Abstract
Background:
Triazole antifungals are widely used as broad-spectrum antifungal activity; however, there are many undetected and unreported adverse events (AEs).
Methods:
Data from the Food and Drug Administration Adverse Event Reporting System (FAERS) from the first quarter (Q1) of 2004 to the third quarter (Q3) of 2021 were selected for disproportionality analysis to assess the connection between antifungal triazoles, and AEs and important medical events (IMEs).
Results:
A total of 22,566 records associated with triazole antifungals were identified, with 9584 triazole antifungal–IME pairs. The following system organ classes (SOCs) appeared as significant signals: ‘Endocrine disorders’ [reported odds ratio (ROR) = 167.94], ‘Metabolism and nutrition disorders’ (ROR = 46.30), and ‘Skin and subcutaneous tissue disorders’ (ROR = 21.37). Strong signals were observed with respiratory failure, rash, hepatic function abnormal, and hypokalemia. Uncommon security signals included a change in the QT interval, neurotoxicity, pseudoaldosteronism, and hallucinations.
Conclusion:
Various triazole antifungals cause AEs of different types and intensities of association. Our results are broadly consistent with prescribing information and previous studies; however, additional pharmacoepidemiological studies are required to verify AEs with modest incidence but high signal.
Plain Language Summary
Keywords
Introduction
Triazole antifungals cease fungal growth by inhibiting lanolin 1-4-alpha-demethylase, the cytochrome P450-dependent membrane protein that inhibits the conversion of lanolin to ergosterol, which is primarily responsible for maintaining the integrity of fungal cell membrane.1–3 Triazole antifungals are broad-spectrum antifungals with clinical indications such as the prevention and treatment of invasive fungal diseases primarily caused by immune deficiency, including the prevention and treatment of invasive fungal diseases caused primarily by immune deficiency.2,4 For the therapy of candidemia in nonneutropenic, invasive pulmonary aspergillosis (IPA), and to prevent invasive candidiasis in the intensive care unit setting, the preferred administration of triazole antifungals as therapeutic agents was proposed by the Clinical Practice Guideline for the Management of Candidiasis and Practice Guidelines for the Diagnosis and Management of Aspergillosis of the Infectious Diseases Society of America (IDSA), updated in 2016.5,6
In 2004, there were already fluconazole and itraconazole in the market. Voriconazole was launched in 2002. Posaconazole and isavuconazole were introduced in 2006 and 2015, respectively. Although voriconazole has been widely used in children aged > 2 years and adults, there is no indication for its use in neonates and children aged < 2 years. The safety and efficacy of isavuconazole in pediatric patients less than 18 years of age have not been established; therefore, there is no indication for pediatric use. Most of the recently reported side effects of the first four antifungals are dryness, drug rash, hair loss, diarrhea, fatigue, nausea, and vomiting, and can also lead to prolonged QT interval.7–13 Although isavuconazole has a similar mechanism of action and belongs to the same triazole class, it causes an adverse reaction opposite to that of the other four drugs, for example, shortening of the QT interval. 14 In addition, the boxed warning of itraconazole indicates drug interactions, cardiac effects, and congestive heart failure (CHF). Therefore, patients with onychomycosis should not be treated with itraconazole when they also have CHF or evidence of ventricular dysfunction (such as a history of CHF).
However, it is unclear whether triazole antifungals may cause other rare or serious adverse events (AEs). Therefore, a comprehensive and in-depth description of important AEs caused by triazole antifungals is critical. This study analyzed data collected from the Food and Drug Administration Adverse Event Reporting System (FAERS) to examine the system organ classes (SOCs), AEs, and important medical events (IMEs) signals associated with triazole antifungals. We also analyzed information on patient characteristics to provide a more comprehensive understanding of the safety of triazole antifungals and to provide evidence and assurance for their appropriate use in the clinical setting.
