Abstract
Background:
Polypharmacy is common in hospitalized older adults. Deprescribing interventions are not well described in the acute-care setting. The objective of this study was to describe a hospital-based, patient-centered deprescribing protocol (Shed-MEDS) and report pilot results.
Methods:
This was a pilot study set in one academic medical center in the United States. Participants consisted of a convenience sample of 40 Medicare-eligible, hospitalized patients with at least five prescribed medications. A deprescribing protocol (Shed-MEDS) was implemented among 20 intervention and 20 usual care control patients during their hospital stay. The primary outcome was the total number of medications deprescribed from hospital enrollment. Deprescribed was defined as medication termination or dose reduction. Enrollment medications reflected all prehospital medications and active in-hospital medications. Baseline characteristics and outcomes were compared between the intervention and usual care groups using simple logistic or linear regression for categorical and continuous measures, respectively.
Results:
There was no significant difference between groups in mean age, sex or Charlson comorbidity index. The intervention and control groups had a comparable number of medications at enrollment, 25.2 (±6.3) and 23.4 (±3.8), respectively. The number of prehospital medications in each group was 13.3 (±4.6) and 15.3 (±4.6), respectively. The Shed-MEDS protocol compared with usual care significantly increased the mean number of deprescribed medications at hospital discharge and reduced the total medication burden by 11.6 versus 9.1 (p = 0.032) medications. The deprescribing intervention was associated with a difference of 4.6 [95% confidence interval (CI) 2.5–6.7, p < 0.001] in deprescribed medications and a 0.5 point reduction (95% CI −0.01 to 1.1) in the drug burden index.
Conclusions:
A hospital-based, patient-centered deprescribing intervention is feasible and may reduce the medication burden in older adults.
Introduction
Polypharmacy, defined as at least five prescribed medications, 1 is common among hospitalized older adults 2 and is associated with multiple poor health outcomes such as decreased medication adherence, 3 increased adverse drug events, 3 and increased health care utilization.4,5 An alarming 45% of older hospitalized patients are discharged on five or more medications, many of which may be harmful or unnecessary.6,7
Polypharmacy and a variety of drug indices that quantify drug burden8,9 have been found to have substantial effects on the development and progression of geriatric syndromes such as long-term cognitive impairment, 10 delirium, 11 falls, 12 frailty, 13 urinary incontinence, 14 and unintentional weight loss. 15 These same syndromes are prevalent in hospitalized older adults,16,17 negatively impact health outcomes and reduce quality of life, even when controlling for illness severity. Recently published research identified 513 medications associated with geriatric syndromes (MAGS) that are commonly prescribed to older adults. 18 Moreover, in a sample of inpatients discharged to skilled nursing facilities (SNFs), patients left the hospital with an average of six MAGS that could be contributing to new or worsening geriatric syndromes in the postacute care period.
Recent attention toward polypharmacy has led to advancements in deprescribing interventions. 19 Few studies, however, have initiated deprescribing interventions in the hospital setting, and only a subset of these interventions incorporated patient and provider preferences as a key element. 19 No studies have focused on high-risk older patients who require inpatient postacute care following their acute care hospitalization. This is a growing population, and allows for continued post-deprescribing monitoring, and potential for continued deprescribing. Moreover, previous studies have focused solely on disease-specific or medication-specific deprescribing such as reducing potentially inappropriate (e.g. antipsychotics) or unnecessary (e.g. proton pump inhibitors) medications. 20 One Australian study 21 evaluated a hospital-based deprescribing intervention designed to reduce total drug burden; however, no published interventions have targeted a broad array of medications, including those associated with geriatric syndromes.
The acute care and inpatient postacute care settings provide the opportunity to review medicines, engage in patient-preference discussions, monitor for drug withdrawal symptoms, and coordinate changes among providers. In addition, it is during this period that both the number of medications and geriatric syndromes are likely to increase. 22 Taken together, these findings suggest that deprescribing among hospitalized older patients to reduce overall drug burden, potentially inappropriate medications (PIMs) and MAGS has tremendous healthcare potential. In this pilot feasibility study, we describe the key features of an algorithm-driven deprescribing framework (Shed-MEDS) that can be implemented in the acute care setting. We also report the pilot results of Shed-MEDS on reducing total medication burden, including medications associated with geriatric syndromes.
