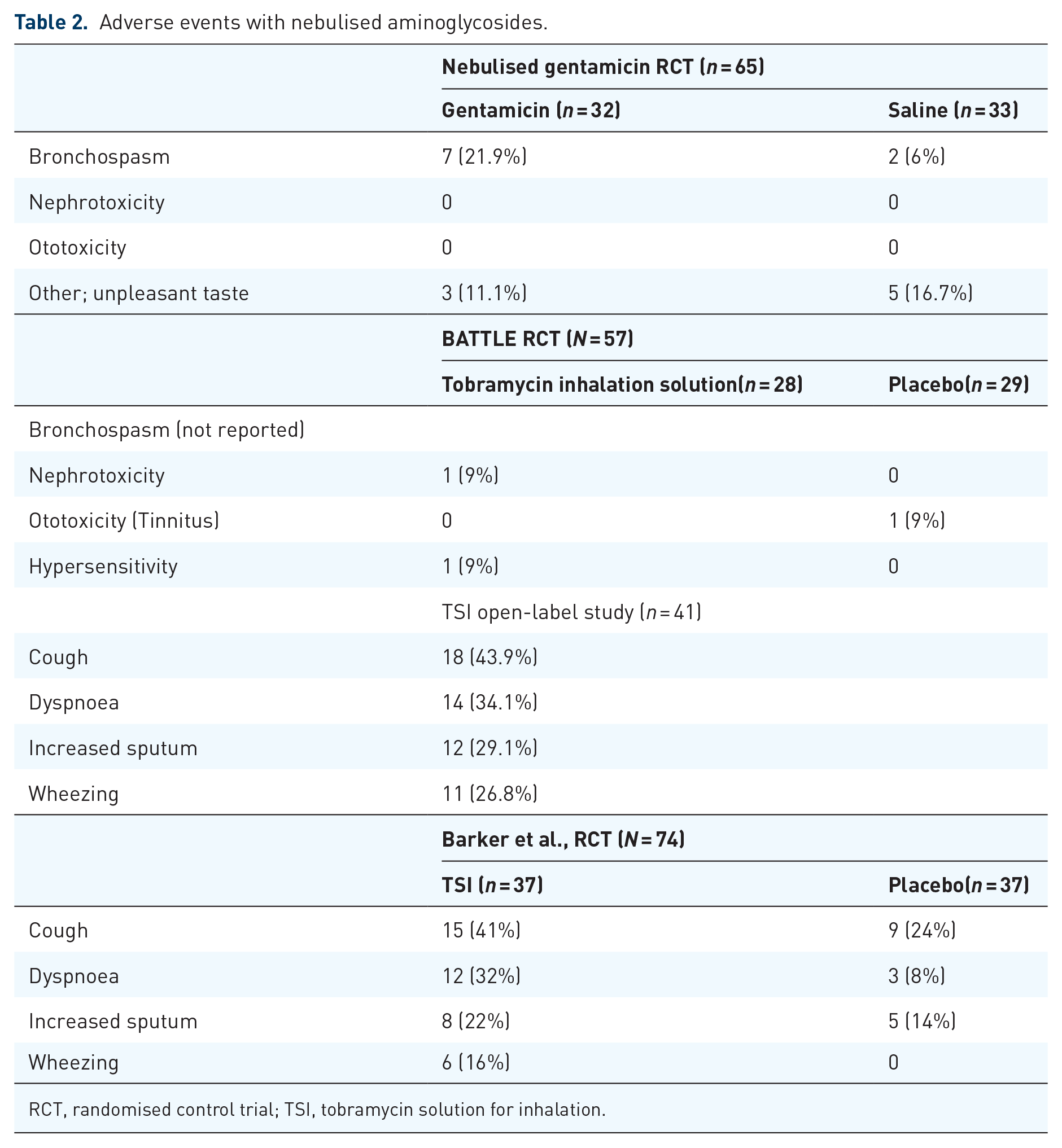
A single-centre RCT by Murray et al. reported on the efficacy of nebulised gentamicin (80 mg twice daily) therapy for 1 year in patients with bronchiectasis. A relatively small group of 65 patients were randomised to either nebulised gentamicin (n = 32, 49%) or saline (n = 33, 51%). In all, 57 patients completed the study, with 27 in the gentamicin group and 30 in the saline group. Bronchospasm was the most common adverse effect in both the nebulised gentamicin (n = 7, 22%) and nebulised saline (n = 2, 6%) group which was treated with nebulised beta-2 agonist. None of the patients in the gentamicin group developed nephrotoxicity or ototoxicity. 31
The BTS guidelines recommend inhaled gentamicin as a second-line therapy to colistin in bronchiectasis patients with chronic Pseudomonas aeruginosa infection and an alternative to oral macrolides in non-Pseudomonas aeruginosa colonised patients. 13 However, the British National Formulary does not recommend concurrent gentamicin and colistimethate as they have neuromuscular blocking effects with an increased risk of ototoxicity/nephrotoxicity.
