Abstract
Introduction:
Many older adults are affected by multimorbidity and subsequent polypharmacy which is associated with adverse outcomes. This is especially relevant for frail older patients. Polypharmacy may be reduced via deprescribing. As part of the complex intervention in the COFRAIL study, we developed a deprescribing manual to be used by general practitioners (GPs) in family conferences, in which GPs, patients and caregivers jointly discuss treatments.
Methods:
We selected indications with a high prevalence in older adults in primary care (e.g. diabetes mellitus, hypertension) and conducted a literature search to identify deprescribing criteria for these indications. We additionally reviewed clinical practice guidelines. Based on the extracted information, we created a deprescribing manual which was then piloted in an expert workshop and in family conferences with volunteer patients according to the inclusion and exclusion criteria of the study protocol.
Results:
Initially, 13 indications/topics were selected. The literature search identified deprescribing guides, reviews and clinical trials as well as lists of potentially inappropriate medication and systematic reviews on the risk and benefits of specific drugs and drug classes in older patients. After piloting and revisions, the deprescribing manual now covers 11 indications/topics. In each chapter, patient- and medication-related deprescribing criteria, monitoring and communication strategies, and information about concerns related to the use of specific drugs in older patients are provided.
Discussion:
We found varying deprescribing strategies in the literature, which we consolidated in our deprescribing manual. Whether this approach leads to successful deprescribing in family conferences is being investigated in the cluster-randomised controlled COFRAIL study.
Plain Language Summary
Many older adults suffer from chronic diseases and take multiple medications concurrently. This can lead to side effects and other undesired events. We developed a manual to help doctors identify which medications can be withdrawn, so that they can discuss this with their patients. This manual was used in the COFRAIL study where doctors, patients and caregivers met in family conferences to discuss their preferences and decide together how future treatments should be handled. The manual contains information on common medications, symptoms and diseases in older patients such as diabetes and high blood pressure. Before the manual was used in the study, it was tested by volunteer patients and their doctors and caregivers to make sure that it is user-friendly.
Key Points
A structured manual was developed to support deprescribing of medicines in frail, older adults in primary care. After piloting with general practitioners and patients, the manual covers 11 indications and/or drug classes, respectively, describing step-by-step the identification of potential drug candidates for deprescribing, the discussion with patient and caregivers as well as monitoring advice for suspected withdrawal effects.
Introduction
Almost half of the population aged 65 years and above is exposed to polypharmacy, usually defined as five or more different medications.1,2 Polypharmacy itself is associated with an increased risk for medication-related problems such as drug–drug interactions, prescribing cascades and potentially inappropriate medications as well as for adverse outcomes such as hospitalisations, falls and mortality.1,3 In addition, frail elderly persons have an even higher risks for adverse outcomes. 4
Different interventions to optimise polypharmacy in older adults have been tested, most of them include a medication review, which is often supported by pharmacists. 5 Medication reviews usually address the identification of drug–drug and drug–disease interactions, dosing errors and potentially inappropriate medications and other drug-related problems, for example, tablet-splitting and swallowing problems.
One approach to reduce polypharmacy is deprescribing, which has been defined as a ‘systematic process of identifying and discontinuing drugs in instances in which existing or potential harms outweigh existing or potential benefits [. . .]’. 6 Moreover, it is ‘the process of withdrawal of an inappropriate medication, supervised by a health care professional with the goal of managing polypharmacy and improving outcomes’. 7 Barriers for deprescribing have been described from the perspective of physicians, patients and caregivers. 8 Uncertainties and knowledge gaps are reinforced, for example, by the scarcity of disease-specific clinical guidelines also addressing the possibility of deprescribing under certain circumstances9,10 as well as the limited number of randomised controlled trials (RCTs) providing evidence for safe deprescribing of specific drugs or drug classes, for example, for antihyperglycemics. 11 However, both patients and caregivers express that they are open to deprescribing one or more medications if this is the advice of the treating physician. 12
To facilitate deprescribing in the COFRAIL study, we developed a deprescribing manual to be used in the context of family conferences. The COFRAIL study is a cluster-randomised intervention trial conducted in primary care practices in Germany, where family conferences are tested as a means to discuss patients’ preferences and the issue of deprescribing in frail older patients exposed to polypharmacy. Briefly, family conferences provide a setting in which physicians, patients and caregivers can jointly discuss treatment options and are engaged in shared decision-making. 13 While a variety of deprescribing tools have been published, 14 none of them cover the relevant requirements for the COFRAIL study and primary care in Germany: for example, medication-specific criteria for common indications in the study population, focus on both potentially inappropriate medication and preventive medication as well as suggestions for patient communication. Thus, in addition to other study tools, 15 a deprescribing manual for general practitioners (GPs) has been developed for the aforementioned conditions. It aims to support the deprescribing process in its several steps: identification of an indication for discontinuing a medication, prioritising the medications targeted for withdrawal, communication with patients and caregivers as well as monitoring for adverse effects of deprescribing. 16
Methods
Context of the development of the deprescribing manual
The deprescribing manual was created as part of a complex intervention in the COFRAIL study. 17 The study’s population are frail patients (according to the Canadian Study of Health and Aging Clinical Frailty Scale 18 ) above the age of 70 years, living at home and receiving at least five different drugs per day.15,17 The complex intervention consists of three family conferences in which deprescribing, nonpharmacological treatment options and patients’ needs are discussed. The deprescribing manual is used by GPs in the intervention group to facilitate the process of deprescribing within the family conferences. Patients in the control group receive regular care. The primary outcome of the study is the rate of hospitalisations after 12 months.