Methods
Study design, setting, and population
We performed a single-site feasibility pilot that included hospital patients admitted to a private, nonprofit, tertiary care teaching hospital located in Nashville, Tennessee between April 2015 and June 2015. We implemented a deprescribing framework (Shed-MEDS) among 20 patients as part of an ongoing quality improvement effort aimed at improving care transitions from the hospital to the postacute care setting. We compared outcomes with 20 patients who were part of the quality improvement effort but who did not receive the deprescribing protocol. We selected all patients from a convenience sample of participants enrolled in a larger Centers for Medicare and Medicaid Services (CMS) funded quality improvement project to improve the care transition from acute care to SNFs. As this was part of a quality improvement project, neither intervention nor control patients were randomized. Inclusion criteria required participants to be Medicare beneficiaries, at least 65 years of age, receiving five or more prescribed medications, and admitted to the hospital with an intended discharge to one of 21 SNFs in the region who had partnered with the medical center as part of the larger CMS study. All prescriptions counted toward the inclusion criterion of five or more medications, including both inhospital and prehospital medications continued during hospitalization. Patients referred to a partner SNF were targeted for this deprescribing pilot study to mitigate potential safety risks related to medication withdrawal. We excluded patients if their anticipated hospital stay was less than 48 h, anticipated mortality was less than 30 days, or they were deemed too ill to participate (e.g. palliative care unit, hospice referral or diagnosis of stage IV metastatic cancer), or were noncommunicative (e.g. severe cognitive impairment or persistent delirium) and did not have a surrogate available. Vanderbilt University’s Institutional Review Board determined the project to be a quality improvement initiative and waived the requirement for written consent. All participants and their surrogates, however, were enrolled using an informed verbal consent process and had the right to refuse participation.
Deprescribing framework
We developed an evidence-based, structured deprescribing framework (Shed-MEDS) to reduce the total number of medications among a subset of patients who were already receiving standardized geriatric assessments and medication reconciliation as part of the larger CMS study. We based Shed-MEDS on a framework by Holmes and colleagues that considers a combination of patient and disease factors, including patient goals of care, appropriate treatment targets, the duration of treatment required for benefit and patients’ life expectancy. 23 We also incorporated medication-specific factors from Scott’s framework 24 for minimizing PIMs use. This includes factors such as drug-specific safety profiles with the potential for drug–drug and drug–disease interactions. Finally, we incorporated patient and prescriber preferences into this model as a key component to inform final deprescribing actions. The deprescribing protocol was implemented in multiple phases to the point of hospital discharge, as described in the intervention section.
Shed-MEDS deprescribing intervention
Shed-MEDS was implemented by an interdisciplinary team consisting of clinical pharmacists, physicians, geriatric nurse practitioners, and qualified research personnel. The MEDS acronym defines the four major phases of the deprescribing framework (Table 1): medication history confirmed for accuracy through chart reviews, patient interviews, and pharmacy calls; evaluate the medication list to identify deprescribing targets and priorities; decide on appropriate deprescribing through a standardized patient-preference interview; synthesize and communicate deprescribing recommendations to prescribers.
Shed-MEDS hospital-based patient-centered deprescribing framework.
