Abstract
Background:
Drug-induced liver injury (DILI) can lead to potentially fatal complications, such as acute liver failure and even death. In clinical practice, glucocorticoids have been considered in some cases of DILI, especially those with hyperbilirubinemia. However, the available evidence remains controversial and its quality is also very limited.
Objectives:
To explore the efficacy and safety of glucocorticoids in patients with acute DILI and hyperbilirubinemia.
Design:
An open-label, multicenter, randomized controlled trial (RCT).
Methods and analysis:
Overall, 232 patients with acute DILI with hyperbilirubinemia will be enrolled, and then, randomly assigned at a ratio of 1:1 to the conventional treatment alone or combined with glucocorticoids groups. The primary endpoint is the improvement of DILI, which is defined as total bilirubin level decreased by 50% after treatment, at the 2nd week. Secondary endpoints include the improvement of DILI at the 4th week and rates of progressive liver injury, liver failure, liver transplantation, survival, and adverse events. As for exploratory endpoints, we will also identify the beneficial population and assess the changes of inflammatory factors following glucocorticoids treatment.
Ethics:
The study has been approved by the Medical Ethical Committee of the General Hospital of Northern Theater Command (ethical approval number Y2024-226) as well as 11 other participating centers.
Discussion:
This RCT will provide solid evidence to clarify the benefits of glucocorticoids in patients with acute DILI and hyperbilirubinemia.
Trial registration:
ClinicalTrials.gov identifier: NCT06922669.
Introduction
Drug-induced liver injury (DILI) is a significant cause of liver damage resulting from exposure to more than 1000 kinds of drugs, such as traditional Chinese medicine, prescription medications, dietary supplements, nutraceuticals, or their metabolites and excipients. 1 The estimated incidence of DILI in China is about 23.8/100,000, which is higher than in other countries. Notably, its true incidence can be higher, because mild DILI cases are not hospitalized and DILI is underdiagnosed or overlooked in some areas with scarce medical resources. 2
In some patients with mild DILI, only nonspecific gastrointestinal symptoms are reported, and often disappear after stopping causative drugs and receiving supportive care; others are asymptomatic. By comparison, severe DILI may lead to acute liver failure (ALF), characterized by significant worsening of liver function, jaundice, coagulopathy, and even death. DILI secondary to drug toxicity and immune-mediated mechanisms has gradually become an important cause of ALF with worse prognosis. 3 Besides, some patients with DILI eventually progress to chronic liver injury, leading to liver fibrosis and even cirrhosis. 4
Currently, international practice guidelines are mostly limited to the management of DILI secondary to several specific drugs, such as acetaminophen, leflunomide, and valproate.5,6 According to the Chinese clinical practice guideline regarding the management of DILI, conventional drugs, including magnesium isoglycyrrhizinate, glutathione, silymarin, and polyene phosphatidylcholine for hepatocellular DILI, and ursodeoxycholic acid capsules and ademetionine 1,4-butanedisulfonate for cholestatic DILI, can be used for relieving hepatic inflammation and intrahepatic cholestasis.1,7 If ALF occurs in severe patients who fail to respond to conventional treatment, liver transplantation should be considered. 8 However, it has been greatly compromised by the shortage of organs, the high cost of medical treatment, and the risks of surgery.
Generally, it is necessary to explore alternative approaches to effectively control the disease progression and improve the prognosis of DILI. It should be noted that total bilirubin (TBIL) is significantly related to the prognosis of patients. 9 As reported in scattered cases,10–12 glucocorticoids should be a promising choice of treatment for acute DILI with severe hyperbilirubinemia due to their anti-inflammatory and immunomodulatory properties. 13 However, the evidence regarding clinical efficacy and safety of glucocorticoids in DILI remains controversial and its quality is very limited from observational studies.9,14–17 Accordingly, they have not been widely recognized by physicians nor formally recommended by practice guidelines.
Herein, we have designed this multicenter randomized controlled trial (RCT) to clarify whether patients with acute DILI with hyperbilirubinemia can benefit from glucocorticoids.