Target groups in terms of use and application
The deprescribing manual was developed to be used by GPs to optimise the medication of their frail older patients in the context of family conferences.
Development process
For the development of the deprescribing manual we used the ‘key elements of CPG [Clinical Practice Guideline] development’, identified by Turner

Steps taken in each development phase.
Phase 1 – preparing for DPM development
In the first phase, we had to determine which topics should be covered in the deprescribing manual, while its scope was already determined by the inclusion criteria of the COFRAIL study, 17 that is, frail older patients.
In order to select medical indications and/or chronic diseases to be covered in the manual, we made use of a published analysis of claims data of a German statutory health insurance. This analysis ranked chronic diseases/indications by their prevalence in a sample of 123,224 primary care patients aged 65 years and older. 20 Indications to be included in the manual needed to have potential for deprescribing in addition to being highly prevalent in older people (i.e. ‘hypertension’ was included, ‘severe vision reduction’ was not considered). During the development process of the manual, recommendations from GPs during pre-testing, an expert workshop and the final piloting were considered to select the final indications/topics for deprescribing. 21 These substeps are described in more detail in the section on phase three.
Phase 2 – systematically reviewing the evidence
Among the key elements for the second phase are ‘establishing clinical questions’ and ‘systematic searching’. In the context of the COFRAIL study, the main clinical question for the development of the manual was ‘which medications can be deprescribed for each of the selected indications’. In 2018, we conducted a literature search in PubMed and Google Scholar focussing on evidence for deprescribing drugs in our selected indications using relevant mesh terms and keywords, for example, ‘deprescribing, deprescribe, aged’. We focussed our search on available systematic reviews for relevant information on benefits and patient safety of deprescribing, as well as on deprescribing trials, clinical practice guidelines and further guidance such as lists of potentially inappropriate medications (PIM). Deprescribing recommendations and other potentially relevant data and information were extracted and collected in tables corresponding to each indication.
Phase 3 – drafting the DPM
Key elements of the third phase are ‘developing recommendations’ and ‘consulting on the draft CPG’ among others. The information collected in the literature search was used to write a first draft of the manual, where each indication was covered in one chapter and deprescribing recommendations were summed up in plain text. According to recommendations obtained through peer review and group discussions of a subset of the members of the COFRAIL study group (two clinical pharmacologists, a pharmacist and two GPs), the draft manual was slightly adapted to the requirements of its target group and to the setting of a family conference. This manual should be used to support the identification of drugs to discontinue as well as the communication and process of stopping drugs.
We piloted the resulting version in a workshop with 41 teaching GPs affiliated with the institutes of general practice of the universities of Düsseldorf and Rostock participating in the COFRAIL trial. Briefly, in their own practices, these GPs act as educators for medical students and young practitioners and are regularly offered workshops and training in didactics for this function. Participants were asked to describe enablers for and barriers to deprescribing in a group discussion, to rate the relevance of all indications covered by the manual using a three-point Likert-type scale and to name missing topics. Decisions about the inclusion of named topics were made via informal consensus within the study group in the aftermath of the workshop, taking the setting of the study and the study population into account. The practicability of the deprescribing manual was tested in this workshop by applying it to a case study of a frail older woman with polypharmacy. The case study is available in the Supplementary Material (Supplement 2).
After the workshop, further changes to the manual were adopted as suggested by the GPs.