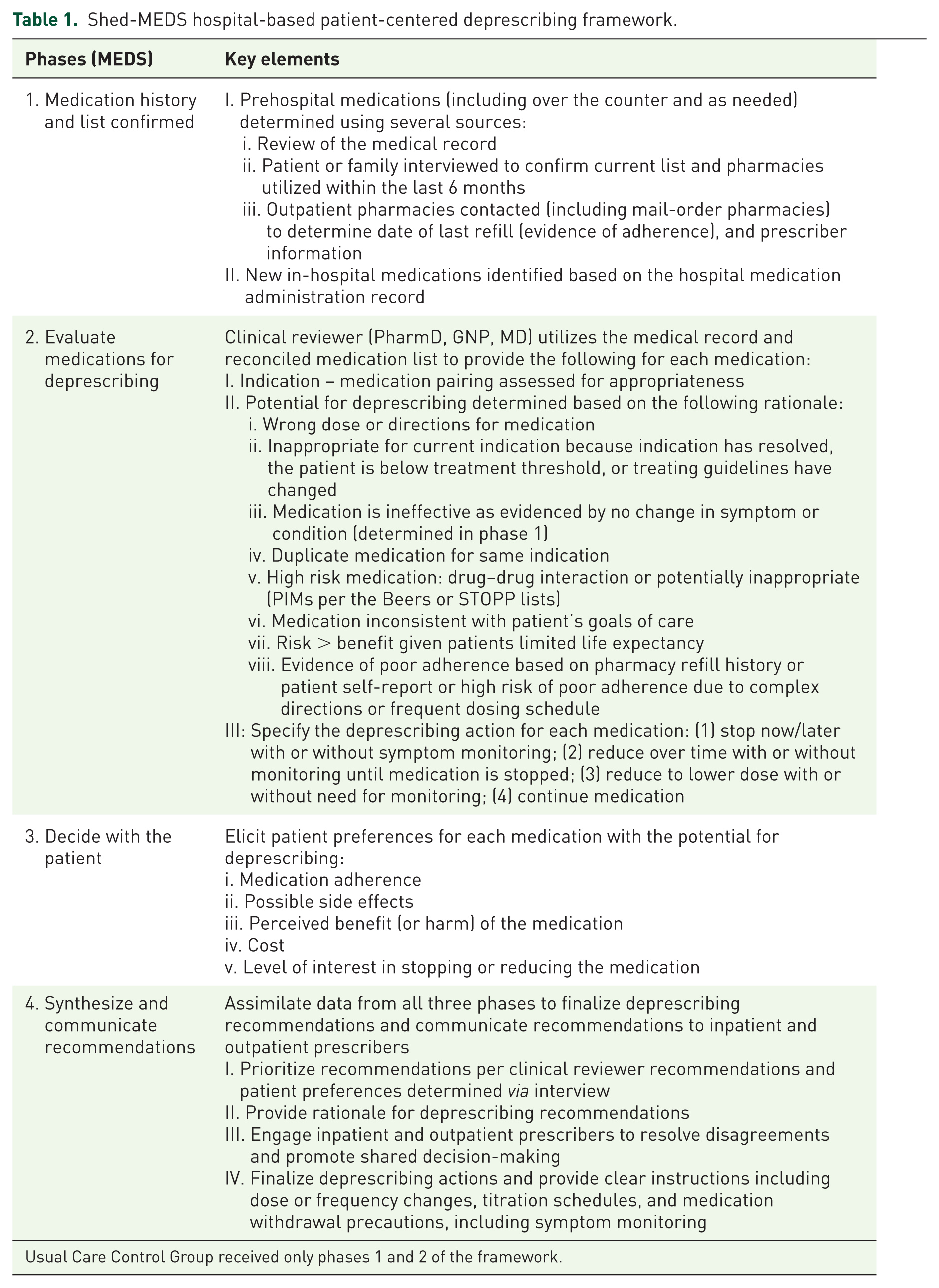
Usual Care Control Group received only phases 1 and 2 of the framework.
Outcome measures
The primary feasiblity outcome measure was the total number of medications deprescribed at hospital discharge compared with the number of medications at hospital enrollment. Hospital enrollment medications reflected any medication with the potential to be continued as a chronic medication at hospital discharge or restarted upon returning home. This included prehospital medications and inhospital medications not on the prehospital medication list. Importantly, it also included medications identified via patient interview and pharmacy refill records that were not on the original prehospital medication list [including over-the-counter and prescribed ‘as needed’ (PRN) medications]. Deprescribed was defined as termination or dose reduction of a medication at the time of hospital discharge. The rationale for including dose reductions in the definition of deprescribing was primarily related to the potential impact on the patient’s anticholinergic and sedative drug burden, which was quantified at enrollment and discharge using a drug burden index (DBI). 8 PIMs were defined as medications contained on the Beers 25 or STOPP 26 lists. MAGS were defined as medications with side effects that may contribute to common geriatric conditions such as delirium, falls and unintentional weight loss. 18
We additionally examined the impact that the patient-preferences interview had on the initial deprescribing recommendations. Initial recommendations were made following medical record review, in the absence of, and prior to, patient and provider input. We then tracked any changes between final discharge medications (after the patient preferences interview) compared with the initial recommendations. We classified each change: those that were changed from deprescribe to continue, those changed from continue to deprescribe, and those where both agreed to deprescribe (or continue the medication), however the final dose or tempo of deprescribing changed (e.g. from stop to decrease dose, or from start as an outpatient to start now).