Methods
Study design
All hospitalized patients with a diagnosis of acute DILI with hyperbilirubinemia will be screened. After signing informed consents, eligible participants will be randomly assigned at a ratio of 1:1. Clinical and laboratory data at admission and during hospitalization and follow-up periods will be collected (Figure 1). The reporting of this study conforms to the SPIRIT statement 18 (Supplemental Material 1).
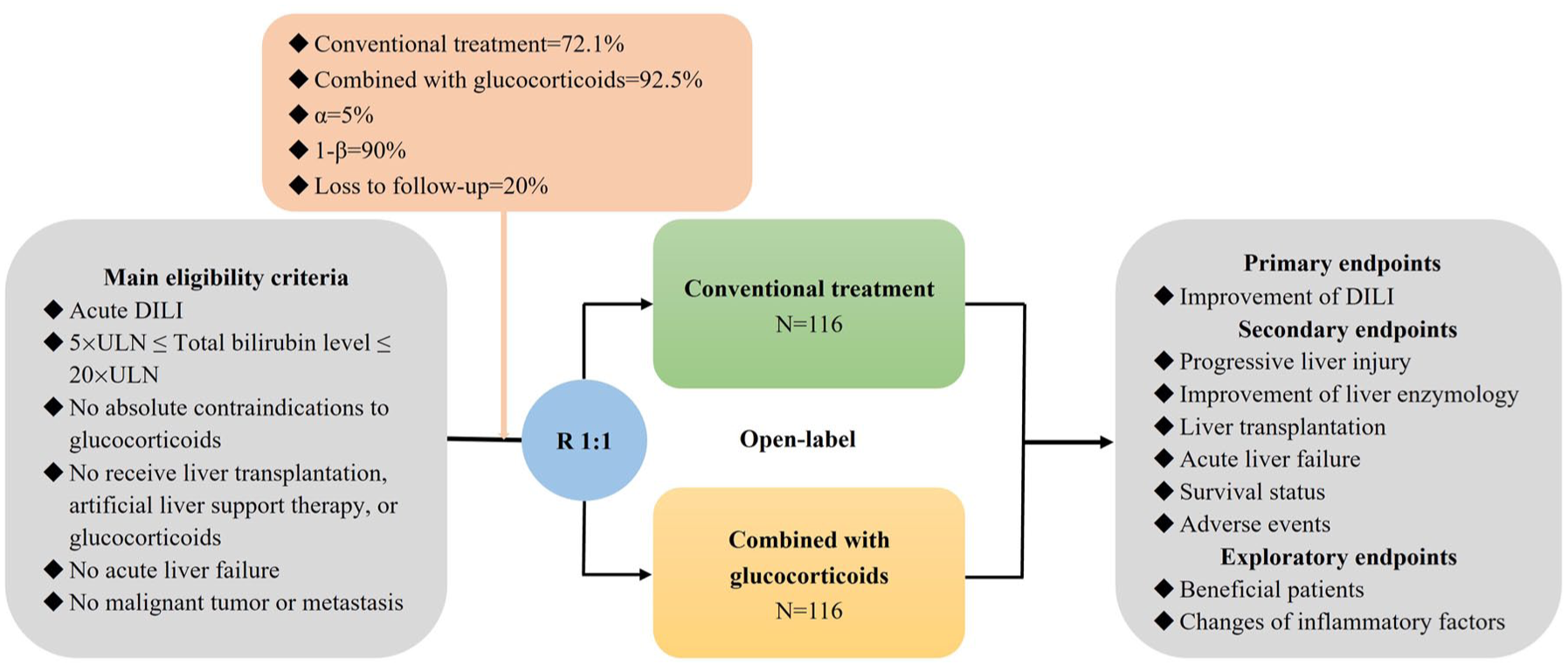
Flowchart of this trial.
Eligibility criteria
Participants will be included, if they meet the following criteria: (1) patients have a definite diagnosis of acute DILI; (2) TBIL level at baseline is 5 × ULN~20 × ULN; (3) patients’ age is 18–80 years old; and (4) patients or their relatives can sign their informed consent forms.
