Abstract
Introduction:
Clozapine, an antipsychotic used in the treatment of schizophrenia, is known for its serious side effects. In order to promote patient safety, drug–drug interaction (DDI) databases can be consulted by clinicians. In this study, the degree of consensus between five sources on DDIs with clozapine is determined.
Methods:
The summary of product characteristics of clozapine, Delphicare interaction database, Stockley’s interaction checker, the Lexicomp interaction database, and the interaction database of Clinical Pharmacology are included. By comparing the original categories assigned to interactions with clozapine by the included DDI sources, a degree of consensus between sources is determined. Furthermore, based on the combined information, an evaluation on the severity of each potential interaction is made.
Results:
One hundred eighty-three potential DDIs with clozapine are retrieved from the five included sources. A consensus between sources is found in 47.5% (
Conclusion:
This study shows major discrepancies between five different sources on DDIs with clozapine. The potential impact of the use of one specific database on patient safety and prescribing behavior could prove to be problematic.
Plain language summary
Clozapine is an antipsychotic agent advised to be used in the treatment of schizophrenia after two failed adequate treatments with other antipsychotics. A reluctance towards prescribing clozapine is described in literature, mainly because of its potential life threatening side effects. In assuring patient safety, evaluating potential interactions with other drugs are a key aspect in the risk assessment of clozapine. Drug–drug interaction (DDI) databases are developed to assist clinicians in this assessment.
In this study we compare five sources on DDI with clozapine. Major discrepancies are found between sources with a consensus found in less than half of all included DDIs. It is hypothesized that these discrepancies can contribute to excessive caution or to negligence in prescribing clozapine. The impact of relying one specific database in the risk assessment of DDIs with clozapine could thus prove to be potentially problematic.
Based on the comparison of the included sources, a conclusion was made on the severity of each unique potential DDI with clozapine. The results are shown in the appendix of supplementary materials.
Introduction
Clozapine is an atypical antipsychotic agent and the treatment of choice in refractory schizophrenia.1–3 Practical guidelines suggest its use after two failed adequate trials with other antipsychotic agents in patients suffering from schizophrenia.4,5 At this moment, there are no alternative treatments available with a comparable efficacy in treatment resistant schizophrenia.6–8 Clozapine is known for its greater tolerability and lower intrinsic risk of extrapyramidal side effects as compared to other antipsychotic agents. 9 However, it is also known to be underused out of fear for other adverse drug reactions (ADRs), some of which are life-threatening.4,6 Among these serious ADRs associated with clozapine are agranulocytosis and myocarditis. The risk of agranulocytosis can be managed by WBC counts and absolute neutrophil counts in monthly blood tests.10,11 Myocarditis can be prevented by slow titration of clozapine. 12 Despite the existence of these safety measures it is described that fear of ADR keeps clinicians from prescribing clozapine to those most in need of an effective antipsychotic agent and literature even suggests the existence of ‘clozaphobia’.4,12,13 To counter this, a rational use of clozapine is promoted based on knowledge about pharmacokinetics, therapeutic drug monitoring, and slow titration. 12
To obtain rational use of clozapine, drug–drug interactions (DDIs) should be taken into account. DDIs can influence the therapeutic efficacy and ADR of clozapine. DDIs involving pharmacokinetics can cause fluctuations in plasma levels of clozapine. Clozapine depends on the cytochrome P450 (CYP) enzymes for its metabolism. More specifically, CYP1A2, CYP2D6, and CYP3A4 are among the enzymes known to be involved in the metabolism of clozapine.14,15 CYP inducing or inhibiting drugs can alter clozapine plasma levels when combined with clozapine.16,17 Alternatively, DDIs with clozapine can occur on a pharmacodynamic level. An example of an additive interaction is the combination of bupropion, a norepinephrine–dopamine reuptake inhibitor, with clozapine. The combination is stated to lower the threshold for seizures. 18 Thus, a clinician should be alert to potential DDIs when combining clozapine with other drugs.
In order to support clinicians in everyday practice with the risk assessment of combining drugs with clozapine, various DDI databases can be consulted. Moreover, DDI databases are often implemented in a hospitals computerized physician order entry (CPOE) as a clinical decision support system to generate alerts when potentially harmful combinations are prescribed. Although designed to assist a clinician or pharmacist in the evaluation of the safety of a drug combination, DDI databases tend to over-alert clinicians. 19
Because of the reluctant attitude toward prescribing clozapine due to potential life-threatening ADR, solid information on potential DDI is vital to support cautious prescribing of clozapine in the vulnerable population suffering from treatment resistant schizophrenia. Over-alerting clinicians could however contribute to ‘clozaphobia’ and lead to poorer outcomes for patients. In our study, we compare the categorization systems of five DDI sources and investigate the degree of consensus between the sources on DDIs with clozapine. Furthermore, an evaluation of the severity of each included DDI with clozapine is made based on information of all sources. In this way, we aim to investigate the potential impact of different sources on risk assessment and contribute to alert prescribing of clozapine.