Statistical analysis
Baseline characteristics and outcomes were compared between the intervention and usual care control groups using simple logistic or linear regression for categorical and continuous measures, respectively. Additionally, propensity score ‘matching weights’ were used to compare groups in a weighted fashion at baseline. 27 This method reweights the convenience sample such that participant allocation resembles that of a randomized trial, and thus reduces the bias in estimating the effect of intervention. Propensity scores were estimated using logistic regression of the intervention assignment variable onto patient factors including age, sex, race, Charlson comorbidity index score, count of medications prior to enrollment, count of medications prescribed while admitted to the hospital, and DBI.
Propensity score weighted linear regression analysis was used to evaluate the effect of intervention on each outcome, adjusting for the corresponding baseline value (e.g. DBI), hospital length of stay, and intervention status. 28 The adjusted effect of intervention was summarized using its estimate and Wald-type 95% confidence intervals (CIs) and p values. For all regression models, the effects of quantitative factors (e.g. hospital length of stay) were modeled linearly, and graphical regression diagnostics (partial residual plots; normal qq plots) were used to examine the appropriateness of modeling assumptions. Finally, we calculated median and interquartile ranges (IQRs) of the medication changes made following the patient preferences interview compared with pre-interview recommendations. We did this for the overall number of changes, and for each category of change described above.
Results
Patient characteristics
In unweighted comparisons, there were no significant differences in the participant characteristics of the intervention and control groups at the time of enrollment, with the exception of hospital length of stay, which was longer in the intervention group (Table 2). However, the two groups were comparable based on the Charlson comorbidity index. 29 Participants in intervention and control groups had been in the hospital a median of 3.5 and 4 days at the time of enrollment, which occurred after a referral to a SNF. The median (IQR) number of prescribed medications at enrollment was 23 (19–29) and 22 (20–24) for intervention and control groups, respectively, with a comparable number of prehospital medications in both groups but a significantly higher number of inhospital medications in the intervention group in the unweighted comparisons (Table 2). In propensity score weighted comparisons, there were no significant differences in participant characteristics or prescribed medications prior to intervention (Table 2).
