Abstract
Nearly all medications carry the risk of drug-induced liver injury (DILI). Idiosyncratic reactions are rare and poorly predictable, and the mechanisms are not always well understood. Pirfenidone is an oral antifibrotic drug used to treat idiopathic pulmonary fibrosis. While elevation of liver enzymes is a common adverse reaction during therapy, it rarely leads to discontinuation or reduction of the drug. Although isolated cases of liver damage or liver failure have been reported, they are infrequent. This report presents the case of a 70-year-old woman with known idiopathic pulmonary fibrosis, depression, hypothyroidism, and hypercholesterolemia who presented at our emergency department with jaundice, anorexia, and asthenia. The patient’s medication regimen included lamotrigine, simvastatin, levothyroxine, and pirfenidone, which had been introduced 6 months prior. Laboratory testing revealed elevated liver enzyme levels consistent with acute hepatocellular hepatitis. Following a medical workup, which included anamnesis, laboratory testing, iconographic investigations, and liver biopsy, we concluded that the patient had suffered from pirfenidone-induced liver injury. Pirfenidone was withdrawn, and liver tests gradually improved. The purpose of this clinical case report is to highlight this rare adverse reaction and to make clinicians aware of its assessment and management. In 2018, only one other case of severe liver failure leading to the death of the patient was reported. Early detection of potential DILI during the workup is crucial to discontinue the suspected medication promptly. Any drug-induced hepatitis must be reported for registration.
Plain language summary
• Drug-induced liver injury (DILI) is a type of hepatitis caused by medication, drugs, or even herbal and dietary supplements.
• There are two types of DILI reactions: intrinsic and idiosyncratic.
○ The intrinsic reaction, such as acute hepatotoxicity due to acetaminophen overdose, is easily predictable and well-known.
○ The idiosyncratic reaction is more complex, unpredictable, and not well-understood. Therefore, diagnosing an idiosyncratic reaction can be challenging.
• Pirfenidone is an immunosuppressive drug used to treat idiopathic pulmonary fibrosis by inhibiting collagen formation through anti-fibrotic and anti-inflammatory mechanisms.
• Several adverse reactions of pirfenidone are well described, including temporary elevation of liver enzymes during treatment. This adverse reaction is mostly asymptomatic and resolves spontaneously with or without dose adjustment.
• However, few cases of severe DILI due to pirfenidone have been reported, which may lead to liver dysfunction.
• This paper reports on a rare idiosyncratic reaction related to pirfenidone that resulted in hepatic adverse reactions.
Background
Drug-induced liver injury (DILI) refers to liver damage caused by drugs, including prescribed and recreational drugs, as well as herbal and dietary supplements. Pirfenidone is an immunosuppressive drug used for the chronic treatment of idiopathic pulmonary fibrosis. It works by inhibiting the formation of collagen using antifibrotic and anti-inflammatory mechanisms. 1 Although temporary elevation of liver enzymes during treatment is a common side-effect, DILI due to pirfenidone is rare. There have been a few reported cases of severe DILI caused by pirfenidone, resulting in liver dysfunction. 2
DILI reactions can be classified into two types: intrinsic and idiosyncratic. Intrinsic reactions are directly linked to the drug and/or its metabolite, are dose-dependent, easily predictable, and have a well-known pathogenesis. Idiosyncratic reactions, on the other hand, are more complex, as their toxicity is not dose-dependent, is unpredictable, and their mechanisms are not fully understood. 3 Thus, the mechanisms underlying the idiosyncratic DILI caused by pirfenidone are not well-established. However, toxic metabolites, immunogenic reactions, or drug-drug interactions may be considered as potential causes. 2 In this report, we present a rare case of idiosyncratic reaction related to pirfenidone.