The second draft of the manual was piloted by four GPs at the two study sites in Düsseldorf and Rostock in eight pilot family conferences with volunteer patients and relatives. GPs, patients and/or their caregivers were interviewed via telephone regarding the usability of the manual (GPs) or their experience (patients, caregivers). Results obtained were further used to improve the manual, for example, formatting and to add more detailed information on preparations for the family conference concerning the medication and deprescribing to other study materials. To report necessary and adequate information about the development process of the deprescribing manual, we used the ‘Conference on Guideline Standardisation Checklist for Reporting Clinical Practice Guidelines’, 22 where applicable (Supplement 3).
Phase 4 – reviewing the DPM
The fourth and last phase consists of the key element ‘planning for evaluating the impact, revising and updating of the DPM’. Usability and impact of the deprescribing manual were investigated as part of the complex intervention of the COFRAIL study.
Results
A brief overview over the development process is provided in Table 1.
Results of the development phases.

DPM, DePrescribing Manual.
Phase 1 – preparing for DPM development
We identified 13 indications/topics for the first draft of the manual (Supplement 4). Certain indications, for example, cardiovascular and cerebrovascular ischaemic diseases, have a significant overlap in the medication used for their prevention and treatment (i.e. statins and platelet inhibitors) and were thus combined leading to a chapter on cardiovascular/cerebrovascular primary and secondary prevention.
Phase 2 – systematically reviewing the evidence
The literature search identified PIM lists and deprescribing guides, reviews and clinical trials, examples are given in Table 2 (Supplement 5 contains the full bibliography of the deprescribing manual and its appendix).
Examples of the types of literature used to develop the deprescribing manual.

PIM, potentially inappropriate medications.
Phase 3 – drafting the DPM
In the first draft of the manual 13, indications/topics were covered. After the workshop, 11 chapters remained for the final manual. Inclusion or exclusion of certain indications/topics was determined via expert consensus within the study group (two clinical pharmacologists, a pharmacist and two GPs), and the reasoning for each decision is provided in Supplement 4. Thus, the deprescribing manual consists of the chapters listed in Box 1.
Indications/topics covered in the deprescribing manual.