The exclusion criteria are as follows: (1) other causes of liver injury, including viral hepatitis, cytomegalovirus infection, Epstein-Barr virus infection, Herpes virus infection, autoimmune liver disease, alcoholic liver disease, hypoxic/ischemic liver disease, Budd–Chiari syndrome, biliary tract disease, Wilson’s disease, hemochromatosis, and α1-antitrypsin deficiency; (2) DILI secondary to the use of immune checkpoint inhibitors (ICIs) or gynura segetum; (3) absolute contraindications to glucocorticoids, such as systemic mold infections or allergies; (4) a history of glucocorticoid therapy within 3 months before enrollment; (5) other diseases requiring glucocorticoids maintenance therapy, such as rheumatoid arthritis, systemic lupus erythematosus, and systemic dermatomyositis, etc.; (6) a history of liver transplantation; (7) artificial liver support therapy before enrollment; (8) malignancy of the liver, bile duct, and pancreas or liver metastasis; (9) ALF; (10) creatinine (Cr) ⩾133 μmol/L; (11) neutrophil count <1 × 109/L; (12) active tuberculosis; (13) severe cardiopulmonary diseases; (14) recent surgery or trauma; (15) mental illness; (16) pregnancy or lactation; (17) patients who participated in other clinical trials within 3 months before enrollment; or (18) other conditions judged by the clinicians to be inappropriate for study.
Sample size calculation
Sample size is calculated by testing for a significant difference in the proportion between two groups. Based on the studies by Hu et al. 9 and Hou et al., 14 the pooled rate of improvement of acute DILI with hyperbilirubinemia should be 92.5% and 75.3% after treatment with and without glucocorticoids, respectively. An alpha value of 0.05, a power of 90%, and a dropout rate of 20% are set. Thus, 116 patients per group are calculated by the PASS 15.0.5 software (NCSS, LLC., Kaysville, UT, USA).
Randomization and blinding
Eligible participants will be randomly assigned to conventional treatment alone or combined with glucocorticoids groups at a ratio of 1:1. The randomization number is generated by an online system (Huifang Inc., Wuxi, China), and all participating centers will compete for enrollment. The trial group assignments are open-label to the participants and clinicians, but are blind to the investigators.
Interventions
According to the Chinese practice guidelines for the management of DILI, 1 conventional treatment primarily includes magnesium isoglycyrrhizinate, glutathione, silymarin, and polyene phosphatidylcholine for hepatocellular DILI, and ursodeoxycholic acid capsules and ademetionine 1,4-butanedisulfonate for cholestatic DILI. Generally, no more than three of these drugs will be combined. Traditional Chinese medicine for DILI will not be recommended.
Except for the above treatment, participants in the conventional treatment combined with glucocorticoids group will also receive 1 mg/kg/day of methylprednisolone intravenously daily for 1 week. The maximal duration of intravenous methylprednisolone treatment can be extended to 2 weeks, if necessary. Then, oral methylprednisolone tablets will be given, starting at a dose of 40 mg/day. The dosage of methylprednisolone tablets will be gradually tapered over a period of 1–3 months. Generally, if TBIL level continues to decrease, the dosage of methylprednisolone tablets will be gradually reduced by 4–8 mg per week.
Regardless of any assigned group, if participants’ conditions worsen, artificial liver support therapy and even liver transplantation will be considered.
Study endpoints
The primary endpoint is the improvement of DILI after treatment at the 2nd week. The secondary endpoints include: (1) improvement of DILI at the 4th week; (2) rates of progressive liver injury at the 2nd and 4th week; (3) rates of 50% reduction from baseline in ALT, aspartate aminotransferase, ALP, and γ-glutamyl transpeptidase levels at the 2nd and 4th week; and (4) rates of liver failure, liver transplantation, survival, and adverse events during the 90-day follow-up period. The exploratory endpoints are (1) the changes of inflammatory factors by glucocorticoids, and (2) the factors associated with a higher probability of achieving clinical benefits after glucocorticoids.
Diagnosis and definitions
The Roussel Uclaf Causality Assessment Method (RUCAM) score will be calculated. 19 Negative anti-HAV IgM, anti-HEV IgM, HBsAg, and anti-HCV are used to rule out liver damage caused by viral hepatitis. DILI will be diagnosed, if there is a RUCAM score of “highly probable” or even a total RUCAM score of “probable” but judged as DILI by the clinicians. According to the R value [R = (ALT/ULN)/(ALP/ULN)], the type of DILI can be divided into hepatocellular (R ⩾ 5), cholestatic (R ⩽ 2), and mixed (2 < R < 5).
