Abstract
Background:
The circulation of falsified medical products is a global threat and is expected to be higher in low- and middle-income countries.
Objective:
This study was conducted to assess the understanding, readiness, and response of Eritrea’s healthcare professionals (HCPs), and identify potential areas of intervention to combat circulation of falsified medical products.
Design:
This was a nationwide population-based cross-sectional survey, conducted in December 2021.
Methods:
This study enrolled representative samples of HCPs working in public and private health facilities. Two-stage stratified cluster sampling was used to select study participants and data were collected through face-to-face interviews. Descriptive statistics, Mann–Whitney U test, Kruskal–Wallis test along with their post hoc tests, Jonckheere-Terpstra, and logistic regression analyses were performed as appropriate.
Results:
The study enrolled 707 HCPs, and 96.6% were successfully surveyed. The majority of the participants (62.5%) encountered products with suspected quality defects and 63.8% claimed that they had reported the incident(s) at least once. About 85% reported that complaints should be submitted to the Eritrean Pharmacovigilance Centre and 74.0% indicated that it should be reported at the earliest time possible even if the reporter lacks details. The standard reporting form for suspected product quality issues was correctly recognized by 13.8%. Overall, the median knowledge and attitude scores were found to be 9 out of 17 (interquartile range, IQR: 4.0) and 30 out of 35 (IQR: 4.0), respectively. Not knowing how to report (55.6%) and what to report (34.9%), no/delayed feedback from the regulatory authority (30.0%), and unavailability of reporting forms (29.0%) were the frequently reported barriers to reporting. In addition, profession (p = 0.027), no/delayed feedback (adjusted odds ratio [AOR]: 4.70; 95% CI: 2.17–10.18; p < 0.001), and not knowing how to report (AOR: 0.12; 95% CI: 0.05–0.28; p < 0.001) were found to be determinants of reporting suspected product quality defects.
Conclusion:
The readiness and response of Eritrea’s HCPs in detecting and reporting falsified medical products seems promising, although a significant knowledge gap was observed.
Plain language summary
Background: Healthcare professionals (HCPs) play a key role in the fight against circulation of falsified medical products, a global threat. Therefore, they are expected to be knowledgeable, vigilant, and responsive enough toward early detection and reporting of suspected falsified medical products. This study was conducted to assess the understanding, readiness and response of Eritrea’s HCPs in combating falsified medical products and identify potential areas of intervention.
that there is a significant association of knowledge and attitude with the level of education; those with low level of education had low knowledge and poor attitude. reporting a suspected product quality problem and hindering factors of reporting were found to be significantly associated with profession. three-fourth of the study participants indicated that cases should be reported at the earliest possible time even if the reporter lacks details. that the majority of the respondents could not recognize the standard reporting form for suspected product quality issues.
Keywords
Introduction
Despite rapid progress in the advancement of healthcare innovations, including optimizing pharmaceutical processes, discoveries, and surveillance systems, the burden of circulation of falsified medical products is becoming overwhelming. 1 Falsified medical products are defined by the World Health Organization (WHO) as ‘medical products that deliberately/fraudulently misrepresent their identity, composition or source’. 1 Falsified medical products may also include products with correct ingredients, wrong ingredients, insufficient active ingredients, or without active ingredients, and/or having fake packaging. 2
Although the true global incidence/prevalence, health impact, and economic burden of falsified medical products have not been measured yet for different reasons, studies conducted so far have reflected that the circulation of such products is a global threat.2–4 According to the WHO estimates, about 73 billion euros worth of counterfeit medical products are traded annually. 5 The extent of the problem varies greatly among regions and individual countries, and the problem is expected to be higher in low- and middle-income countries (LMIC) where there are weak regulatory systems and inadequate supplies of essential medicines to meet local demands.1,6,7 In 2017, it was estimated that 1 in 10 medicines in LMIC were either substandard or falsified, costing an estimated USD 30.5 billion each year. 1
Fighting against the falsification of medical products offers a crucial challenge in the field of public health. The burden is under-reported despite its vast occurrence, and being a cause of unnecessary morbidity, mortality, and loss of public confidence in healthcare systems. 6
Eritrea has been engaged in the fight against substandard and falsified (SF) medical products. Since 2014, around 1688 suspected product quality defects have been reported from healthcare professionals (HCPs) to the Eritrean Pharmacovigilance Center (EPC). Accordingly, 82 medical products were recalled by the National Medicines and Food Administration (NMFA), Eritrea, for either being substandard or falsified. 8 The NMFA, a regulatory body, governed under the Ministry of Health is mandated through Proclamation No. 36/1993 to control medicines, medical supplies, cosmetics, and sanitary items. Its regulatory function includes premarketing evaluation and marketing authorization, licensing establishments, laboratory testing, regulatory inspection, and pharmacovigilance. The EPC and the Regulatory Inspection Unit are responsible for the surveillance of SF medical products in Eritrea.