Originally, each of the chapters started with a short prose section describing treatment and deprescribing criteria for the indication in question, followed by information on adverse drug reactions of those drugs where deprescribing could be considered. In addition, information on drug classes frequently used as treatment and suggestions for monitoring the patient after deprescribing were listed. Considering suggestions from pre-testing and the workshop, we transformed the prose section describing patient and medication criteria and the section on monitoring criteria into bullet point lists, whereas the content remained unchanged. As some of the GPs requested support for communication with patients and specialists, we added examples of arguments for deprescribing including benefits but also potential harms. An excerpt from the chapter on gout can be found in Supplement 6. A new section was introduced containing short extracts from the literature supporting our recommendations.
In order to allow for easy and fast access to relevant indications, we created deprescribing algorithms containing most (but not all) information provided in the written form. For example, specific information on drug classes was usually not included. Each algorithm starts with patient criteria and its path can be followed to either a stopping point (e.g. ‘no deprescribing’) or a monitoring loop. The algorithm for gout is presented in Figure 2.
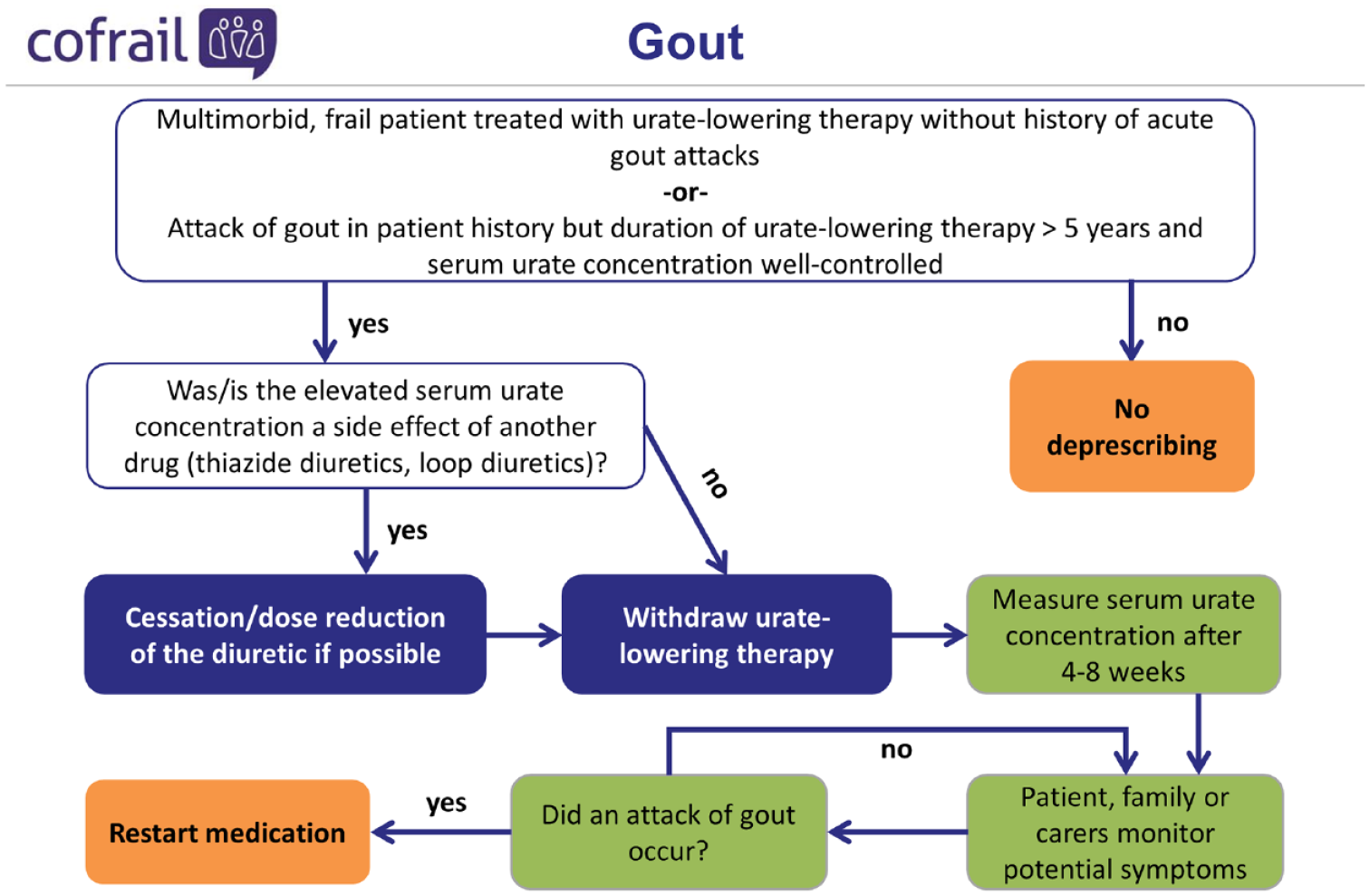
Deprescribing algorithm for gout.
For another quick access point, we summarised the medication criteria for each chapter in a chart and placed it directly after the introduction/user guide presented at the beginning of the manual. The full table is provided in Supplement 7.
The piloting of the second draft (including the deprescribing algorithms and the deprescribing chart) only resulted in small changes to its format, for example, colour coding of the chapters. Regarding content and accessibility, participants of the last pilot testing reported high satisfaction with both.
Phase 4 – reviewing the DPM
Data of the COFRAIL study are being analysed to evaluate the deprescribing manual’s impact as part of the complex intervention. Results will be presented in a number of planned publications.
Discussion
We present a deprescribing manual for frail older patients (⩾70 years
17
) to be used by GPs in the context of family conferences. The manual should also allow for a less formal process outside of family conferences, where patients and caregivers are involved. The topics were selected in two steps: first, an analysis of most frequent indications, and second, an assessment of GPs with respect to the relevance for their needs. The results of the selection process show that even frequent indications were sometimes not suitable for deprescribing in primary care, whereas some drug classes seemed to have a high potential for being withdrawn. Although we used a somewhat different approach for identification of relevant topics than Farrell
As described by Reeves, Scott and others, the first steps towards deprescribing are a review of the patient’s medication and the identification of medicines where risks outweigh benefits for the individual patient.6,31,33,34 We thus designed chapters providing criteria to identify patients who may benefit from deprescribing and the drugs which may be discontinued. In addition, the introduction of the manual contains a brief practical guidance on medication reviews.