The improvement of acute DILI is defined as TBIL level decreased by 50% as compared to the baseline. Progressive liver injury is defined as TBIL level increased compared to the baseline. ALF is diagnosed, if overt hepatic encephalopathy develop with INR ⩾1.5 after treatment. 20
Data collection
The following data will be collected: (1) demographic data, including gender and age; (2) suspected drug(s), start and stop time, and dose and route of administration; (3) height, weight, and vital signs; (4) clinical symptoms related to DILI, including fatigue, anorexia, epigastric discomfort, pruritus, fever, and rash; (5) abnormal findings on physical examination; (6) laboratory data, as shown in Table 1, including liver function, routine blood test, renal function, coagulation function, serum sodium, serum potassium, lipids, serum ammonia, alpha-fetoprotein, inflammatory factors, viral hepatitis, and immunological series; (7) liver histological results; (8) types of DILI; (9) adverse events; (10) treatment options; (11) liver transplantation; (12) artificial liver support; and (13) survival status. Data collection will be completed and stored through an online central system during the entire study period. Data modification can be traced.
Study schedule.

It is not necessary.
DILI, drug-induced liver injury.
Follow-up
Follow-up visits will be scheduled on Day 28 and Day 90. All participants will be reminded by the investigators to undergo follow-up examinations.
Study schedule
The study treatment and assessment plan are listed in Table 1.
Withdrawal
Participants will be withdrawn from the trial at any time due to any of the following reasons: (1) participants’ conditions worsen during the trial, and participants should receive liver transplantation or artificial liver support; (2) participants develop complications and are not appropriate to continue their assigned treatments; (3) participants’ adherence is poor; (4) adverse events occur; (5) other conditions judged by the investigators, which are necessary for participants’ withdrawal from the trial; or (6) participants request to terminate their assigned treatment.
Trial termination
Trial will be discontinued due to any of the following reasons: (1) major errors occur during the trial; (2) sponsors request to terminate the trial for protecting the rights and safety of the participants; (3) the National Medical Products Administration or Ethics committee order the trial termination for any reason; or (4) other reasons for trial termination according to the judgment by the investigators.
Adverse events
Adverse events related to glucocorticoids mainly include infection, water-sodium retention, Cushing syndrome, elevated blood glucose, gastrointestinal ulcer, thromboembolic disease, neuropsychiatric symptoms, osteoporosis, increased intraocular pressure, and withdrawal syndrome. Information related to adverse events will be recorded in detail, including start and end dates, symptoms and signs, severity and frequency, management, and outcomes.
Data analysis
The intention-to-treat set comprises all participants randomized. The per-protocol set consists of participants who complete their assigned treatments and have the primary endpoints observed. The safety set contains participants who receive at least one dose of glucocorticoids and undergo at least one safety evaluation. Continuous variables are described as mean ± standard deviation and median (range), and categorical variables as frequencies (percentages). Whitney U test, Chi-square test, and Fisher’s exact test are used, if appropriate. Post hoc analysis will also be conducted. Factors associated with clinical benefits after glucocorticoids will be analyzed in multivariate logistic regression analyses. A two-sided p-value of <0.05 is considered statistically significant. All statistical analyses are performed using IBM SPSS 26.0 software (IBM Corp, Armonk, NY, USA).
Discussion
The mechanisms of DILI have been widely evaluated. Except for the direct toxicity of drugs themselves, causing liver damage and dysfunction, they may activate innate immunity and T cell responses, thereby leading to the release of inflammatory cytokines.21,22 Glucocorticoids can inhibit the immune response and reduce the production of pro-inflammatory cytokines, which are beneficial for protecting hepatocytes from further damage and mitigating inflammatory responses.