Methods
Data sources and standardization
The FAERS is an adverse reaction spontaneous reporting database which contains AE reports, medication error reports, and product quality complaints resulting in AEs that were submitted to FDA. This study coded reported AEs, product quality complaints, and medication errors using terms from the Medical Dictionary of Regulatory Activities (MedDRA) External Link Disclaimer terminology. It enables quantification of the association between the drug and the reported AEs by signal detection.15–17 In this study, we extracted reports of all triazole antifungal drugs in FAERS between the first quarter of 2004 (first FDA approval) and the third quarter of 2021 (Q3; the latest). Each report in the FAERS database includes information on the following areas: administrative and demographic information and the PRIMARYID; information on the source of the reports; patient outcome information in the case reports; reaction information in the case reports; and medication information in the case reports. At the same time, the FAERS data are processed to remove duplicate data. The information on the demographic information form is processed according to the FDA recommendation; if two or more reported CASEIDs (the number for identifying a FAERS case) were the same, the latest FDA_DT (the date the FDA received the case) was selected. If the CASEID and FDA_DT were the same, the higher PRIMARYID (a unique number for identifying a FAERS report) was selected. 18 The drug names in FAERS have not been standardized and contain various forms of trade names and generic names for each drug. We obtained the trade names of the drugs to be studied by searching the DrugBank database online. The study drugs included fluconazole (Diflucan), itraconazole (Tolsura, Sporanox), voriconazole (Vfend), posaconazole (Noxafil), and isavuconazole (Cresemba). 19 We selected only the role codes set as the primary suspect drug for the initial report. In MedDRA, all AEs were coded as preferred terms (AEs), and in addition, the corresponding master SOCs were assigned according to the AEs, this corresponds to a systematic classification of medical terms. We screened triazole antifungal drug-associated AEs through the list of IMEs and mapped the characteristics of triazole antifungal drug-associated AEs. To identify suspected adverse drug reactions (ADRs) of concern within routine pharmacovigilance activities, the European Medicines Agency (EMA) developed a list of IMEs, which will be used to prioritize its review of reports of suspected ADRs. A complete list of IMEs can be found at https://www.ema.europa.eu/en/documents/other/meddra-important-medical-event-terms-list-version-241_en.xlsx. Serious outcomes include those that result in disability, hospitalization, death, or life-threatening events.
Statistical analysis
In studies of pharmacovigilance, AE signals can indicate potential drug-related AE risk and can be detected using many methods. In this study, we used the most common method of signal detection, disproportionality analysis, which may represent a signal that the frequency of occurrence of a target drug–event pair is disproportionate to other drug–event pairs in the FAERS database.20,21 We verify the stability of the detection signal by the proportional reporting rate method.22,23 The application of the proportional reporting ratio and 95% confidence intervals (CIs) from the two-by-two contingency table are shown in Table 1. According to EMA guideline, to reduce the probability of a false-positive signal, a significant signal was defined as reported odds ratio (ROR) > 2 with at least 10 cases. All analyses were performed in Python 3 (Anaconda, Friedrichstrasse 123 10117 Berlin, Germany) and Microsoft® Excel® 2019 (Microsoft Company, Microsoft Building, No. 5 Danling Street, Haidian District, Beijing, China).
Two-by-two contingency table for the disproportionality analysis.

AEs, adverse events.
‘a’ represents the number of reports containing both the suspect drug and the suspect ADR; ‘b’ represents the number of reports containing the suspect drug with other ADRs (except the event of interest); ‘c’ represents the number of reports containing the suspect ADR with other medications (except the drug of interest); and ‘d’ represents the number of reports containing other medications and other ADRs.
Results
Descriptive analysis
During the study period (from 2004Q1 to 2021Q3), 16,854,646 AE reports were collected in the FAERS database, which contained a total of 78,729 reports of triazole antifungals being reported as the primary suspected drug. Including 21,239 (38.24%) AEs and the distribution of IMEs among the five triazole antifungals and information about the clinical characteristics of the patients are shown in Tables 2 and 3. Figure 1 demonstrates the number and trend of AEs and IMEs for each year from 2004 to 2021. Generally, among AEs and IMEs collected by FAERS related to triazole antifungals, the distribution of such indicators was similar. The majority of reported cases were male (44.76%), and in the information about age, the median age of patients with triazole antifungal-associated AE was 57 years [interquartile range (IQR) 39–69 years], except for unspecified age, and nearly half (45.08%) of the patients were below 65 years of age. Except for unspecified data, the median weight of reported AE-related patients was 64 (IQR: 52–71) kg. There was an overall upward trend in the number of AE and IME reports with each successive reporting year, peaking in 2019 (AE = 2876, IME = 1527). The majority of AE reports were from the United States (40.86%), followed by France (6.56%). The indications were mainly for the prevention and treatment of fungal infections, such as aspergillosis. Among the outcome indicators reported for patients, serious AEs (SAEs) related to hospitalization were the most frequently reported (28.19%), followed by death (19.84%), and life-threatening accounted for 6.33%. It suggested that triazole antifungals may have potentially SAEs.