Overall, there are different approaches and tools for deprescribing. For example, the Good Palliative-Geriatric Practice algorithm34,35 asks questions concerning the validity of the indication, risks and benefits in the individual patient and potential side effects. It can be applied to a patient’s entire medication regimen, but requires the user to have access to extensive knowledge about the patient, the evidence-based use of each medication and its side-effect profile. A group of Canadian researchers, however, developed deprescribing guidelines for specific drugs and drug classes.29,36–39 These guidelines provide evidence-based recommendations which the user may apply. However, the number of drugs for which they are available is limited and thus it is unlikely that a patient’s entire medication regimen is covered. The methodology for developing deprescribing guidelines requires either pre-existing systematic reviews fitting the scope of the guideline or the performance of a systematic review during guideline development. 40 Thus, establishing deprescribing guidelines is time-consuming and not necessarily feasible as part of a trial. PIM lists name drugs which should not be prescribed to older patients because of a negative risk-benefit-ratio in these patients.23–25 PIM lists contain a variety of drugs and drug classes but while a literature review is often conducted during their development, the final rating of the drugs as inappropriate is based on expert consensus. 41 In addition, they do not cover treatment concepts in frail older people such as higher blood pressure targets 42 and the non-PIM medication used to achieve these goals. In summary, there are a variety of advantages and disadvantages of each approach towards deprescribing. We attempted to create a comprehensive and user-friendly deprescribing manual for GPs in the context of family conferences by incorporating strategies and data from these different approaches.
Different strategies exist not only for determining which medication to target for deprescribing but also for the number of medications to be discontinued at any given time. Garfinkel
43
employed a poly-deprescribing approach where they withdrew as many medications as possible at the same time, unless several medications were prescribed for the same indication in which case they were deprescribed sequentially. Scott
Whether deprescribing chronic medication in primary care and nursing homes is successful has been the subject of several RCTs described in a systematic review by Thio
While we took ‘key elements of CPG development’ 19 into account, it was not possible to consider all of them in the development of the deprescribing manual within the scope of the COFRAIL study. For example, the involvement of patients was limited to eight pilot family conferences and while we conducted an extensive literature search, we did not perform systematic reviews. The scope of the literature search, the implementation and evaluation of the deprescribing manual’s effect were pre-determined by its place within the boundaries of the study. To further develop and refine the manual, systematic reviews focussing on deprescribing specific medications should be conducted.
Finally, Blom
Strengths and limitations
Due to the timeline of the COFRAIL study, it was not possible to conduct systematic reviews for each indication covered in the deprescribing manual. In addition, we conducted our literature search in PubMed and Google Scholar, which is why it may be that we missed relevant literature which is only indexed in other databases. A strength of our approach was the strong focus on usability in primary care and the involvement of GPs and to some extent patients in the development process. Using the key elements of CPG development as described by Turner
Conclusion
We developed a deprescribing manual whose impact will be investigated as part of a complex intervention in the COFRAIL study. In case of a positive effect on patient-relevant endpoints, a consensus-based process (e.g. a Delphi survey) within the relevant medical societies is planned to enable the publication of consensus-based deprescribing recommendations.
Supplemental Material
sj-docx-1-taw-10.1177_20420986221122684 – Supplemental material for Development of a deprescribing manual for frail older people for use in the COFRAIL study and in primary care
Supplemental material, sj-docx-1-taw-10.1177_20420986221122684 for Development of a deprescribing manual for frail older people for use in the COFRAIL study and in primary care by Nina-Kristin Mann, Sven Schmiedl, Achim Mortsiefer, Veronika Bencheva, Susanne Löscher, Manuela Ritzke, Eva Drewelow, Gregor Feldmeier, Sara Santos, Stefan Wilm and Petra A. Thürmann in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The COFRAIL study group: Stefan Wilm (principal investigator PI), Achim Mortsiefer (PI), Sara Santos, Susanne Löscher, Yekaterina Pashutina (Institute for General Practice, Centre for Health and Society, Medical Faculty, Heinrich-Heine-University Düsseldorf); Attila Altiner, Anja Wollny, Eva Drewelow, Manuela Ritzke (Institute of General Practice at the University Medical Centre Rostock); Petra Thürmann, Sven Schmiedl, Nina-Kristin Mann, Veronika Bencheva, Matthias Gogolin (Department of Clinical Pharmacology, School of Medicine, Faculty of Health, Witten/Herdecke University); Gabriele Meyer, Steffen Fleischer, Jens Abraham (Institute for Health and Nursing Science, Medical Faculty, Martin Luther University Halle-Wittenberg); Andrea Icks, Markus Vomhof, Joseph Montalbo (Institute for Health Services Research and Health Economics, Centre for Health and Society, Faculty of Medicine, Heinrich-Heine-University Düsseldorf); Birgitt Wiese (WG Medical Statistics and IT-Infrastructure, Institute of General Practice, Hannover Medical School)
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