Currently, the prevailing peace with the neighboring countries is likely to open business opportunities and increase the movement of people which makes the control of SF medical products a challenge. Accordingly, it requires stricter regulation, cross-border collaboration, and increased vigilance of HCPs to win the fight against falsified medical products. The readiness and challenges of Eritrea’s HCPs to prevent, detect, and respond to incidents of falsified medical products are still unknown. This study was, therefore, conducted to assess the understanding, readiness, and response of HCPs in Eritrea in combating falsified medical products and identify the potential areas of intervention.
Methods
Study design and setting
This was a descriptive cross-sectional study using a quantitative approach and it was conducted in December 2021. It involved representative samples of HCPs from all levels of the health facilities (health stations, health centers, community hospitals, primary hospitals, regional referral hospitals, National Referral Hospitals, dental laboratories/clinics, and drug retail outlets), and administrative offices (national and subnational levels) available in public and private sectors of the six administrative regions of the country. During the study period, there were 290 health facilities, 300 drug retail outlets, 5 dental clinics, and 4 dental laboratories in Eritrea 9 .
Study population
During the study period, there were about 6349 HCPs working in health-related institutions in Eritrea. 10 All HCPs who were actively working during the data collection period were the target population. Regardless of their academic qualification, workplace, and work experience, all HCPs had an equal opportunity to take part in the study.
Exclusion criteria
Establishments without renewed licenses, nontechnical staff, internees, and HCPs working in military health institutions were excluded from the study.
Sample size determination
The sample size was calculated using the single proportion formula with finite population correction and taking p (proportion) as 0.5 for no study, to determine the knowledge, attitude, and practice on SF medical products that have been conducted in Eritrea. Statistics for the confidence interval (Z), of 1.96, and margin of error (e) were taken as 0.05. Accordingly, the initial sample size was calculated to be 363, and after adjustment by a design effect of 1.75 due to the two-stage sampling used and 10% nonresponse, the final sample size was 707 HCPs.
Sampling technique
Two-stage stratified cluster sampling was used to select the HCPs. Taking the observational fact that the knowledge, attitude, and practice of HCPs on falsified medical products varies across the administrative zones of Eritrea, each zone was considered as a stratum to obtain a reliable estimate of the main variables. During the first stage of the sampling, health-related institutions (primary sampling units) were selected using probability proportionate to size from each zone/strata (Supplemental Material 1). In the second stage, HCPs were selected from the primary sampling units systematically.
Sample allocation
Although proportional allocation is usually the preferred approach for sample allocation, 11 the square root allocation (power allocation with power equal to 0.5) method was used to allocate the sampled HCPs. This is because the sample size allocated for Debubawi Keih Bahri using proportional allocation to the population was too small to bring reliable estimates. The number of HCPs for each zone, allocation of samples, and formation of clusters are displayed in Supplemental Material 2.
Data collection tool and approach
Data were collected using a self-designed, structured questionnaire (Supplemental Material 3). The questionnaire was developed by authors who have diverse expertise in the field of pharmacy, clinical pharmacy, pharmacoepidemiology, and health system strengthening. The questionnaire was constructed to capture the relevant parameters to measure the knowledge, attitude, and practice of the HCPs on falsified medical products.
The questionnaire entails four domains regarding the demographic characteristics of participants and tries to assess the knowledge of the respondents using 12 questions, attitude was measured using 9 questions (organized in a five-point Likert scale). Lastly, the practice of HCPs toward falsified medical products was assessed by nine questions. The data were collected using face-to-face interviews and, in some instances, a telephone interview was applied when participants could not be reached physically.
Data collectors were trained on the data collection tool and procedure to minimize the intra- and inter-rater variability and potential selection bias. The questionnaire was not validated; however, a pretest was conducted among 30 individuals prior to the study to check the suitability and comprehensiveness of the questionnaire in Edaga Hamus Community Hospital, an institution that was not selected in the survey.
The name list of the randomly selected participants was communicated to each health facility’s administrator. Then, the interviewer explained the objectives of the study and gathered written informed consent. The interviews were conducted in a conducive environment in the participants’ workplace, and no one else was present besides the participant and the interviewer. Enough time was provided to complete the interviews.
Controlling bias and confounders
The knowledge, attitude, and practice of HCPs on falsified medical products could be influenced by the location where the health facility is located. As such, a two-stage stratified cluster sampling design was employed, and weights were computed to minimize the influence of sampling bias. To avoid the selection of the most accessible health facilities which can affect the generalizability of the study findings, all health facilities were given a chance for selection, and telephone interviews were done for hard-to-reach health facilities. Moreover, data collectors were strictly instructed and supervised to enroll only the randomly selected HCPs. Participants were considered as nonresponse without replacement, after making three attempts to reach them. Although it is completely unavoidable, ample time was given to the respondents to remember past details, and thereby minimize recall bias. In addition, to avoid information bias that could arise through the influence of other people or literature search, questionnaires were filled in the form of an interview, instead of self-administration.