Gentamicin is not developed specifically for nebulisation so requires repurposing of an intravenous preparation and in our experience, this is commonly in a glass vial with varying levels of ethanol and/or metabisulphites as preservatives. It is our single-centre experience that although very few patients demonstrated an acute bronchospasm event when given a test dose, relatively few remain on nebulised gentamicin by the end of 12 months. In all, 148 patients were included in the analysis out of which only 64 (43%) patients continued gentamicin at the end of 12 months and these patients had a lower mean age, higher mean absolute FEV1 and lower mean Bronchiectasis Severity Index (BSI. This suggests that older patients with severe bronchiectasis and worsening lung function are at higher risk of treatment cessation. 32
This lack of long-term adherence is likely a complex issue of treatment burden (nebulisation takes some dexterity to reconstitute and 10–15 min to deliver), airway reactivity and variation between gentamicin preparations. It is our experience that patients can tolerate one manufacturer’s gentamicin preparation but when supplied with a different brand, bronchospasm can occur soon after switching (ranging from first dose to within a week). Hence, lack of long-term adherence may reflect patient treatment burden, tolerability and the preparation(s) they have previously used. 32
Administration of antibiotics through nebulisers is time-consuming, cumbersome and costly. Other alternatives include pressurised metered dose inhalers (pMDI) and dry powder inhalers, although pMDI is not viable since antibiotic therapy requires a large dose of drugs to be delivered locally as compared to bronchodilators or steroids. Dry powder inhalation (DPI) are more efficient, cost-effective and rapid alternative to Small Volume Nebulisers (SVN). In terms of drug delivery to the airway, nebulisers deliver higher concentrations as compared to DPI. The drawback for inhalational systems is poor deposition of drugs in pulmonary areas with minimal airflow or maximum focus of inflammation. 33
The BTS recommends inhaled gentamicin as a second-line alternative to inhaled colistin in bronchiectasis patients with chronic Pseudomonas aeruginosa infection. 13 Inhaled aminoglycosides should be avoided if the creatinine clearance is <30 mL/min and with concomitant nephrotoxic medications. Gentamicin should be used with caution if the patient has significant hearing loss/balance issues. Since bronchospasm is a known adverse effect with inhaled antibiotics, inhaled bronchodilators should be used pre-therapy in patients who are prone/known to have this adverse effect. 13 Melani et al. have recorded a case of a paradoxical episode of near-fatal bronchoconstriction after the use of inhaled gentamicin solution in a bronchiectasis patient. 34 In our clinical practice, we have observed large variations in the tolerability of different gentamicin preparations and suggest different brands are not interchangeable. There is a large variation in alcohol and metabisulphite excipient concentrations, and this is notable as they may induce bronchospasm.