Distribution of triazole antifungals across therapies.

AEs, adverse effects; IMEs, important medical events.
Information of patients with reported AEs/ IMEs associated with triazole antifungals.

AEs, adverse effects; IMEs, important medical events; IQR, interquartile range.

The number of AE/IME reports for triazole antifungals between 2004 and 2021.
Association intensity of different SOCs with triazole antifungals-related AEs/IMEs
Considering the triazole antifungals as a whole, the associated AE and IME signal intensities at different SOCs are shown in Table 4. The following SOCs with high signal values were observed: ‘Endocrine disorders’ (ROR of AEs = 167.94; ROR of IMEs = 20.89). In addition, ‘Congenital, familial, and genetic disorders’ (ROR of AEs = 109.91; ROR of IMEs = 27.90), ‘Metabolism and nutrition disorders’ (ROR of AEs = 46.30; ROR of IMEs = 9.84), and ‘Skin and subcutaneous tissue disorders’ (ROR of AEs = 21.37; ROR of IMEs = 7.71). Triazole antifungal-induced AEs also showed significance, among them, ‘Surgical and medical procedures’ had a lower number of reports but relatively no significance in IMEs.
Signal strength at the SOC level in triazole antifungals and AEs/IMEs.

AEs, adverse effects; IMEs, important medical events; ROR, reported odds ratio; SOC, system organ class.
The spectrum of IMEs differs in triazole antifungals regimens
To explore whether there is an association between each specific IME and different drugs, we further analyzed the data. Figure 2 lists the top 50 AEs signals by frequency and also includes whether this adverse reaction was reported in the prescribing information (PI). ROR is set as an indicator by us and it is defined as a strong signal when the value is not below 20. The complete table of PTs for triazole antifungals can be found in Supplementary Table S1. The profiles of AE signals are approximately the same in Figure 2 and Supplementary Table S1. The Preferred Terms (PTs) in Figure 2 are grouped by SOC and further sorted in the corresponding SOCs according to the total number of reports in descending order. For the complete signal spectrum, the signals with ROR ⩾ 10 in triazole antifungal-involved AEs were mainly distributed in ‘General disorders and administration site conditions’ (n = 2853), and accordingly, this is where most signals appeared. This result was in line with the intensity between the relevant SOCs attributed by AEs in Table 4.