Methods
Color labels assigned to the different interaction severity levels of the five original sources.

SmPC, summary of product characteristics.
Determining consensus between sources
Potential DDIs with information from only one source are not assessed. Information from at least two out of five sources on potential DDI is needed in order to decide on a consensus. Considering each unique DDI, a consensus is concluded when the respective categories of each of the original sources are all assigned the same color label (Table 2).
Determination of consensus.
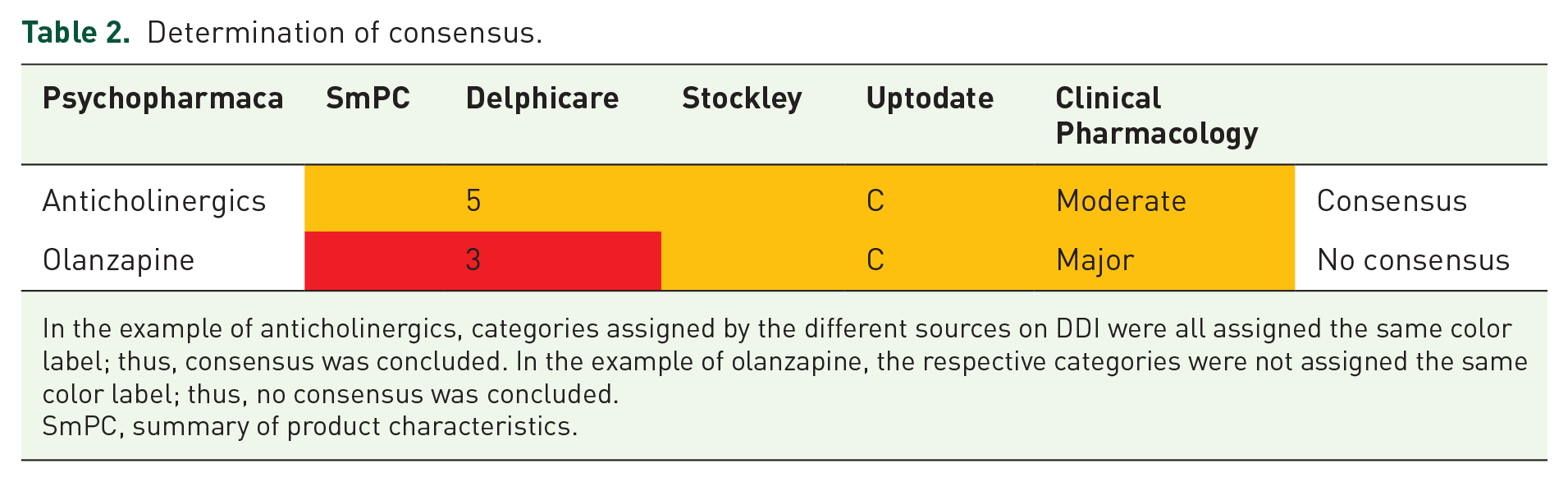
In the example of anticholinergics, categories assigned by the different sources on DDI were all assigned the same color label; thus, consensus was concluded. In the example of olanzapine, the respective categories were not assigned the same color label; thus, no consensus was concluded.
SmPC, summary of product characteristics.
Evaluation of the severity of the DDI
An evaluation on the severity of each unique DDI is made by comparing the respective color categories from the included sources in the excel file, by two separate researchers (Table 3). Sources without information on a DDI are excluded from the evaluation. When at least half of the sources categorize the DDI as a contraindication (red label), the DDI is evaluated as ‘contraindicated’ and a red label is assigned in conclusion. When all sources categorize the DDI as safe, the DDI is evaluated as ‘safe’ and a green label is assigned in conclusion. When at least one source categorizes the DDI as a potential harmful interaction, the DDI is evaluated as ‘caution needed’ and an orange label is assigned in conclusion.
Determination of the level of severity of each DDI based on information from five sources on DDI.

DDI, drug–drug interaction.
The initial analysis is performed by a pharmacist (VA) and repeated by a psychiatrist (DHM) in order to reach consensus.
Results
A total of 183 potential DDIs with clozapine are retrieved from five DDI sources. 27.3% (
The number of sources (of the five included sources on DDI) that reported on a potential DDI (of the 183 DDI included).