Nebulised amikacin
In a different study design, nebulised amikacin twice daily (for 2 weeks) was added to the treatment of acute bronchiectasis exacerbations in patients with Pseudomonas aeruginosa colonisation. In all, 143 patients were randomised to either the intervention group (n = 72) or the placebo group (n = 69). Both groups showed adverse events, including bronchospasm. In the intervention group, two patients (4%) experienced bronchospasm. In the control group, one patient (1.4%) developed bronchospasm. None of the subjects developed ototoxicity or nephrotoxicity. These data are useful as they do inform the potential start of nebulised long-term antibiotics during or soon after an exacerbation. 35
Tobramycin inhalation solution/powder
BATTLE, a double-blind RCT reported the occurrence of bronchial hypersensitivity after the use of tobramycin inhalation solution (TIS). In all, 57 subjects were randomised to either TIS 300 mg once daily (n = 28) or 0.9% saline group for 52 weeks (n = 29). The most common adverse events leading to study discontinuation in the treatment arm were dyspnoea (n = 3, 27%), severe cough (n = 2, 18%) and allergic reaction (n = 1, 9%). One patient (9%) in the treatment group developed reversible renal impairment after treatment for 9 months. No subject in the TIS group developed ototoxicity. One patient (9%) in the placebo group developed ototoxicity with tinnitus during the treatment period which was not related to the medication (Table 2). 36
Adverse events with nebulised aminoglycosides.
RCT, randomised control trial; TSI, tobramycin solution for inhalation.
Three further tobramycin clinical trials showed a similar pattern of adverse events to those identified in the BATTLE trial (cough, dyspnoea, increased sputum and wheezing).37–39
The iBEST RCT was conducted to evaluate the efficacy, safety and tolerability of different doses of tobramycin inhalation powder (TIP) in patients with bronchiectasis and chronic Pseudomonas aeruginosa infection. Three dosing regimens (84 mg daily, 140 mg daily and 224 mg daily) were studied in this trial, with the treatment duration being 16 weeks followed by an 8-week follow-up. Patients were randomised to one of the three treatment cohorts (1:1:1) and within each cohort, patients were randomised in a 2:2:1 ratio to receive continuous TIP, cyclical TIP/placebo (28 days cycle) or placebo only. The most common adverse events reported in this trial were cough (18.7%) and dyspnoea (17.8%; Table 3). 40
Treatment-emergent adverse events leading to study drug discontinuation.

GFR, glomerular filtration rate; OD, once a day; TIP, tobramycin inhalation powder.
Five patients (4.7%) experienced ototoxicity and 12 patients (11.2%) experienced haemoptysis. There was no clinically significant bronchial hypersensitivity observed post-inhalation of TIP. Ototoxicity presented as tinnitus and deafness which was mild, transient, and did not lead to changes in the study drug administration.