Statistical analysis
The data were entered into the computer using the Census and Survey Processing System (CSPro, Version 7.3) software package and exported to SPSS (Version 26) for analyses. To account for the multistage sampled nature of the data, weights were computed before analyses. Descriptive statistics was performed for categorical variables (using frequency and percentage) and numerical variables such as mean (SD) or median (interquartile range, IQR), as appropriate. Percentages of some indicators were expressed along with their 95% confidence intervals. After confirming non-normality using the Kolmogorov–Smirnov test, nonparametric tools, namely Mann–Whitney and Kruskal–Wallis test were used to assess the difference in knowledge score and attitude score across the categories of the predictors. For the significant categorical predictor variables with more than two categories, pairwise comparison was performed using a post hoc test, accounting for the multiple comparison adjustment. The presence of a trend in knowledge and attitude scores across the ordinal predictor categories was assessed using Jonckheere-Terpstra; moreover, effect sizes were computed. To determine the factors affecting the reporting of suspected product quality defects, bivariate and multiple logistic regression were used. p-Values less than 0.05 were considered significant throughout the analysis.
The study was reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 12 as shown in Supplemental Material 4.
Results
Background characteristics of the study participants
A total of 707 HCPs were approached for enrollment and 24 were unable to participate due to refusal (n = 4) and unavailability (n = 17). Moreover, the remaining three partially filled out the questionnaire making the response percentage 96.6%. Out of the final 683 participants, the majority were females (56.8%), and about one-third (37.5%) of the population lie within the age group of 20–29 years. Two-thirds (66.4%) of the respondents were from the Maekel zone, and 38.3% were from National Referral Hospitals. About 44% of the overall participants had a diploma/advanced diploma educational background and 30.4% had 20 years and above of professional experience. The weighted and un-weighted percentage distribution of the characteristics of the study participants is summarized in Table 1.
Background characteristics of the participants.

Public health, physiotherapy, imaging/radiology, optometry, dental therapy, nurse aid.
IQR, interquartile range.
Knowledge of falsified medical products
Almost 90% (89.2%) of the respondents had ever heard about falsified medical products and out of them, 85.3% stated that there is a difference between falsified and substandard medical products.
More than 80% of the respondents were able to correctly distinguish falsified medical products as ‘poor quality of unknown origin’ (86.5%) and ‘intentionally mislabeled products’ (82.6%). About half of the respondents were also able to correctly label ‘products with defects that arise from manufacturing errors’ as non-falsified medical products (51.4%) and ‘products with a good quality of unknown origin’ as falsified medical products (51.9%). Moreover, the majority of the respondents correctly identified that medicines (96.4%), medical supplies (76.4%), vaccines (74.9%), diagnostics (74%), and medical devices (63.9%) could be falsified.
Adverse effects (82.5%) and treatment failure (66.2%) were the most commonly reported consequences of using falsified medical products (Figure 1). Besides, the high demand which exceeds the available supply of medical products (45%) and weak market surveillance (37.8%) were reported as key factors which encourage the circulation of falsified medical products (Figure 2).

Percentage distribution of the participant’s knowledge of the consequences of using falsified medical products.

Percentage distribution of the participant’s knowledge of factors encouraging infiltration/circulation of falsified medical products.
HCPs (74.6%), regulatory bodies (55.3%), and the general public (46.1%) were the most commonly reported stakeholders, who could play a role in preventing infiltration/circulation of falsified medical products (Table 2). Market surveillance/inspection (57.7%) and awareness raising (57.3%) were also the most frequently suggested activities that should be done to prevent infiltration/circulation of falsified medical products.
Percentage distribution of the participant’s knowledge on stakeholders who play a role in preventing infiltration/circulation of falsified medical products and activities needed to prevent further infiltration.

Professional associations and civil societies, religious entities, and UN agencies.
Enhance commitment and motivations of HCPs, research, encourage domestic manufacturing, provide adequate budget for the purchase of medical products, reduce the price of medicines, purchase medical products from governmental facilities, prepare and follow policies, and guidelines for procurement.
The standard form used in Eritrea to report suspected falsified medical products was correctly recognized by only 13.8% of the respondents. The submission of the filled standard reporting form to the EPC of the NMFA (84.7%), respective Zonal Pharmacy (80.2%), and pharmacovigilance focal points (76.4%) were correctly identified by the majority of the respondents. While PHARMECOR Eritrea (pharmaceutical and medical suppliers corporation) (37.4%) and Pharmacy Services Division (27.7%) were incorrectly identified as Centers that could receive suspected incidents.
On assessing the knowledge regarding the best time to report falsified medical products, 74.0% of the respondents reported that suspected cases should be submitted at the earliest possible time even if details are not available. On the other hand, few respondents perceived that the best time to report is after the reporter confirms that the product is falsified (10.5%) when several similar cases are identified (7.5%) if the information on the reporting form is fully complete (4.1%) and/or after completion of investigation by a zonal committee (3.9%).