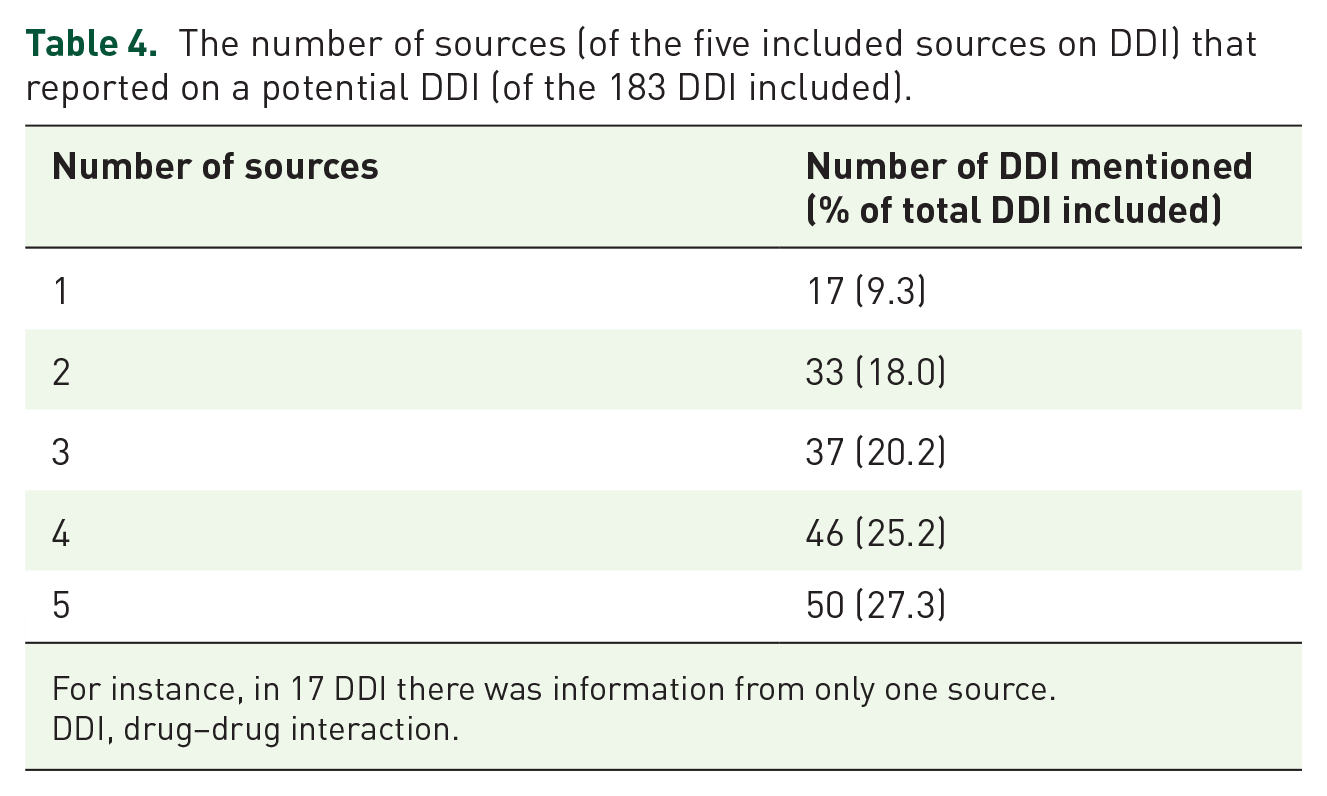
For instance, in 17 DDI there was information from only one source.
DDI, drug–drug interaction.
Determining consensus between sources
In 9.3% (
Prevalence of consensus on the level of severity of an interaction between the included sources on DDI.

Information of at least two sources was needed in order to determine consensus.
DDI, drug–drug interaction.
Prevalence of the combination of different levels of severity assigned to a DDI where no consensus was concluded.

DDI, drug–drug interaction.
Discrepancies in DDI categorization between sources are found. The SmPC of clozapine states that 31.3% (
Evaluation of the severity of the DDI
Of all DDIs, 11.5% (
Discussion
This study compares categorization systems of different DDI sources and investigates the degree of consensus on the severity of potential DDIs with clozapine. Five well-known sources are included; the SmPC of clozapine, the Delphicare interaction database, Stockley’s interaction checker, the Lexicomp interaction database, and the interaction database of Clinical Pharmacology. These sources provide a categorizing system of potential DDIs to support a clinician in the decision making process. Furthermore DDI databases are often implemented in hospital CPOE to alert clinicians in case of a potential DDI.