Most patients who developed renal impairment (raised creatinine/decreased GFR) had multiple pre-existing medical conditions and polypharmacy.
Treatment-emergent adverse events leading to study drug discontinuation were higher in the active treatment cohorts with the greatest number of cases being in cohort C (224 mg TIP daily).
This study showed that TIP was well tolerated but with an increase in renal side effects at high doses.
With regards to drug delivery, TIP is delivered via capsule-based, breath-actuated DPI designed to deliver a reliable dose of drug into the airway with low airflow resistance. Hence, TIP requires slow deep breaths for the active drug to reach the peripheral airways. In addition, the drug administration time for TIP was faster compared to nebulised TIS (Tobramycin inhalation solution). This was seen in the EAGER trial where the mean administration time over three cycles of treatment was significantly lower with TIP 112 mg than with TIS 300 mg/5 mL. 41
Colistimethate sodium
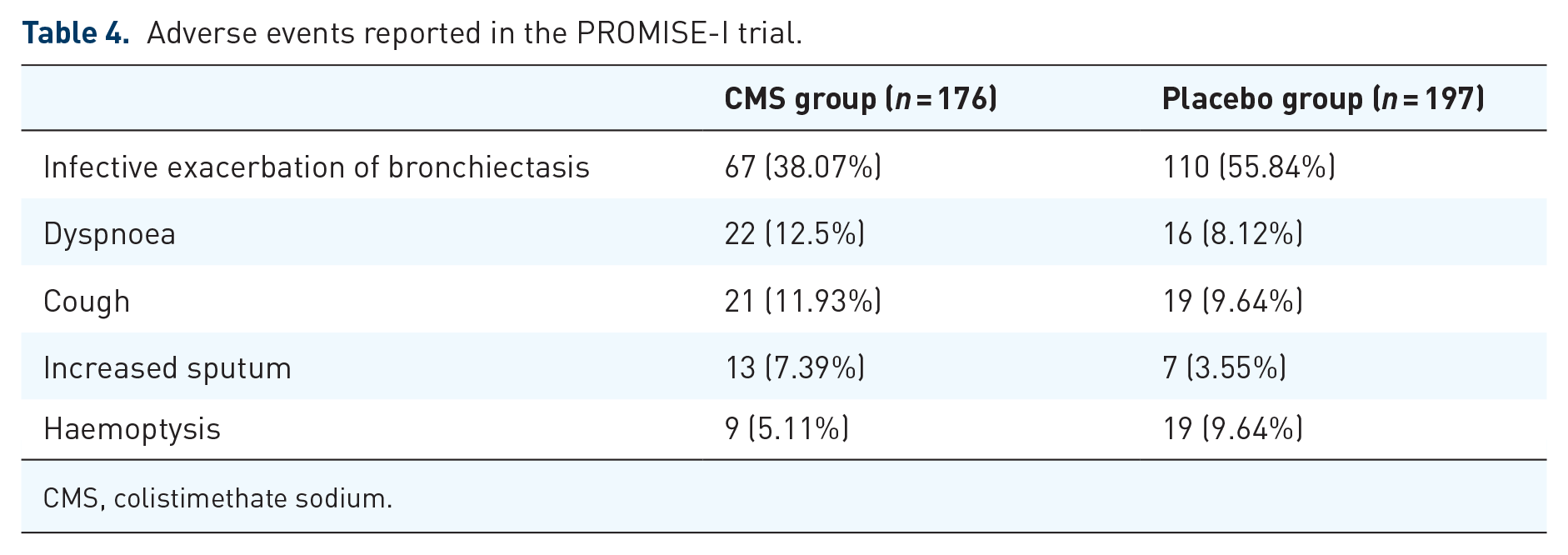
In the PROMIS-I trial, subjects with bronchiectasis and long-term Pseudomonas aeruginosa infection were randomised to inhaled colistimethate sodium (CMS) or placebo group in a 1:1 ratio. A dose of 1 million international units (MIU)/80 g of CMS was administered via an intelligent nebuliser (I-neb) that synchronised drug delivery with patient inhalation used twice daily for 12 months. In all, 176 subjects were randomised to the CMS group and 197 to placebo. In all, 142 subjects (80.68%) in the CMS group and 159 subjects (80.71%) in the placebo group reported any adverse events (Table 4). 42
Adverse events reported in the PROMISE-I trial.
CMS, colistimethate sodium.
Ciprofloxacin
Ciprofloxacin, a fluoroquinolone, has bactericidal activity against multiple organisms affecting the respiratory tract including Pseudomonas aeruginosa. The airway-targeted dry powder formulation allows the drug to penetrate the central and lower airways at lower concentrations as compared to systemic therapy. 43
The RCT by Wilson et al. assessed the safety and efficacy of ciprofloxacin DPI treatment. Subjects were randomised to either ciprofloxacin DPI 32.5 mg or placebo, both twice daily. During the study, 41 subjects (68.3%) in the treatment group and 42 subjects (65.6%) in the placebo group reported treatment-emergent adverse events. 44
The RESPIRE I and II trials then studied the safety and efficacy of 14- and 28-day on/off-ciprofloxacin DPI. These phase III trials had the same design but a different analysis plan. Patients were randomised to twice-daily ciprofloxacin DPI 32.5 mg or placebo in two treatment regimens of 14/28 days on/off cycle for 48 weeks. Adverse events of special interest included bronchospasm, haemoptysis, anaphylaxis and tendon disorder (Table 5). 45
RESPIRE I and II trial.

DPI, dry powder inhalation.