Case report
Case presentation
In January 2022, a 70-year-old Caucasian woman was referred to our emergency department by her general practitioner due to asthenia, nausea, vomiting, anorexia, jaundice, and discoloration of urine and stools for 2 weeks. She also reported experiencing generalized pruritus and mild anterior rhinorrhea. The patient was also known in the pneumology department for idiopathic pulmonary fibrosis, for which she had been receiving treatment with pirfenidone 801 mg three times a day for approximately 6 months. The patient’s medical history includes treatment for depression with lamotrigine, as well as treatment for substituted hypothyroidism and hypercholesterolemia with simvastatin. Upon admission, the patient presented with mucocutaneous jaundice, generalized scratching lesions, and respiratory crackles, which are known symptoms of her pneumopathy. The patient’s vital parameters were normal and there were no signs of encephalopathy. Laboratory testing revealed perturbations in liver enzymology, including alanine transferase (ALT) at 966 U/L (normal range: 7–35), aspartate transferase (AST) at 1558 U/L (normal range: 9–36), alkaline phosphatase (ALP) at 192 U/L (normal range: 35–105), gamma-glutamyl transferase at 306 U/L (normal range: ⩽40), total bilirubin at 8. 9 mg/dL (normal range: ⩽1.2) including 8 mg/dL of direct bilirubin (normal range: ⩽0.3), lactate dehydrogenase at 467 U/L (normal range: ⩽250), and international normalized ratio at 1.32 (normal range: 0.8–1.2). The patient had no history of liver disease, alcohol abuse, or use of hepatotoxic or phytotherapeutic drugs. Serological testing ruled out viral hepatitis, including anti-hepatitis A virus antibodies immunoglobulin (Ig) M and IgG; hepatitis B virus surface antigens, anti-hepatitis B core antibodies, and anti-hepatitis B surface antibodies; and anti-hepatitis C virus (HCV) antibodies. The patient tested negative for anti-hepatitis E virus (HEV) IgM and IgG antibodies, anti-cytomegalovirus IgM and IgG antibodies, anti-Epstein-Barr virus (EBV) IgM and IgG antibodies, anti-Herpes Simplex virus 1 and 2 IgM and IgG antibodies, and anti-Varicella Zoster virus IgM and IgG antibodies. Polymerase chain reaction (PCR) for HEV was also negative. The