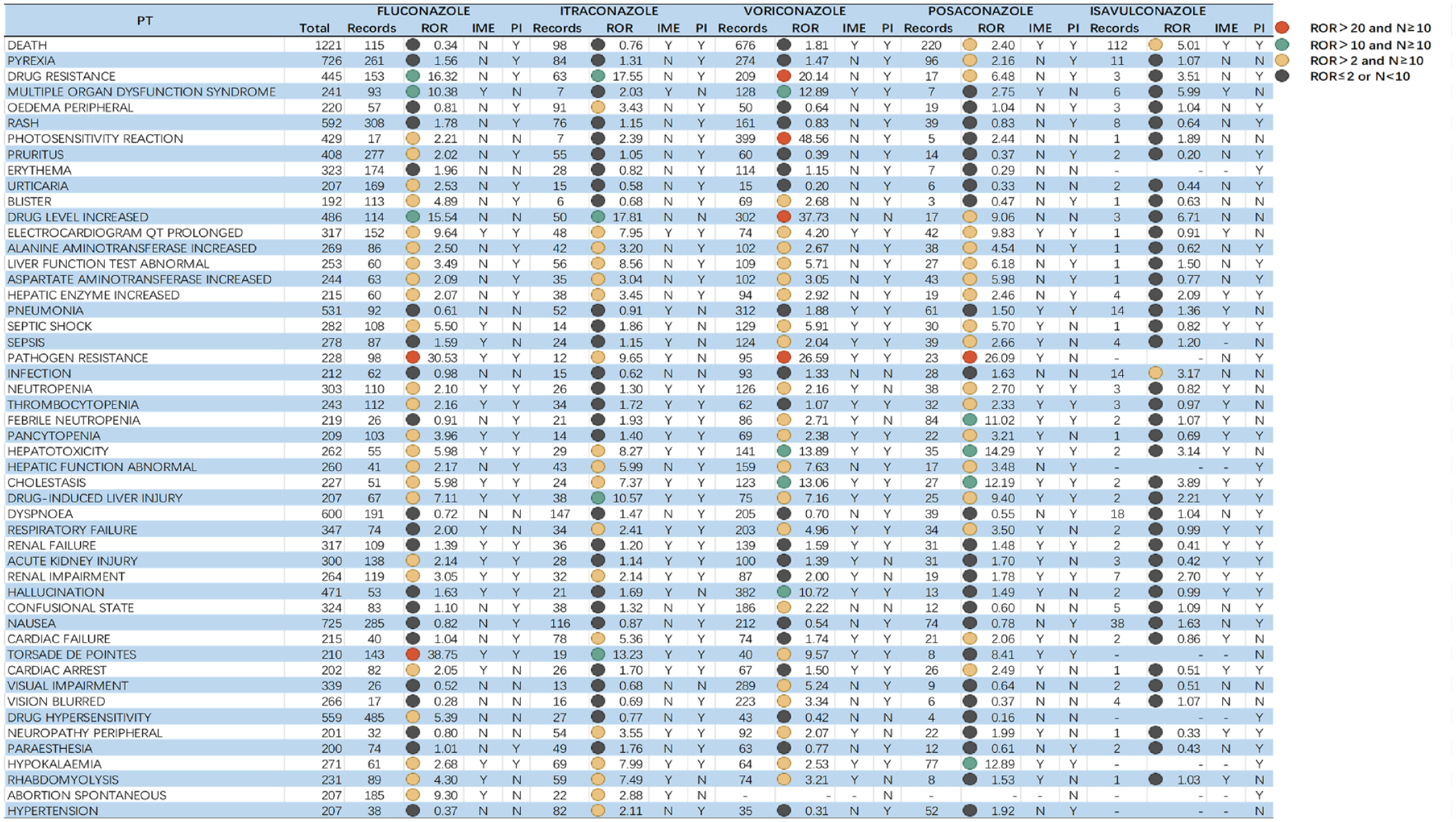
Top 50 AEs induced by triazole antifungals.
From a drug-specific perspective, the broadest spectrum of detected signals was fluconazole, which included a total of 182 signals, ranging from pruritus (ROR = 2.02) to synostosis (ROR = 193.71). Most of the AEs identified with fluconazole were currently known as AEs (rash, neutropenia, etc.); however, some stronger signals were found in it (vaginal mucosal blistering), which was not mentioned in the previous PI for fluconazole. For itraconazole, a total of 72 AE signals were mined, with ROR025 values ranging from 2.01 (cardiac failure congestive) to 1211.90 (pseudohyponatremia). Strong signals emerged in pseudohyponatremia, symmetrical drug-related intertriginous, and flexural exanthema. The ROR signal for adverse reactions of voriconazole ranged from 2.04 (face edema) to 48.56 (photosensitivity reaction). A total of 71 significant signals were observed, in addition to the rash, hepatotoxicity, and other ADRs that are common to both fluconazole and itraconazole. Voriconazole also had some ADRs, such as photosensitivity reaction, hallucination, which are different from the other azoles. A total of 36 PTs were strongly associated with posaconazole, of which pseudoaldosteronism (PIPH), hepatocellular injury, and hepatotoxicity were all strongly signaled by posaconazole. Since isavuconazole has been on the market for a relatively short period of time, fewer AEs have been reported, and therefore, no adverse reactions meeting the threshold criteria have been reported.
Discussion
In this study, the ROR method was used to mine the FAERS database for potential adverse signals regarding triazole antifungals. The method is based on statistical ideas and disproportionate analysis of data using the ROR method, which has the advantage of mutual validation in a quantitative and rapid manner to reveal the association of the target drug with AEs. In contrast to previous studies that analyzed AEs caused by all triazole antifungal drugs,24–27 we concentrated on the IMEs related to triazole antifungals, visually expressing the incidence of an important potential danger signal.