In the RESPIRE-1 trial, only one subject in the placebo group developed tendonitis, but there were more reported musculoskeletal events in the active treatment arms; the causes for this are unclear but systemic ciprofloxacin is associated with joint pains and/or tendinopathy. Treatment-emergent musculoskeletal adverse events were infrequently reported in the RESPIRE II trial (Table 5). Tendon disorders were reported by two patients in each ciprofloxacin DPI treatment group and zero patients in the placebo groups. Ciprofloxacin DPI had a good safety profile in this trial series and was well tolerated with more than 70% of subjects completing the treatment.46,47
The once-daily respiratory bronchiectasis inhalation treatment (ORBIT) trials were designed to overcome the local intolerance adverse effects associated with inhaled antibiotics and improve the local antimicrobial action in the airways. The use of liposome-encapsulated ciprofloxacin and free ciprofloxacin in one solution provides a high peak concentration from the free ciprofloxacin component and a slow release from the liposomal ciprofloxacin. 48
In the ORBIT-2 trial, dual-release ciprofloxacin for inhalation (DRCFI) was used in patients with bronchiectasis and Pseudomonas aeruginosa infection. DRCFI was administered once daily for 24 weeks in the form of three treatment cycles, with each cycle of 28 days ‘on’ inhaled therapy and then 28 days ‘off’ therapy. Twenty subjects were randomised to active treatment and 22 to the placebo group. 48
Both ORBIT-3 and 4 were similar in their design wherein inhaled ciprofloxacin/placebo was administered once daily for six 56-day treatment cycles, for 48 weeks. In terms of adverse events, 21 (5%) of 389 versus 7 (4%) of 196 patients (ciprofloxacin vs placebo) reported any adverse events. The most common adverse events related to airway irritation (cough, dyspnoea, wheezing and oropharyngeal pain) occurred in a similar pattern in both groups. 49
Fluoroquinolone-associated adverse effects (tendonitis, tendon rupture, muscle weakness) were not frequent in the treatment group. 49 This could be because of the low systemic levels of ciprofloxacin that were observed in this study. Antibiotic resistance especially with Pseudomonas aeruginosa is an increasingly worrying concern for clinicians. In this trial, ciprofloxacin Minimum Inhibitory Concentrations (MICs) among Pseudomonas isolates were higher after treatment, but this decreased by the end of the off-ciprofloxacin cycle.
Aztreonam
AIRBX-1 and 2 were two identical RCTs conducted to assess the efficacy and safety of aztreonam for inhalational solution (AZLI) in bronchiectasis and gram-negative bacterial infection. AZLI has a primary antipseudomonal action and improves lung function in cystic fibrosis-related bronchiectasis. 50
Subjects were randomised to either 4 weeks of three daily 75 mg AZLI or placebo. In both trials, discontinuation due to safety or tolerability reasons was higher in the AZLI group as compared to the placebo group (20% vs 3% in AIRBX-1 and 7% vs 4% in AIRBX-2). The most common adverse effects noted in this trial were dyspnoea, cough and increased sputum production, all expected adverse effects with inhaled antibiotics. Dyspnoea was the most common adverse effect despite beta-2 agonist bronchodilation before the administration of the drug/placebo. AZLI failed to show significant clinical improvement in both trials in contrast to positive outcomes seen in AZLI-cystic fibrosis trials. Arguably, these prior trial designs could be improved upon to test exacerbation reduction effects. 50
In summary, inhaled antibiotics, either in powder or nebulised formats, consistently show predictable adverse effects of upper airway irritation, taste disturbance and bronchospasm (aminoglycosides) to varying degrees. Few studies however noted significant increased rates of GI disturbances. Patient withdrawals from the studies were noted with aminoglycosides and aztreonam. 51
Anti-inflammatory therapies
Inflammation is a key component of bronchiectasis. Although neutrophils appear predominantly in most patients, there is a recognition that a population of patients with significant eosinophilia can now be identified. Targeting airway inflammation is therefore an attractive option with an increasing awareness that this may have indirect benefits on mucus retention and even potentially airway bacterial load. 52
Inhaled steroids
Inhaled corticosteroids (ICS) are guidelines recommended for the treatment of asthma and COPD. These are common comorbidities in bronchiectasis and a study in the UK demonstrated that 30% primary-care COPD population had airway abnormalities potentially classifiable as bronchiectasis. 53 ICS was the most commonly used treatment in bronchiectasis patients (n = 8700, 51.3% of overall cohort (n = 16963)) in the EMBARC (European Multicentre Bronchiectasis Audit and Research Collaboration) cohort. ICS was used by 2595 patients (30.4%) without documented COPD or asthma and its use was most common in the UK as compared to other European regions. 4