patient’s IgA, IgM, and IgG levels were normal. It is worth noting that there was a significant antinuclear factor titration at 1/320 with diffuse granular cytoplasmic appearance. Anti-mitochondrial (M2) and anti-smooth muscle antibodies were negative. The liver ultrasound was normal and did not show any evidence of biliary obstruction. A transvenous liver biopsy was performed, covering 24 mm of liver tissue, including 18 portal spaces (Figure 1). The biopsy showed lobular and portal hepatitis characterized by an eosinophilic inflammatory portal infiltrate with hepatocyte ballooning, bilirubinostasis, and the presence of biliary thrombi, which is compatible with a drug-induced origin. It showed moderate portal fibrosis and short fibrotic septa, which is compatible with F3 fibrosis according to the METAVIR classification. The R-factor was calculated to be 15.16, indicating a hepatocellular pattern. After excluding other potential causes and using the Roussel Uclaf Causality Assessment Method (RUCAM) with a score of 9, we arrived at a diagnosis of acute liver injury induced by pirfenidone with a hepatocellular pattern. Following the suspension of pirfenidone, the patient adhered to a treatment plan consisting of colestyramine oral suspension (4 g twice daily) and local application of mentholated talc for pruritus. The treatment was well-tolerated and resulted in the complete resolution of symptoms, with the exception of mild persistent pruritus. Liver tests and bilirubin levels showed slow improvement (Figure 2). Pirfenidone was permanently withdrawn from the patient’s treatment. The reporting of this case report conforms to the CARE guidelines (Supplemental File). 4
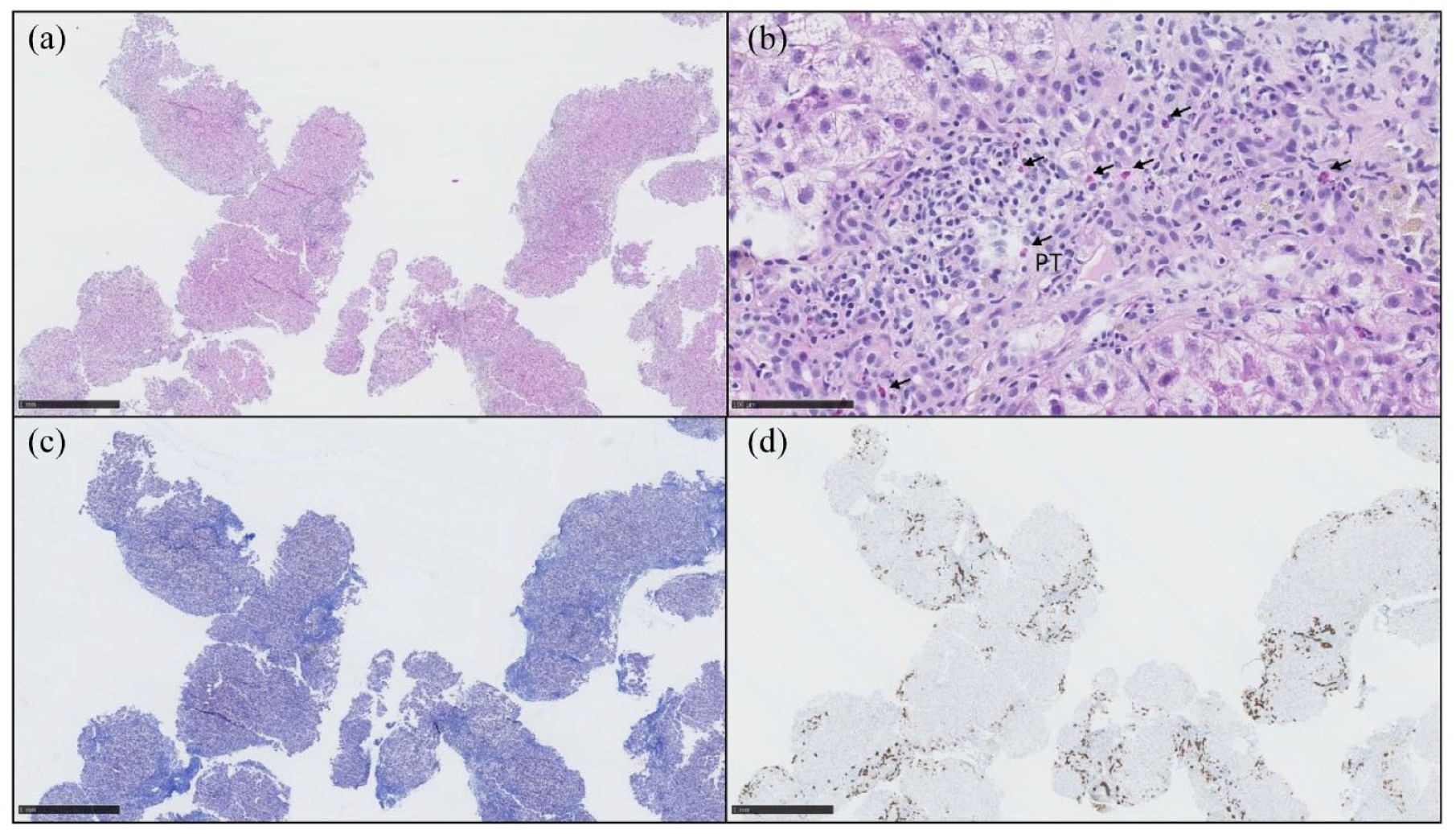
The liver biopsy showed moderate to severe inflammation in PTs (a, hematoxylin and eosin 2.5x). The inflammatory infiltrate was composed of lymphocytes, eosinophils (arrows), and rare plasmocytes (b, hematoxylin and eosin 30x). An active cholangitis was focally observed. A moderate portal fibrosis was observed with initial formation of short septa (Metavir score F3) (c, Masson trichrome 2.5x). A canalicular proliferation was also observed in PTs with discrete cholestasis (d, cytokeratin 7 2.5x).

Biochemical parameters evolution.