Participant characteristics and intervention outcomes: unweighted and weighted comparisons.
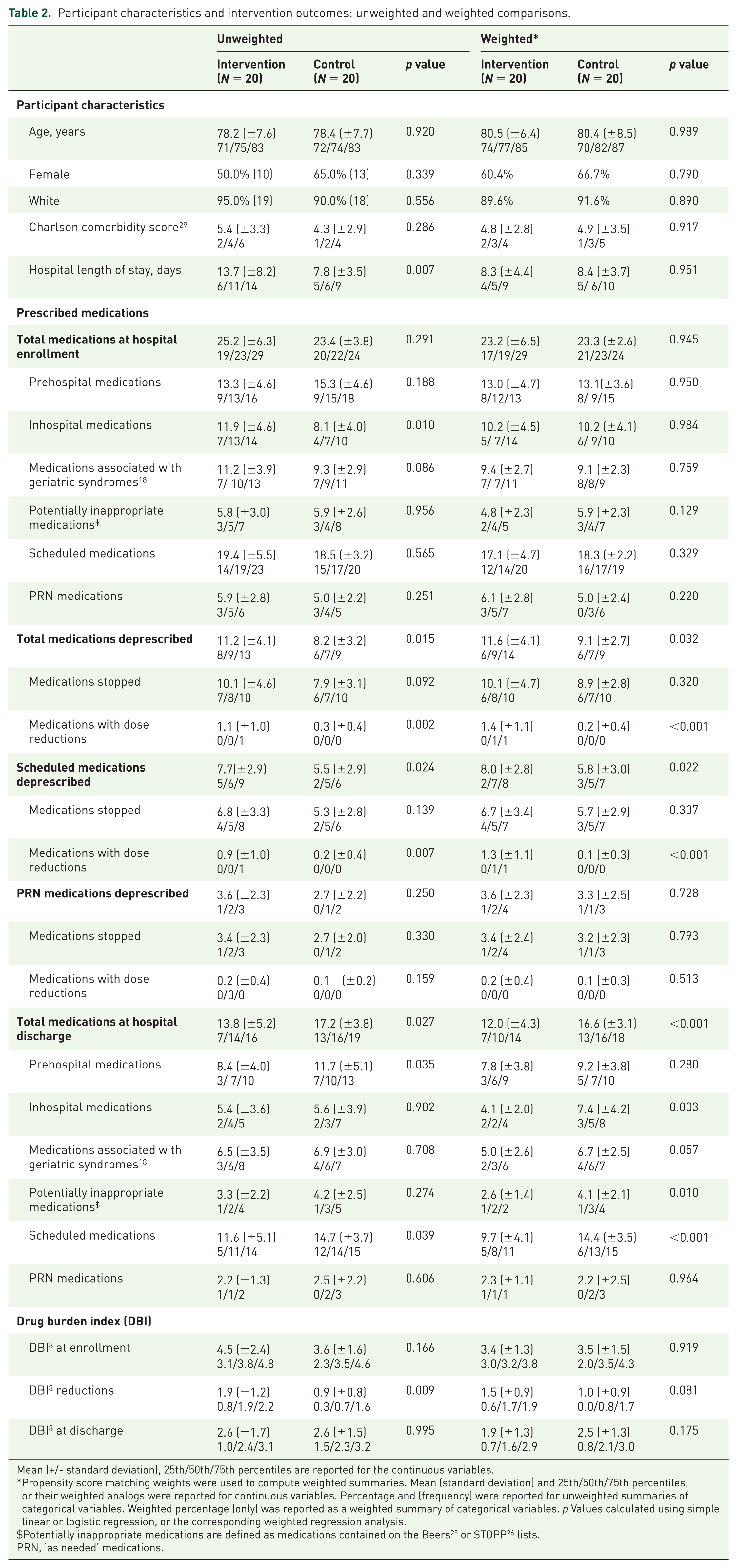
Mean (+/- standard deviation), 25th/50th/75th percentiles are reported for the continuous variables.
Propensity score matching weights were used to compute weighted summaries. Mean (standard deviation) and 25th/50th/75th percentiles, or their weighted analogs were reported for continuous variables. Percentage and (frequency) were reported for unweighted summaries of categorical variables. Weighted percentage (only) was reported as a weighted summary of categorical variables. p Values calculated using simple linear or logistic regression, or the corresponding weighted regression analysis.
Potentially inappropriate medications are defined as medications contained on the Beers 25 or STOPP 26 lists.
PRN, ‘as needed’ medications.
Results of the intervention
The Shed-MEDS protocol, as compared with usual care, significantly increased the number of deprescribed medications at hospital discharge. As shown in Table 2, the mean number of medications deprescribed per patient was significantly higher in the intervention group compared with the control group (11.6 versus 9.1, p = 0.032). Scheduled, as compared with PRN medications, accounted for the majority of this difference. The results of the propensity score weighted linear regression showed that the intervention was associated with an overall reduction of 4.6 medications (95% CI 2.5–6.7, p < 0.001), a reduction in MAGS of 1.8 (95% CI 0.7–2.9, p = 0.002), and a reduction in PIMS of 0.9 (95% CI 0.1–1.7, p = 0.034) after adjusting for corresponding enrollment values and length of hospital stay. There was a reduction in the DBI score of 0.5 point (95% CI −0.01 to 1.1, p value = 0.052), which approached significance, after adjusting for the enrollment DBI score. Using residual diagnostics, there was no evidence of nonlinear associations in any model, nor was there evidence to warrant alternative regression methods (e.g. Poisson regression for the count outcomes).