This is despite the bronchiectasis guidelines recommending avoiding such treatment in the absence of high-quality evidence. No significant improvement in lung function or exacerbation has been identified in long-term studies (>6 months) with ICS. 13 The recognised harms of ICS are best described in asthma and COPD and include, but are not limited to, systemic absorption (skin thinning, bruising and increased rates of osteoporosis) or local immune suppression (oral candidiasis and pulmonary infection). Particularly relevant to bronchiectasis are the additional observations in COPD that ICS treatment increases the risk of pneumonia, particularly in those with poor lung function, prior history of pneumonia and older age. 54 Although similar data have not yet been described in bronchiectasis, the much higher airway bacterial load in bronchiectasis makes this a significant possibility. One of the further concerns is that ICS use has been linked with non-tuberculous mycobacterium (NTM) infection. 55
A recent database study from Taiwan on the effectiveness and safety of fixed-dose combination ICS/long-acting β2-agonists (ICS/LABA) in bronchiectasis as compared to long-acting antimuscarinic antagonist (LAMA) only in 1281 and 455 patients, respectively. The authors found no concerning safety signals with comparable outcomes to LAMA, after propensity matching, with hazard ratios of 1.22 (95% CI 0.81–1.83) for hospitalised respiratory infection, 1.06 (95% CI 0.84–1.33) for acute exacerbation and 1.06 (95% CI 0.66–1.02) for all-cause hospitalisation. The authors also studied whether different ICS preparations may have been associated with different outcomes. Their data suggested beclomethasone/formoterol or budesonide/formoterol led to a lower risk of acute exacerbation compared to fluticasone/salmeterol and that beclomethasone/formoterol resulted in lower risks of hospitalised respiratory infection and all-cause hospitalisation (HR 0.55, 95% 0.37–0.80) compared to fluticasone/salmeterol. These data are intriguing, and the authors suggest a potential difference in potency and higher airway immunosuppression associated with fluticasone/salmeterol. There is a potential for bias by indication in the differential use of these inhaled therapies and observational studies do not provide causality; future randomised trials are needed. 56
It is plausible that ICS use could drive an increased susceptibility to NTM through suppression of local immunity, but it is possible that bias by indication may play a role in this with injudicious use of ICS in a patient with previously undiagnosed NTM infection. A meta-analysis across chronic airway diseases, however, suggests that ICS use is associated with increased NTM risk. 57
Brensocatib
Brensocatib is a novel drug therapy being investigated for use in bronchiectasis. It is a reversible inhibitor of dipeptidyl peptidase 1 (DPP-1) which is involved in activating neutrophil serine proteases (amongst other host-derived proteases) during inflammation in bronchiectasis. Neutrophil elastase has been strongly linked with increased disease severity and risk of exacerbations.
The WILLOW trial examined the incidence of bronchiectasis exacerbations in patients on two dosage regimens of brensocatib (10 or 25 mg once daily) over 24 weeks in 256 patients. Periodontitis and skin hyperkeratosis were the adverse effects of special interest in this trial as the genetic absence of DPP-1 presents as Papillon-Lefevre syndrome (diffuse palmoplantar keratoma and periodontitis). The incidence of skin hyperkeratosis was higher in the 25 mg brensocatib group, while periodontitis was higher in the 10 mg group. 58
In terms of efficacy, both 10 and 25 mg brensocatib prolonged the time to first exacerbation. 59 A phase III trial of 52 weeks of treatment to assess the efficacy and safety of brensocatib in bronchiectasis (ASPEN trial) is underway at this point. The trial has completed recruitment, but safety and efficacy data are not yet available. 59
A risk–benefit assessment of brensocatib based on the WILLOW trial suggests a negative number needed to harm and a potential positive risk-benefit profile of brensocatib. These results indicate that brensocatib could be safely used for treating bronchiectasis patients. 60
A double-blind trial was conducted to determine the efficacy of short-term (28 days) brensocatib 25 mg in patients with COVID-19 infections. Safety analysis from this study shows that none of the subjects developed adverse effects of special interest except for one subject developing dental complications. 61