Discussion
Although recent progress has been made in the field of DILI, its management remains challenging for clinicians. DILI has a wide range of clinical presentations and may manifest as acute hepatitis, chronic liver injury, and rarely cirrhosis. DILI is classified as acute if liver test abnormalities have persisted for less than 1 year and as chronic if they have persisted for more than 1 year. 5 Approximately 10% of all cases of acute hepatitis are caused by DILI. 6 There is a constantly increasing number of drugs that can cause DILI. Currently, over 1500 medications, including drugs, agents, herbal, and dietary supplements, have been reported to cause DILI. 7 Clinicians should be aware of this phenomenon and consider it as a possible cause of liver injury if a substance is recently introduced. Antibiotics and central nervous system agents are responsible for over 60% of DILI cases. In the United States, the use of herbal and dietary supplements is the second leading cause of DILI.8,9 Additionally, DILI is the primary reason for medication withdrawal from the market. 10 Although our patient is being treated with lamotrigine, which is also associated with idiosyncratic DILI, it is highly unlikely that it is the cause in this case, as she has been taking it for many years. Furthermore, lamotrigine was not discontinued despite the improvement in liver enzymes.
There are two main categories of DILI: intrinsic reaction and idiosyncratic reaction. Intrinsic reactions can affect all individuals and are predictable, reproducible, and dose-dependent. An example of a well-known intrinsic reaction is acetaminophen-induced toxicity. 11 Idiosyncratic reaction, on the other hand, affects only some individuals, is uncommon and unpredictable, has varied clinical manifestations, and is not dose-dependent.8,11
Pirfenidone is an orally administered immunosuppressive drug. Originally developed as an antihelmintic and antipyretic agent, it is now primarily used as an antifibrotic agent for the treatment of idiopathic pulmonary fibrosis. 1 The molecule is a phenyl-substituted pyridinone (5-methyl-1-phenyl-2-(1H)-pyridone) that exhibits several properties through a range of mechanisms. First, anti-inflammatory properties by the suppression of pro-inflammatory cytokines, inflammatory cell accumulation, and inflammasome activation. Then, antioxidative stress properties by reducing oxidative stress, blocking antioxidant inhibition, and stimulating antioxidative stress cytokines. Also, antifibrotic properties due to downregulation of profibrotic mediators, including different growth factors, like TFG-β, PDFG, JNK, or MCP1. 12 Large randomized controlled trials have shown that serum aminotransferase perturbations greater than three times the upper limit of normal (ULN) occurred in 4% of patients treated with pirfenidone. The majority of patients were asymptomatic and the enzymes rapidly normalized with or without dose adjustment. Pirfenidone had to be withdrawn in approximately 1% of patients. It is currently recommended to monitor serum aminotransferase levels every month for the first 6 months of treatment with pirfenidone. After this initial period, monitoring should occur every 3 months. Treatment should be discontinued if serum ALT or AST levels exceed five times the ULN or if symptoms of liver injury or jaundice appear. 2
To the best of our knowledge, only one other case of idiosyncratic DILI caused by pirfenidone has been published in 2018. In this case, DILI occurred 1 month after drug exposure and led to acute liver failure (ALF), resulting in the patient’s death on the fourth day of hospitalization. 13 However, in 2020, the European Medicines Agency warned healthcare professionals about several severe cases of DILI associated with pirfenidone and recommended close monitoring of liver enzymes in patients taking this medication. 14 In this case, DILI occurred 6 months after drug exposure and fully recovered after discontinuation. The mechanisms behind how pirfenidone causes DILI are not yet well-established, but potential causes include toxic metabolites, immunogenic reactions, or drug-drug interactions. 2 The liver biopsy describes an inflammatory infiltrate containing lymphocytes, which is consistent with an immunogenic reaction. However, an autoimmune-like reaction cannot be ruled out due to the presence of rare plasmocytes and the antinuclear factor titration of 1/320 in the blood test. Pirfenidone is primarily metabolized by the cytochrome P450 (CYP) system, specifically CYP1A2, and to a lesser extent by CYP2C9, 2C19, 2D6, and 2E1. This results in the production of 5-carboxy-pirfenidone as the main metabolite, which has minimal pharmacological effects. 