Our study showed that some azoles, including fluconazole, itraconazole, voriconazole, and posaconazole, are associated with QT prolongation, this is shown in the PI. The QTc interval is the meaning of the time between depolarization and repolarization of ventricular myocytes, and the interval is related to heart rate, thus is shortened in tachycardia and prolonged in bradycardia. QT intervals > 440 ms are considered pathologically prolonged. 28 There are case report studies supporting the association between QT prolongation and fluconazole.29,30 By retrospectively analyzing patients treated with voriconazole from 2009 to 2015, Gueta et al. compared QT intervals with and without voriconazole, and the results showed a risk of QT prolongation in hematologic patients taking voriconazole, with an AE manifested mainly by QT ⩾ 450 ms and hypokalemia. 31 Alkan et al. found that an adolescent patient with acute lymphoblastic leukemia treated with voriconazole revealed symptoms of QT interval prolongation, nonsustained polymorphic ventricular tachycardia, and asymptomatic bradycardia. In addition, prolonged QT interval was found to be uncorrelated with voriconazole concentration and dose after redosing. 30 By studying the effect of fluconazole on the human ether-a-go-go-related gene (hERG) K+ channels (wild type, F656C, and Y652A) expressed in human embryonic kidney (HEK293) cells, Han et al. found that the mechanism by which fluconazole causes acquired long QT syndrome (LQTS) may be through direct inhibition of hERG currents and disruption of hERG protein transport; the obligatory determinants of hERG current inhibition by fluconazole are the Y652 and F656 molecular mutations. 32 It is stated in the instructions that these four drugs cause QT prolongation by inhibiting the rectifier potassium channel current (Ikr). Therefore, before prescribing these triazoles, a history of heart disease and coadministration of drugs with potential QT prolongation or metabolism by CYP3A4 should be confirmed in patients. Higher frequency ECGs, and laboratory tests during voriconazole administration, can detect QT interval prolongation over time. However, unlike the first four drugs, the instructions for isavuconazole show that it shortens the QT interval of the heart in a dose-dependent manner. Mellinghoff et al. suggested that the QT interval is influenced by electrolytes, and hypocalcemia influences the de- and repolarization phases of the cardiac action potential by altering the potentials across myocyte cellular membranes. 33 Therefore, in clinical practice, physicians should proactively ask patients if they have a history of relevant genetic disorders. For patients with familial short QT syndrome disease, a very rare autosomal dominant inherited channelopathy, isavuconazole should be contraindicated.33,34
In Supplementary Table S1, it can be seen that adverse reactions regarding endocrine system disorders mainly include adrenal insufficiency, PIPH. In a single-center, retrospective, observational study, the adverse effects of posaconazole-induced pseudohyperaldosteronism were shown. 35 Kevin Kuriakose 36 think a potential mechanism is the inhibition of the enzyme 11-hydroxysteroid dehydrogenase 2, with resultant apparent mineralocorticoid excess. Adrenal suppression or insufficiency may also result from azole-mediated inhibition of CYP3A4 during concomitant administration of azole antifungals with oral, intravenous, or inhaled corticosteroids. 27 If PIPH is not resolved, a change to another triazole antifungal or the addition of an aldosterone antagonist is the additional potential interventions to consider. Despite these interventions, PIPH may persist after treatment modification. Also, early diagnosis and continuous monitoring should also be performed. 37
Neurotoxicity, the main feature of the clinical presentation, is a rare and serious adverse reaction. 38 It is shown in the instructions that triazoles may increase plasma levels of vinca alkaloids and cause neurotoxicity due to their inhibitory effect on CYP3A4. The mechanism by which triazole antifungals cause neurotoxicity is unclear. Eşkut et al. 39 speculated that the coexistence of these systemic abnormalities may be related to the interaction of immune-mediated mechanisms, individual factors, or some unknown genetic cause. Baseline neurotoxicity assessment should be performed before initiating triazole therapy and should be reviewed regularly. Early detection of neuropathy is required to prevent the development of potentially irreversible symptoms. 40 Such adverse reactions can also occur when two drugs are coadministered. Since vincristine is a CYP3A4 substrate and azole antifungals are CYP3A4 inhibitors, when triazole antifungals are coadministered with vincristine, they inhibit the metabolism of vincristine, resulting in overexposure to vincristine and serious neurotoxicity.41,42
Thompson et al. found differences in the fluorine content of fluconazole, itraconazole, voriconazole, and posaconazole. Voriconazole is a trifluorinated compound containing three fluorine atoms; fluconazole and posaconazole are difluoro compounds with two fluorine atoms; and itraconazole does not contain fluorine atoms. 43 By analyzing 215 cases, Tailor and Richardson 44 found that only voriconazole caused periosteal reactions and bone pain, and concluded that there is a correlation between skeletal manifestations and hyperfluoridemia. This is also indicated in voriconazole’s PI: fluorosis and periostitis reactions have been reported during long-term voriconazole treatment, and voriconazole should be discontinued if these symptoms occur. In a study by Moon et al., high plasma fluoride concentrations with periostitis and clinical symptoms of skeletal pain were found in patients on voriconazole therapy for a long time. This adverse effect can be prevented by measuring fluoride concentrations. 45 Wermers et al. 46 found that transplant patients on long-term voriconazole produced exophytic bone and warty painful periostitis associated with fluoride overdose, in addition to a decrease in fluoride levels and improvement in skeletal pain when voriconazole was discontinued.