Acute severe DILI is often accompanied by significantly elevated TBIL and poor prognosis. Glucocorticoids have been employed for the treatment of acute DILI with hyperbilirubinemia in clinical practice. However, they have not been widely recommended due to the lack of high-quality evidence. In a retrospective cohort study, Hu et al. 9 suggested the benefit of glucocorticoids in patients with severe DILI and hyperbilirubinemia. There is a shorter duration required to achieve a 50% reduction of TBIL level after methylprednisolone or prednisone in a pulse or reduced-dose regimen, especially in patients with TBIL level >243 µmol/L. Additionally, the recovery rate of DILI after glucocorticoids was further improved to 87.9%, but the difference was not statistically significant. This might be because liver injury was more severe in the glucocorticoid treatment group. Similarly, in another real-world observational study, Wu et al. 15 also suggested that glucocorticoids could decrease TBIL level in severe DILI (TBIL ⩾5 × ULN) at the early stage, which might be beneficial to reduce the probability of progression to ALF. The recovery could be improved in the case where the TBIL level was still elevated in spite of conventional treatment. In another observational study, where a reduced dosage of glucocorticoids was employed in 20 patients with DILI and TBIL >10 × ULN, there was a higher rate of liver enzymes normalization (100% vs 74%) and a shorter course of severe DILI (80.7 vs 184.9 days) after glucocorticoids. 14 In contrast, some studies have concluded that glucocorticoids were not beneficial for the treatment of DILI. Wan et al. 16 defined TBIL <5 × ULN as a reduction in the severity of liver injury, and found that prednisone did not significantly influence the rate of reduction in the severity of DILI, regardless of 40 or 60 mg. 16 Pang et al. 17 also found that glucocorticoids could not significantly improve the resolution of severe DILI. Collectively, there was still controversy in the efficacy of glucocorticoids for severe DILI. This might be attributed to the heterogeneity in the study design among studies. First, the definitions of severe DILI, particularly the severity of hyperbilirubinemia, are different. Second, the study endpoints were obviously different among them. Third, the dosage, formulation, and duration of glucocorticoids were not consistent among them.
To the best of our knowledge, this should be the first RCT designed to clarify the efficacy and safety of glucocorticoids for the treatment of acute DILI with hyperbilirubinemia. It has several features, as follows. First, it will be conducted at multiple centers with a relatively large sample size, increasing the generalizability of the research findings. Second, the risk of selection bias will be minimized to ensure that the patient characteristics at baseline should be comparable between the two groups. Third, DILI patients who had developed ALF will be excluded. This is primarily because ALF carries a high risk of death, and often requires liver transplantation, where glucocorticoids may be potentially ineffective. Fourth, patients with DILI secondary to ICIs will be excluded. This is primarily because glucocorticoids have been clearly recommended for DILI caused by ICIs according to the practice guidelines from various countries. Fifth, patients with DILI secondary to gynura segetum will also be excluded. This is primarily because gynura segetum may cause hepatic sinusoidal obstruction syndrome, which should be treated by the stepwise anticoagulant-TIPS therapy. Collectively, the latter three exclusion critiera will affect our assessment regarding the efficacy of glucocorticoids and conventional hepatoprotective drugs.
In conclusion, our findings will provide more solid evidence to clarify whether glucocorticoids should be considered for the treatment of acute DILI with hyperbilirubinemia and identify a group of patients who are more likely to obtain clinical benefits from glucocorticoids.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261440777 – Supplemental material for Efficacy and safety of glucocorticoids for acute drug-induced liver injury with hyperbilirubinemia: protocol of a multicenter randomized controlled trial
Supplemental material, sj-docx-1-tag-10.1177_17562848261440777 for Efficacy and safety of glucocorticoids for acute drug-induced liver injury with hyperbilirubinemia: protocol of a multicenter randomized controlled trial by Qianqian Li, Yong Lin, Xin Zeng, Lu Zhou, Fengmei Wang, Qing Ye, Yanjing Gao, Lianyi Guo, Jin Zhu, Jia Li, Yiling Li, Lichun Shao, Yiling Hu, Jiancun Xiao, Ao Jia, Dongsheng Wang, Lijun Chang, Jian Wang, Jianzhong Zhang, Ran Wang, Fei Gao, Qinke Wu, Pingfang Hu, Changpeng Zhu, Lingyan Cai, Ying Ran, Yueyue Li, Jiayuan Zhang, Yupeng Ran, Chunyan Wang, Ningning Wang, Jian Zhang, Xiehua Zhang, Juan Li, Jing Sun, Yunxiang Chu, Yali Ma, Tingting Wang, Zheng Zheng, Yue Shen, Xingshun Qi and Weifen Xie in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