12 In our patient’s treatment, only simvastatin is also metabolized by the CYP system, specifically CYP3A4. Currently, there is no known drug interaction between pirfenidone and the other medications taken by our patients. Nintedanib, another antifibrotic drug used for the chronic treatment of idiopathic pulmonary fibrosis, acts as a tyrosine kinase inhibitor. Clinical trials have shown that 13% of patients with progressive fibrosing interstitial lung disease treated with nintedanib experienced elevations of ALT, AST, or both ⩾3x ULN, compared to 1.8% in the placebo group. In 2022, a case report suggests that nintedanib induced DILI in a patient with suspected pulmonary fibrosis after coronavirus disease more than 4 months after its introduction. 15 However, it is important to note that pirfenidone and nintedanib are two different molecules with distinct physiochemical and pharmacokinetic characteristics. 15
It is important to note that DILI is a diagnosis of exclusion. The first step is to rule out common causes of liver injury, such as viral, autoimmune, ischemic hepatitis, or biliary obstruction. The standard workup involves a detailed medical history, including information on recent medication use, both legal and illegal drugs, as well as herbal and dietary supplements taken within the last 12 months. Liver injury often occurs within 6 months of exposure to a new agent, although longer exposure times are possible. In this case, our patient experienced liver injury 6 months after being introduced to pirfenidone. However, with certain substances, DILI may occur after a longer latency period of more than 1 year, such as with nitrofurantoin, minocycline, amiodarone, or some statins. 16 Therefore, it is recommended to conduct a detailed review of all medications taken and their timing of exposure over the past 12 months. Clinicians should conduct routine blood tests, including viral hepatitis serologies, autoimmune hepatitis (AIH) serologies, IgG levels, and ceruloplasmin levels in young patients. If there is a high suspicion of acute hepatitis C, HCV RNA testing may be performed, as serology could be negative in the early stages. 17 Abdominal imaging techniques such as ultrasonography, computed tomography, or nuclear magnetic resonance should be used to investigate the pancreaticobiliary duct and exclude intra or extrahepatic obstruction caused by bile duct stones, malignancies, or rare biliary diseases such as sclerosing cholangitis. 8 Additionally, the imaging workup must evaluate the infra- and supra-hepatic vessels.
DILI is suspected when liver enzymes ALT ⩾ 5x ULN or ALP ⩾ 2x ULN increase following a new medication. 18 DILI is classified based on the R-factor, which is calculated as (ALT/ULN)/(ALP/ULN). A hepatocellular profile is indicated if R ⩾ 5, a mixed profile if 2 < R < 5, and a cholestatic profile if R ⩽ 2. 19 In this case, the R-factor was calculated to be 15.16, indicating a hepatocellular pattern.
The RUCAM is a validated tool that uses clinical, biological, and radiologic data to evaluate the likelihood of causality for suspected DILI. 20 There are two subtypes of RUCAM based on the pattern attributed by the R-factor: one for the acute hepatocellular pattern and the other for acute cholestatic or mixed pattern. The causality classification is based on the RUCAM final score: ⩽0 (excluded), 1–2 (unlikely), 3–5 (possible), 6–8 (probable), and ⩾9 (highly probable). 20 The Revised Electronic Causality Assessment Method (RECAM) is an updated and computerized version of RUCAM, which was published in the Journal of the American Association for the Study of Liver Diseases in 2022. This tool is evidence-based and aims to increase objectivity and reliability among users. It has been shown to be as effective as RUCAM in diagnosing DILI but appears to perform better than RUCAM in cases at the diagnostic extremes. 21 The causality classification is based on the final score obtained from RECAM: < −4, unlikely/excluded; −3 to 3, possible; 4 to 7, probable; and ⩾8, highly probable. Our case had a RUCAM score of 9, which made the diagnosis of acute pirfenidone-induced liver injury highly probable (Table 1). The RECAM score was 3, which made the diagnosis possible. However, we lost 4 points in the final score because PCR HCV was not processed (−3) and due to an antinuclear factor titration >1/80 (−1).
Roussel Uclaf Causality Assessment Method for hepatocellular DILI (completed for this case).