Attitude toward falsified medical products
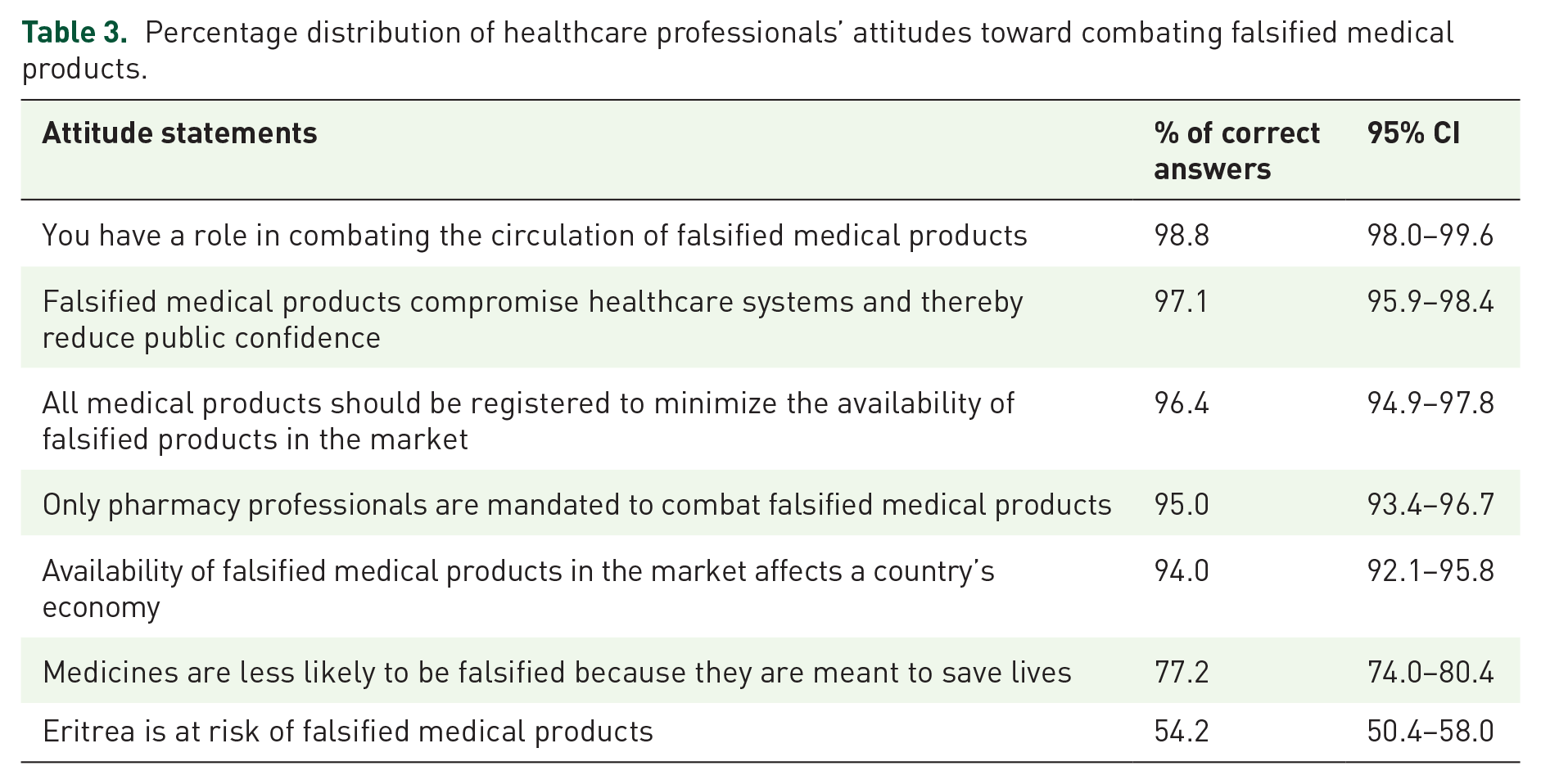
The majority of the respondents had a positive attitude that favors combating falsified medical products with an overall attitude score of 30 out of 35 (IQR: 4.0) (Table 3). About half of the respondents (45.8%, 95% CI: 42.0–49.6) believed that their knowledge and skills for the detection and reporting of falsified medical products were not adequate.
Percentage distribution of healthcare professionals’ attitudes toward combating falsified medical products.