Effect of patient engagement
Calls to pharmacies in phase 1 of the Shed-MEDS protocol (Table 1) revealed an average of 1.1 and 1.0 prehospital medications in the intervention and control groups, respectively, that were unknown to the hospital treatment team. These medications represent prescriptions with potential to be refilled following hospital discharge. Patient preference interviews identified numerous medications that patients were willing to deprescribe (e.g. due to lack of efficacy, unwanted side effects or cost) as well as potential barriers to deprescribing (e.g. belief that medicine is helpful, concerns about worsening of symptoms). The standardized patient preference interview resulted in a median of 10 adjustments (IQR 6.3–13) to the initial describing recommendations per patient (i.e. decision to continue, stop, dose or frequency adjustments). Table 3 categorizes the changes according to the direction of the change as compared with the initial deprescribing recommendations prior to patient engagement. The median times a decision was made to continue a medication that was initially recommended for deprescribing was two fewer than the number of deprescribed medications that were originally recommended to be continued.
Changes in deprescribing recommendations based on patient/provider preferences (intervention group only).
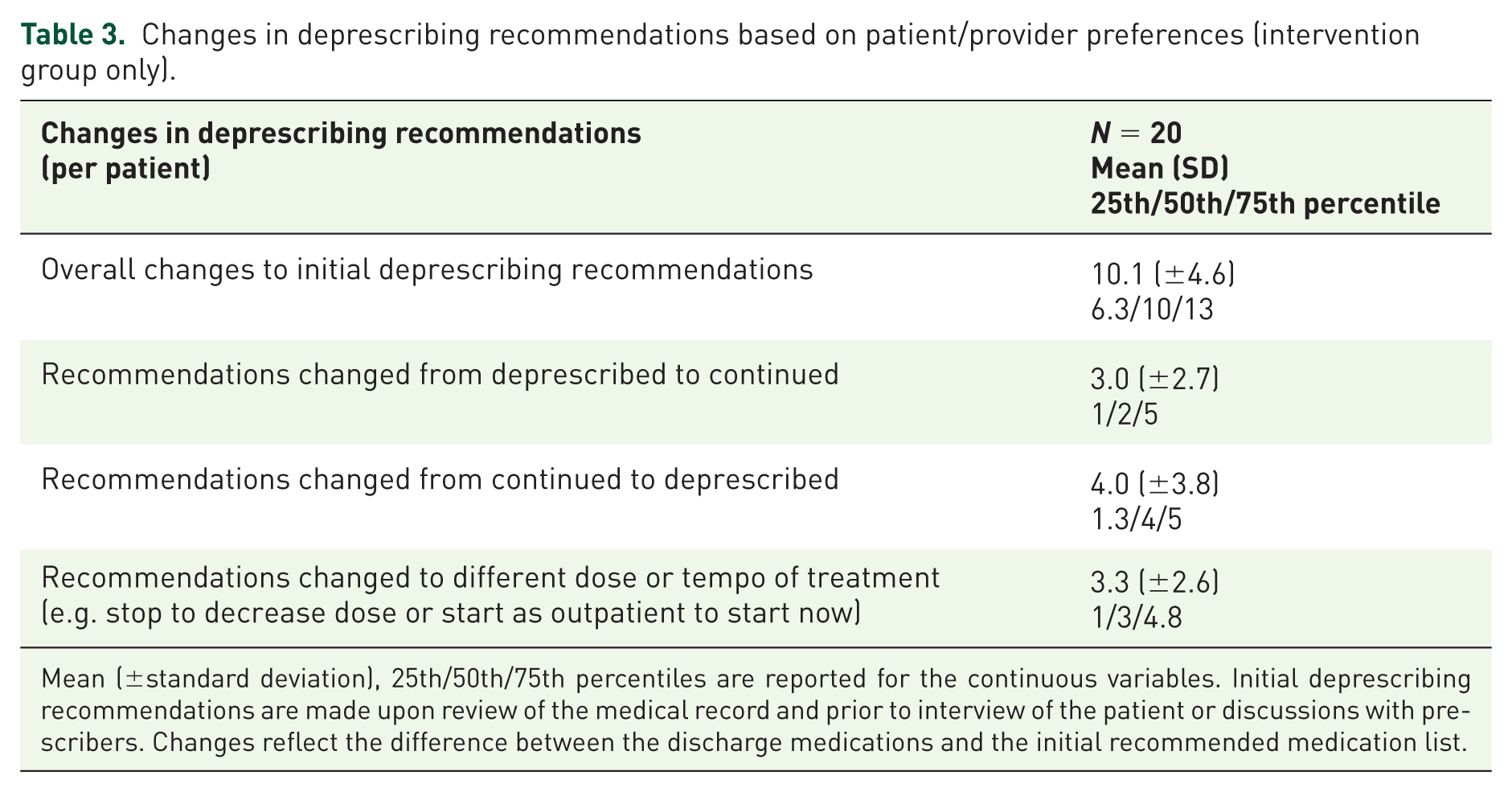
Mean (±standard deviation), 25th/50th/75th percentiles are reported for the continuous variables. Initial deprescribing recommendations are made upon review of the medical record and prior to interview of the patient or discussions with prescribers. Changes reflect the difference between the discharge medications and the initial recommended medication list.
Discussion
In this pilot, a hospital-based patient-centered deprescribing intervention (Shed-MEDS) reduced the total number of medications, number of PIMs, and number of MAGS at hospital discharge. An overall reduction in DBI also approached significance (p = 0.052). Desprescibing efforts have been described in prior studies; however, Shed-MEDS is an innovative hospital-based deprescribing framework that targets a broad array of medications and incorporates patient preferences. The large number of adjustments to the initial deprescribing recommendations after patient engagement underscores the potential for discrepancies between provider and patient preferences. 30
Older hospitalized patients with a planned discharge to SNFs experience substantial polypharmacy. The total number of medications at enrollment was over 20 regardless of intervention, more than half of which were prehospital medications that were not on the active inpatient medication list. Importantly, any one of these medications has the potential to be continued after discharge to SNF and beyond at home. In order to successfully deprescribe, it is important to not only focus on the current inpatient medications, but medications for which the patient has active prescriptions and refills that may be reinitiated at home unless there is an active intervention. For this reason, obtaining an accurate medication list, including pharmacy refills and patient history, is a critical first step in deprescribing.