Itepekimab
Airway epithelial damage signals such as alarmins may contribute to the pathophysiology of bronchiectasis. Interleukin-33 is one such epithelial alarmin. Itepekimab is an IL-33 signal inhibitor monoclonal antibody that inhibits airway inflammation and tissue remodelling. The efficacy and safety of itepekimab have been previously studied in asthma and COPD (AERIFY-I and II). 62 The most common adverse effects noted in the phase II trial for itepekimab (n = 73 in itepekimab group) in asthma were nasopharyngitis (n = 13, 18%), allergic rhinitis (n = 3, 4%), nausea (n = 4, 5%) and back pain (n = 4, 5%). 62 This is similar to the results seen in a phase II COPD trial with itepekimab where the most common adverse events in the active group were nasopharyngitis (n = 28, 16%), bronchitis (n = 18, 10%), headaches (n = 14, 8%) and upper respiratory tract infection (n = 13, 8%). 63
Benralizumab
Benralizumab is a monoclonal antibody that binds to the eosinophilic and basophilic IL-5 receptor and elicits antibody-dependent cell-mediated cytotoxicity. 64 The use of benralizumab has been extensively studied in asthma. The most common adverse effects associated with benralizumab in asthma RCTs included nasopharyngitis, worsening of asthma and bronchitis. 65 Since benralizumab is administered subcutaneously, injection-site-related reactions (urticaria) were seen in 2% of patients in the CALIMA RCT. 66 Two Cochrane reviews studying anti-IL-5 therapy in asthma and COPD, each showed no excess serious adverse events with benralizumab.67,68 Bronchiectasis has recently been noted to have a sub-population of patients where eosinophils may contribute to disease pathogenesis. 69 An RCT studying the efficacy and safety of benralizumab in bronchiectasis (MAHALE RCT) is currently underway. 70
Mucoactive therapy
Mucociliary clearance in bronchiectasis is impaired by excessive mucus production and viscosity, airway dehydration and structural lung changes. This stagnant mucus becomes chronically colonised with bacteria resulting in a neutrophilic response. Mucoactive medications such as hypertonic saline, mannitol and DNase have a direct impact on mucus clearance from the airways.5,12
Hypertonic saline
Hypertonic saline (HS) hydrates the airways, reduces mucus viscosity and may improve chest clearance in bronchiectasis. Both isotonic saline (IS) and HS have been used in bronchiectasis aiming to improve mucus clearance and quality of life. A systematic review of HS in bronchiectasis shows that in most of the RCTs, patients with hypersensitivity to HS were excluded and hence the number of adverse effects caused by HS could be lower than that in the wider patient group where HS may be applied. 71
A Cochrane review on inhaled hyperosmolar agents in bronchiectasis comparing HS versus IS noted conflicting results and it was not possible to draw robust conclusions. 72 There was no significant difference between the adverse effects seen in either group. Local adverse events such as bronchospasm are common with inhaled hyperosmolar agents and hence patients should undergo a challenge test under supervision before commencing on IS/HS. Those with clinical features of bronchospasm are frequently excluded or pre-treated with inhaled bronchodilators. This is in line with the BTS bronchiectasis guidelines, which recommend pre-treatment with bronchodilators in high-risk patients (asthma, bronchial hyperreactivity and severe airflow obstruction-FEV1 < 1 L).13,72 The CLEAR trial comparing hypertonic saline 6% to carbocisteine, to both treatments combined and to standard care has completed recruitment but has not yet been reported. Rates of short-term and long-term intolerance will be reported alongside efficacy. 73
Inhaled mannitol
A 52-week 400 mg versus 50 mg (deemed taste-matched non-active placebo) inhaled mannitol RCT was conducted to determine the impact of mannitol on bronchiectasis. 20.2% versus 16.7% of patients (mannitol vs placebo) developed adverse events related to the study medication. Cough (12.9% vs 9.6% in active vs placebo) and dyspnoea (8.6% and 7% active vs placebo) were two of the most common adverse events reported in this trial. Mannitol therapy was safe and well-tolerated over 12 months; however, there was no significant reduction in the exacerbation rate in patients with mild to moderate bronchiectasis. Hence, the efficacy of mannitol versus the adverse effects/cost burden should be balanced when making clinical decisions. 74
DNase