Practice of reporting of suspected SF medical products
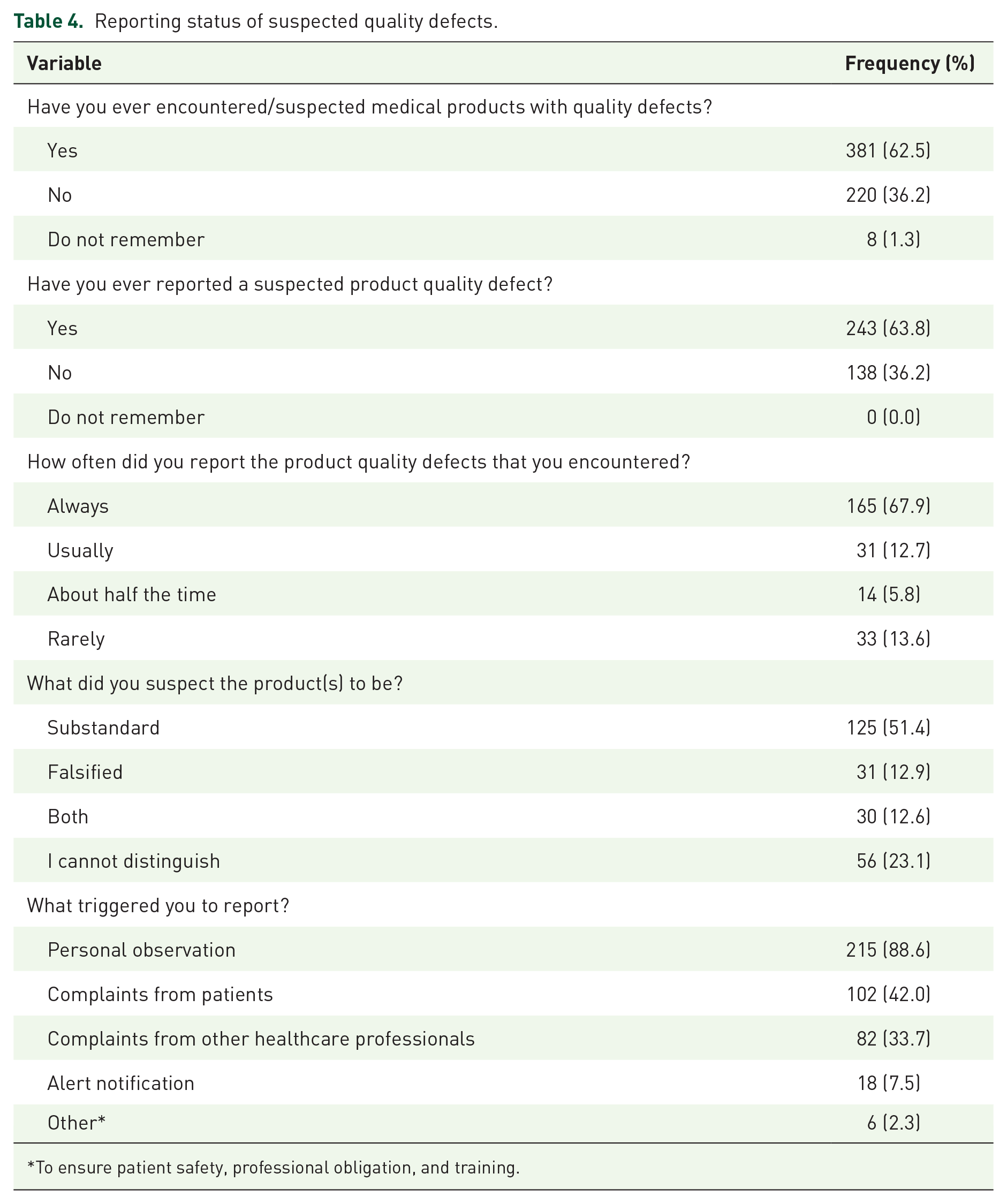
About two-thirds (62.5%) of the study participants claimed that they had encountered products with suspected quality defect(s) and 63.8% of the respondents had reported the quality defect(s) at least once during the period of their practice. About 68% of those who had a history of reporting claimed that they had reported all of the incident(s) encountered. The most triggering factor to report was personal observation (88.6%) followed by patient complaints. Moreover, about 51.4% of the participants suspected the encountered products with quality defects to be substandard products (Table 4).
Reporting status of suspected quality defects.
To ensure patient safety, professional obligation, and training.
When those participants who encountered defective products were asked if there was any hindrance in reporting suspected quality defects, 63.7% (n = 241) reported that they had at least one hindering factor while 36.3% reported no hindrance. Not knowing how to report (55.6%) and what to report (34.9%) followed by no/delayed feedback (30.3%) and unavailability of reporting forms (29.0%) were the most frequently reported hindrances (Table 5).
Percentage distribution of barriers to reporting.

Unavailability of alternatives of medicines, perception that similar cases should not be reported, the belief that contraband medicines are not reportable and/or are out of the scope of regulation.
Regarding the training status of the study participants, less than one-fifth (16.5%) reported that they were trained about falsified medical products, and 83.5% had shared their knowledge of falsified medical products with others. From those who did not share their knowledge (n = 100) on falsified medical products, the main reasons for not sharing were as follows: had no enough knowledge (39.9%), had no interest (28.1%), limited personal experience (27.5%), low perceived risk (7.9%), and had no time to share (4.6%).
Comparison of the knowledge score across the categories of demographics
The median (IQR) knowledge score of the respondents was 9 out of 17 (4.00). Mann–Whitney U test revealed that the median knowledge score among males (M = 9, IQR = 3) was significantly greater than those of females (M = 8.1, IQR = 3) (p < 0.001) with an effect size of 0.185.
Knowledge scores across the various professions revealed significant differences (p < 0.001) with an effect size of 0.122. After employing the multiple comparison adjustment, pharmacists were found to have significantly greater median knowledge scores compared to nurse practitioners (p < 0.001) and laboratory experts (p < 0.001).
A significant trend in knowledge score was observed with an increase in the level of education (p < 0.001), with an effect size of 0.092. HCPs with bachelor/masters degree (M = 10, IQR = 5) had higher knowledge scores compared to those with diploma/advanced diploma (M = 9, IQR = 2, p < 0.001) and those with a certificate (M = 8, IQR = 4, p < 0.001). A summary of the comparison of the knowledge score across the categories of demographic characteristics is shown in Table 6.