Some AEs (e.g. hallucinations) associated with voriconazole have been previously reported in the literature.47–51 Hallucinations may occur after voriconazole and isavuconazole are administered in the PI and may also occur when fluconazole is overdosed. A study addressing the prospective collection of data on AEs and systematic observation of patients using a predefined clinical study format found a 16.6% incidence of hallucinations in patients with hematological and other malignancies after voriconazole administration, which was significantly higher than the levels found in previous studies. This may be because physicians focus more on whether the patient’s illness improves after taking voriconazole and do not ask about AEs (such as hallucinations) that may be considered unrelated. Patients should be warned of this AE before starting treatment with voriconazole; driving should be avoided after administration and patients should be reminded that hallucinations are not a sign of mental illness. 49 A large multicentre study of therapeutic drug monitoring of voriconazole showed that AEs related to neurotoxicity (including visual and auditory hallucinations) are minimized when voriconazole is administered at drug concentrations ⩽ 5 mg/liter. Therefore, voriconazole is a predictor of neurotoxic AEs, and its dose can be reduced or it can be discontinued when this AE occurs. 52
In AEs and IMEs caused by triazole antifungals, we obtained signals of hepatobiliary disorders, gastrointestinal disorders, and nervous system disorders, which were generally in line with the safety data derived from another systematic review and meta-analysis. 53 Therefore, when using such drugs, clinicians and patients should pay attention to whether they have AE symptoms mentioned above and, if necessary, performance measures, such as drug monitoring or drug discontinuation, should be implemented.
This study has some limitations. First, FAERS is the part of the Spontaneous Reporting System, which has inherent flaws, such as incomplete information reported, duplicate cases reported, unverified information in reports, inability to determine incidence through reports, and the presence of reports that do not determine causality. Although we performed duplicate data removal and manual correction of the data, there may still be records for these five drugs that were not retrieved. Second, despite the disproportionate analysis is mostly used in pharmacovigilance, this method still has limitations and lacks criteria to assess the validity and extent of drug safety problems.54,55 To provide clinicians with a quicker and more intuitive understanding of suspected AEs with triazole antifungals, we defined a threshold grading of signal intensity. In addition, monitored AE signals represent only potential associations, not causality. Finally, the single use of these five triazole antifungal drugs was analyzed by us in this study. Therefore, in the follow-up sequence study, we considered comparing other drugs and the safety signals after the combination. Nonetheless, a large amount of data on adverse drug events is provided by FAERS. We systematically mined and analyzed potential AEs and IMEs signals related to triazole antifungals and provided the number of reported AEs and signal values of triazole antifungal-induced AEs, which may provide a valuable real-world basis for further investigative studies of clinical practice and drug–AE relationships in the field.
Conclusion
With the widespread utilization of triazole antifungals, potential safety concerns have emerged. Our comprehensive study of potential IME signatures of triazole antifungals showed that the typology and number of IMEs varied depending on the medication. The majority of results were in line with the PI and previous studies except for hallucinations, osteochondritis, and prolonged ECG QT. Results based on the FAERS database can only suggest that these IMEs were reported in triazole antifungals, and evidence of causality requires extra studies, such as pharmacodynamic and pharmacokinetic plausibility, and pharmacoepidemiological studies, and other safety studies.
Supplemental Material
sj-docx-1-taw-10.1177_20420986221143266 – Supplemental material for Safety of triazole antifungals: a pharmacovigilance study from 2004 to 2021 based on FAERS
Supplemental material, sj-docx-1-taw-10.1177_20420986221143266 for Safety of triazole antifungals: a pharmacovigilance study from 2004 to 2021 based on FAERS by Shuang Chai, Jing-Lun Zhan, Li-Mei Zhao and Xiao-Dong Liu in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The authors thank all the contributors of the FDA Adverse Event Reporting System (FAERS) database, and the 345 Talent Program of Shengjing Hospital and Outstanding Scientific Fund of Shengjing Hospital for the funding support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