Source: Inspired from Danan and Benichou. 20
Group I (6 causes): Recent viral infection with HAV (Anti-HAV IgM) or HBV (Anti-HBc IgM) or HCV (Anti-HCV Ig) and circumstantial arguments for non-A and non-B hepatitis); Biliary obstruction (hepatobiliary sonography); Alcoholism (AST/ALT ⩾ 2); Acute recent hypotension history (particularly if underlying heart disease).
Group II (3 causes): Complications of underlying diseases; clinical and/or biological context suggesting CMV, EBV, or Herpes virus infection.
Score, resulting causality grading: ⩽0, excluded; 1–2, unlikely; 3–5, possible; 6–8, probable; and ⩾9, highly probable.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; CMV, cytomegalovirus; DILI, drug-induced liver injury; EBV, Epstein-Barr virus; HAV, hepatitis A virus; HBV, hepatitis B virus; HCV, hepatitis C virus; Ig, Immunoglobulin; RUCAM, Roussel Uclaf Causality Assessment Method; ULN, upper limit of the normal range.
Liver biopsy is not necessary for diagnosing DILI, but it may be performed if another cause is suspected. 8 The American College of Gastroenterology (AGC) recommends performing a liver biopsy when AIH is suspected and immunosuppressive therapy is planned. AGC suggests performing a liver biopsy in the following situations: if liver tests continue to show elevated levels or if liver function worsens despite discontinuing the suspected agent; for hepatocellular DILI, if the peak of ALT level has not reduced more than 50% at 30–60 days after onset; for cholestatic DILI if the peak of ALP has not reduced more than 50% at 180 days despite withdraw of the suspected agent; if the suspected agent may not be stopped for clinical reasons or if its reintroduction is considered; if liver tests perturbation persists after 180 days with clinical signs of liver dysfunctions, like pruritus or jaundice, to excluded chronic liver disease and chronic DILI. 8 In this case, a liver biopsy was performed mainly to exclude an autoimmune disease due to the presence of abnormal antinuclear factor titration. The liver histology was compatible with DILI. Liver fibrosis was observed, which may be associated with chronic toxicity caused by pirfenidone. However, due to the limitations of interpreting liver fibrosis during acute hepatitis assessment, it cannot be confirmed. Nevertheless, based on the transient elastography processed 17 months after liver biopsy, which estimated a fibrosis stage equivalent to F0–F1 according to METAVIR classification, it appears that the liver fibrosis has recovered. During acute hepatitis assessment, clinicians should interpret liver fibrosis with caution and take into account the patient’s medical history. This is particularly important due to the growing worldwide prevalence of metabolic dysfunction-associated liver disease, which may be responsible for pre-existing liver fibrosis. 22
The initial step in treating DILI is to discontinue the suspected agent to prevent ALF. Although a few studies have shown improvement with the use of corticosteroids in DILI, it should only be considered for suspected DILI with autoimmune features and cannot be systematically recommended. 8 For cholestasis and pruritus, bile salt chelators, ursodeoxycholic acid, or antihistamines should be used. In cases of severe DILI, other treatment options may require intensive care support, N-acetyl cysteine, or even liver transplant. 13 To prevent worsening, pirfenidone was discontinued in our patient as soon as DILI was suspected, resulting in progressive biological and clinical improvement.
In conclusion, this case report describes a rare hepatic adverse reaction associated with the use of pirfenidone, which has been reported for the second time in the literature. Although DILI is uncommon, clinicians should always consider it when a patient presents with acute liver injury, particularly if a new medication has been recently introduced. A complete biological and radiological workup should be performed to exclude other causes, and the suspected drug should be promptly discontinued to prevent further progression. Liver biopsy should be considered when there is doubt or in severe cases. All cases of DILI must be reported to improve understanding of the mechanisms and enhance patient care.
Supplemental Material
sj-docx-1-taw-10.1177_20420986241270866 – Supplemental material for Pirfenidone-induced liver injury, a case report of a rare idiosyncratic reaction
Supplemental material, sj-docx-1-taw-10.1177_20420986241270866 for Pirfenidone-induced liver injury, a case report of a rare idiosyncratic reaction by Florent Fortunati, Antoine Froidure, Pamela Baldin, Yves Horsmans, Nicolas Lanthier, Géraldine Dahlqvist and Bénédicte Delire in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