Comparison of knowledge scores across the categories of demographic characteristics.

B/M, bachelor/masters; C, certificate; D/A, diploma/advanced diploma; IQR, interquartile range; JKT, Jonckheere-Terpstra test; L, laboratory; M, median; M/D, medicines/dentistry; NP, nurse practitioner; NS, not significant; P, pharmacy; Z, standard score.
Comparison of attitude score across the categories of demographics
The median attitude score was found to be 30 (IQR: 4.0) out of 35. Mann–Whitney U test revealed that the median attitude score among vmales (M = 31, IQR = 4) was significantly greater than those of females (M = 29, IQR = 5) (p < 0.048), with an effect size of 0.077. According to the Jonckheere-Terpstra test, no significant trend in attitude was observed with increase in an age (p = 0.736).
When attitude score was compared across the various professions, a significant difference was observed (p < 0.001), with an effect size of 0.104. Using multiple comparison adjustments, pharmacists were found to have significantly greater median attitude scores compared to nurse practitioners (p < 0.001), laboratory experts (p = 0.028), and medicine/dentistry (p = 0.006).
Similarly, with an increase in level of education, a significant trend in attitude score was observed (p < 0.001), with an effect size of 0.192. Respondents with bachelor/masters degree had higher attitude scores compared to those with diploma/advanced diploma (p < 0.001) and certificates (p < 0.001). A summary of the comparison of attitude scores across the categories of demographic characteristics is given in Table 7.
Comparison of the attitude scores across the categories of the demographic characteristics.

B/M, bachelor/masters; C, certificate; D/A, diploma/advanced diploma; IQR, interquartile range; JKT, Jonckheere-Terpstra test; L, laboratory; M, median; M/D, medicines/dentistry; NP, nurse practitioner; NS, not significant; P, pharmacy.
Determinants of reporting suspected product quality defects
The possible factors affecting the reporting of suspected product quality issues were determined using logistic regression. At the multivariable level, profession (p = 0.027), no/delayed feedback from the regulatory authority (p < 0.001), and not knowing how to report (p < 0.001) were the variables found to significantly influence the reporting pattern. Accordingly, the medicine/dentistry and pharmacist professions, respectively, were associated with 81% (AOR: 0.19, 95% CI: 0.05–0.73) and 75% (AOR: 0.25, 95% CI: 0.08–0.80) decrease in reporting when compared with laboratory personnel. Those who do not know how to report were found to be less likely to report (AOR: 0.12, 95% CI: 0.05–0.28). On the other hand, the odds of reporting among those who responded that there is delayed feedback was higher (AOR: 4.70, 95% CI: 2.17–10.18) when compared with their counterparts. Details of the bivariate and multiple logistic regression results are shown in Table 8.
Factors affecting reporting suspected product quality defects using bivariate and multiple logistic regression.
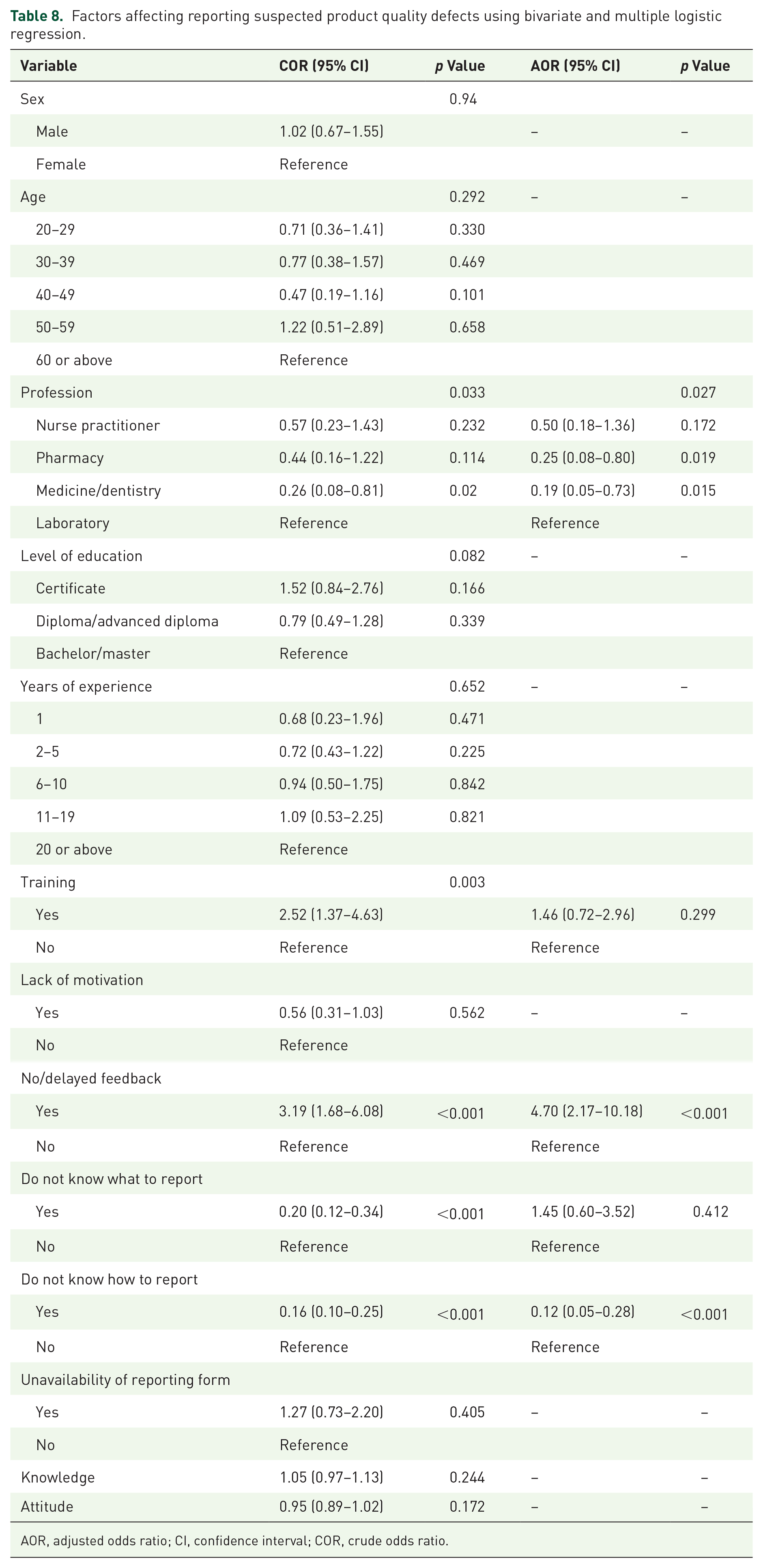
AOR, adjusted odds ratio; CI, confidence interval; COR, crude odds ratio.
Discussion
HCPs’ contribution in reporting suspected SF medical products was found to be substantial as about two-thirds of the respondents claimed that they had reported suspected product quality issues. This, however, needs to be further maximized as several hindering factors have been reported by the respondents. Lack of knowledge on what and how to report products with quality defects, failure of the regulatory authority to provide feedback in a timely manner, and ensure the availability of reporting forms were the main reasons, and these problems were also reported as barriers to reporting in studies done in Kuwait, United Kingdom, and United States.13–15