Because of the potential life-threatening ADR of clozapine, 11 solid data on potential DDIs is essential to ensure patient safety. This information is needed to help clinicians avoid contraindicated combinations or monitor drug combinations with a potentially harmful DDI. At the same time, it is hypothesized that over-alerting as a cause of a DDI categorization system can result in the clinician avoiding a drug. Because of the ‘clozaphobia’ described in recent literature this may especially be a problem when it comes to clozapine treatment. 12 This could prove problematic for the highly vulnerable population of patients suffering from treatment resistant schizophrenia. It is argued that the delicate pharmacological consideration asks for consistent information on DDIs. In what follows we investigate consistency of DDI sources by evaluating the degree of agreement between different sources on DDI with clozapine.
Determining consensus between sources
A first important indication of the discrepancies between sources is that more than a quarter of the included DDIs (
These discrepancies are confirmed by the comparison of categories of the five included DDI sources. A consensus is found in only 47.5% (
Evaluation of the severity of the DDI
In recent literature, clinicians are advised to consult more than one source or database on DDI when assessing the risk of a drug–drug combination.
25
In our study, two researchers separately evaluate each unique potential DDI with clozapine based on the assigned categories from the five included sources. A conclusion on the risk of each potential DDI is made and labeled as ‘contraindicated’, ‘caution needed’ or ‘safe’ (see Appendix of Supplemental Materials). In conclusion, 11.5% (
Our findings underline the impact of the choice of a DDI database on the evaluation of a drug combination with clozapine or on the hospital CPOE alert system. It is clear that the SmPC and the APB Delphicare interaction database are more conservative in the categorization of a potential DDI with clozapine. This contributes to a poor degree of consensus between sources. The lacking consistency of DDI databases and poor agreement among DDI databases on DDI with clozapine is in line with recent findings on the matter.25,27
It is hypothesized that reasons for these discrepancies lie in the lack of solid evidence on DDI with clozapine. Most literature on DDIs with clozapine addresses pharmacokinetic interactions through the CYP enzyme metabolism. Some drugs, such as fluoroquinolones,15,28 fluvoxamine, 29 and omeprazole, 30 have extensive literature on their CYP inducing/inhibiting properties. Some DDI result in conflicting findings, such as mirtazapine 31 and valproate 32 or in mere case reports such as olanzapine 33 and flupentixol. 34 Interestingly, literature on potential pharmacodynamical interactions of clozapine with drugs such as benzodiazepines is scarce. Moreover, recent research shows none of the potential psychotropic DDIs with clozapine are supported by primary studies containing a combined sample size of more than 100 patients. 35 Although these combinations are common in psychiatric care, supporting evidence is lacking and is predominantly based on case reports.
Strengths and limitations
This study is the first to compare five different sources on DDIs with clozapine. The inclusion of five international sources can be considered as a strength. A total of 183 DDIs are included; however, this list may not be exhaustive. Furthermore, some databases render no detailed information on certain interactions. This affects the assessment of some DDIs and may lead to an overestimation of the risk of certain DDI with clozapine.
Conclusion
Clozapine is important in the management of treatment resistant schizophrenia though it is known to be under prescribed due to its potential harmful ADR. DDIs can have an important impact on the efficacy as well as the ADR of clozapine. Physicians across specialties and pharmacists can rely on DDI databases for information on a potential DDI with clozapine. Moreover, these databases are often implemented in the CPOE software in hospitals to warn clinicians when potentially harmful combinations are prescribed.
Our study shows major discrepancies between five different sources on DDI with clozapine. There are large differences in the number of reported interactions by DDI sources. Furthermore, researchers find a poor agreement between sources, with a consensus in less than half of all included DDIs. The impact of relying one specific database in the risk assessment of DDIs with clozapine proves to be potentially problematic.
Furthermore, substantial evidence on potential DDIs with clozapine is lacking. Although primary literature on DDIs with clozapine is much needed, our comparison of different sources on DDI may help clinicians in evaluating the safety of specific drug combinations and managing further monitoring of a potential DDI with clozapine.
Supplemental Material
sj-xlsx-1-taw-10.1177_20420986241233842 – Supplemental material for Alert prescribing of clozapine: a comparison of five drug–drug interaction sources
Supplemental material, sj-xlsx-1-taw-10.1177_20420986241233842 for Alert prescribing of clozapine: a comparison of five drug–drug interaction sources by Jeroen Govaerts, Annelies Verluyten, Filip Bouckaert, Marc A. F. De Hert and Franciska A. M. Desplenter in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.