Airway secretions have been found to contain large amounts of extracellular DNA likely from neutrophil extracellular traps and dead bacteria. This DNA mesh may limit mucus flow and nebulised DNase I has been successfully trialled in cystic fibrosis-related bronchiectasis. The European Respiratory Society guidelines, however, do not recommend recombinant human DNase in bronchiectasis. Notably one early trial of DNase I in bronchiectasis where 349 patients were randomised to placebo (176) versus treated (173) over 24 weeks was associated with more frequent pulmonary exacerbations and greater FEV1 decline in patients who received rh-DNase. The potential reasons for an increased signal of harm in this population are unclear given its role in treating cystic fibrosis-related bronchiectasis. 75
Carbocisteine
The BTS guidelines for bronchiectasis recommend a 6-month trial of carbocisteine followed by long-term therapy if clinical benefits are noted. The evidence base for this is limited. Larger-scale trials in COPD have shown that this medication is generally well tolerated with GI disturbances (nausea, dyspepsia and/or peptic ulceration) and skin adverse effects (pruritis/rash) amongst the most commonly reported. 76 As noted above, the CLEAR trial should provide more safety and efficacy data on this within bronchiectasis. Notably, cardiac co-morbidities are common in bronchiectasis and the risk of peptic ulceration with anti-platelet agents on top of mucolytics needs to be factored into risk–benefit discussions with patients.
Multimorbidity and polypharmacy in bronchiectasis
Bronchiectasis was historically considered an orphan disease but with an increasing prevalence in the UK and an ageing population, clinicians need to focus on the impact of multimorbidity and polypharmacy. The most common co-morbid conditions associated with bronchiectasis include COPD, cardiovascular disease, hypertension, asthma and diabetes. 10 The concurrence of bronchiectasis with other lung pathologies (asthma/COPD) further complicates the management pathway for these patients due to different pathophysiology. Multimorbidity is also associated with higher rates of exacerbations and hospitalisations, thereby increasing the pressure on an already overstretched health system.
Bronchiectasis–COPD overlap syndrome requires to be studied in detail as COPD is the most common co-morbid condition. In addition, asthma (n = 5267, 31%) and COPD (n = 4324, n = 25.5%) were frequently reported as the cause of bronchiectasis in the EMBARC cohort (n = 16,963), reflecting the overlap syndromes that have been associated with worse outcomes. 4
In terms of mortality, with an increase in co-morbidities, the mortality rate also increases particularly with COPD, Gastroesophageal Reflux Disease (GORD) and rheumatoid arthritis. 9 With these conditions, a common theme is there is a higher risk of polypharmacy and its complications with increasing co-morbidities in bronchiectasis. Polypharmacy is associated with higher mortality, falls, emergency readmissions, adverse effects, drug interactions, lower quality of life and increased medication errors.9,10,77
Conclusion
Patients with bronchiectasis suffer from a lack of evidence-based therapies arising from large-scale clinical trials with robust safety data. Many drugs recommended in current guidelines are repurposed and have limited efficacy data and scantier safety data specific to this population. Certain scenarios within bronchiectasis are associated with an increased risk of adverse events including the potential for drug–drug interactions for therapies targeting the pathophysiology of bronchiectasis and the comorbidities/bronchiectasis aetiologies. Large database studies that capture the adverse events/harms that may be common in co-morbid ‘real-world’ patients with polypharmacy regiments are needed to understand the key safety signals and major drug–drug interactions that are important in bronchiectasis.
The field of bronchiectasis has had to adapt to being a somewhat neglected condition and most drugs used in bronchiectasis lack strong effectiveness and safety data in bronchiectasis. Many of the drugs used have been developed and tested for other conditions (e.g. inhaled therapy for asthma and COPD) or nebulised antibiotics (e.g. cystic fibrosis). As new drugs are developed, and larger national or international registries emerge linking research to routine care, the clinical care community should adopt practicable pharmacovigilance approaches where possible. This is increasingly important with gaining populations with multimorbidity and polypharmacy that may lead to drug–drug interactions and new safety challenges.
Footnotes
Acknowledgements
Henil Upadhyay’s role was funded by the NIHR research capacity funding grant administered through the Newcastle Upon Tyne Hospitals NHS Foundation Trust. The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