This study also revealed that there was confusion about the reporting channel and limited knowledge of the national reporting form, that is, the blue form (Supplemental Material 5), used for reporting suspected SF medical products. To address such problems, in collaboration with partners, well-designed risk communication and HCP engagement strategies, and massive training are recommended to be shortly implemented. Although the majority of the respondents correctly understand that suspected quality defects should be reported at the earliest possible time, however, some respondents had a misconception that cases should only be reported once they are confirmed. This misunderstanding should, therefore, be corrected as it could be a potential cause of delayed reporting and/or not reporting at all.
The study also indicated that several of the respondents, especially the lower-level HCPs, had limited knowledge of the detection and reporting of falsified medical products. Since 2015, the EPC has incorporated topics related to monitoring SF medical products into its basic pharmacovigilance training. This training, however, was mainly targeted toward pharmacists, general practitioners, and holders of bachelors of Science in Nursing. This might be one possible explanation for the low knowledge score among the lower-level HCPs.
The fact that a significant proportion of the respondents believe that they have a role to play in combating falsified medical products and that fighting falsified medical products is every HCP’s responsibility, gives confidence in their future engagement. Besides, the substantial transfer of knowledge observed in this study and the history of reporting suspected SF products are all good signs of readiness of the HCPs to contribute to the fight against the circulation of falsified medical products. If this good attitude of HCPs that favors combating falsified medical products is complemented with massive awareness-raising programs, and strengthening the existing surveillance system in the country, then it is anticipated to attain success in the effort to fight against the global threat.
Considering that Eritrea’s medical products procurement system is centralized, some respondents believe that ‘Eritrea is not at risk of falsified medical products’. Similar misconceptions were also reported in HCPs residing in high-income countries. 16 It is, therefore, important to aware HCPs that no country is immune to the circulation of falsified medical products.3,17–19
Having HCPs with inadequate knowledge of detecting and reporting falsified medical products is an opportunity for the circulation of falsified medical products in the country. This could, in turn, expose consumers to potentially serious public health risks. In this regard, this study has outlined several key implications. As raising awareness is the first step in fighting falsified medical products, organizing massive sensitization programs, especially targeted to the lower-level HCPs, on preventing, detecting, and reporting product quality defects has paramount importance. Such training has been delivered by the EPC of the NMFA in collaboration with major stakeholders such as zonal medical offices, professional societies, UN partners, and academia. This should, however, be strengthened and efforts need to be made by the major stakeholders mentioned above to reach the untrained HCPs. In the long run, taking into consideration the demanding nature of in-service training, courses on medicine quality monitoring need to be incorporated into academia.
In addition, to tackle no/delayed feedback by the regulatory authority as a potential barrier, the NMFA should acknowledge and motivate HCPs who report any suspected product quality defects at the earliest possible time.