Our pilot successfully deprescribed medications among patients with an intended discharge from the hospital to a SNF, who represent a cohort of patients at higher risk for loss of independence relative to patients discharged to home. 31 Within our sample, the majority of described medications were scheduled, and nearly half of the medications deprescribed had known associations with geriatric syndromes, which are rarely a medical priority for the primary hospital team. Only 58% of medications identified as MAGS overlapped with the American Geriatric Society’s 2015 Beer’s criteria. 25 Although the negative health effects of Beer’s medications are well known, our pilot intervention data suggest that inpatient providers are less likely to consider additional MAGS, which are, in turn, known to contribute to poor postdischarge health outcomes. 22 Broadening the scope of medications to include MAGS represents a further opportunity to potentially delay onset or progression of a number of geriatric syndromes. This feasibility pilot was too small to specifically examine outcomes beyond medication changes, however future trials should examine whether deprescribing PIMs and MAGs positively impact health outcomes, including geriatric syndromes.
The Shed-MEDS intervention was successful in deprescribing other medications as well. Half of the medications deprescribed were neither MAGSs nor on the Beer’s/STOPP list. Although not related to geriatric syndromes, these medications represent additional exposures to unnecessary medications that may have alternative side effects beyond geriatric syndromes and come with an unnecessary cost burden. 32 In addition, there is evidence that polypharmacy alone is a predictor of adverse drug events, 33 readmissions, 5 and future additional inappropriate prescribing. 34
Our pilot intervention additionally demonstrates the importance of involving the patient/surrogate in the decisionmaking process along with the outpatient providers and the hospital treatment team. Our deprescribing framework reinforces the practice of initiating treatment decisions by first understanding the patients’ treatment goals. The large number of changes to the initial deprescribing recommendations after patient engagement underscores the well described discrepancy between provider and patient goals of care. 30 Interestingly, the median number of changes that led to more new deprescribing actions was greater than those that led to fewer. This suggests a willingness of patients to reduce or stop medications when they are engaged in a conversation about the efficacy, side effects, and cost of their current medications. Moreover, the patient-preference interview responses suggest that hospitalized older adults are willing to both stop and titrate medications with clinical guidance and reassurance during their acute care stay.
Initiating deprescribing in the hospital setting provides distinct advantages over previously reported outpatient-based interventions. In contrast to the outpatient setting, our hospital-based intervention utilized widely available interdisciplinary staff (nurses, pharmacists, physicians, social workers) required for safe and effective broad-based deprescribing efforts. Additionally, the hospital stay affords the time for in-depth standardized patient-preference interviews necessary to overcome deprescribing barriers, incorporate outpatient and inpatient provider input, and aligns medication therapies with patients’ goals of care. 35 Moreover, deprescribing among patients transitioning to postacute care treatment facilities provides greater oversight to monitor for the unintended consequences of deprescribing, such as drug withdrawal or adverse events. As a result, the Shed-MEDS hospital-based intervention facilitated coordinated medication discontinuation across the continuum of acute and postacute care.
This pilot study is limited in generalizability as it was a nonrandomized, single site study among hospitalized older adults referred to SNFs in one geographic area. The use of a convenience sample from a larger quality improvement project and nonrandomized groups resulted in significant differences between intervention and control participants in unweighted comparisons; namely, intervention participants had a longer length of hospital stay and more in-hospital medications at the time of enrollment, allowing for greater time for deprescribing in the Shed-Meds group. The propensity score weighted analyses, however, adjusted for these group differences; and the Charlson comorbidity index score was comparable between the two groups. Another limitation is the intensive nature of the intervention itself. As implemented in this pilot study, the intervention requires a significant amount of time (estimated 2–3 h per patient). This time-intensive protocol, however, was targeted toward a patient population at high risk for poor clinical outcomes, not all hospitalized patients. Lastly, this pilot study was focused on the feasibility of performing intensive deprescribing among hospitalized patients transitioning to inpatient postacute care, and therefore, did not have the resources to determine long-term health effects or sustainability of the intervention following SNF discharge to home.
As awareness grows regarding the burden of geriatric syndromes in hospitalized adults, there is increasing attention toward the development of interventions that simultaneously address multiple chronic conditions. Although polypharmacy is strongly associated with geriatric syndromes, it is unknown whether reducing medications will improve geriatric syndromes or other health outcomes. Future investigation of similar hospital-based deprescribing interventions is needed to determine the feasibility and efficacy of addressing multiple geriatric syndromes within one intervention framework directed toward polypharmacy. A randomized controlled trial [ClinicalTrials.gov identifier: NCT02979353] is currently underway to address the effects of the Shed-MEDS deprescribing intervention on these and other important health outcomes.
Footnotes
Acknowledgements
The authors would like to acknowledge the following team members for their contribution to collecting data for this project: Christopher Simon Coelho, Emily A. Long, Carole H. Bartoo, and Dr Linda M. Beuscher. The contents of this publication are solely the responsibility of the authors and do not necessarily represent the official views of the US Department of Health and Human Services or any of its agencies, the National Center for Advancing Translation Science, the National Institutes of Health or the Department of Veterans’ Affairs
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Department of Health and Human Services, Centers for Medicare & Medicaid Services grant #1C1CMS331006 awarded to Principal Investigator, John F. Schnelle, PhD. Dr Vasilevskis was supported by the National Institute on Aging of the National Institutes of Health award K23AG040157 and the Geriatric Research, Education and Clinical Center (GRECC). Dr Bell was supported by NIA-K award K23AG048347-01A1. Dr Mixon is supported by a VA HSR&D Career Development Award (12-168). This research was also supported by CTSA award UL1TR000445 from the National Center for Advancing Translational Sciences.
Conflict of interest statement
Dr Kripalani has received stock/stock options from Bioscape Digital, LLC.