Limitation
In this study, the proportion of reporting SF medical products by HCPs presented in this study was as reported by the respondents and could not be confirmed by the research team. HCPs who verbally notified the pharmacy department, whenever they encountered a suspected SF medical product, were considered as if they had actually reported it. Moreover, as the reported information might have been affected by recall bias, the proportion of reporting could be slightly under or over-estimated.
On the questions related to practice, all the respondents were asked if they had ever encountered and reported suspected SF medical products rather than falsified products. As it is impractical for the respondents to accurately differentiate the nature of the quality defect, even if, they conducted an independent investigation. Thus, readers should be well aware that the proportion of reported suspected quality defects in this study does not represent only falsified products.
Conclusion
The overall readiness and response of Eritrea’s HCPs in detecting and reporting falsified medical products seems to be promising, although a significant knowledge gap was observed especially in the lower-level HCPs. Developing strategies, mobilizing resources, and establishing zonal regulatory offices as well as facility-based vigilance systems are critical interventions for success. Such approaches are imperative to facilitate cascade training, identify untrained professionals, and ensure the availability of reporting forms, that can ultimately result in timely reporting of incidents. Moreover, the regulatory body should also provide timely feedback and motivate HCPs.
Supplemental Material
sj-docx-1-taw-10.1177_20420986231213713 – Supplemental material for Understanding, readiness, and response of healthcare professionals in combating falsified medical products in Eritrea: a population-based survey
Supplemental material, sj-docx-1-taw-10.1177_20420986231213713 for Understanding, readiness, and response of healthcare professionals in combating falsified medical products in Eritrea: a population-based survey by Yodit Fitsum, Azania Werede, Abubeker Mahmud Saleh, Eyasu H. Tesfamariam, Francis Magombo, Freminatos Misghina, Hermella Yemane, Iyassu Bahta, Liya Abraham, Merhawi Bahta, Merhawi Debesai, Meron Tesfagaber, Michael Ghirmai, Sirak Tesfamariam and Mulugeta Russom in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986231213713 – Supplemental material for Understanding, readiness, and response of healthcare professionals in combating falsified medical products in Eritrea: a population-based survey
Supplemental material, sj-docx-2-taw-10.1177_20420986231213713 for Understanding, readiness, and response of healthcare professionals in combating falsified medical products in Eritrea: a population-based survey by Yodit Fitsum, Azania Werede, Abubeker Mahmud Saleh, Eyasu H. Tesfamariam, Francis Magombo, Freminatos Misghina, Hermella Yemane, Iyassu Bahta, Liya Abraham, Merhawi Bahta, Merhawi Debesai, Meron Tesfagaber, Michael Ghirmai, Sirak Tesfamariam and Mulugeta Russom in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-3-taw-10.1177_20420986231213713 – Supplemental material for Understanding, readiness, and response of healthcare professionals in combating falsified medical products in Eritrea: a population-based survey
Supplemental material, sj-docx-3-taw-10.1177_20420986231213713 for Understanding, readiness, and response of healthcare professionals in combating falsified medical products in Eritrea: a population-based survey by Yodit Fitsum, Azania Werede, Abubeker Mahmud Saleh, Eyasu H. Tesfamariam, Francis Magombo, Freminatos Misghina, Hermella Yemane, Iyassu Bahta, Liya Abraham, Merhawi Bahta, Merhawi Debesai, Meron Tesfagaber, Michael Ghirmai, Sirak Tesfamariam and Mulugeta Russom in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-4-taw-10.1177_20420986231213713 – Supplemental material for Understanding, readiness, and response of healthcare professionals in combating falsified medical products in Eritrea: a population-based survey
Supplemental material, sj-docx-4-taw-10.1177_20420986231213713 for Understanding, readiness, and response of healthcare professionals in combating falsified medical products in Eritrea: a population-based survey by Yodit Fitsum, Azania Werede, Abubeker Mahmud Saleh, Eyasu H. Tesfamariam, Francis Magombo, Freminatos Misghina, Hermella Yemane, Iyassu Bahta, Liya Abraham, Merhawi Bahta, Merhawi Debesai, Meron Tesfagaber, Michael Ghirmai, Sirak Tesfamariam and Mulugeta Russom in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-5-taw-10.1177_20420986231213713 – Supplemental material for Understanding, readiness, and response of healthcare professionals in combating falsified medical products in Eritrea: a population-based survey
Supplemental material, sj-docx-5-taw-10.1177_20420986231213713 for Understanding, readiness, and response of healthcare professionals in combating falsified medical products in Eritrea: a population-based survey by Yodit Fitsum, Azania Werede, Abubeker Mahmud Saleh, Eyasu H. Tesfamariam, Francis Magombo, Freminatos Misghina, Hermella Yemane, Iyassu Bahta, Liya Abraham, Merhawi Bahta, Merhawi Debesai, Meron Tesfagaber, Michael Ghirmai, Sirak Tesfamariam and Mulugeta Russom in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The authors sincerely thank the Zonal Medical Offices in all zones for their remarkable support in facilitating the study. Besides, the authors would like to acknowledge the Personnel Recruitment and Deployment Unit, and Health Information System of the Ministry of Health for providing the required information on distribution of health facilities and healthcare professionals. Above all, the study participants are highly appreciated for their time and honest answers.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
